372:
624:
56:, the intramolecular esterification of the corresponding hydroxycarboxylic acids, which takes place spontaneously when the ring that is formed is five- or six-membered. Lactones with three- or four-membered rings (α-lactones and β-lactones) are very reactive, making their isolation difficult. Special methods are normally required for the laboratory synthesis of small-ring lactones as well as those that contain rings larger than six-membered.
768:
532:
175:
118:
570:
206:
152:
241:
1187:(It's desirable for this group of compounds — whose simplest representative until now has been the substance that's described in the preceding — to have a general designation, and since the name "lactide" isn't applicable because then the archetypal lactide would not be a lactide, we therefore suggest the name "lactone" as the designation of this group .)
1185:"Es ist wünschenswerth, für diese Gruppe von Verbindungen, deren bis jetzt einfachster Repräsentant der im Vorstehenden beschriebene Körper ist, eine allgemeine Bezeichnungsweise zu haben, und da der Name "Lactide" nicht anwendbar ist, weil dann das Lactid κατ εξοχην kein Lactid sein wurde, so schlagen wir als Gruppenbezeichnung den Namen "Lactone" vor".
273:
suffix and a Greek letter prefix that specifies the number of carbon atoms in the heterocycle — that is, the distance between the relevant -OH and the -COOH groups along said backbone. The first carbon atom after the carbon in the -COOH group on the parent compound is labelled α, the second will be
714:
of the hydrolysis of lactones is less than the entropy of straight-chained esters. Straight-chained esters give two products upon hydrolysis, making the entropy change more favorable than in the case of lactones which gives only a single product.
1076:"Indépendamment de la lactide dont je viens de rappeler l'existence dans les produits de la distllation de l'acide lactique, celui-ci donne encore, par sa décomposition, une autre substance, que je propose d'appeler
1082:(Independently of the lactide of which I have just recalled the existence in the products of the distillation of lactic acid, this gives further, by its decomposition, another substance, which I propose to call
706:
of the hydrolysis reaction of the lactone is lower than that of the straight-chained ester i.e. the products (hydroxyacids) are less favored in the case of the lactones. This is because although the
274:
labeled β, and so forth. Therefore, the prefixes also indicate the size of the lactone ring: α-lactone = 3-membered ring, β-lactone = 4-membered, γ-lactone = 5-membered, δ-lactone = 6-membered, etc.
320:, lactones are named as heterocyclic pseudoketones by adding the suffix 'one', 'dione', 'thione', etc. and the appropriate multiplicative prefixes to the name of the heterocyclic parent hydride.
831:. Replacement of a methylene unit by oxygen barely affects the odor of these compounds, and oxalactones with 15 – 17-membered rings are produced in addition to cyclopentadecanolide (e. g.,
784:
Lactones contribute significantly to the flavor of fruit, and of unfermented and fermented dairy products, and are therefore used as flavors and fragrances. Some examples are
389:
5-Membered γ-lactones and 6-membered δ-lactones are prevalent. β-lactones appear in a number of natural products. α‑Lactones can be detected as transient species in
344:-CH(OH)-COOH. Lactic acid, in turn, derives its name from its original isolation from soured milk (Latin: lac, lactis). The name was coined in 1844 by the French chemist
1388:
1257:
976:
694:
the lactone to its parent compound, the straight chained bifunctional compound. Like straight-chained esters, the hydrolysis-condensation reaction of lactones is a
792:(5-decanolide), which has a creamy coconut/peach flavour; γ-dodecalactone (4-dodecanolide), which also has a coconut/fruity flavor, a description which also fits
543:
for longer hydroxy acids, or the strained β‑lactones. γ‑Lactones, on the other hand, are so stable that 4-hydroxy acids (R-CH(OH)-(CH
1477:
1044:
1010:
667:
971:
1493:
Odile Dechy-Cabaret; Blanca Martin-Vaca; Didier
Bourissou (2004). "Controlled Ring-Opening Polymerization of Lactide and Glycolide".
1614:
1580:
1546:
1143:
609:
1197:
Danheiser, Rick L.; Nowick, James S. (1991) . "A practical and efficient method for the synthesis of β‑lactones".
613:
1092:
865:
1162:
1707:
1028:
345:
727:
605:
812:
617:
601:
1231:. International Journal of Mass Spectrometry and Ion Processes, Volumes 165-166, November issue, Pages 71-82.
885:
832:
895:
660:
590:
360:
38:
904:
762:(polylactide). The resulting polylactic acid has been heavily investigated for commercial applications.
375:
1639:"Polyesters as a Model System for Building Primitive Biologies from Non-Biological Prebiotic Chemistry"
371:
1650:
1416:
1322:
870:
808:
703:
699:
644:
539:
Many methods in ester synthesis can also be applied to that of lactones. Lactonization competes with
460:
397:
349:
317:
1061:
1350:
1025:
Nomenclature of
Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book)
695:
353:
1354:
196:
1403:
Chandru, Kuhan; Jia, Tony Z.; Mamajanov, Irena; Bapat, Niraja; Cleaves, H. James (2020-10-16).
216:
185:
130:
103:
1702:
1678:
1610:
1604:
1576:
1542:
1536:
1510:
1473:
1450:
1432:
1366:
1342:
1334:
1139:
1129:
1040:
944:
578:
520:
390:
1570:
1405:"Prebiotic oligomerization and self-assembly of structurally diverse xenobiological monomers"
1111:
1006:
1668:
1658:
1502:
1440:
1424:
1338:
1326:
1298:
1232:
1206:
1174:
1032:
843:
824:
800:, which has an intense coconut flavor of this series, despite not occurring in coconut, and
797:
793:
789:
785:
687:
648:
640:
636:
632:
563:
500:
472:
429:
34:
1080:, parce qu'elle me paraît être à l'acide lactique ce que l'acétone est à l'acide acétique."
767:
623:
759:
594:
516:
421:
117:
81:
1654:
1420:
1673:
1638:
1445:
1404:
1224:
652:
540:
531:
496:
456:
448:
174:
64:
42:
1236:
846:
is an important plastic. Its formation has even been considered in the context of the
1696:
512:
476:
444:
436:
401:
309:
1273:
859:
656:
492:
205:
111:
1637:
Chandru, Kuhan; Mamajanov, Irena; Cleaves, H. James; Jia, Tony Z. (January 2020).
1470:
Poly(lactic acid): Synthesis, Structures, Properties, Processing, and
Applications
801:
162:
1229:
Generation of α-acetolactone and the acetoxyl diradical •CH2COO• in the gas phase
1314:
1036:
847:
569:
559:
508:
337:
303:
275:
1428:
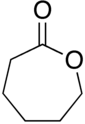
1374:
1086:, because it seems to me to be to lactic acid what acetone is to acetic acid.)
1318:
504:
484:
480:
405:
291:
87:
46:
1436:
1178:
949:
828:
691:
488:
468:
297:
1682:
1514:
1454:
754:
Some lactones convert to polyesters: For example the double lactone called
718:
Lactones also react with amines to give the ring-opened alcohol and amide.
151:
1293:
John E. Cabaj, David Kairys, and Thomas R. Benson Org. Process Res. Dev.;
240:
1663:
1569:
Mehta, Bhavbhuti M.; Kamal-Eldin, Afaf; Iwanski, Robert Z., eds. (2012).
1346:
820:
707:
452:
417:
382:
1210:
593:
with the cationic intermediate captured intramolecularly by an adjacent
17:
935:
880:
816:
755:
711:
586:
464:
333:
1506:
1302:
116:
926:
917:
582:
440:
413:
409:
363:
extended the name "lactone" to all intramolecular carboxylic esters.
249:
Lactones are usually named according to the precursor acid molecule (
1386:
1255:
Karl-Georg
Fahlbusch; et al. (2007), "Flavors and Fragrances",
435:
Lactone rings occur widely as building blocks in nature, such as in
348:, who first obtained it as a derivative of lactic acid. An internal
1538:
Flavours and fragrances chemistry, bioprocessing and sustainability
336:, which is formed from the dehydration of 2-hydroxypropanoic acid (
1330:
939:
930:
921:
622:
568:
530:
370:
730:. For instance, gamma-lactones is reduced to butane-1,4-diol, (CH
710:
of the hydrolysis of esters and lactones are about the same, the
1165:[Investigations into unsaturated acids, third article].
1136:
Dictionary of
Chemistry: an etymological and historical approach
1131:
Dictionnaire de chimie: Une approche étymologique et historique
815:) have odors similar to macrocyclic ketones of animal origin (
425:
1138:] (in French). Brussels, Belgium: de boeck. p. 183.
796:(4-octanolide), although it also has a herbaceous character;
352:
within the same molecule of lactic acid would have produced
1163:"Untersuchungen über ungesättige Säuren, dritte Abhandlung"
666:
An alternative radical reaction yielding γ-lactones is the
1291:
Development of a
Commercial Process to Produce Oxandrolone
396:
Macrocyclic lactones are also important natural products.
1609:. Boca Raton, FL: CRC/Taylor & Francis. p. 242.
1274:"The Oxford Companion to Beer definition of barrel-aging"
823:), but they can be prepared more easily, for example, by
788:(4-decanolide), which has a characteristic peach flavor;
678:
Lactones exhibit the reactions characteristic of esters.
627:γ-Lactone synthesis from fatty alcohols and acrylic acid
647:
can be prepared in good yield in a one-step process by
1468:
R. Auras; L.-T. Lim; S. E. M. Selke; H. Tsuji (2010).
45:. They can be saturated or unsaturated. Some contain
1223:
Detlef Schröder, Norman
Goldberg, Waltraud Zummack,
428:
wood, and they contribute to the flavour profile of
404:oil. Of the naturally occurring bicyclic lactones,
977:International Union of Pure and Applied Chemistry
1575:. Boca Raton: Taylor & Francis. p. 74.
1227:, John C. Poutsma and Robert R. Squires (1997),
49:replacing one or more carbon atoms of the ring.
1345:with ring-opening and finally reduction of the
1389:Ullmann's Encyclopedia of Industrial Chemistry
1369:, Coll. Vol. 7, p.164 (1990); Vol. 64, p.175 (
1258:Ullmann's Encyclopedia of Industrial Chemistry
1009:. Department of Chemistry & Biochemistry,
994:(8th ed.), McGraw-Hill, pp. 798–799
990:Francis A. Carey; Robert M. Giuliano (2011),
285:The other suffix used to denote a lactone is
8:
1530:
1528:
1526:
1524:
1007:"Illustrated Glossary of Organic Chemistry"
67:in alphabetical order indicate ring size.
37:. They are derived from the corresponding
1672:
1662:
1444:
400:is responsible for the musklike odor of
69:
1572:Fermentation effects on food properties
961:
726:Lactones can be reduced to diols using
907:(Musk T), a widely used synthetic musk
898:can be isolated from the heartwood of
332:derives from the ring compound called
1261:(7th ed.), Wiley, pp. 74‒78
1250:
1248:
1246:
1244:
289:, used in substance class names like
7:
1357:and intramolecular lactone formation
862:(Hexahydroxydiphenic acid dilactone)
356:, a lactone with a 3-membered ring.
562:the key lactone-forming step is an
1313:The complete reaction sequence is
972:Compendium of Chemical Terminology
25:
1095:[Memoir on lactic acid].
1064:[Memoir on lactic acid].
610:Corey-Nicolaou macrolactonization
408:are responsible for the odors of
1606:Sensory-directed flavor analysis
1167:Annalen der Chemie und Pharmacie
1110:Pelouze, J. (January 15, 1845).
1097:Annales de Chimie et de Physique
766:
239:
204:
173:
150:
1060:Pelouze, J. (9 December 1844).
686:Heating a lactone with a base (
558:In one industrial synthesis of
1297:; 11(3) pp 378–388; (Article)
1093:"Mémoire sur l'acide lactique"
1062:"Mémoire sur l'acide lactique"
1029:The Royal Society of Chemistry
1:
1237:10.1016/S0168-1176(97)00150-X
1199:Journal of Organic Chemistry
866:Flavogallonic acid dilactone
827:of the corresponding linear
1112:"Researches on lactic acid"
1037:10.1039/9781849733069-00648
668:manganese-mediated coupling
359:In 1880 the German chemist
1724:
1603:Marsili, Ray, ed. (2007).
1535:Berger, R.G., ed. (2007).
1429:10.1038/s41598-020-74223-5
1128:Menten, Pierre de (2013).
1099:. 3rd series (in French).
555:H) spontaneously cyclize.
424:. Lactones are present in
979:, 2014-02-24, p. 817
813:15-pentadec-11/12-enolide
728:lithium aluminium hydride
682:Hydrolysis and aminolysis
614:Baeyer–Villiger oxidation
606:Shiina macrolactonization
600:Specific methods include
1179:10.1002/jlac.18802000102
1161:Fittig, Rudolph (1880).
886:Tergallic acid dilactone
833:12-oxa-16-hexadecanolide
618:nucleophilic abstraction
602:Yamaguchi esterification
896:Valoneic acid dilactone
346:Théophile-Jules Pelouze
52:Lactones are formed by
39:hydroxycarboxylic acids
27:Cyclic carboxylic ester
1278:Craft Beer and Brewing
1108:English translation:
807:Macrocyclic lactones (
780:Flavors and fragrances
661:di-tert-butyl peroxide
628:
591:electrophilic addition
574:
536:
386:
361:Wilhelm Rudolph Fittig
278:lactones are known as
121:
1392:(7th ed.), Wiley
1031:. 2014. p. 822.
1005:Steven A. Hardinger.
626:
572:
535:Oxandrolone synthesis
534:
374:
318:preferred IUPAC names
120:
1664:10.3390/life10010006
1541:. Berlin: Springer.
1323:elimination reaction
1116:The Chemical Gazette
1091:Pelouze, J. (1845).
956:References and notes
890:Rhynchosia volubilis
871:Rhynchosia volubilis
809:cyclopentadecanolide
704:equilibrium constant
398:Cyclopentadecanolide
350:dehydration reaction
1655:2020Life...10....6C
1421:2020NatSR..1017560C
1211:10.1021/jo00003a047
1105: ; see p. 262.
905:Ethylene brassylate
696:reversible reaction
354:alpha-propiolactone
269:= 6, etc.), with a
95:Structure, comment
1409:Scientific Reports
1355:sodium borohydride
1124: ; see p. 31.
629:
575:
566:– esterification.
537:
521:cardiac glycosides
387:
379:-glucono-δ-lactone
253:= 2 carbon atoms,
122:
1708:Functional groups
1507:10.1021/cr040002s
1479:978-0-470-29366-9
1367:Organic Syntheses
1343:lead tetraacetate
1335:organic oxidation
1321:(not displayed),
1303:10.1021/op060231b
1272:Oliver, Garrett.
1046:978-0-85404-182-4
992:Organic Chemistry
945:Halolactonization
900:Shorea laeviforia
876:Shorea laeviforia
585:is attacked by a
579:halolactonization
573:iodolactonization
473:neurotransmitters
430:barrel-aged beers
391:mass spectrometry
378:
247:
246:
35:carboxylic esters
16:(Redirected from
1715:
1687:
1686:
1676:
1666:
1634:
1628:
1627:
1625:
1623:
1600:
1594:
1593:
1591:
1589:
1566:
1560:
1559:
1557:
1555:
1532:
1519:
1518:
1490:
1484:
1483:
1465:
1459:
1458:
1448:
1400:
1394:
1393:
1383:
1377:
1364:
1358:
1339:osmium tetroxide
1327:lithium chloride
1311:
1305:
1288:
1282:
1281:
1269:
1263:
1262:
1252:
1239:
1221:
1215:
1214:
1205:(3): 1176–1185.
1194:
1188:
1182:
1158:
1152:
1149:
1123:
1104:
1073:
1057:
1051:
1050:
1021:
1015:
1014:
1002:
996:
995:
987:
981:
980:
966:
888:can be found in
868:can be found in
844:Polycaprolactone
825:depolymerization
770:
688:sodium hydroxide
649:radical addition
564:organic reaction
501:anticancer drugs
376:
243:
208:
177:
170:γ-Butyrolactone
154:
84:
75:(number of atoms
70:
21:
1723:
1722:
1718:
1717:
1716:
1714:
1713:
1712:
1693:
1692:
1691:
1690:
1636:
1635:
1631:
1621:
1619:
1617:
1602:
1601:
1597:
1587:
1585:
1583:
1568:
1567:
1563:
1553:
1551:
1549:
1534:
1533:
1522:
1501:(12): 6147–76.
1492:
1491:
1487:
1480:
1467:
1466:
1462:
1402:
1401:
1397:
1385:
1384:
1380:
1365:
1361:
1312:
1308:
1289:
1285:
1271:
1270:
1266:
1254:
1253:
1242:
1222:
1218:
1196:
1195:
1191:
1160:
1159:
1155:
1146:
1127:
1109:
1090:
1074:From p. 1223:
1059:
1058:
1054:
1047:
1023:
1022:
1018:
1004:
1003:
999:
989:
988:
984:
968:
967:
963:
958:
914:
856:
841:
802:γ-undecalactone
782:
777:
760:polylactic acid
758:polymerizes to
752:
745:
741:
737:
733:
724:
702:. However, the
684:
676:
663:as a catalyst.
645:γ-undecalactone
631:The γ-lactones
595:carboxylic acid
554:
550:
546:
529:
517:resorcylic acid
461:mevalonolactone
369:
343:
326:
236:
201:
197:δ-Valerolactone
147:
141:β-Propiolactone
92:Parent lactone
82:Systematic name
80:
76:
74:
62:
28:
23:
22:
15:
12:
11:
5:
1721:
1719:
1711:
1710:
1705:
1695:
1694:
1689:
1688:
1629:
1615:
1595:
1581:
1561:
1547:
1520:
1485:
1478:
1460:
1395:
1378:
1359:
1306:
1283:
1264:
1240:
1225:Helmut Schwarz
1216:
1189:
1153:
1151:
1150:
1144:
1125:
1106:
1066:Comptes rendus
1052:
1045:
1016:
997:
982:
960:
959:
957:
954:
953:
952:
947:
942:
933:
924:
913:
910:
909:
908:
902:
893:
883:
878:
863:
855:
852:
848:origin of life
840:
837:
781:
778:
776:
773:
772:
771:
751:
750:Polymerization
748:
743:
739:
735:
731:
723:
720:
683:
680:
675:
672:
653:fatty alcohols
552:
548:
544:
541:polymerization
528:
525:
513:phytoestrogens
497:amphotericin B
457:spironolactone
449:gluconolactone
393:experiments.
368:
365:
341:
325:
322:
316:To obtain the
245:
244:
237:
235:
234:
231:
228:
227:ε-Caprolactone
224:
222:
219:
214:
210:
209:
202:
200:
199:
193:
191:
188:
183:
179:
178:
171:
168:
165:
160:
156:
155:
148:
146:
145:
142:
138:
136:
133:
128:
124:
123:
114:
109:
106:
101:
97:
96:
93:
90:
85:
78:
65:Greek prefixes
61:
58:
43:esterification
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
1720:
1709:
1706:
1704:
1701:
1700:
1698:
1684:
1680:
1675:
1670:
1665:
1660:
1656:
1652:
1648:
1644:
1640:
1633:
1630:
1618:
1616:9781420017045
1612:
1608:
1607:
1599:
1596:
1584:
1582:9781439853351
1578:
1574:
1573:
1565:
1562:
1550:
1548:9783540493396
1544:
1540:
1539:
1531:
1529:
1527:
1525:
1521:
1516:
1512:
1508:
1504:
1500:
1496:
1489:
1486:
1481:
1475:
1471:
1464:
1461:
1456:
1452:
1447:
1442:
1438:
1434:
1430:
1426:
1422:
1418:
1414:
1410:
1406:
1399:
1396:
1391:
1390:
1382:
1379:
1376:
1372:
1368:
1363:
1360:
1356:
1352:
1348:
1344:
1340:
1336:
1332:
1328:
1324:
1320:
1316:
1310:
1307:
1304:
1300:
1296:
1292:
1287:
1284:
1279:
1275:
1268:
1265:
1260:
1259:
1251:
1249:
1247:
1245:
1241:
1238:
1234:
1230:
1226:
1220:
1217:
1212:
1208:
1204:
1200:
1193:
1190:
1186:
1183:From p. 62:
1180:
1176:
1172:
1169:(in German).
1168:
1164:
1157:
1154:
1147:
1145:9782804181758
1141:
1137:
1133:
1132:
1126:
1121:
1117:
1113:
1107:
1102:
1098:
1094:
1088:
1087:
1085:
1081:
1077:
1071:
1068:(in French).
1067:
1063:
1056:
1053:
1048:
1042:
1038:
1034:
1030:
1027:. Cambridge:
1026:
1020:
1017:
1012:
1008:
1001:
998:
993:
986:
983:
978:
974:
973:
965:
962:
955:
951:
948:
946:
943:
941:
938:, a cyclic di
937:
934:
932:
928:
925:
923:
919:
916:
915:
911:
906:
903:
901:
897:
894:
891:
887:
884:
882:
879:
877:
874:seeds and in
873:
872:
867:
864:
861:
858:
857:
853:
851:
849:
845:
838:
836:
834:
830:
826:
822:
818:
814:
810:
805:
803:
799:
798:γ-nonalactone
795:
794:γ-octalactone
791:
790:δ-decalactone
787:
786:γ-decalactone
779:
774:
769:
765:
764:
763:
761:
757:
749:
747:
729:
721:
719:
716:
713:
709:
705:
701:
697:
693:
689:
681:
679:
673:
671:
669:
664:
662:
658:
654:
650:
646:
642:
641:γ-decalactone
638:
637:γ-nonalactone
634:
633:γ-octalactone
625:
621:
619:
615:
611:
607:
603:
598:
596:
592:
588:
584:
580:
571:
567:
565:
561:
556:
542:
533:
526:
524:
522:
518:
514:
510:
506:
502:
498:
494:
490:
486:
482:
478:
477:butyrolactone
474:
470:
466:
462:
458:
454:
450:
446:
445:nepetalactone
442:
438:
437:ascorbic acid
433:
431:
427:
423:
419:
415:
411:
407:
403:
402:angelica root
399:
394:
392:
384:
380:
373:
366:
364:
362:
357:
355:
351:
347:
339:
335:
331:
323:
321:
319:
314:
312:
311:
310:bufadienolide
306:
305:
300:
299:
294:
293:
288:
283:
281:
280:macrolactones
277:
272:
268:
264:
260:
256:
252:
242:
238:
232:
229:
226:
225:
223:
221:Oxepan-2-one
220:
218:
215:
212:
211:
207:
203:
198:
195:
194:
192:
189:
187:
184:
181:
180:
176:
172:
169:
167:Oxolan-2-one
166:
164:
161:
158:
157:
153:
149:
144:Propiolactone
143:
140:
139:
137:
135:Oxetan-2-one
134:
132:
129:
126:
125:
119:
115:
113:
110:
108:Oxiran-2-one
107:
105:
102:
99:
98:
94:
91:
89:
86:
83:
79:
77:in the ring)
72:
71:
68:
66:
59:
57:
55:
54:lactonization
50:
48:
44:
40:
36:
32:
19:
1646:
1642:
1632:
1620:. Retrieved
1605:
1598:
1586:. Retrieved
1571:
1564:
1552:. Retrieved
1537:
1498:
1494:
1488:
1469:
1463:
1415:(1): 17560.
1412:
1408:
1398:
1387:
1381:
1375:Article link
1370:
1362:
1309:
1294:
1290:
1286:
1277:
1267:
1256:
1228:
1219:
1202:
1198:
1192:
1184:
1170:
1166:
1156:
1135:
1130:
1122:(54): 29–35.
1119:
1115:
1100:
1096:
1089:Reprinted:
1083:
1079:
1075:
1072:: 1219–1227.
1069:
1065:
1055:
1024:
1019:
1000:
991:
985:
970:
969:"lactones",
964:
899:
889:
875:
869:
860:Ellagic acid
842:
806:
783:
753:
725:
717:
685:
677:
665:
657:acrylic acid
630:
599:
576:
557:
538:
493:erythromycin
434:
395:
388:
358:
329:
327:
315:
308:
302:
296:
290:
286:
284:
279:
270:
266:
262:
258:
254:
250:
248:
230:Caprolactone
112:Acetolactone
63:
60:Nomenclature
53:
51:
30:
29:
1315:bromination
929:, a cyclic
920:, a cyclic
700:equilibrium
651:of primary
560:oxandrolone
509:epothilones
485:antibiotics
481:avermectins
338:lactic acid
304:cardenolide
276:Macrocyclic
190:Oxan-2-one
47:heteroatoms
33:are cyclic
1697:Categories
1319:haloketone
1103:: 257–268.
854:Dilactones
829:polyesters
708:enthalpies
698:, with an
519:lactones,
505:vernolepin
489:macrolides
416:oils, and
406:phthalides
367:Occurrence
292:butenolide
233:Hexanolide
88:IUPAC name
1495:Chem. Rev
1472:. Wiley.
1437:2045-2322
975:, 2.3.3,
950:Phthalein
722:Reduction
692:hydrolyse
674:Reactions
527:Synthesis
469:lactonase
328:The name
324:Etymology
298:macrolide
217:ε-lactone
186:δ-lactone
163:γ-lactone
131:β-lactone
104:α-lactone
73:Ring size
1703:Lactones
1683:31963928
1649:(1): 6.
1515:15584698
1455:33067516
1347:aldehyde
1173:: 1–96.
912:See also
839:Plastics
821:civetone
734:(OH)-(CH
659:, using
453:hormones
422:woodruff
418:coumarin
271:-lactone
31:Lactones
18:Lactones
1674:7175156
1651:Bibcode
1446:7567815
1417:Bibcode
1351:alcohol
1349:to the
1084:lactone
1078:lactone
936:Lactide
881:Lactide
817:muscone
756:lactide
712:entropy
690:) will
587:halogen
465:enzymes
334:lactide
330:lactone
1681:
1671:
1622:2 July
1613:
1588:2 July
1579:
1554:2 July
1545:
1513:
1476:
1453:
1443:
1435:
1329:to an
1142:
1043:
927:Lactim
918:Lactam
746:(OH).
583:alkene
441:kavain
414:lovage
410:celery
287:-olide
263:valero
259:butyro
255:propio
1353:with
1331:enone
1325:with
1317:to a
1134:[
940:ester
931:imide
922:amide
892:seeds
581:, an
491:like
267:capro
265:= 5,
261:= 4,
257:= 3,
251:aceto
1679:PMID
1643:Life
1624:2015
1611:ISBN
1590:2015
1577:ISBN
1556:2015
1543:ISBN
1511:PMID
1474:ISBN
1451:PMID
1433:ISSN
1371:1986
1341:and
1295:2007
1140:ISBN
1041:ISBN
1011:UCLA
775:Uses
616:and
589:via
420:for
412:and
383:E575
340:) CH
1669:PMC
1659:doi
1503:doi
1499:104
1441:PMC
1425:doi
1337:by
1299:doi
1233:doi
1207:doi
1175:doi
1171:200
1033:doi
835:).
742:-CH
655:to
577:In
551:-CO
523:).
511:),
499:),
483:),
471:),
463:),
426:oak
307:or
41:by
1699::
1677:.
1667:.
1657:.
1647:10
1645:.
1641:.
1523:^
1509:.
1497:.
1449:.
1439:.
1431:.
1423:.
1413:10
1411:.
1407:.
1373:)
1333:,
1276:.
1243:^
1203:56
1201:.
1118:.
1114:.
1101:13
1070:19
1039:.
850:.
819:,
811:,
804:.
670:.
643:,
639:,
635:,
620:.
612:,
608:,
604:,
597:.
507:,
495:;
479:,
459:,
451:,
447:,
443:,
439:,
432:.
313:.
301:,
295:,
213:7
182:6
159:5
127:4
100:3
1685:.
1661::
1653::
1626:.
1592:.
1558:.
1517:.
1505::
1482:.
1457:.
1427::
1419::
1301::
1280:.
1235::
1213:.
1209::
1181:.
1177::
1148:.
1120:3
1049:.
1035::
1013:.
744:2
740:2
738:)
736:2
732:2
553:2
549:2
547:)
545:2
515:(
503:(
487:(
475:(
467:(
455:(
385:)
381:(
377:D
342:3
282:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.