51:
316:), hence the bridgehead carbon in front gets number 1, the carbonyl gets number 2 and numbering continues along the carbon chain following the longest path, until the doubly substituted top carbon (number 7). Equal to norbornane, this molecule also has two paths of 2 carbon atoms and one path of 1 carbon atom between the two bridgehead carbons, so the numbers within the brackets stay . Combining the brackets and suffix (now filling in the position of the carbonyl as well) gives us heptan-2-one. Besides
43:
31:
202:
330:
When naming simple fused bicyclic compounds, the same method as for bridged bicyclic compounds is applied, except the third path between the two bridgehead atoms now consists of zero atoms. Therefore, fused bicyclic compounds have a "0" included in the brackets. For example,
281:. In between the prefix and the suffix, a pair of brackets with numerals denotes the number of carbon atoms between each of the bridgehead atoms. These numbers are arranged in descending order and are separated by periods. For example, the carbon frame of
262:. Numbering of the carbon chain always begins at one bridgehead atom (where the rings meet) and follows the carbon chain along the longest path, to the next bridgehead atom. Then numbering is continued along the second longest path and so on. Fused and
289:. This molecule has two paths of 2 carbon atoms and a third path of 1 carbon atom between the two bridgehead carbons, so the brackets are filled in descending order: . Addition of the prefix
558:
Moss GP, The
Working Party of the International Union of Pure and Applied Chemistry , Organic Chemistry Division, Commission on Nomenclature of Organic Chemistry (III.1) (1998).
305:
259:
247:
542:
452:
397:
335:
is named bicyclodecane. The numbers are sometimes omitted in unambiguous cases. For example, bicyclobutane is typically called simply
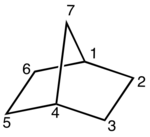
185:, the two rings share three or more atoms, separating the two bridgehead atoms by a bridge containing at least one atom. For example,
612:
559:
496:
658:
653:
564:
526:
304:
in this case, hence the suffix heptanone. We start with numbering the carbon frame at the bridgehead atom with the
250:. The root of the compound name depends on the total number of atoms in all rings together, possibly followed by a
50:
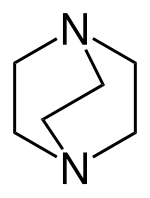
57:(1,4-diazabicyclooctane) is often incorrectly depicted with one skewed ethylene group for the sake of clarity.
633:
522:
Nomenclature of
Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book)
582:
538:
448:
393:
263:
138:
77:. Bicyclic structures occur widely, for example in many biologically important molecules like
42:
572:
530:
476:
421:
255:
274:
134:
74:
638:
159:, two rings share two adjacent atoms. In other words, the rings share one covalent bond,
301:
647:
355:
336:
313:
560:"Nomenclature of fused and bridged fused ring systems (IUPAC Recommendations 1998)"
321:
309:
190:
142:
30:
520:
442:
387:
85:. A bicyclic compound can be carbocyclic (all of the ring atoms are carbons), or
534:
343:
324:
116:
86:
413:
137:, the two rings share only one single atom, the spiro atom, which is usually a
590:
282:
201:
186:
146:
105:
35:
586:
577:
481:
426:
94:
327:
so the complete, official name becomes 1,7,7-trimethylbicycloheptan-2-one.
237:
bonds radiate, like spokes:bicyclooctane, bicyclo-nonane, bicycloundecane.
123:
109:
468:
17:
332:
297:
286:
194:
175:
171:
101:
82:
78:
363:
359:
351:
350:
has a total of 8 atoms in its bridged structure, hence the root name
267:
251:
226:
129:
Three modes of ring junction are possible for a bicyclic compound:
347:
200:
90:
54:
49:
41:
29:
497:"Aromatic Hydrocarbon - an overview | ScienceDirect Topics"
189:, also known as bicycloheptane, can be viewed as a pair of
89:(the rings' atoms consist of at least two elements), like
362:
atoms. Therefore, the official name gets the additional
320:, the prefix should also specify the positions of all
193:
rings each sharing three of their five carbon atoms.
73: 'ring') is a molecule that features two joined
369:and the total name becomes 1,4-diazabicyclooctane.
285:contains a total of 7 atoms, hence the root name
300:also counts 7 atoms, but is substituted with a
119:), or a combination of aliphatic and aromatic (
141:. An example of a spirocyclic compound is the
613:"Bridged-bicyclic-rings-and-how-to-name-them"
8:
576:
480:
425:
389:Organic Chemistry: An Acid—Base Approach
221:
214:
207:
378:
93:. Moreover, the two rings can both be
46:The spirocyclic compound spiroundecane
293:gives the total name bicycloheptane.
7:
354:. Here the two bridgehead atoms are
246:Bicyclic molecules are described by
25:
386:Smith, Michael B. (2011-06-29).
27:Molecule with two joined rings
1:
447:. University Science Books.
197:is a more elaborate example.
535:10.1039/9781849733069-FP001
441:Sorrell, Thomas N. (2006).
233:are the carbons from which
675:
529:. 2014. pp. 155–157.
527:Royal Society of Chemistry
264:bridged bicyclic compounds
183:bridged bicyclic compounds
38:, formally bicycloheptane
414:"heterocyclic compounds"
167:are directly connected (
69: 'two' and
578:10.1351/pac199870010143
482:10.1351/goldbook.S05881
427:10.1351/goldbook.H02798
617:MasterOrganicChemistry
238:
58:
47:
39:
501:www.sciencedirect.com
275:spirocyclic compounds
204:
53:
45:
34:The bridged bicyclic
33:
296:The carbon frame of
634:Polycyclic compound
659:Bicyclic compounds
654:Molecular geometry
619:. August 14, 2014.
248:IUPAC nomenclature
239:
157:bicyclic compounds
59:
48:
40:
544:978-0-85404-182-4
469:"spiro compounds"
454:978-1-891389-38-2
444:Organic Chemistry
399:978-1-4200-7921-0
139:quaternary carbon
63:bicyclic molecule
16:(Redirected from
666:
621:
620:
609:
603:
602:
600:
598:
589:. Archived from
580:
565:Pure Appl. Chem.
555:
549:
548:
517:
511:
510:
508:
507:
493:
487:
486:
484:
465:
459:
458:
438:
432:
431:
429:
410:
404:
403:
383:
306:highest priority
260:highest priority
256:functional group
231:bridgehead atoms
224:
217:
210:
165:bridgehead atoms
21:
674:
673:
669:
668:
667:
665:
664:
663:
644:
643:
630:
625:
624:
611:
610:
606:
596:
594:
557:
556:
552:
545:
519:
518:
514:
505:
503:
495:
494:
490:
473:IUPAC GOLD BOOK
467:
466:
462:
455:
440:
439:
435:
418:IUPAC GOLD BOOK
412:
411:
407:
400:
385:
384:
380:
375:
277:get the prefix
244:
223:
219:
216:
212:
209:
205:
154:fused/condensed
135:spiro compounds
28:
23:
22:
15:
12:
11:
5:
672:
670:
662:
661:
656:
646:
645:
642:
641:
636:
629:
626:
623:
622:
604:
571:(1): 143–216.
550:
543:
512:
488:
460:
453:
433:
405:
398:
377:
376:
374:
371:
243:
240:
199:
198:
179:
150:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
671:
660:
657:
655:
652:
651:
649:
640:
637:
635:
632:
631:
627:
618:
614:
608:
605:
593:on 2016-10-12
592:
588:
584:
579:
574:
570:
567:
566:
561:
554:
551:
546:
540:
536:
532:
528:
525:. Cambridge:
524:
523:
516:
513:
502:
498:
492:
489:
483:
478:
474:
470:
464:
461:
456:
450:
446:
445:
437:
434:
428:
423:
419:
415:
409:
406:
401:
395:
392:. CRC Press.
391:
390:
382:
379:
372:
370:
368:
365:
361:
357:
353:
349:
345:
340:
338:
337:bicyclobutane
334:
328:
326:
323:
319:
315:
311:
307:
303:
299:
294:
292:
288:
284:
280:
276:
272:
269:
265:
261:
257:
254:denoting the
253:
249:
241:
236:
232:
228:
203:
196:
192:
188:
184:
180:
177:
173:
170:
166:
162:
158:
155:
151:
148:
144:
140:
136:
132:
131:
130:
127:
125:
122:
118:
115:
111:
108:), or can be
107:
103:
100:
96:
92:
88:
84:
80:
76:
72:
68:
64:
56:
52:
44:
37:
32:
19:
639:Bredt's rule
616:
607:
595:. Retrieved
591:the original
568:
563:
553:
521:
515:
504:. Retrieved
500:
491:
472:
463:
443:
436:
417:
408:
388:
381:
366:
344:heterocyclic
341:
329:
325:substituents
317:
312:goes before
295:
290:
278:
270:
245:
242:Nomenclature
234:
230:
191:cyclopentane
182:
168:
164:
160:
156:
153:
143:photochromic
128:
120:
113:
98:
87:heterocyclic
70:
66:
62:
60:
358:instead of
117:naphthalene
648:Categories
506:2021-05-06
373:References
283:norbornane
273:, whereas
187:norbornane
147:spiropyran
106:norbornane
65:(from
36:norbornane
587:1365-3075
367:1,4-diaza
346:molecule
258:with the
225:bicyclic
172:α-thujene
95:aliphatic
79:α-thujene
628:See also
475:. 2014.
420:. 2014.
356:nitrogen
302:carbonyl
266:get the
124:tetralin
110:aromatic
18:Bicyclic
597:7 March
333:decalin
318:bicyclo
298:camphor
291:bicyclo
287:heptane
271:bicyclo
227:alkanes
195:Camphor
176:decalin
145:switch
102:decalin
83:camphor
585:
541:
451:
396:
364:prefix
360:carbon
352:octane
322:methyl
314:proton
310:methyl
268:prefix
252:suffix
229:. The
218:, and
348:DABCO
279:spiro
235:three
91:DABCO
75:rings
71:cycle
55:DABCO
599:2016
583:ISSN
539:ISBN
449:ISBN
394:ISBN
342:The
174:and
169:e.g.
163:the
161:i.e.
121:e.g.
114:e.g.
104:and
99:e.g.
81:and
573:doi
531:doi
477:doi
422:doi
181:In
152:In
133:In
126:).
650::
615:.
581:.
569:70
562:.
537:.
499:.
471:.
416:.
339:.
222:11
211:,
178:).
67:bi
61:A
601:.
575::
547:.
533::
509:.
485:.
479::
457:.
430:.
424::
402:.
308:(
220:C
215:9
213:C
208:8
206:C
149:.
112:(
97:(
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.