335:
237:
44:
664:
832:
35:
468:
727:
Additional diagenetic processes can further alter the cholestane molecule. For instance, cholestane is susceptible to stereochemical shifts over time from its natural isomer. These changes can be the effect of thermal or microbial alteration. Thermal alteration can cause changes in stereochemistry at
790:
will elute from the column based on molecular weight and various stereochemistry, which makes traditional mass spectrometry challenging due to close co-elution of isomers. Alternatively, one can measure cholestane using GC/MS/MS experiments which target the m/z fragment 217 (from molecular ion 372).
630:
in general). Molecular oxygen is required to produce cholesterol; thus, the presence of cholestane suggests some trace of oxygen in the paleoenvironment. Cholestane is not exclusively derived from diagenesis of animal-derived steroid molecules; cholestane may also be associated with the presence of
802:
values of cholestane reflect the carbon isotope composition of the animals that created the original cholesterol molecules. Animal carbon isotope composition is typically understood to be a function of their diet; therefore, carbon isotope composition of cholestane would reflect this original diet
736:
position from α to β configuration reflects anaerobic microbial activity, and can be understood through isotope labeling experiments on controlled microbe experiments metabolizing the steroid of interest. One study demonstrated that there are two reactions that can produce loss of the cholesterol
755:
1264:
Summons, Roger E; Brassell, Simon C; Eglinton, Geoffrey; Evans, Evan; Horodyski, Robert J; Robinson, Neil; Ward, David M (November 1988). "Distinctive hydrocarbon biomarkers from fossiliferous sediment of the Late
Proterozoic Walcott Member, Chuar Group, Grand Canyon, Arizona".
842:
Presence of cholestane does not necessarily indicate presence of animals, but is often used in conjunction with other biomarkers to note the rise of distinct taxa in the fossil record; with regard to this, a study measured relative abundance in cholestane versus other
854:
Tracing the actual origins of cholestane within the fossil record is challenging, as most of the rocks from that time period are heavily metamorphosed and thus potential biomarkers are thermally altered. A study linked the source of cholestane to a specific
677:, but only one of them is formed naturally in production of cholesterol (3β-ol, 5α (H), 14α (H), 17α (H), 20R) and is therefore the primary stereoisomer of interest for cholestane measurements. Deviations from this stereochemistry often reflects
174:
601:
orientation (3β-ol, 5α (H), 14α (H), 17α (H), 20R). This stereochemical orientation is typically maintained throughout diagenetic processes, but cholestane can be found in the fossil record with many stereochemical configurations.
1732:
Hoshino, Yosuke; Poshibaeva, Aleksandra; Meredith, William; Snape, Colin; Poshibaev, Vladimir; Versteegh, Gerard J. M.; Kuznetsov, Nikolay; Leider, Arne; van
Maldegem, Lennart; Neumann, Mareike; Naeher, Sebastian (2017).
167:
691:
typically leads to the loss of functional groups and double bonds in organic molecules. For cholestane specifically, diagenesis of cholesterol to cholestane produces a molecule that is fully saturated compared to its
1100:
Sonawane, Prashant D.; Pollier, Jacob; Panda, Sayantan; Szymanski, Jedrzej; Massalha, Hassan; Yona, Meital; Unger, Tamar; Malitsky, Sergey; Arendt, Philipp; Pauwels, Laurens; Almekias-Siegl, Efrat (2016-12-22).
1205:
Brocks, Jochen J.; Jarrett, Amber J. M.; Sirantoine, Eva; Hallmann, Christian; Hoshino, Yosuke; Liyanage, Tharika (August 2017). "The rise of algae in
Cryogenian oceans and the emergence of animals".
791:
This specific method first looks for the 372 molecular ion of cholestane, and then fragments that molecular ion further to its m/z 217 fragment in order to improve identification of specific isomers.
525:
in the rock record. Presence of cholestane, its derivatives and related chemical compounds in environmental samples is commonly interpreted as an indicator of animal life and/or traces of O
732:
chiral center, as well as the hydrogen atoms. The ratio of R/S stereoisomers is typically reported as a measure of “thermal maturity”. In contrast, conversion of the hydrogen atom at the C
529:, as animals are known for exclusively producing cholesterol, and thus has been used to draw evolutionary relationships between ancient organisms of unknown phylogenetic origin and modern
1622:
Mermoud, F.; Wünsche, L.; Clerc, O.; Gülaçar, F.O.; Buchs, A. (January 1984). "Steroidal ketones in the early diagenetic transformations of Δ5 sterols in different types of sediments".
696:
counterpart. This process occurs without the loss or gain of carbon atoms and therefore can serve as an indicator of the original steroid produced by the organism in the environment.
668:
Cholesterol degrades to cholestane by loss of OH functional group and saturation of double bond (indicated in pink). Stereochemistry of the molecule is maintained in this degradation.
541:), but because these other organisms produce a variety of sterols it cannot be used as a conclusive indicator of any one taxon. It is often found in analysis of organic compounds in
481:
737:
double bond—(1) direct reduction of double bond or (2) production of ketone prior to reduction of double bond—resulting in distinct isomerization of the hydrogen atom at the C
1461:
Grantham, P.J.; Wakefield, L.L. (January 1988). "Variations in the sterane carbon number distributions of marine source rock derived crude oils through geological time".
956:
960:
1657:
Taylor, Craig D.; Smith, Steven O.; Gagosian, Robert B. (November 1981). "Use of microbial enrichments for the study of the anaerobic degradation of cholesterol".
1550:
Schoell, M.; Schouten, S.; Damste, J. S. S.; de Leeuw, J. W.; Summons, R. E. (1994-02-25). "A Molecular
Organic Carbon Isotope Record of Miocene Climate Changes".
358:
InChI=1S/C27H48/c1-19(2)9-8-10-20(3)23-14-15-24-22-13-12-21-11-6-7-17-26(21,4)25(22)16-18-27(23,24)5/h19-25H,6-18H2,1-5H3/t20-,21?,22+,23-,24+,25+,26+,27-/m1/s1
368:
InChI=1/C27H48/c1-19(2)9-8-10-20(3)23-14-15-24-22-13-12-21-11-6-7-17-26(21,4)25(22)16-18-27(23,24)5/h19-25H,6-18H2,1-5H3/t20-,21?,22+,23-,24+,25+,26+,27-/m1/s1
384:
639:, although the abundance of such non-metaozan cholestane is unknown. Embryophytes generally produce a variety of sterols, which are collectively known as
767:
741:
position. The C14 and C17α hydrogen atoms are more stable and undergo changes to β configuration in much lower abundances than the 5 hydrogen atom.
707:. An experiment demonstrated that over 4 weeks at 300 °C, cholestane underwent 17% decomposition of its alkane side chain. In contrast, the
783:
554:
504:
1164:
1717:
932:
349:
983:
Bobrovskiy, Ilya; Hope, Janet M.; Ivantsov, Andrey; Nettersheim, Benjamin J.; Hallmann, Christian; Brocks, Jochen J. (2018-09-20).
770:
to quantify relative abundance to other organic compounds. This measurement is done by extraction of the steranes into a non-polar
565:, which is an organic molecule made primarily by animals and make up ~30% of animal cell membranes. Cholesterol is responsible for
806:
More generally, steranes can be used as an indicator of environmental shifts. A study has presented δC values of steranes versus
614:) of ancient animal life and is often used by geochemists and geobiologists to reconstruct animal evolution (particularly in the
906:
708:
255:
1309:
488:
291:
814:, as changes in the isotope value must be either a result of dissolved inorganic carbon within the water or biological
313:
1188:
1797:
570:
1700:
Hayes, John M. (2001-12-31), "3. Fractionation of Carbon and
Hydrogen Isotopes in Biosynthetic Processes",
232:
1051:
815:
611:
70:
60:
1823:
1818:
1746:
1666:
1559:
1513:
1470:
1342:
1274:
1214:
1063:
996:
330:
1333:
Summons, Roger E.; Erwin, Douglas H. (2018-09-20). "Chemical clues to the earliest animal fossils".
1813:
875:. Cholestane is not a specific marker for animals though and is found in most eukaryotic lineages.
116:
754:
1601:
1384:
1246:
1182:
1138:
950:
864:
799:
1772:
1713:
1682:
1639:
1593:
1585:
1529:
1486:
1443:
1425:
1376:
1368:
1290:
1238:
1230:
1170:
1160:
1130:
1122:
1079:
1032:
1024:
938:
928:
682:
214:
1762:
1754:
1705:
1674:
1631:
1575:
1567:
1521:
1478:
1433:
1415:
1358:
1350:
1282:
1222:
1114:
1071:
1014:
1004:
985:"Ancient steroids establish the Ediacaran fossil Dickinsonia as one of the earliest animals"
578:
412:
300:
126:
910:
868:
775:
674:
598:
334:
236:
142:
134:
1750:
1670:
1563:
1517:
1474:
1346:
1278:
1218:
1067:
1000:
194:
1793:
1767:
1734:
1438:
1403:
848:
803:
value as well. δC values can be measured using a gas chromatograph coupled to an IRMS.
574:
459:
1075:
903:
663:
1807:
1678:
1635:
1525:
1482:
1286:
1250:
872:
716:
225:
1605:
1388:
280:
43:
1142:
590:
1571:
831:
715:) is very thermally stable. Diagenetic processes can also cause methyl shifts and
1504:
Mango, Frank D. (January 1990). "The origin of light cycloalkanes in petroleum".
1102:
1159:. Sadava, David E. (9th ed.). Sunderland, Mass.: Sinauer Associates. 2011.
640:
636:
627:
615:
562:
538:
518:
507:
1402:
Darnet, Sylvain; Blary, Aurélien; Chevalier, Quentin; Schaller, Hubert (2021).
838:
fossil was proved to be ancient animal via cholestane biomarker identification.
1709:
942:
844:
779:
688:
678:
623:
558:
514:
510:
434:
245:
205:
1686:
1643:
1589:
1533:
1490:
1429:
1420:
1372:
1294:
1234:
1174:
1126:
1118:
1103:"Plant cholesterol biosynthetic pathway overlaps with phytosterol metabolism"
1083:
1028:
1354:
1009:
984:
856:
648:
632:
619:
542:
534:
522:
1776:
1758:
1597:
1447:
1380:
1363:
1242:
1134:
1036:
758:
Cholestane isomers elute at different times in GC/MS/MS experiments in the
17:
1310:"Biosynthesis and Regulation of Cholesterol (with Animation) | Animations"
810:
and used it to propose changes in the photic zone over the course of the
644:
594:
586:
566:
530:
1226:
1019:
884:
811:
771:
693:
582:
447:
267:
34:
610:
Cholestane in the fossil record is often interpreted as an indicator (
1580:
807:
787:
700:
652:
533:
taxa. Cholesterol is made in low abundance by other organisms (e.g.,
458:
Except where otherwise noted, data are given for materials in their
830:
786:” fraction using silica gel column gas chromatography. Cholestane
753:
662:
185:
173:
166:
156:
557:
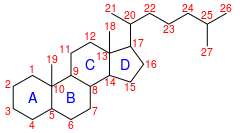
C-27 animal biomarker often found in petroleum deposits. It is a
766:
Cholestane can be extracted from samples and measured on the
318:
643:, and cholesterol remains a minor component. In contrast,
1404:"Phytosterol Profiles, Genomes and Enzymes – An Overview"
847:
biomarkers to demonstrate the rise of algae during the
762:
372→217 fragment. Figure adapted from
Bobrovskiy et al.
476:
1052:"Sterols of the amansieae (rhodomelaceae: Rhodophyta)"
1735:"Cryogenian evolution of stigmasteroid biosynthesis"
1545:
1543:
254:
923:Peters, Kenneth E. (Kenneth Eric), 1950- (2007).
279:
1050:Combaut, Georges; Saenger, Peter (April 1984).
331:DTXSID701357332 DTXSID20880725, DTXSID701357332
141:
133:
125:
699:Thermal alteration can also cause loss of the
8:
955:: CS1 maint: multiple names: authors list (
647:produce other cyclic triterpenoids such as
959:) CS1 maint: numeric names: authors list (
581:. In humans, it is also the precursor for
513:. This 27-carbon biomarker is produced by
333:
235:
213:
26:
1796:at the U.S. National Library of Medicine
1766:
1579:
1437:
1419:
1362:
1018:
1008:
299:
397:C41CCCC1(3(2CC(2(C)CC3)(C)CCCC(C)C)CC4)C
896:
389:
354:
329:
1180:
948:
655:are utilized as bacterial biomarkers.
226:
1617:
1615:
361:Key: XIIAYQZJNBULGD-LDHZKLTISA-N
193:
7:
1200:
1198:
1095:
1093:
978:
976:
974:
972:
970:
392:C(CCCC(C)C)1CC21(CC32CCC43(CCCC4)C)C
371:Key: XIIAYQZJNBULGD-LDHZKLTIBN
270:
99:)-9a,11a-Dimethyl-1-hexadecahydro-1
1157:Life : the science of biology
569:rigidity and fluidity, as well as
25:
597:and naturally assumes a specific
1704:, De Gruyter, pp. 225–278,
521:and is one of the most abundant
466:
424:
42:
33:
1659:Geochimica et Cosmochimica Acta
1506:Geochimica et Cosmochimica Acta
1267:Geochimica et Cosmochimica Acta
659:Natural preservation in fossils
462:(at 25 °C , 100 kPa).
927:. Cambridge University Press.
863:to provide constraints to the
651:and their diagenetic products
418:
1:
1572:10.1126/science.263.5150.1122
1076:10.1016/s0031-9422(00)85025-6
1679:10.1016/0016-7037(81)90068-5
1636:10.1016/0146-6380(84)90023-8
1526:10.1016/0016-7037(90)90191-m
1483:10.1016/0146-6380(88)90115-5
1308:Mehta, Sweety (2013-09-17).
1287:10.1016/0016-7037(88)90031-2
904:The Nomenclature of Steroids
871:as evolutionary preludes to
1702:Stable Isotope Geochemistry
1840:
1408:Frontiers in Plant Science
1710:10.1515/9781501508745-006
723:Stereochemical alteration
681:, thermal maturation and
593:). It is synthesized via
456:
405:
380:
345:
109:
69:
59:
54:
41:
32:
1798:Medical Subject Headings
1421:10.3389/fpls.2021.665206
1119:10.1038/nplants.2016.205
1355:10.1126/science.aau9710
1010:10.1126/science.aat7228
782:) and purified into a “
571:intracellular transport
103:-cyclopentaphenanthrene
1759:10.1126/sciadv.1700887
1187:: CS1 maint: others (
839:
763:
745:Measurement techniques
670:
834:
827:Early life biomarkers
816:isotope fractionation
757:
666:
618:Earth history; e.g.,
71:Systematic IUPAC name
1624:Organic Geochemistry
1463:Organic Geochemistry
673:Cholesterol has 256
1751:2017SciA....3E0887H
1671:1981GeCoA..45.2161T
1564:1994Sci...263.1122S
1558:(5150): 1122–1125.
1518:1990GeCoA..54...23M
1475:1988OrGeo..12...61G
1347:2018Sci...361.1198S
1341:(6408): 1198–1199.
1279:1988GeCoA..52.2625S
1227:10.1038/nature23457
1219:2017Natur.548..578B
1068:1984PChem..23..781C
1001:2018Sci...361.1246B
995:(6408): 1246–1249.
925:The biomarker guide
442: g·mol
29:
1314:PharmaXChange.info
909:2011-05-14 at the
867:classification of
840:
764:
671:
489:Infobox references
27:
1665:(11): 2161–2168.
1273:(11): 2625–2637.
1213:(7669): 578–581.
795:δC isotope ratios
683:preservation bias
497:Chemical compound
495:
494:
314:CompTox Dashboard
175:Interactive image
168:Interactive image
16:(Redirected from
1831:
1781:
1780:
1770:
1739:Science Advances
1729:
1723:
1722:
1697:
1691:
1690:
1654:
1648:
1647:
1619:
1610:
1609:
1583:
1547:
1538:
1537:
1501:
1495:
1494:
1458:
1452:
1451:
1441:
1423:
1399:
1393:
1392:
1366:
1330:
1324:
1323:
1321:
1320:
1305:
1299:
1298:
1261:
1255:
1254:
1202:
1193:
1192:
1186:
1178:
1153:
1147:
1146:
1097:
1088:
1087:
1047:
1041:
1040:
1022:
1012:
980:
965:
964:
954:
946:
920:
914:
901:
579:nerve conduction
553:Cholestane is a
479:
473:
470:
469:
441:
426:
420:
413:Chemical formula
338:
337:
322:
320:
303:
283:
272:
258:
239:
228:
217:
197:
177:
170:
145:
137:
129:
46:
37:
30:
21:
1839:
1838:
1834:
1833:
1832:
1830:
1829:
1828:
1804:
1803:
1790:
1785:
1784:
1745:(9): e1700887.
1731:
1730:
1726:
1720:
1699:
1698:
1694:
1656:
1655:
1651:
1621:
1620:
1613:
1549:
1548:
1541:
1503:
1502:
1498:
1460:
1459:
1455:
1401:
1400:
1396:
1364:1721.1/134716.2
1332:
1331:
1327:
1318:
1316:
1307:
1306:
1302:
1263:
1262:
1258:
1204:
1203:
1196:
1179:
1167:
1155:
1154:
1150:
1099:
1098:
1091:
1049:
1048:
1044:
982:
981:
968:
947:
935:
922:
921:
917:
911:Wayback Machine
902:
898:
893:
881:
869:Ediacaran biota
829:
824:
797:
776:dichloromethane
752:
747:
740:
735:
731:
725:
714:
706:
703:side-chain at C
661:
608:
551:
528:
498:
491:
486:
485:
484: ?)
475:
471:
467:
463:
439:
429:
423:
415:
401:
398:
393:
388:
387:
376:
373:
372:
369:
363:
362:
359:
353:
352:
341:
323:
316:
307:
286:
273:
261:
248:
220:
200:
180:
160:
149:
119:
105:
104:
65:
50:
49:IUPAC numbering
47:
23:
22:
15:
12:
11:
5:
1837:
1835:
1827:
1826:
1821:
1816:
1806:
1805:
1802:
1801:
1789:
1788:External links
1786:
1783:
1782:
1724:
1718:
1692:
1649:
1611:
1539:
1496:
1453:
1394:
1325:
1300:
1256:
1194:
1166:978-1429219624
1165:
1148:
1089:
1062:(4): 781–782.
1056:Phytochemistry
1042:
966:
933:
915:
895:
894:
892:
889:
888:
887:
880:
877:
849:Neoproterozoic
828:
825:
823:
820:
796:
793:
751:
748:
746:
743:
738:
733:
729:
724:
721:
712:
704:
660:
657:
607:
604:
599:stereochemical
575:cell signaling
550:
547:
526:
496:
493:
492:
487:
465:
464:
460:standard state
457:
454:
453:
450:
444:
443:
437:
431:
430:
427:
421:
416:
411:
408:
407:
403:
402:
400:
399:
396:
394:
391:
383:
382:
381:
378:
377:
375:
374:
370:
367:
366:
364:
360:
357:
356:
348:
347:
346:
343:
342:
340:
339:
326:
324:
312:
309:
308:
306:
305:
296:
294:
288:
287:
285:
284:
276:
274:
266:
263:
262:
260:
259:
251:
249:
244:
241:
240:
230:
222:
221:
219:
218:
210:
208:
202:
201:
199:
198:
190:
188:
182:
181:
179:
178:
171:
163:
161:
154:
151:
150:
148:
147:
139:
131:
122:
120:
115:
112:
111:
107:
106:
74:
73:
67:
66:
63:
57:
56:
52:
51:
48:
39:
38:
24:
14:
13:
10:
9:
6:
4:
3:
2:
1836:
1825:
1822:
1820:
1817:
1815:
1812:
1811:
1809:
1799:
1795:
1792:
1791:
1787:
1778:
1774:
1769:
1764:
1760:
1756:
1752:
1748:
1744:
1740:
1736:
1728:
1725:
1721:
1719:9781501508745
1715:
1711:
1707:
1703:
1696:
1693:
1688:
1684:
1680:
1676:
1672:
1668:
1664:
1660:
1653:
1650:
1645:
1641:
1637:
1633:
1629:
1625:
1618:
1616:
1612:
1607:
1603:
1599:
1595:
1591:
1587:
1582:
1577:
1573:
1569:
1565:
1561:
1557:
1553:
1546:
1544:
1540:
1535:
1531:
1527:
1523:
1519:
1515:
1511:
1507:
1500:
1497:
1492:
1488:
1484:
1480:
1476:
1472:
1468:
1464:
1457:
1454:
1449:
1445:
1440:
1435:
1431:
1427:
1422:
1417:
1413:
1409:
1405:
1398:
1395:
1390:
1386:
1382:
1378:
1374:
1370:
1365:
1360:
1356:
1352:
1348:
1344:
1340:
1336:
1329:
1326:
1315:
1311:
1304:
1301:
1296:
1292:
1288:
1284:
1280:
1276:
1272:
1268:
1260:
1257:
1252:
1248:
1244:
1240:
1236:
1232:
1228:
1224:
1220:
1216:
1212:
1208:
1201:
1199:
1195:
1190:
1184:
1176:
1172:
1168:
1162:
1158:
1152:
1149:
1144:
1140:
1136:
1132:
1128:
1124:
1120:
1116:
1112:
1108:
1107:Nature Plants
1104:
1096:
1094:
1090:
1085:
1081:
1077:
1073:
1069:
1065:
1061:
1057:
1053:
1046:
1043:
1038:
1034:
1030:
1026:
1021:
1016:
1011:
1006:
1002:
998:
994:
990:
986:
979:
977:
975:
973:
971:
967:
962:
958:
952:
944:
940:
936:
934:9780521039987
930:
926:
919:
916:
912:
908:
905:
900:
897:
890:
886:
883:
882:
878:
876:
874:
873:metazoan life
870:
866:
862:
858:
852:
850:
846:
837:
833:
826:
821:
819:
817:
813:
809:
804:
801:
794:
792:
789:
785:
781:
777:
773:
769:
761:
756:
749:
744:
742:
722:
720:
718:
717:aromatization
710:
702:
697:
695:
690:
686:
684:
680:
676:
675:stereoisomers
669:
665:
658:
656:
654:
650:
646:
642:
638:
634:
629:
625:
621:
617:
613:
605:
603:
600:
596:
592:
588:
584:
580:
576:
572:
568:
564:
560:
556:
548:
546:
544:
540:
536:
532:
524:
520:
516:
512:
509:
506:
502:
490:
483:
478:
461:
455:
451:
449:
446:
445:
438:
436:
433:
432:
417:
414:
410:
409:
404:
395:
390:
386:
379:
365:
355:
351:
344:
336:
332:
328:
327:
325:
315:
311:
310:
302:
298:
297:
295:
293:
290:
289:
282:
278:
277:
275:
269:
265:
264:
257:
253:
252:
250:
247:
243:
242:
238:
234:
231:
229:
227:ECHA InfoCard
224:
223:
216:
212:
211:
209:
207:
204:
203:
196:
192:
191:
189:
187:
184:
183:
176:
172:
169:
165:
164:
162:
158:
153:
152:
144:
140:
136:
132:
128:
124:
123:
121:
118:
114:
113:
108:
102:
98:
94:
90:
86:
82:
78:
72:
68:
62:
58:
53:
45:
40:
36:
31:
19:
1742:
1738:
1727:
1701:
1695:
1662:
1658:
1652:
1627:
1623:
1555:
1551:
1512:(1): 23–27.
1509:
1505:
1499:
1469:(1): 61–73.
1466:
1462:
1456:
1411:
1407:
1397:
1338:
1334:
1328:
1317:. Retrieved
1313:
1303:
1270:
1266:
1259:
1210:
1206:
1156:
1151:
1113:(1): 16205.
1110:
1106:
1059:
1055:
1045:
992:
988:
924:
918:
899:
861:Dickinsonia)
860:
853:
845:triterpenoid
841:
835:
822:Case studies
805:
798:
765:
759:
726:
711:structure (C
698:
687:
672:
667:
641:phytosterols
637:embryophytes
609:
591:testosterone
552:
500:
499:
110:Identifiers
100:
96:
92:
88:
84:
80:
76:
1824:Triterpenes
1819:Cholestanes
1794:Cholestanes
1020:1885/230014
836:Dickinsonia
633:rhodophytes
628:Proterozoic
616:Precambrian
563:cholesterol
561:product of
539:land plants
535:rhodophytes
519:cholesterol
508:tetracyclic
452:0.911 g/ml
406:Properties
233:100.035.496
195:CHEBI:35516
28:Cholestane
18:Cholestanes
1814:Biomarkers
1808:Categories
1319:2019-06-04
943:1015511618
891:References
780:chloroform
728:both the C
709:polycyclic
689:Diagenesis
679:diagenesis
624:Cryogenian
559:diagenetic
549:Background
523:biomarkers
515:diagenesis
511:triterpene
501:Cholestane
435:Molar mass
304: (5α)
301:U260HWN305
246:IUPHAR/BPS
206:ChemSpider
155:3D model (
130: 5α/β
127:14982-53-7
117:CAS Number
64:Cholestane
61:IUPAC name
1687:0016-7037
1644:0146-6380
1630:: 25–29.
1590:0036-8075
1581:1874/4185
1534:0016-7037
1491:0146-6380
1430:1664-462X
1373:0036-8075
1295:0016-7037
1251:205258987
1235:0028-0836
1183:cite book
1175:368046231
1127:2055-0278
1084:0031-9422
1029:0036-8075
951:cite book
865:taxonomic
857:Ediacaran
784:saturates
649:hopanoids
620:Ediacaran
612:biomarker
606:Biomarker
555:saturated
543:petroleum
505:saturated
1777:28948220
1606:40960698
1598:17831625
1448:34093623
1389:52306517
1381:30237342
1243:28813409
1135:28005066
1037:30237355
907:Archived
879:See also
859:fossil (
645:bacteria
595:squalene
587:estrogen
583:hormones
567:membrane
531:metazoan
146: 5β
143:481-20-9
138: 5α
135:481-21-0
1768:5606710
1747:Bibcode
1667:Bibcode
1560:Bibcode
1552:Science
1514:Bibcode
1471:Bibcode
1439:8172173
1414:: 878.
1343:Bibcode
1335:Science
1275:Bibcode
1215:Bibcode
1143:5518449
1064:Bibcode
997:Bibcode
989:Science
913:, IUPAC
885:Sterane
812:Miocene
808:hopanes
788:isomers
774:(e.g.,
772:solvent
694:steroid
653:hopanes
585:(i.e.,
482:what is
480: (
448:Density
440:372.681
281:6857534
268:PubChem
215:5256870
1800:(MeSH)
1775:
1765:
1716:
1685:
1642:
1604:
1596:
1588:
1532:
1489:
1446:
1436:
1428:
1387:
1379:
1371:
1293:
1249:
1241:
1233:
1207:Nature
1173:
1163:
1141:
1133:
1125:
1082:
1035:
1027:
941:
931:
701:alkane
631:e.g.,
477:verify
474:
385:SMILES
55:Names
1602:S2CID
1385:S2CID
1247:S2CID
1139:S2CID
768:GC/MS
750:GC/MS
503:is a
350:InChI
186:ChEBI
157:JSmol
1773:PMID
1714:ISBN
1683:ISSN
1640:ISSN
1594:PMID
1586:ISSN
1530:ISSN
1487:ISSN
1444:PMID
1426:ISSN
1377:PMID
1369:ISSN
1291:ISSN
1239:PMID
1231:ISSN
1189:link
1171:OCLC
1161:ISBN
1131:PMID
1123:ISSN
1080:ISSN
1033:PMID
1025:ISSN
961:link
957:link
939:OCLC
929:ISBN
713:1-17
635:and
626:and
577:and
292:UNII
256:2802
95:,11a
1763:PMC
1755:doi
1706:doi
1675:doi
1632:doi
1576:hdl
1568:doi
1556:263
1522:doi
1479:doi
1434:PMC
1416:doi
1359:hdl
1351:doi
1339:361
1283:doi
1223:doi
1211:548
1115:doi
1072:doi
1015:hdl
1005:doi
993:361
778:or
760:m/z
517:of
319:EPA
271:CID
91:,9b
87:,9a
83:,3b
79:,3a
1810::
1771:.
1761:.
1753:.
1741:.
1737:.
1712:,
1681:.
1673:.
1663:45
1661:.
1638:.
1626:.
1614:^
1600:.
1592:.
1584:.
1574:.
1566:.
1554:.
1542:^
1528:.
1520:.
1510:54
1508:.
1485:.
1477:.
1467:12
1465:.
1442:.
1432:.
1424:.
1412:12
1410:.
1406:.
1383:.
1375:.
1367:.
1357:.
1349:.
1337:.
1312:.
1289:.
1281:.
1271:52
1269:.
1245:.
1237:.
1229:.
1221:.
1209:.
1197:^
1185:}}
1181:{{
1169:.
1137:.
1129:.
1121:.
1109:.
1105:.
1092:^
1078:.
1070:.
1060:23
1058:.
1054:.
1031:.
1023:.
1013:.
1003:.
991:.
987:.
969:^
953:}}
949:{{
937:.
851:.
818:.
800:δC
730:20
719:.
705:17
685:.
622:,
589:,
573:,
545:.
537:,
428:48
422:27
75:(1
1779:.
1757::
1749::
1743:3
1708::
1689:.
1677::
1669::
1646:.
1634::
1628:6
1608:.
1578::
1570::
1562::
1536:.
1524::
1516::
1493:.
1481::
1473::
1450:.
1418::
1391:.
1361::
1353::
1345::
1322:.
1297:.
1285::
1277::
1253:.
1225::
1217::
1191:)
1177:.
1145:.
1117::
1111:3
1086:.
1074::
1066::
1039:.
1017::
1007::
999::
963:)
945:.
739:5
734:5
527:2
472:Y
425:H
419:C
321:)
317:(
159:)
101:H
97:R
93:S
89:S
85:R
81:S
77:R
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.