421:
398:
40:
683:
codon mutation for cystic fibrosis demonstrated increased CFTR protein production and function. Extending on this work, a mechanistic study with yeast and human cells has elucidated the details of ataluren-mediated nonstandard codon-anticodon base pairings which result in specific amino acid substitutions at specific codon positions in the CFTR protein.
31:
2172:
742:(EMA) and received market authorization from the European Commission to treat people with nonsense mutation Duchenne muscular dystrophy in August 2014; a confirmatory phase III clinical trial was required. By December it was on the market in Germany, France, Italy, Denmark, Spain and a number of other European Union countries.
682:
Studies have demonstrated that ataluren treatment increases expression of full-length dystrophin protein in human and mouse primary muscle cells containing the premature stop codon mutation for
Duchenne muscular dystrophy and rescues striated muscle function. Studies in mice with the premature stop
752:
In July 2016, NHS England agreed a
Managed Access Agreement (MAA) for Translarna providing reimbursed patient access to Translarna in England via a five-year MAA. This followed a positive recommendation from the National Institute for Health and Care Excellence (NICE) in April 2016, subject to PTC
1436:
Haas M, Vlcek V, Balabanov P, Salmonson T, Bakchine S, Markey G, et al. (January 2015). "European
Medicines Agency review of ataluren for the treatment of ambulant patients aged 5 years and older with Duchenne muscular dystrophy resulting from a nonsense mutation in the dystrophin gene".
686:
The
European Medicines Agency review on the approval of ataluren concluded that "the non-clinical data available were considered sufficient to support the proposed mechanism of action and to alleviate earlier concerns on the selectivity of ataluren for premature stop codons."
607:
More than 10% of people taking ataluren in clinical trials experienced vomiting; more than 5% experienced diarrhea, nausea, headache, upper abdominal pain, and flatulence; between 1% and 5% of people experienced decreased appetite and weight loss,
1891:
730:
In 2010, PTC Therapeutics released preliminary results of its phase IIb clinical trial for
Duchenne muscular dystrophy, with participants not showing a significant improvement in the six minute walk distance after the 48 weeks of the trial.
1884:
1536:
719:
to identify compounds that increased the amount of protein expressed by mutated genes, and then optimized one of the hits in the screen to create this drug. As with the results of many cell-based screens, the
1570:
1877:
1537:"PTC Therapeutics, Inc. and Genzyme Corporation Announce Preliminary Results from the Phase 2b Clinical Trial of Ataluren; Primary Endpoint Does Not Reach Statistical Significance within Duration of Study"
678:
processing, stability of the mRNA or the resultant protein, thereby making a functional protein similar to the non-mutated endogenous product. It seems to work particularly well for the stop codon 'UGA'.
1011:
1234:
1044:
1603:
1851:
612:, high blood pressure, cough, nosebleeds, abdominal discomfort, constipation, rashes, pain in their arms, legs, and chest muscles, blood in their urine, urinary incontinence, and fever.
102:
551:
651:
of ataluren, it was discovered to bind and stabilize firefly luciferase, thus explaining the mechanism by which it created a false positive effect on the read through assay.
753:
and NHS England finalizing the terms of the MAA. NICE issued its final guidance later in July with implementation of the MAA for patients following within two months.
1562:
770:
735:
1003:
70:
1816:
1634:
1790:
507:
1510:
1326:
Welch EM, Barton ER, Zhuo J, Tomizawa Y, Friesen WJ, Trifillis P, et al. (May 2007). "PTC124 targets genetic disorders caused by nonsense mutations".
749:
for ataluren, which was based on a clinical trial in which ataluren missed its primary endpoint; PTC appealed and the FDA declined again in
October 2016.
1036:
1595:
1843:
777:
recommended not renewing the conditional marketing authorization for
Translarna, a medicine for treating people with Duchenne muscular dystrophy.
1833:
Text was copied from this source which is copyright
European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
623:
should not be given to someone taking ataluren, as they interfere with its mechanism of action. Caution should be used with drugs that induce
2126:
932:
1951:
1674:
843:
1183:
2097:
527:
493:
1379:"PTC124 is an orally bioavailable compound that promotes suppression of the human CFTR-G542X nonsense allele in a CF mouse model"
711:'s lab at the University of Pennsylvania, which was initially funded in part by Parent Project Muscular Dystrophy. The team used
175:
132:
807:
1743:"Ataluren and similar compounds (specific therapies for premature termination codon class I mutations) for cystic fibrosis"
756:
In March 2017, PTC terminated development of ataluren for cystic fibrosis due to lack of efficacy in the phase III trials.
2058:
2162:
1977:
1481:"Press release: PTC Therapeutics Announces $ 15.4 Million NIH Research Grant for Duchenne Muscular Dystrophy | Evaluate"
297:
2143:
568:
377:
2212:
1626:
1992:
1869:
837:
774:
739:
1657:"NHS England successfully negotiates access to new drug treatment for children with duchenne muscular dystrophy"
2197:
1982:
1004:"Researchers question 'read-through' mechanism of muscular dystrophy drug ataluren : Spoonful of Medicine"
671:
246:
2131:
2082:
1901:
1480:
88:
416:
2192:
867:"Therapeutic suppression of premature termination codons: mechanisms and clinical considerations (review)"
366:
2207:
609:
237:
1488:
1037:"Press Release: Questions Raised About Process Used to Identify Experimental Drug for Genetic Disease"
2202:
1390:
1335:
1275:
1198:
1136:
1079:
746:
712:
1844:"Meeting highlights from the Committee for Medicinal Products for Human Use (CHMP) 24-27 June 2024"
583:
Ataluren is used in the
European Union to treat people with Duchenne muscular dystrophy who have a
393:
192:
95:
535:
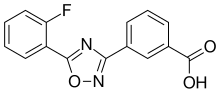
InChI=1S/C15H9FN2O3/c16-12-7-2-1-6-11(12)14-17-13(18-21-14)9-4-3-5-10(8-9)15(19)20/h1-8H,(H,19,20)
1723:
1462:
1359:
1184:"Molecular basis for the high-affinity binding and stabilization of firefly luciferase by PTC124"
1105:
1817:"EMA recommends non-renewal of authorisation of Duchenne muscular dystrophy medicine Translarna"
1791:"EMA recommends non-renewal of authorisation of Duchenne muscular dystrophy medicine Translarna"
39:
1264:"Ataluren stimulates ribosomal selection of near-cognate tRNAs to promote nonsense suppression"
832:
1772:
1715:
1454:
1418:
1351:
1303:
1226:
1164:
1097:
984:
928:
922:
896:
721:
667:
648:
584:
346:
147:
52:
286:
1762:
1754:
1705:
1446:
1408:
1398:
1343:
1293:
1283:
1216:
1206:
1154:
1144:
1125:"Mechanism of PTC124 activity in cell-based luciferase assays of nonsense codon suppression"
1087:
974:
964:
886:
878:
716:
572:
433:
201:
306:
2176:
2102:
1262:
Roy B, Friesen WJ, Tomizawa Y, Leszyk JD, Zhuo J, Johnson B, et al. (November 2016).
620:
1511:"PTC Therapeutics, Inc. Initiates Phase 2 Study Of PTC124 In Duchenne Muscular Dystrophy"
662:(an effect referred to as "read-through") by promoting insertion of certain near-cognate
1394:
1339:
1279:
1202:
1140:
1083:
420:
397:
2137:
2035:
1923:
1767:
1742:
1413:
1378:
1298:
1263:
1221:
1159:
1124:
979:
952:
891:
866:
2186:
1727:
1656:
675:
409:
226:
1466:
1109:
80:
2107:
1969:
1905:
1758:
1363:
1182:
Auld DS, Lovell S, Thorne N, Lea WA, Maloney DJ, Shen M, et al. (March 2010).
921:
Pace A, Buscemi S, Piccionello AP, Pibiri I (2015). Scriven EF, Ramsden CA (eds.).
663:
115:
110:
803:
707:
Ataluren was discovered by scientists at PTC Therapeutics in a collaboration with
969:
2043:
2025:
2005:
1946:
1563:"PTC Therapeutics Receives Positive Opinion from CHMP for Translarna (ataluren)"
708:
1383:
Proceedings of the
National Academy of Sciences of the United States of America
1268:
Proceedings of the National Academy of Sciences of the United States of America
1191:
Proceedings of the National Academy of Sciences of the United States of America
1129:
Proceedings of the National Academy of Sciences of the United States of America
2066:
2048:
2020:
2015:
2010:
2000:
1941:
1450:
696:
659:
588:
469:
277:
2112:
2092:
1403:
1288:
1211:
1149:
74:
1776:
1719:
1458:
1422:
1355:
1307:
1230:
1168:
1101:
988:
900:
882:
655:
257:
22:
1377:
Du M, Liu X, Welch EM, Hirawat S, Peltz SW, Bedwell DM (February 2008).
1347:
1092:
1067:
266:
1959:
1914:
1596:"PTC Therapeutics Announces Launch of Translarna (ataluren) in Germany"
636:
212:
1933:
1710:
1693:
624:
357:
599:
People who are pregnant or breast feeding should not take ataluren.
30:
927:. Advances in Heterocyclic Chemistry. Academic Press. p. 127.
326:
492:
483:
337:
632:
628:
317:
154:
141:
1873:
1485:
PTC, University of Pennsylvania, and the NIH via Evaluate Group
1675:"Drug Company Ends Ataluren Program for CF Nonsense Mutations"
382:
734:
In May 2014, ataluren received a positive opinion from the
745:
In February 2016, FDA declined to accept PTC Therapeutics
924:
3. Recent Advances in the Chemistry of 1,2,4-Oxadiazoles
1123:
Auld DS, Thorne N, Maguire WF, Inglese J (March 2009).
647:
While a large number of studies failed to identify the
2160:
806:. UK Electronic Medicines Compendium. 24 April 2017.
2075:
2057:
2034:
1991:
1968:
1932:
1913:
481:
468:
432:
427:
408:
376:
356:
336:
316:
296:
276:
256:
245:
236:
211:
191:
166:
131:
126:
101:
87:
69:
61:
51:
46:
591:gene, can walk, and are more than five years old.
827:
825:
804:"Translarna - Summary of Product Characteristics"
225:
200:
1741:Aslam AA, Sinha IP, Southern KW (March 2023).
771:Committee for Medicinal Products for Human Use
736:Committee for Medicinal Products for Human Use
1885:
1321:
1319:
1317:
8:
21:
1747:The Cochrane Database of Systematic Reviews
1257:
1255:
953:"A read-through drug put through its paces"
871:International Journal of Molecular Medicine
1892:
1878:
1870:
419:
396:
285:
38:
29:
1766:
1709:
1590:
1588:
1412:
1402:
1297:
1287:
1220:
1210:
1158:
1148:
1091:
1031:
1029:
978:
968:
946:
944:
890:
798:
796:
794:
792:
790:
727:Phase I clinical trials started in 2004.
305:
1627:"FDA snubs PTC appeal for Duchenne drug"
1041:NIH via Drug Discovery & Development
2167:
1068:"Chemical biology: ignore the nonsense"
786:
699:; its chemical name is 3-benzoic acid.
670:with no apparent effects on downstream
567:, is a medication for the treatment of
532:
512:
392:
265:
180:
916:
914:
912:
910:
410:
20:
16:Duchenne muscular dystrophy medication
1606:from the original on 27 December 2014
1008:Nature Medicine: Spoonful of Medicine
365:
345:
79:
7:
1637:from the original on 28 January 2017
846:from the original on 29 October 2020
114:
1491:from the original on 23 August 2023
1240:from the original on 23 August 2023
1047:from the original on 7 October 2017
865:Karijolich J, Yu YT (August 2014).
325:
216:
515:Fc3ccccc3c1nc(no1)c2cc(ccc2)C(=O)O
14:
1900:Other drugs for disorders of the
1854:from the original on 12 July 2024
1066:Schmitz A, Famulok M (May 2007).
810:from the original on 15 July 2017
2170:
1625:Pagliarulo N (17 October 2016).
1573:from the original on 30 May 2019
1517:(Press release). 27 January 2006
1014:from the original on 16 May 2017
453:
450:
444:
540:Key:OOUGLTULBSNHNF-UHFFFAOYSA-N
2059:Histone deacetylase inhibitors
1759:10.1002/14651858.CD012040.pub3
459:
438:
1:
1543:(Press release). 3 March 2010
1978:Delandistrogene moxeparvovec
970:10.1371/journal.pbio.1001458
658:less sensitive to premature
654:Ataluren is thought to make
627:, or that are substrates of
610:high levels of triglycerides
563:, sold under the brand name
951:Roberts RG (25 June 2013).
569:Duchenne muscular dystrophy
2229:
1993:Antisense oligonucleotides
1692:DeFrancesco L (May 2017).
1679:Cystic Fibrosis Foundation
724:of ataluren is not known.
428:Chemical and physical data
2121:
1848:European Medicines Agency
1821:European Medicines Agency
1795:European Medicines Agency
1451:10.1016/j.nmd.2014.11.011
1002:Devitt L (25 June 2013).
838:European Medicines Agency
775:European Medicines Agency
740:European Medicines Agency
548:
523:
503:
171:
37:
28:
1983:Onasemnogene abeparvovec
1954:clostridium histolyticum
81:International Drug Names
1902:musculo-skeletal system
1439:Neuromuscular Disorders
1404:10.1073/pnas.0711795105
1289:10.1073/pnas.1605336113
1212:10.1073/pnas.0909141107
1150:10.1073/pnas.0813345106
883:10.3892/ijmm.2014.1809
1694:"Drug pipeline: 1Q17"
842:. 17 September 2018.
571:. It was designed by
1698:Nature Biotechnology
747:new drug application
713:phenotypic screening
1797:. 15 September 2023
1602:. 3 December 2014.
1395:2008PNAS..105.2064D
1348:10.1038/nature05756
1340:2007Natur.447...87W
1280:2016PNAS..11312508R
1274:(44): 12508–12513.
1203:2010PNAS..107.4878A
1141:2009PNAS..106.3585A
1093:10.1038/nature05715
1084:2007Natur.447...42S
1043:. 3 February 2009.
760:Society and culture
150:(Prescription only)
25:
2148:Never to phase III
769:In June 2024, the
2213:Muscle protectors
2158:
2157:
1197:(11): 4878–4883.
934:978-0-12-802874-2
833:"Translarna EPAR"
722:biological target
649:biological target
595:Contraindications
585:nonsense mutation
558:
557:
494:Interactive image
378:CompTox Dashboard
158:
145:
2220:
2175:
2174:
2173:
2166:
2083:Aceneuramic acid
1894:
1887:
1880:
1871:
1864:
1863:
1861:
1859:
1850:. 28 June 2024.
1840:
1834:
1832:
1830:
1828:
1813:
1807:
1806:
1804:
1802:
1787:
1781:
1780:
1770:
1738:
1732:
1731:
1713:
1711:10.1038/nbt.3874
1689:
1683:
1682:
1671:
1665:
1664:
1653:
1647:
1646:
1644:
1642:
1622:
1616:
1615:
1613:
1611:
1592:
1583:
1582:
1580:
1578:
1559:
1553:
1552:
1550:
1548:
1533:
1527:
1526:
1524:
1522:
1507:
1501:
1500:
1498:
1496:
1487:. 10 July 2007.
1477:
1471:
1470:
1433:
1427:
1426:
1416:
1406:
1389:(6): 2064–2069.
1374:
1368:
1367:
1323:
1312:
1311:
1301:
1291:
1259:
1250:
1249:
1247:
1245:
1239:
1224:
1214:
1188:
1179:
1173:
1172:
1162:
1152:
1135:(9): 3585–3590.
1120:
1114:
1113:
1095:
1063:
1057:
1056:
1054:
1052:
1033:
1024:
1023:
1021:
1019:
999:
993:
992:
982:
972:
948:
939:
938:
918:
905:
904:
894:
862:
856:
855:
853:
851:
829:
820:
819:
817:
815:
800:
717:chemical library
573:PTC Therapeutics
554:
496:
476:
461:
455:
452:
446:
440:
423:
412:
401:
400:
386:
384:
369:
349:
329:
309:
289:
269:
249:
229:
219:
218:
204:
156:
153:
143:
140:
118:
83:
42:
33:
26:
24:
2228:
2227:
2223:
2222:
2221:
2219:
2218:
2217:
2198:Cystic fibrosis
2183:
2182:
2181:
2171:
2169:
2161:
2159:
2154:
2153:
2138:Clinical trials
2117:
2103:Hyaluronic acid
2071:
2053:
2036:Glucocorticoids
2030:
1987:
1964:
1928:
1917:and derivatives
1909:
1898:
1868:
1867:
1857:
1855:
1842:
1841:
1837:
1826:
1824:
1815:
1814:
1810:
1800:
1798:
1789:
1788:
1784:
1753:(3): CD012040.
1740:
1739:
1735:
1691:
1690:
1686:
1681:. 3 March 2017.
1673:
1672:
1668:
1655:
1654:
1650:
1640:
1638:
1624:
1623:
1619:
1609:
1607:
1594:
1593:
1586:
1576:
1574:
1561:
1560:
1556:
1546:
1544:
1535:
1534:
1530:
1520:
1518:
1509:
1508:
1504:
1494:
1492:
1479:
1478:
1474:
1435:
1434:
1430:
1376:
1375:
1371:
1334:(7140): 87–91.
1325:
1324:
1315:
1261:
1260:
1253:
1243:
1241:
1237:
1186:
1181:
1180:
1176:
1122:
1121:
1117:
1078:(7140): 42–43.
1065:
1064:
1060:
1050:
1048:
1035:
1034:
1027:
1017:
1015:
1001:
1000:
996:
963:(6): e1001458.
950:
949:
942:
935:
920:
919:
908:
864:
863:
859:
849:
847:
831:
830:
823:
813:
811:
802:
801:
788:
783:
767:
762:
705:
695:Ataluren is an
693:
668:nonsense codons
666:at the site of
645:
621:Aminoglycosides
618:
605:
603:Adverse effects
597:
581:
552:(what is this?)
549:
544:
541:
536:
531:
530:
519:
516:
511:
510:
499:
474:
464:
458:
449:
443:
404:
380:
372:
352:
332:
312:
292:
272:
252:
232:
215:
207:
187:
184:
179:
178:
162:
122:
90:
17:
12:
11:
5:
2226:
2224:
2216:
2215:
2210:
2205:
2200:
2195:
2185:
2184:
2180:
2179:
2156:
2155:
2152:
2151:
2150:
2149:
2146:
2135:
2129:
2123:
2122:
2119:
2118:
2116:
2115:
2110:
2105:
2100:
2095:
2090:
2085:
2079:
2077:
2073:
2072:
2070:
2069:
2063:
2061:
2055:
2054:
2052:
2051:
2046:
2040:
2038:
2032:
2031:
2029:
2028:
2023:
2018:
2013:
2008:
2003:
1997:
1995:
1989:
1988:
1986:
1985:
1980:
1974:
1972:
1970:Gene therapies
1966:
1965:
1963:
1962:
1957:
1949:
1944:
1938:
1936:
1930:
1929:
1927:
1926:
1920:
1918:
1911:
1910:
1899:
1897:
1896:
1889:
1882:
1874:
1866:
1865:
1835:
1823:. 28 June 2024
1808:
1782:
1733:
1684:
1666:
1648:
1631:BioPharma Dive
1617:
1584:
1554:
1528:
1502:
1472:
1428:
1369:
1313:
1251:
1174:
1115:
1058:
1025:
994:
940:
933:
906:
877:(2): 355–362.
857:
821:
785:
784:
782:
779:
766:
763:
761:
758:
738:(CHMP) of the
704:
701:
692:
689:
644:
641:
617:
614:
604:
601:
596:
593:
580:
577:
556:
555:
546:
545:
543:
542:
539:
537:
534:
526:
525:
524:
521:
520:
518:
517:
514:
506:
505:
504:
501:
500:
498:
497:
489:
487:
479:
478:
472:
466:
465:
462:
456:
447:
441:
436:
430:
429:
425:
424:
414:
406:
405:
403:
402:
389:
387:
374:
373:
371:
370:
362:
360:
354:
353:
351:
350:
342:
340:
334:
333:
331:
330:
322:
320:
314:
313:
311:
310:
302:
300:
294:
293:
291:
290:
282:
280:
274:
273:
271:
270:
262:
260:
254:
253:
251:
250:
242:
240:
234:
233:
231:
230:
222:
220:
209:
208:
206:
205:
197:
195:
189:
188:
186:
185:
183:3-benzoic acid
182:
174:
173:
172:
169:
168:
164:
163:
161:
160:
151:
137:
135:
129:
128:
124:
123:
121:
120:
107:
105:
99:
98:
93:
91:administration
85:
84:
77:
67:
66:
63:
59:
58:
55:
49:
48:
44:
43:
35:
34:
15:
13:
10:
9:
6:
4:
3:
2:
2225:
2214:
2211:
2209:
2206:
2204:
2201:
2199:
2196:
2194:
2193:Benzoic acids
2191:
2190:
2188:
2178:
2168:
2164:
2147:
2145:
2142:
2141:
2139:
2136:
2133:
2130:
2128:
2125:
2124:
2120:
2114:
2111:
2109:
2106:
2104:
2101:
2099:
2096:
2094:
2091:
2089:
2086:
2084:
2081:
2080:
2078:
2074:
2068:
2065:
2064:
2062:
2060:
2056:
2050:
2047:
2045:
2042:
2041:
2039:
2037:
2033:
2027:
2024:
2022:
2019:
2017:
2014:
2012:
2009:
2007:
2004:
2002:
1999:
1998:
1996:
1994:
1990:
1984:
1981:
1979:
1976:
1975:
1973:
1971:
1967:
1961:
1958:
1956:
1955:
1950:
1948:
1945:
1943:
1940:
1939:
1937:
1935:
1931:
1925:
1922:
1921:
1919:
1916:
1912:
1907:
1903:
1895:
1890:
1888:
1883:
1881:
1876:
1875:
1872:
1853:
1849:
1845:
1839:
1836:
1822:
1818:
1812:
1809:
1796:
1792:
1786:
1783:
1778:
1774:
1769:
1764:
1760:
1756:
1752:
1748:
1744:
1737:
1734:
1729:
1725:
1721:
1717:
1712:
1707:
1703:
1699:
1695:
1688:
1685:
1680:
1676:
1670:
1667:
1662:
1658:
1652:
1649:
1636:
1632:
1628:
1621:
1618:
1605:
1601:
1597:
1591:
1589:
1585:
1572:
1568:
1564:
1558:
1555:
1542:
1538:
1532:
1529:
1516:
1512:
1506:
1503:
1490:
1486:
1482:
1476:
1473:
1468:
1464:
1460:
1456:
1452:
1448:
1444:
1440:
1432:
1429:
1424:
1420:
1415:
1410:
1405:
1400:
1396:
1392:
1388:
1384:
1380:
1373:
1370:
1365:
1361:
1357:
1353:
1349:
1345:
1341:
1337:
1333:
1329:
1322:
1320:
1318:
1314:
1309:
1305:
1300:
1295:
1290:
1285:
1281:
1277:
1273:
1269:
1265:
1258:
1256:
1252:
1236:
1232:
1228:
1223:
1218:
1213:
1208:
1204:
1200:
1196:
1192:
1185:
1178:
1175:
1170:
1166:
1161:
1156:
1151:
1146:
1142:
1138:
1134:
1130:
1126:
1119:
1116:
1111:
1107:
1103:
1099:
1094:
1089:
1085:
1081:
1077:
1073:
1069:
1062:
1059:
1046:
1042:
1038:
1032:
1030:
1026:
1013:
1009:
1005:
998:
995:
990:
986:
981:
976:
971:
966:
962:
958:
954:
947:
945:
941:
936:
930:
926:
925:
917:
915:
913:
911:
907:
902:
898:
893:
888:
884:
880:
876:
872:
868:
861:
858:
845:
841:
839:
834:
828:
826:
822:
809:
805:
799:
797:
795:
793:
791:
787:
780:
778:
776:
772:
764:
759:
757:
754:
750:
748:
743:
741:
737:
732:
728:
725:
723:
718:
714:
710:
702:
700:
698:
690:
688:
684:
680:
677:
673:
672:transcription
669:
665:
661:
657:
652:
650:
642:
640:
638:
634:
630:
626:
622:
615:
613:
611:
602:
600:
594:
592:
590:
586:
578:
576:
574:
570:
566:
562:
553:
547:
538:
533:
529:
522:
513:
509:
502:
495:
491:
490:
488:
485:
480:
473:
471:
467:
437:
435:
431:
426:
422:
418:
415:
413:
411:ECHA InfoCard
407:
399:
395:
394:DTXSID5046776
391:
390:
388:
379:
375:
368:
364:
363:
361:
359:
355:
348:
344:
343:
341:
339:
335:
328:
324:
323:
321:
319:
315:
308:
304:
303:
301:
299:
295:
288:
284:
283:
281:
279:
275:
268:
264:
263:
261:
259:
255:
248:
244:
243:
241:
239:
235:
228:
224:
223:
221:
214:
210:
203:
199:
198:
196:
194:
190:
181:
177:
170:
165:
159: Rx-only
152:
149:
139:
138:
136:
134:
130:
125:
117:
112:
109:
108:
106:
104:
100:
97:
94:
92:
86:
82:
78:
76:
72:
68:
64:
60:
56:
54:
50:
47:Clinical data
45:
41:
36:
32:
27:
19:
2208:Orphan drugs
2108:Palovarotene
2098:Chondrocytes
2087:
1953:
1952:Collagenase
1924:Hydroquinine
1856:. Retrieved
1847:
1838:
1825:. Retrieved
1820:
1811:
1799:. Retrieved
1794:
1785:
1750:
1746:
1736:
1701:
1697:
1687:
1678:
1669:
1660:
1651:
1639:. Retrieved
1630:
1620:
1608:. Retrieved
1599:
1575:. Retrieved
1566:
1557:
1545:. Retrieved
1540:
1531:
1519:. Retrieved
1514:
1505:
1493:. Retrieved
1484:
1475:
1442:
1438:
1431:
1386:
1382:
1372:
1331:
1327:
1271:
1267:
1242:. Retrieved
1194:
1190:
1177:
1132:
1128:
1118:
1075:
1071:
1061:
1049:. Retrieved
1040:
1016:. Retrieved
1007:
997:
960:
957:PLOS Biology
956:
923:
874:
870:
860:
850:29 September
848:. Retrieved
836:
812:. Retrieved
768:
765:Legal status
755:
751:
744:
733:
729:
726:
706:
694:
685:
681:
653:
646:
643:Pharmacology
619:
616:Interactions
606:
598:
582:
564:
560:
559:
550:
367:ChEMBL256997
133:Legal status
127:Legal status
18:
2203:Oxadiazoles
2134:from market
2044:Deflazacort
2026:Viltolarsen
2006:Drisapersen
1947:Chymopapain
1661:NHS England
1610:27 December
1600:MarketWatch
1567:MarketWatch
1445:(1): 5–13.
709:Lee Sweeney
660:stop codons
579:Medical use
477: g·mol
417:100.132.097
347:CHEBI:94805
202:775304-57-9
167:Identifiers
62:Other names
53:Trade names
2187:Categories
2067:Givinostat
2049:Vamorolone
2021:Nusinersen
2016:Golodirsen
2011:Eteplirsen
2001:Casimersen
1942:Bromelains
1704:(5): 400.
1244:4 November
781:References
697:oxadiazole
589:dystrophin
565:Translarna
482:3D model (
470:Molar mass
307:K16AME9I3V
278:ChemSpider
238:IUPHAR/BPS
193:CAS Number
176:IUPAC name
57:Translarna
2144:Phase III
2132:Withdrawn
2113:Risdiplam
2093:Branaplam
1728:205284732
1547:22 August
1521:22 August
691:Chemistry
656:ribosomes
89:Routes of
75:Drugs.com
2177:Medicine
2088:Ataluren
1852:Archived
1777:36866921
1720:28486449
1635:Archived
1604:Archived
1571:Archived
1541:BioSpace
1515:BioSpace
1489:Archived
1467:41468577
1459:25497400
1423:18272502
1356:17450125
1308:27702906
1235:Archived
1231:20194791
1169:19208811
1110:29789135
1102:17450128
1045:Archived
1012:Archived
989:23824301
901:24939317
844:Archived
808:Archived
561:Ataluren
258:DrugBank
227:11219835
103:ATC code
96:By mouth
23:Ataluren
1960:Trypsin
1934:Enzymes
1915:Quinine
1858:12 July
1827:29 June
1801:29 June
1768:9983356
1641:18 June
1495:18 June
1414:2538881
1391:Bibcode
1364:4423529
1336:Bibcode
1299:5098639
1276:Bibcode
1222:2841876
1199:Bibcode
1160:2638738
1137:Bibcode
1080:Bibcode
1051:18 June
1018:18 June
980:3692443
892:4094583
814:18 June
773:of the
703:History
637:OATP1B3
587:in the
475:284.246
434:Formula
287:9394889
267:DB05016
213:PubChem
119:)
113: (
111:M09AX03
2163:Portal
2127:WHO-EM
1775:
1765:
1726:
1718:
1577:30 May
1465:
1457:
1421:
1411:
1362:
1354:
1328:Nature
1306:
1296:
1229:
1219:
1167:
1157:
1108:
1100:
1072:Nature
987:
977:
931:
899:
889:
625:UGT1A9
508:SMILES
358:ChEMBL
327:D09323
146:
65:PTC124
2076:Other
1724:S2CID
1463:S2CID
1360:S2CID
1238:(PDF)
1187:(PDF)
1106:S2CID
840:(EMA)
715:of a
635:, or
528:InChI
484:JSmol
338:ChEBI
1860:2024
1829:2024
1803:2024
1773:PMID
1751:2023
1716:PMID
1643:2017
1612:2014
1579:2019
1549:2023
1523:2023
1497:2017
1455:PMID
1419:PMID
1352:PMID
1304:PMID
1246:2018
1227:PMID
1165:PMID
1098:PMID
1053:2017
1020:2017
985:PMID
929:ISBN
897:PMID
852:2020
816:2017
676:mRNA
664:tRNA
633:OAT3
629:OAT1
318:KEGG
298:UNII
247:7341
71:AHFS
1906:M09
1763:PMC
1755:doi
1706:doi
1447:doi
1409:PMC
1399:doi
1387:105
1344:doi
1332:447
1294:PMC
1284:doi
1272:113
1217:PMC
1207:doi
1195:107
1155:PMC
1145:doi
1133:106
1088:doi
1076:447
975:PMC
965:doi
887:PMC
879:doi
383:EPA
217:CID
148:POM
116:WHO
2189::
2140::
1846:.
1819:.
1793:.
1771:.
1761:.
1749:.
1745:.
1722:.
1714:.
1702:35
1700:.
1696:.
1677:.
1659:.
1633:.
1629:.
1598:.
1587:^
1569:.
1565:.
1539:.
1513:.
1483:.
1461:.
1453:.
1443:25
1441:.
1417:.
1407:.
1397:.
1385:.
1381:.
1358:.
1350:.
1342:.
1330:.
1316:^
1302:.
1292:.
1282:.
1270:.
1266:.
1254:^
1233:.
1225:.
1215:.
1205:.
1193:.
1189:.
1163:.
1153:.
1143:.
1131:.
1127:.
1104:.
1096:.
1086:.
1074:.
1070:.
1039:.
1028:^
1010:.
1006:.
983:.
973:.
961:11
959:.
955:.
943:^
909:^
895:.
885:.
875:34
873:.
869:.
835:.
824:^
789:^
674:,
639:.
631:,
575:.
442:15
155:EU
142:UK
2165::
1908:)
1904:(
1893:e
1886:t
1879:v
1862:.
1831:.
1805:.
1779:.
1757::
1730:.
1708::
1663:.
1645:.
1614:.
1581:.
1551:.
1525:.
1499:.
1469:.
1449::
1425:.
1401::
1393::
1366:.
1346::
1338::
1310:.
1286::
1278::
1248:.
1209::
1201::
1171:.
1147::
1139::
1112:.
1090::
1082::
1055:.
1022:.
991:.
967::
937:.
903:.
881::
854:.
818:.
486:)
463:3
460:O
457:2
454:N
451:F
448:9
445:H
439:C
385:)
381:(
157::
144::
73:/
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.