236:
124:. The type of equilibrium defects in non-stoichiometric compounds can vary with attendant variation in bulk properties of the material. Non-stoichiometric compounds also exhibit special electrical or chemical properties because of the defects; for example, when atoms are missing, electrons can move through the solid more rapidly. Non-stoichiometric compounds have applications in
31:
509:
catalysed by solid catalysts. These considerations also highlight the fact that stoichiometry is determined by the interior of crystals: the surfaces of crystals often do not follow the stoichiometry of the bulk. The complex structures on surfaces are described by the term "surface reconstruction".
339:
and others). These materials are always iron-deficient owing to the presence of lattice defects, namely iron vacancies. Despite those defects, the composition is usually expressed as a ratio of large numbers and the crystals symmetry is relatively high. This means the iron vacancies are not randomly
518:
The migration of atoms within a solid is strongly influenced by the defects associated with non-stoichiometry. These defect sites provide pathways for atoms and ions to migrate through the otherwise dense ensemble of atoms that form the crystals. Oxygen sensors and solid state batteries are two
226:
is a small number (0.05 in the previous example) representing the deviation from the "ideal" formula. Nonstoichiometry is especially important in solid, three-dimensional polymers that can tolerate mistakes. To some extent, entropy drives all solids to be non-stoichiometric. But for practical
340:
scattered over the crystal, but form certain regular configurations. Those vacancies strongly affect the magnetic properties of pyrrhotite: the magnetism increases with the concentration of vacancies and is absent for the stoichiometric
614:, respectively, who in the 19th century advocated rival theories of the composition of substances. Although Dalton "won" for the most part, it was later recognized that the law of definite proportions had important exceptions.
496:
by metal oxides. The process operates via the transfer of "lattice" oxygen to the hydrocarbon substrate, a step that temporarily generates a vacancy (or defect). In a subsequent step, the missing oxygen is replenished by
376:
It is sometimes difficult to determine if a material is non-stoichiometric or if the formula is best represented by large numbers. The oxides of tungsten illustrate this situation. Starting from the idealized material
501:. Such catalysts rely on the ability of the metal oxide to form phases that are not stoichiometric. An analogous sequence of events describes other kinds of atom-transfer reactions including
211:
ions to balance the charge. The composition of a non-stoichiometric compound usually varies in a continuous manner over a narrow range. Thus, the formula for wüstite is written as
96:
chemical compounds. Since the solids are overall electrically neutral, the defect is compensated by a change in the charge of other atoms in the solid, either by changing their
606:
depending on whether their properties showed monotonic behavior with respect to composition or not. The term berthollide was accepted by IUPAC in 1960. The names come from
463:
and their analogs are well known to form in non-stoichiometric proportions. The non-stoichiometric phases exhibit useful properties vis-à-vis their ability to bind
45:
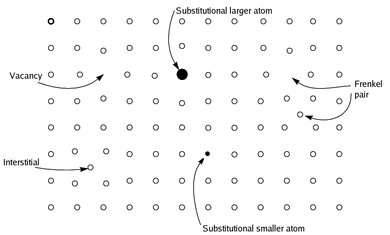
showing the regular square array of atoms on one face (open circles, o), and with these, places where atoms are missing from a regular site to create
976:
762:
735:
381:, one can generate a series of related materials that are slightly deficient in oxygen. These oxygen-deficient species can be described as
648:
Geng, Hua Y.; et al. (2012). "Anomalies in nonstoichiometric uranium dioxide induced by a pseudo phase transition of point defects".
262:
The monosulfides of the transition metals are often nonstoichiometric. Best known perhaps is nominally iron(II) sulfide (the mineral
947:
925:
872:
851:
794:
92:
Contrary to earlier definitions, modern understanding of non-stoichiometric compounds view them as homogeneous, and not mixtures of
85:); most often, in such materials, some small percentage of atoms are missing or too many atoms are packed into an otherwise perfect
227:
purposes, the term describes materials where the non-stoichiometry is measurable, usually at least 1% of the ideal composition.
550:
966:
903:
843:
819:
786:
710:
971:
595:
591:
546:
981:
303:
705:
N. N. Greenwood & A. Earnshaw, 2012, "Chemistry of the
Elements," 2nd Edn., Amsterdam, NH, NLD:Elsevier,
607:
368:< 0.58). This solid conducts hydrogen by virtue of the mobility of the hydrogen atoms within the solid.
36:
540:
285:
814:
5th Edn., pp. 65, 75, 99f, 268, 271, 277, 287, 356, 409, Oxford, OXF, GBR: Oxford
University Press,
133:
669:
598:
was shown to have merit for many solid compounds. Kurnakov divided non-stoichiometric compounds into
506:
446:
42:
898:
6th Edn., pp. 202, 271, 316, 777, 888. 897, and 1145, New York, NY, USA:Wiley-Interscience,
685:
659:
240:
70:
943:
921:
899:
868:
847:
839:
815:
790:
782:
758:
752:
731:
706:
378:
352:
86:
82:
66:
57:
in each case resulting in a material that is moved toward being measurably non-stoichiometric.
891:
677:
439:
74:
17:
412:= 20, 24, 25, 40. Thus, the last species can be described with the stoichiometric formula
531:, the sensor allows the introduction of increased air to effect more thorough combustion.
160:
97:
951:
673:
628:
172:
168:
129:
109:
93:
78:
391:, but in fact they are stoichiometric species with large unit cells with the formulas
100:, or by replacing them with atoms of different elements with a different charge. Many
960:
810:
Atkins, P. W.; Overton, T. L.; Rourke, J. P.; Weller, M. T.; Armstrong, F. A., 2010,
689:
502:
449:
55:, or substituted by a smaller or larger atom not usually seen (closed circles, • ),
650:
51:
235:
907:
823:
714:
611:
485:
101:
681:
574:. The critical temperature of the superconductor depends on the exact value of
299:
263:
164:
493:
295:
116:, whereas the more common material is nonstoichiometric, with the formula
934:
J. S. Anderson, 1963, "Current problems in nonstoichiometry (Ch. 1)," in
929:
623:
468:
290:
77:
composition whose proportions cannot be represented by a ratio of small
30:
834:
The Rare Earth
Trifluorides, Part 2 Arxius de les Seccions de Ciències
464:
125:
105:
894:, Geoffrey Wilkinson, Carlos A. Murillo & Manfred Bochmann, 1999,
489:
156:
27:
Chemical compounds that cannot be represented by an empirical formula
527:
sensor in automotive exhaust systems. At low partial pressures of O
664:
520:
153:
29:
942:
series, Vol. 39, Washington, DC, USA: American
Chemical Society,
920:
series, Vol. 39, Washington, DC, USA: American
Chemical Society,
836:
Dmitrii N. Khitarov, Boris
Pavlovich Sobolev, Irina V. Alexeeva
519:
applications that rely on oxide vacancies. One example is the
355:
is a nonstoichiometric material of the approximate composition
108:
have non-stoichiometric examples; for example, stoichiometric
545:
Many superconductors are non-stoichiometric. For example,
187:. The non-stoichiometry reflect the ease of oxidation of
49:, displaced to an adjacent acceptable space to create a
484:
Many useful compounds are produced by the reactions of
430:
implies a more random distribution of oxide vacancies.
701:
699:
41:
Shown is a two-dimensional slice through a primitive
728:
Solid State
Chemistry: An Introduction, 3rd edition
294:. Pyrrhotite is remarkable in that it has numerous
553:, is a non-stoichiometric solid with the formula Y
442:present a series of non-stoichiometric compounds.
159:, especially when the metal is not in its highest
594:and his students that Berthollet's opposition to
298:, i.e. crystalline forms differing in symmetry (
239:Pyrrhotite, an example of a non-stoichiometric
423:, whereas the non-stoichiometric description
8:
838:, Institut d'Estudis Catalans, 2000, p75ff.
777:Metal-Organic and Organic Molecular Magnets
867:. Courier Dover Publications. p. 153.
582:= 0, but this value can be as great as 1.
757:. John Wiley and Sons. pp. 382–390.
754:Geochemistry of hydrothermal ore deposits
663:
195:effectively replacing a small portion of
806:
804:
802:
459:
455:
426:
419:
415:
400:
394:
384:
358:
335:
331:
324:
320:
313:
309:
269:
246:
234:
214:
182:
179:, the actual stoichiometry is closer to
119:
812:Shriver and Atkins' Inorganic Chemistry
640:
132:material and in electrochemical (i.e.,
950:, DOI 10.1021/ba-1964-0039.ch001, see
865:The Historical Background of Chemistry
438:At high temperatures (1000 °C),
281:= 0 to 0.2). The rare stoichiometric
7:
938:(Roland Ward, Ed.), pp. 1–22,
781:Royal Society of Chemistry, 2007,
590:It was mainly through the work of
578:. The stoichiometric species has
203:. Thus for every three "missing"
152:Nonstoichiometry is pervasive for
25:
863:Henry Marshall Leicester (1971).
112:, which is rare, has the formula
928:, DOI 10.1021/ba-1964-0039, see
199:with two thirds their number of
551:high-temperature superconductor
207:ions, the crystal contains two
35:Origin of title phenomenon in
1:
896:Advanced Inorganic Chemistry,
977:Non-stoichiometric compounds
751:Hubert Lloyd Barnes (1997).
549:, arguably the most notable
63:Non-stoichiometric compounds
18:Non-stoichiometric compounds
936:Nonstoichiometric Compounds
914:Nonstoichiometric Compounds
779:Peter Day, Alan E Underhill
592:Nikolai Semenovich Kurnakov
547:yttrium barium copper oxide
998:
730:. CRC Press. p. 214.
682:10.1103/PhysRevB.85.144111
538:
726:Lesley E. Smart (2005).
716:, accessed 8 July 2015.
288:is known as the mineral
163:. For example, although
37:crystallographic defects
953:, accessed 8 July 2015.
931:, accessed 8 July 2015.
909:, accessed 8 July 2015.
825:, accessed 8 July 2015.
608:Claude Louis Berthollet
492:, a conversion that is
541:Cuprate superconductor
259:
69:, almost always solid
59:
967:Solid-state chemistry
940:Advances in Chemistry
918:Advances in Chemistry
266:) with a composition
238:
33:
507:hydrodesulfurization
447:coordination polymer
43:cubic crystal system
972:Inorganic chemistry
912:Roland Ward, 1963,
674:2012PhRvB..85n4111G
480:Oxidation catalysis
306:) and composition (
71:inorganic compounds
348:Palladium hydrides
260:
241:inorganic compound
136:) system designs.
67:chemical compounds
60:
982:General chemistry
764:978-0-471-57144-5
737:978-0-7487-7516-3
535:Superconductivity
440:titanium sulfides
379:tungsten trioxide
353:Palladium hydride
83:empirical formula
16:(Redirected from
989:
892:F. Albert Cotton
879:
878:
860:
854:
837:
832:
826:
808:
797:
780:
775:
769:
768:
748:
742:
741:
723:
717:
703:
694:
693:
667:
645:
462:
429:
422:
407:
390:
363:
343:
338:
327:
316:
284:
276:
253:
221:
210:
206:
202:
198:
194:
190:
186:
178:
171:) has an ideal (
123:
115:
21:
997:
996:
992:
991:
990:
988:
987:
986:
957:
956:
888:
886:Further reading
883:
882:
875:
862:
861:
857:
835:
833:
829:
809:
800:
778:
776:
772:
765:
750:
749:
745:
738:
725:
724:
720:
704:
697:
647:
646:
642:
637:
620:
588:
573:
566:
562:
558:
543:
537:
530:
524:
516:
500:
482:
477:
461:
457:
453:
436:
428:
424:
421:
417:
413:
406:
398:
392:
389:
382:
374:
372:Tungsten oxides
362:
356:
350:
341:
337:
333:
329:
326:
322:
318:
315:
311:
307:
282:
274:
267:
251:
244:
243:, with formula
233:
219:
212:
208:
204:
200:
196:
192:
188:
184:
180:
176:
161:oxidation state
150:
145:
139:
130:superconductive
121:
117:
113:
98:oxidation state
79:natural numbers
28:
23:
22:
15:
12:
11:
5:
995:
993:
985:
984:
979:
974:
969:
959:
958:
955:
954:
932:
910:
887:
884:
881:
880:
873:
855:
827:
798:
770:
763:
743:
736:
718:
695:
658:(14): 144111.
639:
638:
636:
633:
632:
631:
629:Vacancy defect
626:
619:
616:
587:
584:
568:
564:
560:
554:
539:Main article:
536:
533:
528:
522:
515:
514:Ion conduction
512:
498:
481:
478:
476:
473:
435:
432:
373:
370:
349:
346:
232:
229:
173:stoichiometric
149:
146:
144:
141:
110:iron(II) oxide
94:stoichiometric
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
994:
983:
980:
978:
975:
973:
970:
968:
965:
964:
962:
952:
949:
948:9780841222076
945:
941:
937:
933:
930:
927:
926:9780841222076
923:
919:
915:
911:
908:
905:
901:
897:
893:
890:
889:
885:
876:
874:9780486610535
870:
866:
859:
856:
853:
852:9788472836105
849:
845:
841:
831:
828:
824:
821:
817:
813:
807:
805:
803:
799:
796:
795:9781847551399
792:
788:
784:
774:
771:
766:
760:
756:
755:
747:
744:
739:
733:
729:
722:
719:
715:
712:
708:
702:
700:
696:
691:
687:
683:
679:
675:
671:
666:
661:
657:
653:
652:
644:
641:
634:
630:
627:
625:
622:
621:
617:
615:
613:
609:
605:
601:
597:
593:
585:
583:
581:
577:
572:
557:
552:
548:
542:
534:
532:
526:
513:
511:
508:
504:
503:hydrogenation
495:
491:
487:
479:
474:
472:
470:
466:
451:
450:Prussian blue
448:
443:
441:
433:
431:
411:
404:
397:
388:
380:
371:
369:
367:
361:
354:
347:
345:
305:
301:
297:
293:
292:
287:
280:
273:
265:
257:
250:
242:
237:
231:Iron sulfides
230:
228:
225:
218:
174:
170:
169:ferrous oxide
166:
162:
158:
155:
147:
142:
140:
137:
135:
131:
127:
111:
107:
103:
99:
95:
90:
88:
84:
80:
76:
72:
68:
64:
58:
54:
53:
48:
44:
40:
38:
32:
19:
939:
935:
917:
913:
895:
864:
858:
830:
811:
773:
753:
746:
727:
721:
655:
651:Phys. Rev. B
649:
643:
603:
600:berthollides
599:
596:Proust's law
589:
579:
575:
570:
555:
544:
517:
486:hydrocarbons
483:
475:Applications
452:, nominally
444:
437:
409:
402:
395:
386:
375:
365:
359:
351:
289:
278:
271:
261:
258:= 0 to 0.2).
255:
248:
223:
216:
151:
138:
102:metal oxides
91:
62:
61:
56:
52:Frenkel pair
50:
46:
34:
612:John Dalton
434:Other cases
364:(0.02 <
148:Iron oxides
961:Categories
904:0471199575
844:847283610X
820:0199236178
787:1847551394
711:0080501095
635:References
604:daltonides
300:monoclinic
264:pyrrhotite
175:) formula
143:Occurrence
690:119288531
665:1204.4607
494:catalyzed
304:hexagonal
296:polytypes
286:endmember
81:(i.e. an
75:elemental
73:, having
47:vacancies
624:F-Center
618:See also
469:thallium
408:, where
291:troilite
222:, where
106:sulfides
670:Bibcode
586:History
465:caesium
165:wüstite
134:battery
126:ceramic
87:lattice
946:
924:
906:, see
902:
871:
850:
842:
822:, see
818:
793:
785:
761:
734:
713:, see
709:
688:
525:-based
490:oxygen
471:ions.
157:oxides
89:work.
686:S2CID
660:arXiv
488:with
154:metal
944:ISBN
922:ISBN
900:ISBN
869:ISBN
848:ISBN
840:ISBN
816:ISBN
791:ISBN
783:ISBN
759:ISBN
732:ISBN
707:ISBN
610:and
602:and
505:and
467:and
458:(CN)
445:The
427:2.95
183:0.95
128:and
120:0.95
104:and
65:are
678:doi
521:CeO
420:118
357:PdH
342:FeS
302:or
283:FeS
191:to
177:FeO
114:FeO
963::
916:,
846:,
801:^
789:,
698:^
684:.
676:.
668:.
656:85
654:.
569:7−
563:Cu
559:Ba
460:18
454:Fe
425:WO
416:40
405:−2
385:3−
383:WO
344:.
336:12
332:11
330:Fe
328:,
325:10
319:Fe
317:,
308:Fe
270:1−
268:Fe
247:1−
245:Fe
215:1−
213:Fe
209:Fe
205:Fe
201:Fe
197:Fe
193:Fe
189:Fe
181:Fe
118:Fe
877:.
767:.
740:.
692:.
680::
672::
662::
580:x
576:x
571:x
567:O
565:3
561:2
556:x
529:2
523:2
499:2
497:O
456:7
418:O
414:W
410:n
403:n
401:3
399:O
396:n
393:W
387:x
366:x
360:x
334:S
323:S
321:9
314:8
312:S
310:7
279:x
277:(
275:S
272:x
256:x
254:(
252:S
249:x
224:x
220:O
217:x
185:O
167:(
122:O
39:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.