437:
diethylaluminum 2,2,6,6-tetramethylpiperidide in 10 mL of benzene at 0 °C was added dropwise over 5 minutes a solution of 0.18 g (0.001 mol) of epoxide in 3 mL of benzene. The mixture was stirred at 0 °C until analysis indicated the absence of starting material. The reaction was quenched by the addition of ice-cold 1 N
436:
A benzene solution of 1 eq of diethylaluminum chloride was added dropwise at 0° to a solution of 1 eq of lithium 2,2,6,6-tetramethylpiperidide prepared in the usual fashion in benzene. The resulting slurry was stirred for 30 minutes and used immediately. To a stirred mixture of 0.004 mol of
337:
Suitably substituted unsaturated epoxides may undergo vinylogous elimination, which leads to conjugated allylic alcohols. Substituted vinyl epoxides undergo 1,4-elimination in some but not all cases; however, epoxides with β unsaturation eliminate cleanly to give conjugated allylic alcohols.
387:(ether/hexane solvent mixtures derived from the synthesis of the lithium amide are usually used directly for isomerization reactions). An excess of the base is employed to account for impurities that consume base and reaction of the base with the ether
378:
to a solution of the amine in ether. Dry glassware and inert atmosphere are required for these reactions. Alternatively, lithium amides may be prepared by the direct action of lithium on the corresponding amine. Typical temperatures for
441:. The organic layer was separated, and the aqueous layer was extracted with ether. The organic layers were combined, washed with brine, dried, and concentrated. The residue was purified by preparative TLC (R
77:, non-racemic epoxides, the configuration of the allylic alcohol product matches that of the epoxide substrate at the carbon whose C–O bond does not break (the starred carbon below). Besides
247:
addition of the base to the unsubstituted epoxide carbon may take place. Non-nucleophilic, sterically hindered bases have been used to isomerize terminal epoxides successfully, however.
287:
Five- and six-membered rings containing epoxides afford allylic alcohols upon treatment with amide bases; however, reactions of medium-ring epoxides may be complicated by competitive
136:
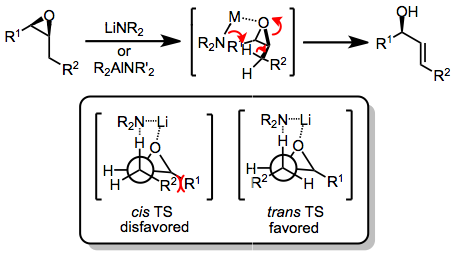
conditions proceeds by a β-elimination process. A model has been advanced that invokes an initial complex between the lithium amide base and epoxide. Concerted C–O bond cleavage and
264:
Acyclic disubstituted epoxides undergo deprotonation at the least substituted site (unless a conjugated double bond can be formed; see equation (9) below) with high selectivity for
167:
Other processes may take place competitively under basic conditions, particularly when β-elimination is slow or not possible. These pathways likely begin with lithiation of a
200:(HMPA) is used as an additive with lithium amide bases, selectivity for the formation of allylic alcohols increases. These reactions are believed to proceed through
100:
products (see equation (2) below). The strongly basic conditions required for most isomerizations of this type represent the reaction's primary disadvantage.
144:
transition state to give an allylic alkoxide, which is protonated upon workup. Deprotonation typically occurs at the exist in the transition state for
408:
Aluminum amides, which are bulkier and sometimes more selective than lithium amides, are prepared from the corresponding lithium amides and
332:
305:
282:
259:
233:
89:
eliminations are possible when the epoxide substrate is substituted with vinyl or dienyl groups. Unconstrained systems tend to form
352:
191:
162:
431:
314:
without competitive carbenoid transformations. The use of bulky aluminum amide bases facilitates elimination at the substituent
114:
310:
Trisubstituted epoxides do not easily undergo metalation in the ring. As a result, allylic alcohols are formed from these
722:
453:) δ 3.73–4.20 (1, m), 4.97–5.82 (2, m); mass spectrum (m/z) 182 (16), 164 (13), 139 (32), 125 (46), and 98 (100).
409:
197:
179:, while intramolecular C–H insertion affords cyclic alcohols with the formation of a new carbon-carbon bond.
311:
412:. Reactions are usually carried out at 0 °C in an inert atmosphere, with benzene as the solvent.
96:, as significant non-bonding interactions are avoided in the transition state for the formation of
78:
438:
391:. Care should be taken when HMPA is added to lithium amide reactions, as it is a known animal
370:
amides are usually prepared in the laboratory through the addition of a titrated solution of
482:
133:
31:
699:
Thies, R. W.; Gasic, M.; Whalen, D.; Grutzner, J. D.; Sakai, M.; Johnson, B.; Winstein, S.
62:
39:
27:
201:
716:
402:
398:
380:
137:
129:
35:
288:
244:
318:
to hydrogen, which occurs from the less sterically hindered epoxide-base complex.
486:
470:
93:
392:
82:
401:
reagents may also be used; however, lower temperatures are required to avoid
449:)-2-cyclododecenol: IR (neat) 3330–3370, 1465, 1450, 970 cm; NMR (CCl
69:
adjacent to the epoxide, elimination, and neutralization of the resulting
86:
70:
55:
351:
331:
304:
281:
258:
232:
388:
375:
367:
190:
176:
172:
161:
73:
lead to synthetically useful allylic alcohol products. In reactions of
51:
23:
384:
212:
168:
74:
66:
20:
113:
430:
219:
epoxides to chiral allylic alcohols with high enantioselectivity.
58:
243:
Terminal epoxides suffer from the limitation that competitive
539:
Yasuda, A.; Tanaka, S.; Oshima, K.; Yamamoto, H.; Nozaki, H.
383:
reactions employing lithium amides are between 0 °C and
405:
of the base. These reactions are most often run in hexanes.
171:
in the epoxide ring, followed by α-elimination to afford a
215:
amide bases may be used in catalytic amounts to isomerize
61:bases, epoxides may open to give the corresponding
85:of the epoxide ring can take place competitively.
132:of epoxides to allylic alcohols under strongly
38:of this type are most often used to synthesize
175:intermediate. 1,2-hydrogen migration leads to
619:Bertillson, S.; Sodergren, M.; Andersson, P.
8:
527:, Academic Press, New York, 1971, Chapter 7.
445:0.22 in 1:2 ether–hexane) to give 99% of (
42:, although other products are possible.
535:
533:
461:
519:
517:
81:some other reactions are possible, as
30:products through the action of strong
639:Yasuda, A.; Yamamoto, H.; Nozaki, H.
496:
494:
358:Experimental conditions and procedure
7:
291:C–H insertion or ketone formation.
17:Base-promoted epoxide isomerization
14:
429:
350:
330:
303:
280:
257:
231:
189:
160:
112:
579:Cope, C.; Lee, H.; Petree, E.
1:
120:Mechanism and stereochemistry
468:Crandall, J. K.; Apparu, M.
679:Crandall, J. K.; Chang, H.
599:Bartsch, R. A.; Závada, J.
561:Angew. Chem. Int. Ed. Engl.
487:10.1002/0471264180.or029.03
739:
659:Thummel, P.; Rickborn, B.
525:Carbene Chemistry, 2nd Ed.
410:diethylaluminum chloride
208:Stereoselective variants
500:Bond, F. T.; Ho, C. Y.
198:hexamethylphosphoramide
148:double bond formation.
641:Bull. Chem. Soc. Jpn.
239:Scope and limitations
19:is the conversion of
125:Prevailing mechanism
196:In many cases when
50:In the presence of
363:Typical conditions
723:Organic reactions
701:J. Am. Chem. Soc.
581:J. Am. Chem. Soc.
541:J. Am. Chem. Soc.
439:hydrochloric acid
416:Example procedure
374:-butyllithium in
730:
708:
697:
691:
677:
671:
657:
651:
637:
631:
617:
611:
597:
591:
577:
571:
557:
551:
537:
528:
521:
512:
498:
489:
466:
433:
426:
354:
347:
334:
327:
307:
300:
284:
277:
261:
254:
235:
228:
193:
186:
164:
157:
116:
109:
63:allylic alcohols
40:allylic alcohols
738:
737:
733:
732:
731:
729:
728:
727:
713:
712:
711:
698:
694:
678:
674:
658:
654:
638:
634:
618:
614:
598:
594:
578:
574:
558:
554:
538:
531:
522:
515:
499:
492:
467:
463:
459:
452:
444:
434:
420:
418:
365:
360:
355:
341:
335:
321:
308:
294:
285:
271:
262:
248:
241:
236:
222:
210:
194:
180:
165:
151:
140:proceeds via a
127:
122:
117:
103:
65:. Removal of a
48:
12:
11:
5:
736:
734:
726:
725:
715:
714:
710:
709:
692:
672:
652:
632:
612:
592:
572:
552:
529:
513:
490:
460:
458:
455:
450:
442:
428:
417:
414:
364:
361:
359:
356:
349:
329:
302:
279:
268:double bonds.
256:
240:
237:
230:
209:
206:
202:E2 elimination
188:
159:
126:
123:
121:
118:
111:
47:
44:
36:Isomerizations
13:
10:
9:
6:
4:
3:
2:
735:
724:
721:
720:
718:
706:
702:
696:
693:
689:
685:
682:
681:J. Org. Chem.
676:
673:
669:
665:
662:
661:J. Org. Chem.
656:
653:
649:
645:
642:
636:
633:
629:
625:
622:
621:J. Org. Chem.
616:
613:
609:
605:
602:
596:
593:
589:
585:
582:
576:
573:
569:
565:
562:
556:
553:
549:
545:
542:
536:
534:
530:
526:
520:
518:
514:
510:
506:
503:
502:J. Org. Chem.
497:
495:
491:
488:
484:
480:
476:
473:
472:
465:
462:
456:
454:
448:
440:
432:
427:
425:
424:
415:
413:
411:
406:
404:
403:decomposition
400:
399:Organolithium
396:
394:
390:
386:
382:
381:isomerization
377:
373:
369:
362:
357:
353:
348:
346:
345:
339:
333:
328:
326:
325:
319:
317:
313:
306:
301:
299:
298:
292:
290:
283:
278:
276:
275:
269:
267:
260:
255:
253:
252:
246:
238:
234:
229:
227:
226:
220:
218:
214:
207:
205:
203:
199:
192:
187:
185:
184:
178:
174:
170:
163:
158:
156:
155:
149:
147:
143:
139:
138:deprotonation
135:
131:
130:Isomerization
124:
119:
115:
110:
108:
107:
101:
99:
95:
92:
88:
84:
80:
79:β-elimination
76:
72:
68:
64:
60:
57:
53:
45:
43:
41:
37:
33:
29:
25:
22:
18:
704:
700:
695:
687:
683:
680:
675:
667:
663:
660:
655:
647:
643:
640:
635:
627:
623:
620:
615:
607:
603:
600:
595:
587:
583:
580:
575:
567:
563:
560:
555:
547:
543:
540:
524:
508:
504:
501:
478:
474:
469:
464:
446:
435:
422:
421:
419:
407:
397:
371:
366:
343:
342:
340:
336:
323:
322:
320:
315:
309:
296:
295:
293:
289:transannular
286:
273:
272:
270:
265:
263:
250:
249:
245:nucleophilic
242:
224:
223:
221:
216:
211:
195:
182:
181:
166:
153:
152:
150:
145:
141:
128:
105:
104:
102:
97:
94:double bonds
90:
49:
46:Introduction
16:
15:
559:Sicher, J.
523:Kirmse, W.
471:Org. React.
28:ring-opened
601:Chem. Rev.
457:References
393:carcinogen
312:substrates
87:Vinylogous
83:metalation
717:Category
71:alkoxide
56:aluminum
24:epoxides
707:, 2262.
670:, 1365.
650:, 1705.
630:, 1567.
590:, 2849.
550:, 6513.
511:, 1421.
481:, 345.
389:solvent
376:hexanes
368:Lithium
177:ketones
173:carbene
52:lithium
703:1972,
690:, 435.
610:, 453.
570:, 200.
385:reflux
213:Chiral
169:carbon
75:chiral
67:proton
266:trans
134:basic
98:trans
91:trans
59:amide
21:alkyl
684:1967
664:1971
644:1979
624:2002
604:1980
584:1959
564:1972
544:1974
505:1976
475:1983
423:(10)
217:meso
32:base
483:doi
344:(9)
324:(8)
316:cis
297:(7)
274:(6)
251:(5)
225:(4)
183:(3)
154:(2)
146:cis
142:syn
106:(1)
54:or
26:to
719::
705:94
688:32
686:,
668:36
666:,
648:52
646:,
628:67
626:,
608:80
606:,
588:80
586:,
568:11
566:,
548:96
546:,
532:^
516:^
509:41
507:,
493:^
479:29
477:,
395:.
204:.
34:.
485::
451:4
447:E
443:f
372:n
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.