300:
185:
208: – a type of nuclear fission in which cosmic rays impact larger isotopes and fragment them. Spallation does not require high temperature and pressure of the stellar environment but can occur on Earth. Though the lighter products of spallation are relatively rare, the odd-mass-number isotopes in this class occur in greater relative abundance compared to even-number isotopes, in contravention of the Oddo–Harkins rule.
88:
926:, which is highly abundant in spite of having an unpaired proton. Additionally, even-parity isotopes that have exactly two more neutrons than protons are not particularly abundant despite their even parity. Each of the light elements oxygen, neon, magnesium, silicon, and sulfur, have two isotopes with even isospin (nucleon) parity. As shown in the plot above, the isotope with an
892:
The Oddo–Harkins rule may suggest that elements with odd atomic numbers have a single, unpaired proton and may swiftly capture another in order to achieve an even atomic number and proton parity. Protons are paired in elements with even atomic numbers, with each member of the pair balancing the spin
930:
number of protons and neutrons is one to two orders of magnitude more abundant than the isotope with even parity but two additional neutrons. This may leave open the role of parity in abundance. The structural or subatomic basis of the unusual abundances of equinucleonic isotopes in baryonic matter
200:. The process involves the fusion of alpha particles (helium-4 nuclei) under high temperature and pressure within the stellar environment. Each step in the alpha process adds two protons (and two neutrons), favoring synthesis of even-numbered elements. Carbon itself is a product of a
75:
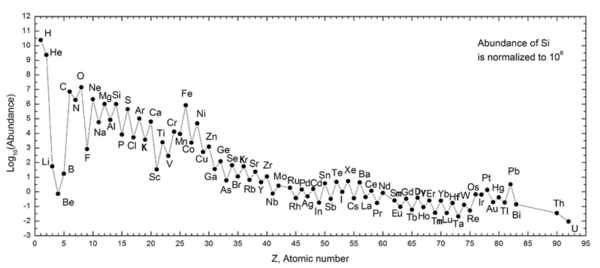
51:(7). Generally, the relative abundance of an even atomic numbered element is roughly two orders of magnitude greater than the relative abundances of the immediately adjacent odd atomic numbered elements to either side. This pattern was first reported by
151:
The early form of the rule derived from Harkin's 1917 study of meteorites. He reasoned as others at the time, that meteorites are more representative of the cosmological abundance of the elements. Harkins observed that elements with even atomic numbers
307:
The elemental basis of the Oddo–Harkins has direct roots in the isotopic compositions of the elements. While even-atomic-numbered elements are more abundant than odd, the spirit of Oddo–Harkins rule extends to the most abundant
255:
nuclei but was too short for every H ion to be reconstituted into heavier elements. In this case, helium, atomic number 2, remains the even-numbered counterpart to hydrogen. Thus, neutral hydrogen—or hydrogen paired with an
303:
A plot of the stable isotopic compositions of the first 16 elements, which make up 99.9% of ordinary matter in the universe. Isotopes with equal numbers of protons and neutrons are particularly abundant.
885:) are not predicted by the shell model. "That nuclei of this type are unusually abundant indicates that the excess stability must have played a part in the process of the creation of elements", stated
188:
Nucleosynthetic origins of light nuclides. The most abundant nuclides have equal numbers of protons and neutrons (box around isotopic symbol). Products of cosmic-ray spallation are the least abundant.
172:. The nuclear core of helium is the same as an alpha particle. This early work connection geochemistry with nuclear physics and cosmology was greatly expanded by the Norwegian group created by
220:, with an atomic number of 1. This may be because, in its ionized form, a hydrogen atom becomes a single proton, of which it is theorized to have been one of the first major conglomerates of
232:. In this period, when inflation of the universe had brought it from an infinitesimal point to about the size of a modern galaxy, temperatures in the particle soup fell from over a trillion
939:
Depending on the mass of a star, the Oddo–Harkins pattern arises from the burning of progressively more massive elements within a collapsing dying star by fusion processes such as the
850:
817:
784:
541:
508:
475:
883:
574:
924:
751:
718:
677:
644:
611:
442:
409:
376:
343:
164:. In addition, he observed that 90% of the material consisted of only 15 different isotopes, with atomic weights in multiples of four, the approximate weight of
977:
972:
291:, and beryllium has only one stable isotope, causing it to lag in abundance with regard to its neighbors, each of which has two stable isotopes.
168:. Three years earlier, Oddo made a similar observation for elements in the Earth's crust, speculating that elements are condensation products of
1183:
1076:
1066:
32:
1271:
1203:
299:
889:
in her acceptance lecture for the Nobel Prize in
Physics in 1963 for discoveries concerning nuclear shell structure.
1276:
143:
to form atoms. The number of protons in the nucleus, called atomic number, uniquely identifies a chemical element.
28:
184:
64:
940:
288:
197:
60:
680:
312:
as well. Isotopes containing an equal number of protons and neutrons are the most abundant. These include
56:
822:
789:
756:
513:
480:
447:
855:
546:
284:
205:
160:. The first seven elements, making up almost 99% of the material in a meteorite, were all even-numbered
68:
896:
723:
690:
649:
616:
583:
414:
381:
348:
315:
1148:
948:
886:
204:
from helium, a process that skips Li, Be, and B. These nuclides (including helium-3) are produced by
201:
960:
684:
985: – Branch of chemistry dealing with radioactivity, transmutation and other nuclear processes
683:
of either protons or neutrons (2, 8, 20, 28, 50, 82, and 126) and are therefore predicted by the
173:
1223:
1179:
1114:
1072:
982:
216:
This postulate, however, does not apply to the universe's most abundant and simplest element:
1215:
1156:
1106:
1047:
1016:
893:
of the other, thus enhancing nucleon stability. A challenge to this explanation is posed by
225:
92:
24:
1241:
165:
112:
59:
in 1917. The Oddo–Harkins rule is true for all elements beginning with carbon produced by
99:
axis is logarithmic); the Oddo–Harkins rule is visible for most of the metallic elements.
1152:
240:
136:
74:
1265:
577:
193:
52:
36:
1094:
283:). This is because most of the universe's lithium, beryllium, and boron are made by
87:
952:
931:
is one of the simplest and most profound unsolved mysteries of the atomic nucleus.
265:
1136:
120:
1227:
1118:
1110:
1020:
275:, which, despite an even atomic number (4), is rarer than adjacent elements (
956:
944:
272:
244:
1160:
257:
229:
217:
140:
132:
124:
108:
48:
1051:
309:
276:
252:
128:
116:
1219:
261:
248:
233:
221:
169:
40:
1035:
1004:
687:
to be unusually abundant. The high abundances of the remaining six (
192:
The Oddo–Harkins rule for elements from C to Fe is explained by the
1071:(Rev. and updated ed.). Univ. of Chicago Press. p. 602.
1036:"The Evolution of the Elements and the Stability of Complex Atoms"
280:
104:
73:
44:
78:
Estimated abundances of the chemical elements in the solar system
63:
but not true for the lightest elements below carbon produced by
1176:
Introduction to planetary science: the geological perspective
1137:"An Unlikely Connection: Geochemistry and Nuclear Structure"
268:
portions of matter following the conclusion of inflation.
1068:
Cosmos an illustrated history of astronomy and cosmology
156:) were about 70 times more abundant than those with odd
1091:
This secondary reference only calls it
Harkins rule.
899:
858:
825:
792:
759:
726:
693:
652:
619:
586:
549:
516:
483:
450:
417:
384:
351:
318:
1130:
1128:
951:. The newly formed elements are ejected slowly as
918:
877:
844:
811:
778:
745:
712:
671:
638:
605:
568:
535:
502:
469:
436:
403:
370:
337:
1202:Rosman, K. J. R.; Taylor, P. D. P. (1998-11-01).
264:—constituted the vast majority of the remaining
1208:Journal of Physical and Chemical Reference Data
1093:Suess, Hans E.; Urey, Harold C. (1956-01-01).
1005:"Die Molekularstruktur der radioaktiven Atome"
43:, with atomic number 6, is more abundant than
959:and eventually join the rest of the galaxy's
8:
1204:"Isotopic Compositions of the Elements 1997"
35:than the elements with immediately adjacent
580:containing whole multiples of He-4 nuclei (
1174:Faure, Gunter; Mensing, Teresa M. (2007).
111:are formed in stars or supernovae through
978:List of elements by stability of isotopes
900:
898:
859:
857:
826:
824:
793:
791:
760:
758:
727:
725:
694:
692:
653:
651:
620:
618:
587:
585:
550:
548:
517:
515:
484:
482:
451:
449:
418:
416:
385:
383:
352:
350:
319:
317:
1040:Journal of the American Chemical Society
298:
183:
135:together. Protons and neutrons form the
86:
995:
16:Relative abundance of chemical elements
973:Abundance of elements in Earth's crust
613:is the exception). Two of the eight (
7:
1197:
1195:
845:{\displaystyle {\ce {^{28}_{14}Si}}}
812:{\displaystyle {\ce {^{24}_{12}Mg}}}
779:{\displaystyle {\ce {^{20}_{10}Ne}}}
536:{\displaystyle {\ce {^{28}_{14}Si}}}
503:{\displaystyle {\ce {^{24}_{12}Mg}}}
470:{\displaystyle {\ce {^{20}_{10}Ne}}}
1009:Zeitschrift für Anorganische Chemie
878:{\displaystyle {\ce {^{32}_{16}S}}}
569:{\displaystyle {\ce {^{32}_{16}S}}}
180:Relation to stellar nucleosynthesis
919:{\displaystyle {\ce {^{14}_{7}N}}}
746:{\displaystyle {\ce {^{14}_{7}N}}}
713:{\displaystyle {\ce {^{12}_{6}C}}}
672:{\displaystyle {\ce {^{16}_{8}O}}}
639:{\displaystyle {\ce {^{4}_{2}He}}}
606:{\displaystyle {\ce {^{14}_{7}N}}}
437:{\displaystyle {\ce {^{16}_{8}O}}}
404:{\displaystyle {\ce {^{14}_{7}N}}}
371:{\displaystyle {\ce {^{12}_{6}C}}}
338:{\displaystyle {\ce {^{4}_{2}He}}}
14:
1242:"The Nobel Prize in Physics 1963"
271:Another exception to the rule is
224:during the initial second of the
127:reach levels high enough to fuse
1:
1095:"Abundances of the Elements"
1034:Harkins, William D. (1917).
236:to several million kelvins.
226:Universe's inflation period
1293:
1099:Reviews of Modern Physics
955:or in the explosion of a
914:
873:
840:
807:
774:
741:
708:
667:
634:
601:
576:. Seven of the eight are
564:
531:
498:
465:
432:
399:
366:
333:
91:Abundance of elements in
1272:Eponymous chemical rules
1111:10.1103/RevModPhys.28.53
1021:10.1002/zaac.19140870118
907:
901:
866:
860:
833:
827:
800:
794:
767:
761:
734:
728:
701:
695:
660:
654:
627:
621:
594:
588:
557:
551:
524:
518:
491:
485:
458:
452:
425:
419:
392:
386:
359:
353:
326:
320:
239:This period allowed the
65:big bang nucleosynthesis
1178:. Dordrecht: Springer.
1003:Oddo, Giuseppe (1914).
289:stellar nucleosynthesis
198:stellar nucleosynthesis
61:stellar nucleosynthesis
1141:Physics in Perspective
935:Relationship to fusion
920:
879:
846:
813:
780:
747:
714:
673:
640:
607:
570:
537:
504:
471:
438:
405:
372:
339:
304:
243:of single protons and
212:Exceptions to the rule
189:
100:
95:per million Si atoms (
79:
57:William Draper Harkins
1161:10.1007/s000160050051
1135:Kragh, Helge (2000).
921:
880:
847:
814:
781:
748:
715:
674:
641:
608:
571:
538:
505:
472:
439:
406:
373:
340:
302:
285:cosmic ray spallation
206:cosmic ray spallation
187:
90:
77:
69:cosmic ray spallation
1065:North, John (2008).
949:triple-alpha process
897:
887:Maria Goeppert Mayer
856:
823:
790:
757:
724:
691:
650:
617:
584:
547:
514:
481:
448:
415:
382:
349:
316:
202:triple-alpha process
139:, which accumulates
1153:2000PhP.....2..381K
1052:10.1021/ja02250a002
961:interstellar medium
941:proton–proton chain
685:nuclear shell model
916:
875:
842:
809:
776:
743:
710:
669:
636:
603:
566:
533:
500:
467:
434:
401:
368:
335:
305:
295:Isotopic abundance
260:, the only stable
190:
174:Victor Goldschmidt
101:
80:
29:even atomic number
1277:Nuclear chemistry
1185:978-1-4020-5544-7
1078:978-0-226-59441-5
983:Nuclear chemistry
906:
905:
904:
865:
864:
863:
832:
831:
830:
799:
798:
797:
766:
765:
764:
733:
732:
731:
700:
699:
698:
659:
658:
657:
626:
625:
624:
593:
592:
591:
556:
555:
554:
523:
522:
521:
490:
489:
488:
457:
456:
455:
424:
423:
422:
391:
390:
389:
358:
357:
356:
325:
324:
323:
21:Oddo–Harkins rule
1284:
1256:
1255:
1253:
1252:
1238:
1232:
1231:
1220:10.1063/1.556031
1214:(6): 1275–1287.
1199:
1190:
1189:
1171:
1165:
1164:
1132:
1123:
1122:
1089:
1083:
1082:
1062:
1056:
1055:
1031:
1025:
1024:
1000:
925:
923:
922:
917:
915:
902:
884:
882:
881:
876:
874:
861:
851:
849:
848:
843:
841:
828:
818:
816:
815:
810:
808:
795:
785:
783:
782:
777:
775:
762:
752:
750:
749:
744:
742:
729:
719:
717:
716:
711:
709:
696:
678:
676:
675:
670:
668:
655:
645:
643:
642:
637:
635:
622:
612:
610:
609:
604:
602:
589:
575:
573:
572:
567:
565:
552:
542:
540:
539:
534:
532:
519:
509:
507:
506:
501:
499:
486:
476:
474:
473:
468:
466:
453:
443:
441:
440:
435:
433:
420:
410:
408:
407:
402:
400:
387:
377:
375:
374:
369:
367:
354:
344:
342:
341:
336:
334:
321:
228:, following the
1292:
1291:
1287:
1286:
1285:
1283:
1282:
1281:
1262:
1261:
1260:
1259:
1250:
1248:
1240:
1239:
1235:
1201:
1200:
1193:
1186:
1173:
1172:
1168:
1134:
1133:
1126:
1092:
1090:
1086:
1079:
1064:
1063:
1059:
1033:
1032:
1028:
1002:
1001:
997:
992:
969:
937:
895:
894:
854:
853:
821:
820:
788:
787:
755:
754:
722:
721:
689:
688:
648:
647:
615:
614:
582:
581:
545:
544:
512:
511:
479:
478:
446:
445:
413:
412:
380:
379:
347:
346:
314:
313:
297:
287:, not ordinary
247:nuclei to form
214:
182:
166:alpha particles
149:
113:nucleosynthesis
85:
39:. For example,
17:
12:
11:
5:
1290:
1288:
1280:
1279:
1274:
1264:
1263:
1258:
1257:
1246:NobelPrize.org
1233:
1191:
1184:
1166:
1124:
1084:
1077:
1057:
1046:(5): 856–879.
1026:
994:
993:
991:
988:
987:
986:
980:
975:
968:
965:
936:
933:
913:
910:
872:
869:
839:
836:
806:
803:
773:
770:
740:
737:
707:
704:
666:
663:
633:
630:
600:
597:
578:alpha nuclides
563:
560:
530:
527:
497:
494:
464:
461:
431:
428:
398:
395:
365:
362:
332:
329:
296:
293:
213:
210:
181:
178:
148:
145:
137:atomic nucleus
84:
81:
37:atomic numbers
23:holds that an
15:
13:
10:
9:
6:
4:
3:
2:
1289:
1278:
1275:
1273:
1270:
1269:
1267:
1247:
1243:
1237:
1234:
1229:
1225:
1221:
1217:
1213:
1209:
1205:
1198:
1196:
1192:
1187:
1181:
1177:
1170:
1167:
1162:
1158:
1154:
1150:
1146:
1142:
1138:
1131:
1129:
1125:
1120:
1116:
1112:
1108:
1104:
1100:
1096:
1088:
1085:
1080:
1074:
1070:
1069:
1061:
1058:
1053:
1049:
1045:
1041:
1037:
1030:
1027:
1022:
1018:
1014:
1011:(in German).
1010:
1006:
999:
996:
989:
984:
981:
979:
976:
974:
971:
970:
966:
964:
962:
958:
954:
950:
946:
942:
934:
932:
929:
911:
908:
890:
888:
870:
867:
837:
834:
804:
801:
771:
768:
738:
735:
705:
702:
686:
682:
681:magic numbers
664:
661:
631:
628:
598:
595:
579:
561:
558:
528:
525:
495:
492:
462:
459:
429:
426:
396:
393:
363:
360:
330:
327:
311:
301:
294:
292:
290:
286:
282:
278:
274:
269:
267:
266:unannihilated
263:
259:
254:
250:
246:
242:
237:
235:
231:
227:
223:
219:
211:
209:
207:
203:
199:
195:
194:alpha process
186:
179:
177:
175:
171:
167:
163:
159:
155:
146:
144:
142:
138:
134:
130:
126:
122:
118:
114:
110:
106:
98:
94:
93:Earth's crust
89:
82:
76:
72:
70:
66:
62:
58:
54:
53:Giuseppe Oddo
50:
46:
42:
38:
34:
33:more abundant
30:
26:
22:
1249:. Retrieved
1245:
1236:
1211:
1207:
1175:
1169:
1144:
1140:
1102:
1098:
1087:
1067:
1060:
1043:
1039:
1029:
1012:
1008:
998:
953:stellar wind
938:
927:
891:
306:
270:
238:
215:
191:
161:
157:
153:
150:
107:bigger than
102:
96:
55:in 1914 and
20:
18:
1015:: 253–268.
121:temperature
83:Definitions
1266:Categories
1251:2024-02-01
1147:(4): 381.
990:References
947:, and the
679:) contain
1228:0047-2689
1119:0034-6861
1105:: 53–74.
957:supernova
945:CNO cycle
273:beryllium
245:deuterium
141:electrons
967:See also
310:isotopes
258:electron
230:Big Bang
218:hydrogen
147:The rule
133:neutrons
125:pressure
109:hydrogen
49:nitrogen
47:(5) and
27:with an
1149:Bibcode
852:, and
277:lithium
253:lithium
234:kelvins
129:protons
117:gravity
115:, when
25:element
1226:
1182:
1117:
1075:
943:, the
543:, and
262:lepton
249:helium
241:fusion
222:quarks
170:helium
41:carbon
928:equal
281:boron
105:atoms
45:boron
1224:ISSN
1180:ISBN
1115:ISSN
1073:ISBN
646:and
279:and
251:and
131:and
123:and
103:All
67:and
19:The
1216:doi
1157:doi
1107:doi
1048:doi
1017:doi
196:of
31:is
1268::
1244:.
1222:.
1212:27
1210:.
1206:.
1194:^
1155:.
1143:.
1139:.
1127:^
1113:.
1103:28
1101:.
1097:.
1044:39
1042:.
1038:.
1013:87
1007:.
963:.
912:14
871:32
868:16
838:28
835:14
829:Si
819:,
805:24
802:12
796:Mg
786:,
772:20
769:10
763:Ne
753:,
739:14
720:,
706:12
665:16
623:He
599:14
562:32
559:16
529:28
526:14
520:Si
510:,
496:24
493:12
487:Mg
477:,
463:20
460:10
454:Ne
444:,
430:16
411:,
397:14
378:,
364:12
345:,
322:He
176:.
119:,
71:.
1254:.
1230:.
1218::
1188:.
1163:.
1159::
1151::
1145:2
1121:.
1109::
1081:.
1054:.
1050::
1023:.
1019::
909:7
903:N
862:S
736:7
730:N
703:6
697:C
662:8
656:O
632:4
629:2
596:7
590:N
553:S
427:8
421:O
394:7
388:N
361:6
355:C
331:4
328:2
162:Z
158:Z
154:Z
152:(
97:y
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.