275:
180:
31:
439:
117:
298:
InChI=1S/C28H38N2O4/c1-5-17-16-30-9-7-19-13-27(33-3)28(34-4)15-22(19)24(30)11-20(17)10-23-21-14-26(32-2)25(31)12-18(21)6-8-29-23/h12-15,17,20,23-24,29,31H,5-11,16H2,1-4H3/t17-,20-,23+,24-/m0/s1
506:
Cephaeline in the form of syrup of ipecac was once commonly recommended as an emergency treatment for accidental poisoning, but its use has been phased out due to its ineffectiveness.
452:
314:
289:
622:
524:
409:
632:
459:
232:
253:
549:
490:. Cephaeline induces vomiting by stimulating the stomach lining and is found in commercial products such as
175:
137:
480:
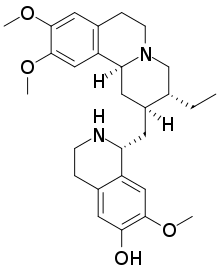
53:
43:
270:
83:
598:
637:
627:
588:
337:
157:
241:
491:
274:
179:
93:
430:
616:
168:
221:
575:
American
Academy Of Pediatrics Committee On Injury, Violence (November 2003).
550:"Pharma Japan: Approval of 4 drugs including Seroquel recommended: CPAC panel"
523:
Lara, Alfonso; Valverde, Roberto; Gomez, Luis; Hidalgo, Nancy (July 1, 2003).
392:
371:
148:
593:
576:
486:
602:
62:)-1-{isoquinolin-2-yl]methyl}-7-methoxy-1,2,3,4-tetrahydroisoquinolin-6-ol
475:
17:
495:
208:
30:
128:
429:
Except where otherwise noted, data are given for materials in their
196:
116:
106:
187:
525:"Micropropagacion de la planta medicinal psychotria acuminata"
258:
447:
577:"Policy statement: Poison treatment in the home"
322:CC1CN2CCc3cc(c(cc32C1C4c5cc(c(cc5CCN4)O)OC)OC)OC
220:
92:
71:Cepheline; Desmethylemetine; Dihydropsychotrine
8:
273:
178:
156:
22:
592:
240:
494:. Chemically, it is closely related to
515:
319:
294:
269:
169:
301:Key: DTGZHCFJNDAHEN-OZEXIGSWSA-N
136:
7:
211:
195:
484:and other plant species including
14:
437:
355:
349:
29:
433:(at 25 °C , 100 kPa).
47:7′,10,11-Trimethoxyemetan-6′-ol
410:Occupational safety and health
361:
343:
1:
654:
554:Chemical Business Newsbase
15:
427:
407:
402:
330:
310:
285:
76:
68:
52:
42:
37:
28:
16:Not to be confused with
594:10.1542/peds.112.5.1182
529:Agronomía Costarricense
623:Isoquinoline alkaloids
481:Cephaelis ipecacuanha
387:White silky crystals
54:Systematic IUPAC name
633:Norsalsolinol ethers
487:Psychotria acuminata
556:. November 14, 2000
423:Emetic / poisonous
379: g·mol
25:
460:Infobox references
23:
478:that is found in
468:Chemical compound
466:
465:
254:CompTox Dashboard
118:Interactive image
645:
607:
606:
596:
587:(5): 1182–1185.
572:
566:
565:
563:
561:
546:
540:
539:
537:
535:
520:
502:Poison treatment
450:
444:
441:
440:
378:
363:
357:
351:
345:
338:Chemical formula
278:
277:
262:
260:
244:
224:
213:
199:
182:
171:
160:
140:
120:
96:
33:
26:
653:
652:
648:
647:
646:
644:
643:
642:
613:
612:
611:
610:
574:
573:
569:
559:
557:
548:
547:
543:
533:
531:
522:
521:
517:
512:
504:
492:syrup of ipecac
469:
462:
457:
456:
455: ?)
446:
442:
438:
434:
420:
376:
366:
360:
354:
348:
340:
326:
323:
318:
317:
306:
303:
302:
299:
293:
292:
281:
271:DTXSID501016520
263:
256:
247:
227:
214:
202:
163:
143:
123:
110:
99:
86:
72:
64:
63:
48:
21:
12:
11:
5:
651:
649:
641:
640:
635:
630:
625:
615:
614:
609:
608:
567:
541:
514:
513:
511:
508:
503:
500:
467:
464:
463:
458:
436:
435:
431:standard state
428:
425:
424:
421:
418:
415:
414:
405:
404:
400:
399:
396:
389:
388:
385:
381:
380:
374:
368:
367:
364:
358:
352:
346:
341:
336:
333:
332:
328:
327:
325:
324:
321:
313:
312:
311:
308:
307:
305:
304:
300:
297:
296:
288:
287:
286:
283:
282:
280:
279:
266:
264:
252:
249:
248:
246:
245:
237:
235:
229:
228:
226:
225:
217:
215:
207:
204:
203:
201:
200:
192:
190:
184:
183:
173:
165:
164:
162:
161:
153:
151:
145:
144:
142:
141:
133:
131:
125:
124:
122:
121:
113:
111:
104:
101:
100:
98:
97:
89:
87:
82:
79:
78:
74:
73:
70:
66:
65:
57:
56:
50:
49:
46:
40:
39:
35:
34:
13:
10:
9:
6:
4:
3:
2:
650:
639:
636:
634:
631:
629:
626:
624:
621:
620:
618:
604:
600:
595:
590:
586:
582:
578:
571:
568:
555:
551:
545:
542:
530:
526:
519:
516:
509:
507:
501:
499:
497:
493:
489:
488:
483:
482:
477:
473:
461:
454:
449:
432:
426:
422:
417:
416:
412:
411:
406:
401:
397:
394:
391:
390:
386:
383:
382:
375:
373:
370:
369:
342:
339:
335:
334:
329:
320:
316:
309:
295:
291:
284:
276:
272:
268:
267:
265:
255:
251:
250:
243:
239:
238:
236:
234:
231:
230:
223:
219:
218:
216:
210:
206:
205:
198:
194:
193:
191:
189:
186:
185:
181:
177:
174:
172:
170:ECHA InfoCard
167:
166:
159:
155:
154:
152:
150:
147:
146:
139:
135:
134:
132:
130:
127:
126:
119:
115:
114:
112:
108:
103:
102:
95:
91:
90:
88:
85:
81:
80:
75:
67:
61:
55:
51:
45:
41:
36:
32:
27:
19:
584:
580:
570:
558:. Retrieved
553:
544:
532:. Retrieved
528:
518:
505:
485:
479:
471:
470:
419:Main hazards
408:
138:ChEMBL255708
77:Identifiers
69:Other names
59:
560:26 December
534:26 December
413:(OHS/OSH):
395:in ethanol
384:Appearance
331:Properties
176:100.006.902
24:Cephaeline
617:Categories
581:Pediatrics
510:References
472:Cephaeline
393:Solubility
372:Molar mass
242:QA971541A1
149:ChemSpider
105:3D model (
84:CAS Number
44:IUPAC name
603:14595067
476:alkaloid
403:Hazards
398:Soluble
94:483-17-0
18:cephalin
638:Emetics
628:Phenols
496:emetine
453:what is
451: (
377:466.622
209:PubChem
601:
474:is an
448:verify
445:
315:SMILES
222:442195
197:C09390
158:390702
129:ChEMBL
38:Names
290:InChI
107:JSmol
599:PMID
562:2009
536:2009
233:UNII
188:KEGG
589:doi
585:112
259:EPA
212:CID
619::
597:.
583:.
579:.
552:.
527:.
498:.
353:38
347:28
58:(1
605:.
591::
564:.
538:.
443:Y
365:4
362:O
359:2
356:N
350:H
344:C
261:)
257:(
109:)
60:R
20:.
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.