347:
324:
1010:
29:
577:
Clodronic acid is approved for human use in Canada and
Australia, the United Kingdom, where it is marketed as Bonefos, Loron, Clodron and in Italy as Clasteon, Difosfonal, Osteostab and several generics. In other countries is prescribed as a bone resorption inhibitor and antihypercalcemic agent. It
590:. It is given by intramuscular injection at one point in time, with the total dose divided into 2-3 sites on the horse. Clinical effects (e.g. improvement of lameness) after a single treatment can be seen up to 6 months post-treatment.
751:
635:
Pennanen N, Lapinjoki S, Urtti A, Mönkkönen J (June 1995). "Effect of liposomal and free bisphosphonates on the IL-1 beta, IL-6 and TNF alpha secretion from RAW 264 cells in vitro".
59:
744:
737:
41:
433:
419:
957:
492:
453:
678:
Rovetta G, Monteforte P, Balestra V (2000). "Intravenous clodronate for acute pain induced by osteoporotic vertebral fracture".
121:
89:
567:
in osteoporotic vertebral fractures showed that clodronic acid provided more analgesia than 3 grams/day of acetaminophen.
1000:
862:
711:
707:"FDA Provides Equine Veterinarians with Important Information about TILDREN and OSPHOS for Navicular Syndrome in Horses"
586:
Clodronic acid is approved for use in horses under the trade name Osphos, for treatment of bone resorptive processes of
223:
1035:
552:
303:
543:
and fracture related pain because of its anti-inflammatory effects shown as a reduction in inflammatory markers like
854:
520:
1040:
1030:
895:
342:
292:
706:
615:
532:
319:
138:
905:
660:
587:
544:
272:
960:
687:
652:
103:
729:
1014:
644:
540:
359:
232:
212:
147:
867:
842:
822:
817:
812:
807:
802:
797:
792:
787:
51:
346:
323:
832:
774:
578:
is not approved for use in the United States, because it has too many adverse effects.
524:
1024:
965:
564:
548:
536:
335:
664:
970:
765:
761:
528:
72:
67:
172:
943:
935:
888:
603:
571:
527:. It is an anti-osteoporotic drug approved for the prevention and treatment of
20:
977:
950:
928:
648:
598:
Clodronic acid has been shown to have several adverse effects. These include:
395:
203:
987:
920:
570:
Clodronic acid is also used in experimental medicine to selectively deplete
45:
28:
691:
656:
782:
183:
192:
982:
158:
283:
252:
913:
461:
InChI=1S/CH4Cl2O6P2/c2-1(3,10(4,5)6)11(7,8)9/h(H2,4,5,6)(H2,7,8,9)
418:
409:
263:
563:
A study comparing the analgesic effect of clodronic acid versus
531:
in post-menopausal women and men to reduce vertebral fractures,
243:
733:
308:
98:
478:
998:
876:
853:
773:
407:
394:
358:
353:
334:
302:
282:
262:
242:
222:
202:
182:
157:
137:
112:
88:
83:
58:
40:
35:
171:
146:
680:Drugs Under Experimental and Clinical Research
745:
8:
19:
752:
738:
730:
523:) is a first generation (non-nitrogenous)
345:
322:
211:
129:(Dichloro-phosphono-methyl)phosphonic acid
27:
231:
1005:
627:
458:
438:
318:
191:
126:
606:, usually within 2 hours of treatment.
336:
18:
291:
271:
50:
7:
71:
958:Parathyroid hormone-related protein
705:U.S. Food and Drug Administration.
602:Signs of discomfort, agitation, or
251:
162:
385:
14:
1008:
373:
367:
466:Key:ACSIXWWBWUQEHA-UHFFFAOYSA-N
379:
364:
1:
712:Food and Drug Administration
855:Bone morphogenetic proteins
441:ClC(Cl)(P(=O)(O)O)P(=O)(O)O
1057:
354:Chemical and physical data
474:
449:
429:
117:
26:
52:International Drug Names
896:Aluminium chlorohydrate
760:Drugs for treatment of
649:10.1023/A:1016281608773
637:Pharmaceutical Research
901:Dual action bone agent
884:Resorption inhibitor
533:hyperparathyroidism
497:clodronate disodium
23:
1036:Equine medications
906:Strontium ranelate
588:navicular syndrome
996:
995:
486:
485:
420:Interactive image
304:CompTox Dashboard
101:
16:Chemical compound
1048:
1013:
1012:
1011:
1004:
829:Non-nitrogenous
754:
747:
740:
731:
724:
723:
721:
719:
702:
696:
695:
675:
669:
668:
632:
541:multiple myeloma
482:
481:
422:
402:
387:
381:
375:
369:
366:
349:
338:
327:
326:
312:
310:
295:
275:
255:
235:
215:
195:
175:
165:
164:
150:
100:
97:
75:
54:
31:
24:
22:
1056:
1055:
1051:
1050:
1049:
1047:
1046:
1045:
1041:Organochlorides
1031:Bisphosphonates
1021:
1020:
1019:
1009:
1007:
999:
997:
992:
872:
868:Eptotermin alfa
863:Dibotermin alfa
849:
843:Tiludronic acid
823:Zoledronic acid
818:Risedronic acid
813:Pamidronic acid
808:Neridronic acid
803:Minodronic acid
798:Incadronic acid
793:Ibandronic acid
788:Alendronic acid
775:Bisphosphonates
769:
758:
728:
727:
717:
715:
704:
703:
699:
677:
676:
672:
634:
633:
629:
624:
596:
594:Adverse effects
584:
582:Veterinary uses
561:
539:in malignancy,
518:
514:
510:
506:
502:
477:
475:
470:
467:
462:
457:
456:
445:
442:
437:
436:
425:
400:
390:
384:
378:
372:
330:
306:
298:
278:
258:
238:
218:
198:
178:
161:
153:
133:
130:
125:
124:
108:
79:
17:
12:
11:
5:
1054:
1052:
1044:
1043:
1038:
1033:
1023:
1022:
1018:
1017:
994:
993:
991:
990:
985:
980:
975:
974:
973:
968:
955:
954:
953:
940:
939:
938:
925:
924:
923:
910:
909:
908:
898:
893:
892:
891:
880:
878:
874:
873:
871:
870:
865:
859:
857:
851:
850:
848:
847:
846:
845:
840:
838:Clodronic acid
835:
833:Etidronic acid
827:
826:
825:
820:
815:
810:
805:
800:
795:
790:
779:
777:
771:
770:
759:
757:
756:
749:
742:
734:
726:
725:
697:
670:
626:
625:
623:
620:
619:
618:
613:
610:
607:
595:
592:
583:
580:
560:
557:
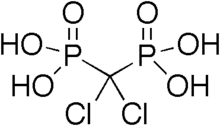
525:bisphosphonate
516:
512:
508:
504:
500:
489:Clodronic acid
484:
483:
472:
471:
469:
468:
465:
463:
460:
452:
451:
450:
447:
446:
444:
443:
440:
432:
431:
430:
427:
426:
424:
423:
415:
413:
405:
404:
398:
392:
391:
388:
382:
376:
370:
362:
356:
355:
351:
350:
340:
332:
331:
329:
328:
315:
313:
300:
299:
297:
296:
288:
286:
280:
279:
277:
276:
268:
266:
260:
259:
257:
256:
248:
246:
240:
239:
237:
236:
228:
226:
220:
219:
217:
216:
208:
206:
200:
199:
197:
196:
188:
186:
180:
179:
177:
176:
168:
166:
155:
154:
152:
151:
143:
141:
135:
134:
132:
131:
128:
120:
119:
118:
115:
114:
110:
109:
107:
106:
94:
92:
86:
85:
81:
80:
78:
77:
64:
62:
56:
55:
48:
38:
37:
33:
32:
21:Clodronic acid
15:
13:
10:
9:
6:
4:
3:
2:
1053:
1042:
1039:
1037:
1034:
1032:
1029:
1028:
1026:
1016:
1006:
1002:
989:
986:
984:
981:
979:
976:
972:
969:
967:
966:Abaloparatide
964:
963:
962:
959:
956:
952:
949:
948:
947:
945:
941:
937:
934:
933:
932:
930:
926:
922:
919:
918:
917:
915:
911:
907:
904:
903:
902:
899:
897:
894:
890:
887:
886:
885:
882:
881:
879:
875:
869:
866:
864:
861:
860:
858:
856:
852:
844:
841:
839:
836:
834:
831:
830:
828:
824:
821:
819:
816:
814:
811:
809:
806:
804:
801:
799:
796:
794:
791:
789:
786:
785:
784:
781:
780:
778:
776:
772:
767:
763:
762:bone diseases
755:
750:
748:
743:
741:
736:
735:
732:
714:
713:
708:
701:
698:
693:
689:
685:
681:
674:
671:
666:
662:
658:
654:
650:
646:
643:(6): 916–22.
642:
638:
631:
628:
621:
617:
616:Renal failure
614:
611:
608:
605:
601:
600:
599:
593:
591:
589:
581:
579:
575:
573:
568:
566:
565:acetaminophen
558:
556:
554:
550:
546:
542:
538:
537:hypercalcemia
534:
530:
526:
522:
498:
494:
490:
480:
473:
464:
459:
455:
448:
439:
435:
428:
421:
417:
416:
414:
411:
406:
399:
397:
393:
363:
361:
357:
352:
348:
344:
341:
339:
337:ECHA InfoCard
333:
325:
321:
320:DTXSID8046959
317:
316:
314:
305:
301:
294:
290:
289:
287:
285:
281:
274:
270:
269:
267:
265:
261:
254:
250:
249:
247:
245:
241:
234:
230:
229:
227:
225:
221:
214:
210:
209:
207:
205:
201:
194:
190:
189:
187:
185:
181:
174:
170:
169:
167:
160:
156:
149:
145:
144:
142:
140:
136:
127:
123:
116:
111:
105:
96:
95:
93:
91:
87:
82:
74:
69:
66:
65:
63:
61:
57:
53:
49:
47:
43:
39:
36:Clinical data
34:
30:
25:
971:Teriparatide
942:
927:
912:
900:
883:
837:
716:. Retrieved
710:
700:
686:(1): 25–30.
683:
679:
673:
640:
636:
630:
609:Head shaking
597:
585:
576:
569:
562:
559:Medical uses
529:osteoporosis
496:
488:
487:
476:
273:CHEBI:110423
90:Legal status
84:Legal status
944:Cathepsin K
936:Romosozumab
889:Ipriflavone
783:Nitrogenous
612:Lip licking
572:macrophages
403: g·mol
343:100.031.090
293:ChEMBL12318
113:Identifiers
1025:Categories
978:Calcitonin
951:Odanacatib
929:Sclerostin
622:References
408:3D model (
396:Molar mass
233:0813BZ6866
204:ChemSpider
148:10596-23-3
139:CAS Number
122:IUPAC name
1015:Chemistry
988:Vitamin D
961:analogues
946:inhibitor
931:inhibitor
921:Denosumab
916:inhibitor
718:3 January
46:Drugs.com
692:10761534
665:46332840
479:(verify)
184:DrugBank
60:ATC code
983:Calcium
657:7667201
360:Formula
193:DB00720
159:PubChem
102::
76:)
70: (
68:M05BA02
1001:Portal
690:
663:
655:
551:, and
434:SMILES
401:244.88
284:ChEMBL
253:D03545
104:℞-only
914:RANKL
877:Other
661:S2CID
604:colic
553:TNF-α
545:IL-1β
495:) or
454:InChI
410:JSmol
264:ChEBI
213:23731
173:25419
720:2015
688:PMID
653:PMID
549:IL-6
521:USAN
244:KEGG
224:UNII
42:AHFS
766:M05
645:doi
555:.
519:) (
499:(Na
493:INN
309:EPA
163:CID
73:WHO
1027::
709:.
684:26
682:.
659:.
651:.
641:12
639:.
574:.
547:,
535:,
507:Cl
503:CH
374:Cl
99:CA
1003::
768:)
764:(
753:e
746:t
739:v
722:.
694:.
667:.
647::
517:2
515:P
513:6
511:O
509:2
505:2
501:2
491:(
412:)
389:2
386:P
383:6
380:O
377:2
371:4
368:H
365:C
311:)
307:(
44:/
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.