2297:
1636:
container then solid condensate with irregular impurity distribution is forming. Then most pure part of the condensate may be extracted as product. The process may be iterated many times by moving (without turnover) the received condensate to the bottom part of the container on the place of refined matter. The irregular impurity distribution in the condensate (that is efficiency of purification) increases with the number of iterations. Zone distillation is the distillation analog of zone recrystallization. Impurity distribution in the condensate is described by known equations of zone recrystallization – with the replacement of the distribution co-efficient k of crystallization - for the separation factor α of distillation.
1081:
thus, are concentrated in the vapor, but heavier volatile components also have a (smaller) partial pressure and necessarily vaporize also, albeit at a lower concentration in the vapor. Indeed, batch distillation and fractionation succeed by varying the composition of the mixture. In batch distillation, the batch vaporizes, which changes its composition; in fractionation, liquid higher in the fractionation column contains more lights and boils at lower temperatures. Therefore, starting from a given mixture, it appears to have a boiling range instead of a boiling point, although this is because its composition changes: each intermediate mixture has its own, singular boiling point.
786:
2149:(HETP) will be greater than expected. The problem is not the packing itself but the mal-distribution of the fluids entering the packed bed. Liquid mal-distribution is more frequently the problem than vapor. The design of the liquid distributors used to introduce the feed and reflux to a packed bed is critical to making the packing perform to it maximum efficiency. Methods of evaluating the effectiveness of a liquid distributor to evenly distribute the liquid entering a packed bed can be found in references. Considerable work has been done on this topic by Fractionation Research, Inc. (commonly known as FRI).
2226:
2246:
1894:, easily form azeotropes. Commonly, these azeotropes are referred to as a low boiling azeotrope because the boiling point of the azeotrope is lower than the boiling point of either pure component. The temperature and composition of the azeotrope is easily predicted from the vapor pressure of the pure components, without use of Raoult's law. The azeotrope is easily broken in a distillation set-up by using a liquid–liquid separator (a decanter) to separate the two liquid layers that are condensed overhead. Only one of the two liquid layers is refluxed to the distillation set-up.
1168:
packing. Reflux is a flow from the condenser back to the column, which generates a recycle that allows a better separation with a given number of trays. Equilibrium stages are ideal steps where compositions achieve vapor–liquid equilibrium, repeating the separation process and allowing better separation given a reflux ratio. A column with a high reflux ratio may have fewer stages, but it refluxes a large amount of liquid, giving a wide column with a large holdup. Conversely, a column with a low reflux ratio must have a large number of stages, thus requiring a taller column.
2296:
2021:
1441:
1132:
A and B. The ratio between A and B in the vapor will be different from the ratio in the liquid. The ratio in the liquid will be determined by how the original mixture was prepared, while the ratio in the vapor will be enriched in the more volatile compound, A (due to Raoult's Law, see above). The vapor goes through the condenser and is removed from the system. This, in turn, means that the ratio of compounds in the remaining liquid is now different from the initial ratio (i.e., more enriched in B than in the starting liquid).
4051:
1554:
1422:
1584:
the need for a condenser separating the two chambers. This technique is often used for compounds which are unstable at high temperatures or to purify small amounts of compound. The advantage is that the heating temperature can be considerably lower (at reduced pressure) than the boiling point of the liquid at standard pressure, and the distillate only has to travel a short distance before condensing. A short path ensures that little compound is lost on the sides of the apparatus. The
828:
1009:, the composition of the source material, the vapors of the distilling compounds, and the distillate change during the distillation. In batch distillation, a still is charged (supplied) with a batch of feed mixture, which is then separated into its component fractions, which are collected sequentially from most volatile to less volatile, with the bottoms – remaining least or non-volatile fraction – removed at the end. The still can then be recharged and the process repeated.
1269:
66:
446:
2262:
2277:
4394:
812:
1966:
1831:
material, which had been wetted with water or a clear liquid with each step dripping down through the wetted cloth through capillary action in succeeding steps, creating a "purification" of the liquid, leaving solid materials behind in the upper bowls and purifying the succeeding product through capillary action through the moistened cloth. This was called "distillatio" by filtration by those using the method.
820:
1600:. A simple vacuum distillation system as exemplified above can be used, whereby the vacuum is replaced with an inert gas after the distillation is complete. However, this is a less satisfactory system if one desires to collect fractions under a reduced pressure. To do this a "cow" or "pig" adaptor can be added to the end of the condenser, or for better results or for very air sensitive compounds a
1124:
801:
1539:
transport is governed by molecular dynamics rather than fluid dynamics. Thus, a short path between the hot surface and the cold surface is necessary, typically by suspending a hot plate covered with a film of feed next to a cold plate with a line of sight in between. Molecular distillation is used industrially for purification of oils.
1339:. As it rises, it cools, condensing on the condenser walls and the surfaces of the packing material. Here, the condensate continues to be heated by the rising hot vapors; it vaporizes once more. However, the composition of the fresh vapors is determined once again by Raoult's law. Each vaporization-condensation cycle (called a
1868:. Some techniques achieve this by "jumping" over the azeotropic composition (by adding another component to create a new azeotrope, or by varying the pressure). Others work by chemically or physically removing or sequestering the impurity. For example, to purify ethanol beyond 95%, a drying agent (or
1131:
Heating an ideal mixture of two volatile substances, A and B, with A having the higher volatility, or lower boiling point, in a batch distillation setup (such as in an apparatus depicted in the opening figure) until the mixture is boiling results in a vapor above the liquid that contains a mixture of
1080:
An implication of one boiling point is that lighter components never cleanly "boil first". At boiling point, all volatile components boil, but for a component, its percentage in the vapor is the same as its percentage of the total vapor pressure. Lighter components have a higher partial pressure and,
2144:
required to make a given separation is calculated using a specific vapor to liquid ratio. If the liquid and vapor are not evenly distributed across the superficial tower area as it enters the packed bed, the liquid to vapor ratio will not be correct in the packed bed and the required separation will
1999:
To control and optimize such industrial distillation, a standardized laboratory method, ASTM D86, is established. This test method extends to the atmospheric distillation of petroleum products using a laboratory batch distillation unit to quantitatively determine the boiling range characteristics of
1830:
Distillation by filtration: In early alchemy and chemistry, otherwise known as natural philosophy, a form of "distillation" by capillary filtration was known as a form of distillation at the time. In this, a series of cups or bowls were set upon a stepped support with a "wick" of cotton or felt-like
1753:
a short path distillation apparatus is typically used (generally in combination with a (high) vacuum) to distill high boiling (> 300 °C) compounds. The apparatus consists of an oven in which the compound to be distilled is placed, a receiving portion which is outside of the oven, and a means
1499:
Some compounds have very high boiling points. To boil such compounds, it is often better to lower the pressure at which such compounds are boiled instead of increasing the temperature. Once the pressure is lowered to the vapor pressure of the compound (at the given temperature), boiling and the rest
1382:
and allows a high rate of heat transfer without heating at a very high temperature. This process involves bubbling steam through a heated mixture of the raw material. By Raoult's law, some of the target compound will vaporize (in accordance with its partial pressure). The vapor mixture is cooled and
910:
Early forms of distillation involved batch processes using one vaporization and one condensation. Purity was improved by further distillation of the condensate. Greater volumes were processed by simply repeating the distillation. Chemists reportedly carried out as many as 500 to 600 distillations in
373:
According to
British chemist T. Fairley, neither the Greeks nor the Romans had any term for the modern concept of distillation. Words like "distill" would have referred to something else, in most cases a part of some process unrelated to what now is known as distillation. In the words of Fairley and
2111:
are: presence of solids in feed, high liquid rates, large column diameters, complex columns, columns with wide feed composition variation, columns with a chemical reaction, absorption columns, columns limited by foundation weight tolerance, low liquid rate, large turn-down ratio and those processes
1635:
Zone distillation is a distillation process in a long container with partial melting of refined matter in moving liquid zone and condensation of vapor in the solid phase at condensate pulling in cold area. The process is worked in theory. When zone heater is moving from the top to the bottom of the
1583:
Short path distillation is a distillation technique that involves the distillate travelling a short distance, often only a few centimeters, and is normally done at reduced pressure. A classic example would be a distillation involving the distillate travelling from one glass bulb to another, without
1538:
of molecules is comparable to the size of the equipment. The gaseous phase no longer exerts significant pressure on the substance to be evaporated, and consequently, rate of evaporation no longer depends on pressure. That is, because the continuum assumptions of fluid dynamics no longer apply, mass
1180:
on top of the distillation flask. The column improves separation by providing a larger surface area for the vapor and condensate to come into contact. This helps it remain at equilibrium for as long as possible. The column can even consist of small subsystems ('trays' or 'dishes') which all contain
1135:
The result is that the ratio in the liquid mixture is changing, becoming richer in component B. This causes the boiling point of the mixture to rise, which results in a rise in the temperature in the vapor, which results in a changing ratio of A : B in the gas phase (as distillation continues,
866:
the distillate and let it drip downward for collection. Later, copper alembics were invented. Riveted joints were often kept tight by using various mixtures, for instance a dough made of rye flour. These alembics often featured a cooling system around the beak, using cold water, for instance, which
2213:
are prepared by distilling these dilute solutions of ethanol. Components other than ethanol, including water, esters, and other alcohols, are collected in the condensate, which account for the flavor of the beverage. Some of these beverages are then stored in barrels or other containers to acquire
1909:
The boiling points of components in an azeotrope overlap to form a band. By exposing an azeotrope to a vacuum or positive pressure, it is possible to bias the boiling point of one component away from the other by exploiting the differing vapor pressure curves of each; the curves may overlap at the
1717:
Membrane distillation is a type of distillation in which vapors of a mixture to be separated are passed through a membrane, which selectively permeates one component of mixture. Vapor pressure difference is the driving force. It has potential applications in seawater desalination and in removal of
1284:
As a result, simple distillation is effective only when the liquid boiling points differ greatly (rule of thumb is 25 °C) or when separating liquids from non-volatile solids or oils. For these cases, the vapor pressures of the components are usually different enough that the distillate may be
1243:
A completely sealed distillation apparatus could experience extreme and rapidly varying internal pressure, which could cause it to burst open at the joints. Therefore, some path is usually left open (for instance, at the receiving flask) to allow the internal pressure to equalize with atmospheric
1154:
Continuous distillation is an ongoing distillation in which a liquid mixture is continuously (without interruption) fed into the process and separated fractions are removed continuously as output streams occur over time during the operation. Continuous distillation produces a minimum of two output
1139:
If the difference in vapour pressure between the two components A and B is large – generally expressed as the difference in boiling points – the mixture in the beginning of the distillation is highly enriched in component A, and when component A has distilled off, the boiling liquid is enriched in
1071:
Dalton's law states that the total pressure is the sum of the partial pressures of each individual component in the mixture. When a multi-component liquid is heated, the vapor pressure of each component will rise, thus causing the total vapor pressure to rise. When the total vapor pressure reaches
1856:
which behaves as if it were a pure compound (i.e., boils at a single temperature instead of a range). At an azeotrope, the solution contains the given component in the same proportion as the vapor, so that evaporation does not change the purity, and distillation does not result in separation. For
1043:
It is a misconception that in a liquid mixture at a given pressure, each component boils at the boiling point corresponding to the given pressure, allowing the vapors of each component to collect separately and purely. However, this does not occur, even in an idealized system. Idealized models of
1167:
for an arbitrary amount of time. For any source material of specific composition, the main variables that affect the purity of products in continuous distillation are the reflux ratio and the number of theoretical equilibrium stages, in practice determined by the number of trays or the height of
221:
molecules into smaller hydrocarbon molecules. Moreover, a partial distillation results in partial separations of the mixture's components, which process yields nearly-pure components; partial distillation also realizes partial separations of the mixture to increase the concentrations of selected
2135:
takes place. Unlike conventional tray distillation in which every tray represents a separate point of vapor–liquid equilibrium, the vapor–liquid equilibrium curve in a packed column is continuous. However, when modeling packed columns, it is useful to compute a number of "theoretical stages" to
2032:
to achieve a more complete separation of products. Reflux refers to the portion of the condensed overhead liquid product from a distillation or fractionation tower that is returned to the upper part of the tower as shown in the schematic diagram of a typical, large-scale industrial distillation
2106:
In modern industrial uses, a packing material is used in the column instead of trays when low pressure drops across the column are required. Other factors that favor packing are: vacuum systems, smaller diameter columns, corrosive systems, systems prone to foaming, systems requiring low liquid
1916:
Under negative pressure, power for a vacuum source is needed and the reduced boiling points of the distillates requires that the condenser be run cooler to prevent distillate vapors being lost to the vacuum source. Increased cooling demands will often require additional energy and possibly new
1658:
involves using the reaction vessel as the still. In this process, the product is usually significantly lower boiling than its reactants. As the product is formed from the reactants, it is vaporized and removed from the reaction mixture. This technique is an example of a continuous vs. a batch
1659:
process; advantages include less downtime to charge the reaction vessel with starting material, and less workup. Distillation "over a reactant" could be classified as a reactive distillation. It is typically used to remove volatile impurity from the distillation feed. For example, a little
2003:
Industrial distillation is typically performed in large, vertical cylindrical columns known as distillation towers or distillation columns with diameters ranging from about 0.65 to 16 metres (2 ft 2 in to 52 ft 6 in) and heights ranging from about 6 to 90 metres (20 to
1920:
Alternatively, if positive pressures are required, standard glassware can not be used, energy must be used for pressurization and there is a higher chance of side reactions occurring in the distillation, such as decomposition, due to the higher temperatures required to effect boiling.
1331:
must be taken into consideration. Therefore, fractional distillation must be used to separate the components by repeated vaporization-condensation cycles within a packed fractionating column. This separation, by successive distillations, is also referred to as rectification.
3458:
1827:. Unlike distillation, freeze distillation concentrates poisonous congeners rather than removing them; As a result, many countries prohibit such applejack as a health measure. Also, distillation by evaporation can separate these since they have different boiling points.
378:
The Latin "distillo," from de-stillo, from stilla, a drop, referred to the dropping of a liquid by human or artificial means, and was applied to any process where a liquid was separated in drops. To distil in the modern sense could only be expressed in a roundabout
3424:
1308:
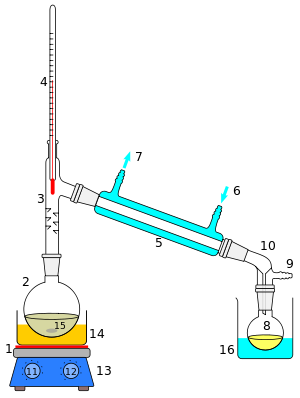
5, is cooled by water (blue) that circulates through ports 6 and 7. The condensed liquid drips into the receiving flask 8, sitting in a cooling bath (blue, 16). The adapter 10 has a connection 9 that may be fitted to a vacuum pump. The components are connected by
1016:, the source materials, vapors, and distillate are kept at a constant composition by carefully replenishing the source material and removing fractions from both vapor and liquid in the system. This results in a more detailed control of the separation process.
1076:
occurs and liquid turns to gas throughout the bulk of the liquid. A mixture with a given composition has one boiling point at a given pressure when the components are mutually soluble. A mixture of constant composition does not have multiple boiling points.
1276:
In simple distillation, the vapor is immediately channeled into a condenser. Consequently, the distillate is not pure but rather its composition is identical to the composition of the vapors at the given temperature and pressure. That concentration follows
1979:
Large scale industrial distillation applications include both batch and continuous fractional, vacuum, azeotropic, extractive, and steam distillation. The most widely used industrial applications of continuous, steady-state fractional distillation are in
1159:
distillate fraction, which has boiled and been separately captured as a vapor and then condensed to a liquid. There is always a bottoms (or residue) fraction, which is the least volatile residue that has not been separately captured as a condensed vapor.
2016:
or boiling ranges. The "lightest" products (those with the lowest boiling point) exit from the top of the columns and the "heaviest" products (those with the highest boiling point) exit from the bottom of the column and are often called the bottoms.
1627:) and can then be stoppered and removed. A fresh collection vessel can then be added to the system, evacuated and linked back into the distillation system via the taps to collect a second fraction, and so on, until all fractions have been collected.
1910:
azeotropic point, but are unlikely to remain identical further along the pressure axis to either side of the azeotropic point. When the bias is great enough, the two boiling points no longer overlap and so the azeotropic band disappears.
2098:
models are used both for design and operation. Moreover, the efficiencies of the vapor–liquid contact devices (referred to as "plates" or "trays") used in distillation towers are typically lower than that of a theoretical 100% efficient
782:, the first major English compendium on the practice, but it has been claimed that much of it derives from Braunschweig's work. This includes diagrams with people in them showing the industrial rather than bench scale of the operation.
2165:. The number of effects is inversely proportional to the kW·h/m of water recovered figure and refers to the volume of water recovered per unit of energy compared with single-effect distillation. One effect is roughly 636 kW·h/m:
2136:
denote the separation efficiency of the packed column with respect to more traditional trays. Differently shaped packings have different surface areas and void space between packings. Both these factors affect packing performance.
2033:
tower. Inside the tower, the downflowing reflux liquid provides cooling and condensation of the upflowing vapors thereby increasing the efficiency of the distillation tower. The more reflux that is provided for a given number of
871:. Today, the retorts and pot stills have been largely supplanted by more efficient distillation methods in most industrial processes. However, the pot still is still widely used for the elaboration of some fine alcohols, such as
2037:, the better the tower's separation of lower boiling materials from higher boiling materials. Alternatively, the more reflux that is provided for a given desired separation, the fewer the number of theoretical plates required.
1004:
The main difference between laboratory scale distillation and industrial distillation are that laboratory scale distillation is often performed on a batch basis, whereas industrial distillation often occurs continuously. In
1945:
This improves the selectivity of the distillation and allows a chemist to optimize distillation by avoiding extremes of pressure and temperature that waste energy. This is particularly important in commercial applications.
2307:. The small holdup volume prevents losses. A "pig" is used to channel the various distillates into three receiving flasks. If necessary the distillation can be carried out under vacuum using the vacuum adapter at the pig.
1644:
Non-condensable gas can be expelled from the apparatus by the vapor of relatively volatile co-solvent, which spontaneously evaporates during initial pumping, and this can be achieved with regular oil or diaphragm pump.
2225:
2302:
Distillation using semi-microscale apparatus. The jointless design eliminates the need to fit pieces together. The pear-shaped flask allows the last drop of residue to be removed, compared with a similarly sized
2182:
There are many other types of multi-effect distillation processes, including one referred to as simply multi-effect distillation (MED), in which multiple chambers, with intervening heat exchangers, are employed.
1184:
There are differences between laboratory-scale and industrial-scale fractionating columns, but the principles are the same. Examples of laboratory-scale fractionating columns (in increasing efficiency) include:
3015:
The earliest possible period seems to be the
Eastern Han dynasty ... the most likely period for the beginning of true distillation of spirits for drinking in China is during the Jin and Southern Song dynasties
1673:
is the process by which the reactants are catalyzed while being distilled to continuously separate the products from the reactants. This method is used to assist equilibrium reactions in reaching completion.
1059:
Raoult's law states that the vapor pressure of a solution is dependent on 1) the vapor pressure of each chemical component in the solution and 2) the fraction of solution each component makes up, a.k.a. the
2139:
Another factor in addition to the packing shape and surface area that affects the performance of random or structured packing is the liquid and vapor distribution entering the packed bed. The number of
1248:
may be used to keep the apparatus at a lower than atmospheric pressure. If the substances involved are air- or moisture-sensitive, the connection to the atmosphere can be made through one or more
427:
Letting seawater evaporate and condense into freshwater can not be called "distillation" for distillation involves boiling, but the experiment may have been an important step towards distillation.
2276:
2874:
2103:. Hence, a distillation tower needs more trays than the number of theoretical vapor–liquid equilibrium stages. A variety of models have been postulated to estimate tray efficiencies.
1092:. In other cases, severe deviations from Raoult's law and Dalton's law are observed, most famously in the mixture of ethanol and water. These compounds, when heated together, form an
273:, a partial distillation to reduce the vapor pressure of crude oil, which thus is safe to store and to transport, and thereby reduces the volume of atmospheric emissions of volatile
1345:) will yield a purer solution of the more volatile component. In reality, each cycle at a given temperature does not occur at exactly the same position in the fractionating column;
2245:
2261:
1864:
If the azeotrope is not considered sufficiently pure for use, there exist some techniques to break the azeotrope to give a more pure distillate. These techniques are known as
2508:
As already mentioned, the textual evidence for Sumero-Babylonian distillation is disclosed in a group of
Akkadian tablets describing perfumery operations, dated ca. 1200 B.C.
582:
3433:
767:), the first book solely dedicated to the subject of distillation, followed in 1512 by a much expanded version. Right after that, in 1518, the oldest distillery in Europe,
713:
started to appear in a number of Latin works, and by the end of the thirteenth century it had become a widely known substance among
Western European chemists. The works of
717:(1223–1296) describe a method for concentrating alcohol involving repeated distillation through a water-cooled still, by which an alcohol purity of 90% could be obtained.
1236:, and a receiver in which the concentrated or purified liquid, called the distillate, is collected. Several laboratory scale techniques for distillation exist (see also
2945:
2902:
629:. The Jabirian experiments with fractional distillation of animal and vegetable substances, and to a lesser degree also of mineral substances, is the main topic of the
1256:
that provide a movable liquid barrier. Finally, the entry of undesired air components can be prevented by pumping a low but steady flow of suitable inert gas, like
1115:
must be applied. When a binary mixture is vaporized and the other component, e.g., a salt, has zero partial pressure for practical purposes, the process is simpler.
2041:
must choose what combination of reflux rate and number of plates is both economically and physically feasible for the products purified in the distillation column.
4210:
3258:
3561:
1163:
Continuous distillation differs from batch distillation in the respect that concentrations should not change over time. Continuous distillation can be run at a
2086:
Design and operation of a distillation tower depends on the feed and desired products. Given a simple, binary component feed, analytical methods such as the
1500:
of the distillation process can commence. This technique is referred to as vacuum distillation and it is commonly found in the laboratory in the form of the
1378:, steam distillation is a method for distilling compounds which are heat-sensitive. The temperature of the steam is easier to control than the surface of a
918:, were developed. In 1822, Anthony Perrier developed one of the first continuous stills, and then, in 1826, Robert Stein improved that design to make his
997:. The latter two are distinctively different from the former two in that distillation is not used as a true purification method but more to transfer all
3521:
2461:
1619:. To do this, the sample is first isolated from the vacuum by means of the taps, the vacuum over the sample is then replaced with an inert gas (such as
384:
Distillation had a broader meaning in ancient and medieval times because nearly all purification and separation operations were subsumed under the term
3765:
3032:
Jâbir ibn Hayyân: Contribution à l'histoire des idées scientifiques dans l'Islam. I. Le corpus des écrits jâbiriens. II. Jâbir et la science grecque
1707:
or other throttling device. This process is one of the simplest unit operations, being equivalent to a distillation with only one equilibrium stage.
1107:
It is not possible to completely purify a mixture of components by distillation, as this would require each component in the mixture to have a zero
757:
2480:
1846:
Interactions between the components of the solution create properties unique to the solution, as most processes entail non-ideal mixtures, where
1781:
atmosphere and any volatile fractions, containing high-boiling liquids and products of pyrolysis, are collected. The destructive distillation of
1288:
A cutaway schematic of a simple distillation operation is shown at right. The starting liquid 15 in the boiling flask 2 is heated by a combined
3982:
1218:
Laboratory scale distillations are almost exclusively run as batch distillations. The device used in distillation, sometimes referred to as a
423:
I have proved by experiment that salt water evaporated forms fresh, and the vapour does not, when it condenses, condense into sea water again.
3898:
3747:
3722:
3531:
3497:
3237:
3008:
2754:
2699:
2649:
2601:
2570:
2390:
2318:
1901:
in water, also exist. As implied by the name, the boiling point of the azeotrope is greater than the boiling point of either pure component.
3830:
Kunesh, John G.; Lahm, Lawrence; Yanagi, Takashi (1987). "Commercial scale experiments that provide insight on packed tower distributors".
3351:
3166:
2079:
2846:
1938:
Pressure-swing distillation is essentially the same as the unidirectional distillation used to break azeotropic mixtures, but here both
1714:
is used for this purpose to remove water from synthesis products. The
Bleidner apparatus is another example with two refluxing solvents.
785:
4203:
4050:
2116:
1689:
is defined as distillation in the presence of a miscible, high boiling, relatively non-volatile component, the solvent, that forms no
352:
describing perfumery operations. The tablets provided textual evidence that an early, primitive form of distillation was known to the
3430:
3918:
3403:
3206:
3136:
3039:
2978:
1360:
or metal to force the rising vapors into close contact with the descending condensate, increasing the number of theoretical plates.
1615:, without the main body of the distillation being removed from either the vacuum or heat source, and thus can remain in a state of
1136:
there is an increasing proportion of B in the gas phase). This results in a slowly changing ratio of A : B in the distillate.
1710:
Codistillation is distillation which is performed on mixtures in which the two compounds are not miscible. In the laboratory, the
1068:, or solutions that have different components but whose molecular interactions are the same as or very similar to pure solutions.
3546:
4368:
3278:
254:
733:(12th–13th century) dynasties, according to archaeological evidence. A still was found in an archaeological site in Qinglong,
4578:
4378:
2942:
2899:
2169:
3473:"Making the Deserts Bloom: Harnessing nature to deliver us from drought, Distillations Podcast and transcript, Episode 239"
2205:-containing plant materials are allowed to ferment, producing a dilute solution of ethanol in the process. Spirits such as
1032:
of the liquid equals the pressure around the liquid, enabling bubbles to form without being crushed. A special case is the
4573:
4568:
4358:
4196:
3086:"Min al-kīmiyāʾ ad alchimiam. The Transmission of Alchemy from the Arab-Muslim World to the Latin West in the Middle Ages"
2859:
2175:
1804:
945:
as a discipline at the end of the 19th century, scientific rather than empirical methods could be applied. The developing
31:
4333:
4040:
1101:
1053:
3375:
2333:
1511:
at atmospheric pressure and which would therefore be decomposed by any attempt to boil them under atmospheric pressure.
4408:
4393:
730:
4593:
3558:
2178:– Commercial large-scale units can achieve around 72 effects with electrical energy input, according to manufacturers.
930:
of modern petrochemical units. The French engineer Armand
Savalle developed his steam regulator around 1846. In 1877,
39:
2871:
1588:
is a kind of short path distillation method which often contains multiple chambers to collect distillate fractions.
949:
industry in the early 20th century provided the impetus for the development of accurate design methods, such as the
4583:
4428:
4111:
3975:
3056:
Burnett, Charles (2001). "The
Coherence of the Arabic-Latin Translation Program in Toledo in the Twelfth Century".
1557:
Short path vacuum distillation apparatus with vertical condenser (cold finger), to minimize the distillation path;
1353:
1206:
531:
35:
4588:
4468:
4353:
1508:
961:. The first industrial plant in the United States to use distillation as a means of ocean desalination opened in
859:
768:
4238:
4025:
3472:
2087:
2020:
1100:
that can be used to estimate the behavior of a mixture of arbitrary components, the only way to obtain accurate
950:
4563:
4511:
4167:
4142:
1877:
1766:
487:
210:
1913:
This method can remove the need to add other chemicals to a distillation, but it has two potential drawbacks.
2458:
1703:
that occurs when a saturated liquid stream undergoes a reduction in pressure by passing through a throttling
4278:
4157:
4152:
4132:
4070:
3254:
2343:
2123:
This packing material can either be random or dumped packing (25–76 millimetres (1–3 in) wide) such as
1974:
1924:
A unidirectional distillation will rely on a pressure change in one direction, either positive or negative.
1841:
1686:
1608:
1548:
1440:
1357:
1322:
1149:
1112:
1097:
1013:
794:
750:
601:
570:
280:
270:
57:
17:
3456:, "Improvement in the Ammonia-Soda Manufacture", published 2 June 1876, issued 25 December 1877
903:
in various countries. Small pot stills are also sold for use in the domestic production of flower water or
4373:
4303:
4137:
3762:
1993:
1670:
1519:
1189:
1156:
998:
175:
of two or more chemically discrete substances; the separation process is realized by way of the selective
2623:(in Ancient Greek and English). Translated by Lee, H. D. P. Harvard University Press. pp. 2.3, 358b.
2521:
1750:
1585:
4558:
4493:
4488:
4293:
4253:
4162:
3968:
2736:
2477:
2353:
1655:
1237:
726:
543:
393:
2131:. Liquids tend to wet the surface of the packing and the vapors pass across this wetted surface, where
1949:
One example of the application of pressure-swing distillation is during the industrial purification of
1421:
3783:
Random
Packing, Vapor and Liquid Distribution: Liquid and gas distribution in commercial packed towers
1811:, and does not produce products equivalent to distillation. This process is used in the production of
1553:
4443:
4328:
4273:
4075:
3804:
3449:
3422:, "Apparatus for Brewing and Distilling", published 5 August 1830, issued 5 February 1831
3304:
3027:
2742:
A Short
History of the Art of Distillation: From the Beginnings up to the Death of Cellier Blumenthal
2637:
A Short
History of the Art of Distillation: From the Beginnings Up to the Death of Cellier Blumenthal
2589:
A Short History of the Art of Distillation: From the Beginnings Up to the Death of Cellier Blumenthal
2328:
2236:
2158:
1824:
1812:
1711:
1336:
1177:
1037:
1033:
942:
938:
distillation, and the same and subsequent years saw developments in this theme for oils and spirits.
926:
got a patent for improving the design even further. Coffey's continuous still may be regarded as the
855:
775:
495:
450:
222:
components. In either method, the separation process of distillation exploits the differences in the
214:
1327:
For many cases, the boiling points of the components in the mixture will be sufficiently close that
827:
4263:
4177:
3712:
3415:
3175:
1873:
1796:
1680:
1679:
is a method for the separation of mixtures of liquids by partial vaporization through a non-porous
1531:
1494:
1375:
970:
507:
242:
223:
1096:, which is when the vapor phase and liquid phase contain the same composition. Although there are
445:
4323:
4219:
4172:
4065:
3877:
3223:
3194:
3153:
3124:
3073:
2714:
2618:
2457:
2019. Distillation: The Historical Symbol of Chemical Engineering. The University of Toledo. URL
2304:
2197:
2128:
1731:
1663:
may be added to remove carbon dioxide from water followed by a second distillation with a little
1425:
1369:
1310:
1006:
401:
321:
168:
3359:
1268:
639:
that was translated into Latin and would go on to form the most important alchemical source for
65:
604:
of organic substances plays an important role in the works attributed to Jābir, such as in the
4463:
4338:
4298:
4106:
4030:
3914:
3894:
3743:
3718:
3674:
3527:
3399:
3233:
3202:
3163:
3132:
3116:
3045:
3035:
3004:
2996:
2974:
2970:
2768:
2760:
2750:
2695:
2645:
2597:
2566:
2551:
Kockmann, Norbert (2014). "History of Distillation". In Andrzej, Górak; Sorensen, Eva (eds.).
2404:
2396:
2386:
2283:
2146:
2141:
2100:
2038:
2034:
1898:
1770:
1696:
1501:
1395:
1341:
990:
613:
469:
388:, such as filtration, crystallization, extraction, sublimation, or mechanical pressing of oil.
339:
238:
50:
3227:
3120:
2964:
2834:
737:
province, China, dating back to the 12th century. Distilled beverages were common during the
4147:
4035:
3936:
3869:
3839:
3812:
3664:
3656:
3333:
3097:
3065:
2815:
2784:
2746:
2641:
2593:
2558:
2533:
2435:
2420:
1762:
1739:
1305:
1293:
1108:
714:
590:
586:
190:
3931:
4521:
4458:
4383:
4363:
4343:
4318:
4258:
4020:
3769:
3565:
3437:
3394:
Othmer, D. F. (1982) "Distillation – Some Steps in its Development", in W. F. Furter (ed)
3379:
3170:
2949:
2906:
2878:
2776:
2484:
2465:
2412:
2338:
2162:
2091:
2056:
1881:
1660:
1601:
1411:
1379:
962:
958:
491:
454:
440:
405:
43:
4010:
4005:
2691:
2684:
2161:
of the process, for use in desalination, or in some cases one stage in the production of
1847:
1328:
1278:
1049:
1045:
3808:
2172:
can achieve more than 20 effects with thermal energy input, as mentioned in the article.
2004:
295 ft) or more. When the process feed has a diverse composition, as in distilling
4537:
4248:
3669:
3644:
3156:(2009). "Alcohol and the Distillation of Wine in Arabic Sources from the 8th Century".
2562:
2537:
2048:
2013:
1989:
1535:
1399:
1301:
1233:
1194:
1065:
1029:
966:
904:
872:
811:
519:
315:
299:
249:. These are some applications of the chemical separation process that is distillation:
246:
234:
1965:
1001:
from the source materials to the distillate in the processing of beverages and herbs.
914:
In the early 19th century, the basics of modern techniques, including pre-heating and
899:. Pot stills made of various materials (wood, clay, stainless steel) are also used by
4552:
4478:
4453:
4080:
3942:
3453:
3419:
3077:
3054:
Vol. II, p. 5. On the attribution of the Latin translation to Gerard of Cremona, see
2718:
2379:
2132:
2072:
2052:
1985:
1950:
1808:
1754:
of rotating the sample. The vacuum is normally generated by using a high vacuum pump.
1743:
1676:
1664:
1597:
1391:
1061:
1025:
954:
931:
923:
880:
876:
605:
148:
3579:
2286:
is able to distill solvents more quickly at lower temperatures through the use of a
2082:
Section of an industrial distillation tower showing detail of trays with bubble caps
4438:
4423:
4313:
3602:
2792:
2448:
Schaschke, C., 2014. A Dictionary of Chemical Engineering. Oxford University Press.
2268:
2202:
2124:
2108:
2064:
1981:
1700:
1407:
1403:
1297:
1164:
1084:
The idealized model is accurate in the case of chemically similar liquids, such as
919:
863:
819:
738:
436:
292:
284:
264:
180:
2008:, liquid outlets at intervals up the column allow for the withdrawal of different
1181:
an enriched, boiling liquid mixture, all with their own vapor–liquid equilibrium.
3908:
2740:
2666:
2635:
2587:
2497:
4413:
4268:
2960:
2348:
1887:
1723:
1527:
1249:
1245:
640:
558:
473:
357:
274:
218:
194:
3158:
Studies in al-Kimya': Critical Issues in Latin and Arabic Alchemy and Chemistry
2078:
201:) is the heating of solid materials to produce gases that condense either into
4433:
4418:
4308:
4243:
3816:
3308:
3069:
2819:
2772:
2267:
Diagram of an industrial-scale vacuum distillation column as commonly used in
2095:
1200:
900:
892:
858:
with long necks pointing to the side at a downward angle to act as air-cooled
832:
680:
539:
3660:
3625:
3049:
2917:
Javed Husain, "The So-Called 'Distillery' at Shaikhan Dheri - A Case Study",
1252:
packed with materials that scavenge the undesired air components, or through
600:) experimented extensively with the distillation of various substances. The
4516:
4101:
3693:
3372:
2788:
2780:
2424:
2408:
2045:
2005:
1939:
1933:
1869:
1852:
1774:
1690:
1415:
1093:
946:
927:
868:
843:
687:
669:
523:
416:
353:
198:
3678:
2416:
2115:
1203:(packed with glass beads, metal pieces, or other chemically inert material)
1176:
Both batch and continuous distillations can be improved by making use of a
490:
described the process. Work on distilling other liquids continued in early
2459:
https://www.utoledo.edu/engineering/chemical-engineering/distillation.html
1123:
546:
says these terracotta distill tubes were "made to imitate bamboo". These "
4473:
4448:
4233:
3860:
Allchin, F. R. (March 1979). "India: The Ancient Home of Distillation?".
3101:
2503:
2145:
not be achieved. The packing will appear to not be working properly. The
1816:
1800:
1786:
1620:
1507:
This technique is also very useful for compounds which boil beyond their
1289:
1257:
981:
The application of distillation can roughly be divided into four groups:
969:
to the region. The availability of powerful computers has allowed direct
658:
636:
547:
527:
307:
3843:
3795:
Spiegel, L (2006). "A new method to assess liquid distributor quality".
3337:
1228:
in which the source material is heated, a condenser in which the heated
554:, as there was no efficient means of collecting the vapors at low heat.
4348:
4188:
4096:
3881:
2323:
2252:
2206:
2068:
1954:
1891:
1858:
1735:
1253:
1089:
1085:
1073:
935:
884:
851:
839:
465:
258:
257:
products to yield alcoholic beverages with a high content by volume of
176:
172:
3085:
800:
4288:
4015:
2665:
Gildemeister, E.; Hoffman, Fr.; translated by Edward Kremers (1913).
2478:
https://www.ranken-energy.com/index.php/products-made-from-petroleum/
2287:
2232:
2029:
1616:
915:
847:
805:
576:
551:
515:
511:
303:
3873:
3692:
Kolesnichenko, V.L.; Goloverda, G.Z.; Kolesnichenko, I.V.; Wang, G.
3279:"These Are 5 Oldest Companies In Europe. Ever Heard Of Any Of Them?"
2552:
237:
that identifies and denotes a process of physical separation, not a
2476:
2017. Products made from petroleum. Ranken Energy Corporation. URL
1897:
High boiling azeotropes, such as a 20 percent by weight mixture of
867:
made the condensation of alcohol more efficient. These were called
229:
In the industrial applications of classical distillation, the term
4483:
4116:
3643:
Kolesnichenko, I.V.; Goloverda, G.Z.; Kolesnichenko, V.L. (2020).
3501:
2933:
Frank Raymond Allchin, "India: the ancient home of distillation?"
2890:
Frank Raymond Allchin, "India: the ancient home of distillation?"
2114:
2077:
2060:
2019:
1964:
1850:
does not hold. Such interactions can result in a constant-boiling
1820:
1778:
1704:
1624:
1612:
1552:
1439:
1420:
1387:
1267:
1220:
1122:
896:
826:
818:
810:
799:
784:
734:
535:
444:
311:
202:
184:
1383:
condensed, usually yielding a layer of oil and a layer of water.
1335:
As the solution to be purified is heated, its vapors rise to the
2764:
2400:
1782:
1523:
1432:°C. Under a vacuum, it distills off into the receiver at only 70
710:
654:
557:
Distillation in China may have begun at the earliest during the
288:
267:
to produce potable water and for medico-industrial applications.
4192:
3964:
3960:
2107:
holdup, and batch distillation. Conversely, factors that favor
1905:
Breaking an azeotrope with unidirectional pressure manipulation
419:
knew that water condensing from evaporating seawater is fresh:
3694:"Methods of Solvent Removal at Ambient Temperatures – Cryovap"
2377:
Harwood, Laurence M.; Moody, Christopher J. (1 January 1990).
2210:
1229:
888:
348:
3785:, Chemical Plants & Processing, Edition Europe, pp. 11–15
3628:[Design of advanced processes of zone distillation].
3265:
The Book of the Art of Distillation out of Simple Ingredients
1611:
taps to allows fractions to be isolated from the rest of the
765:
The Book of the Art of Distillation out of Simple Ingredients
1803:
instead of evaporation. It is not truly distillation, but a
1769:, despite the name, is not truly distillation, but rather a
1036:, where the vapor pressure of the liquid equals the ambient
3645:"A Versatile Method of Ambient-Temperature Solvent Removal"
3129:
Instruments and Experimentation in the History of Chemistry
2385:(Illustrated ed.). Blackwell Scientific Publications.
1876:) can be added to convert the soluble water into insoluble
3772:. Resources.schoolscience.co.uk. Retrieved on 2014-04-20.
2157:
The goal of multi-effect distillation is to increase the
1596:
Some compounds have high boiling points as well as being
709:("burning water", i.e., ethanol) by distilling wine with
701:). In the twelfth century, recipes for the production of
464:
Early evidence of distillation has been found related to
449:
Distillation equipment used by the 3rd century alchemist
3568:. B/R Instrument Corporation (accessed 8 September 2006)
2499:
Chemistry and Chemical Technology in Ancient Mesopotamia
2381:
Experimental Organic Chemistry: Principles and Practice
374:
German chemical engineer Norbert Kockmann respectively:
3582:[Zone distillation: a new method of refining].
1861:(by mass) in water forms an azeotrope at 78.1 °C.
1734:
a vacuum distillation apparatus is used to remove bulk
1607:
The Perkin triangle has means via a series of glass or
1406:
while the watery distillates have many applications in
1349:
is thus a concept rather than an accurate description.
2044:
Such industrial fractionating towers are also used in
1738:
from a sample. Typically the vacuum is generated by a
1352:
More theoretical plates lead to better separations. A
989:, distillation of herbs for perfumery and medicinals (
82:
round-bottom flask containing the mixture to be boiled
3891:
Transport Processes and Separation Process Principles
3498:
ST07 Separation of liquid–liquid mixtures (solutions)
3034:. Cairo: Institut Français d'Archéologie Orientale.
1777:
in which solid substances are heated in an inert or
1111:. If ultra-pure products are the goal, then further
994:
606:
4530:
4502:
4401:
4226:
4125:
4089:
4058:
3998:
1300:bath (orange, 14). The vapor flows through a short
226:of the component substances of the heated mixture.
3948:. Chemical Engineering Research Information Center
3626:"Разработка перспективных схем зонной дистилляции"
3160:. Hildesheim: Georg Olms Verlag. pp. 283–298.
2683:
2378:
2214:more flavor compounds and characteristic flavors.
2119:Large-scale, industrial vacuum distillation column
2024:Diagram of a typical industrial distillation tower
1823:content, respectively. It is also used to produce
1522:is vacuum distillation below the pressure of 0.01
982:
635:, an originally Arabic work falsely attributed to
612:('The Book of Seventy'), translated into Latin by
30:Several terms redirect here. For other uses, see
2806:Taylor, F. (1945). "The evolution of the still".
1390:herbs and flowers can result in two products: an
550:stills" were only capable of producing very weak
1561:Still pot with stirrer bar/anti-bumping granules
1127:A batch still showing the separation of A and B.
934:was granted a U.S. Patent for a tray column for
241:; thus an industrial installation that produces
3832:Industrial & Engineering Chemistry Research
3580:"Зонная дистилляция: новый метод рафинирования"
3182:. Vol. I–III. Paris: Imprimerie nationale.
2372:
2370:
2368:
2251:A simple set-up to distill dry and oxygen-free
1940:positive and negative pressures may be employed
479:Distilled water has been in use since at least
421:
382:
376:
324:to separate impurities and unreacted materials.
4204:
3976:
2966:Economic History of Medieval India, 1200–1500
1799:is an analogous method of purification using
8:
2682:Bryan H. Bunch; Alexander Hellemans (2004).
2671:. Vol. 1. New York: Wiley. p. 203.
1934:Azeotrope § Pressure swing distillation
1526:. 0.01 torr is one order of magnitude above
1285:sufficiently pure for its intended purpose.
1028:of a liquid is the temperature at which the
986:
685:
400:(to drip off) when used by the Romans, e.g.
338:Early evidence of distillation was found on
18:Rectification (chemical/process engineering)
2835:"The Discovery of Alcohol and Distillation"
2067:also enables the production of high-purity
1640:Closed-system vacuum distillation (cryovap)
755:
725:The distillation of beverages began in the
703:
695:
630:
624:
4211:
4197:
4189:
3983:
3969:
3961:
3649:Organic Process Research & Development
2919:Journal of the Pakistan Historical Society
657:is attested in Arabic works attributed to
506:Distillation was practiced in the ancient
205:products or into solid products. The term
3738:Seader, J. D.; Henley, Ernest J. (1998).
3668:
3607:Problems of Atomic Science and Technology
3584:Problems of Atomic Science and Technology
3390:
3388:
2731:
2729:
2554:Distillation: Fundamentals and Principles
2094:can be used. For a multi-component feed,
1884:are often used for this purpose as well.
1693:with the other components in the mixture.
1272:Schematic of a simple distillation setup.
1224:, consists at a minimum of a reboiler or
1044:distillation are essentially governed by
107:the cooling-water outlet of the condenser
3520:Perry, Robert H.; Green, Don W. (1984).
3515:
3513:
3511:
3509:
3260:Liber de arte distillandi de simplicibus
3131:. Cambridge: MIT Press. pp. 35–54.
2720:Collection des anciens alchimistes grecs
2147:height equivalent to a theoretical plate
1699:(or partial evaporation) is the partial
758:Liber de arte distillandi de simplicibus
147:the stirring mechanism (not shown, e.g.
127:the heat control for heating the mixture
102:the cooling-water inlet of the condenser
64:
2364:
2239:exclusively for distillation processes.
2221:
1564:Cold finger – bent to direct condensate
791:Liber de arte Distillandi de Compositis
283:used in the midstream operations of an
3943:"Binary Vapor-Liquid Equilibrium Data"
3781:Moore, F., Rukovena, F. (August 1987)
3326:Industrial & Engineering Chemistry
2990:
2988:
2986:
2849:from the original on 29 November 2017.
1969:Typical industrial distillation towers
571:Fractional distillation § History
392:According to Dutch chemical historian
2690:. Houghton Mifflin Harcourt. p.
2686:The History of Science and Technology
2319:Atmospheric distillation of crude oil
1457:Thermometer/Boiling point temperature
1072:the pressure surrounding the liquid,
577:Liquor § History of distillation
538:dating to the early centuries of the
209:includes the separation processes of
171:the component substances of a liquid
142:heating bath (oil/sand) for the flask
7:
3523:Perry's Chemical Engineers' Handbook
850:became used for distillations. Both
137:stirring mechanism and heating plate
3797:Chemical Engineering and Processing
3121:"Alchemy, Assaying, and Experiment"
2526:Journal of the Institute of Brewing
2522:"The Early History of Distillation"
1953:after its catalytic synthesis from
1726:may also be called "distillation":
1444:Perkin triangle distillation setup
510:, which is evident from baked clay
77:The heat source to boil the mixture
3937:Case Study: Petroleum Distillation
3889:Geankoplis, Christie John (2003).
3603:"Zone distillation: justification"
2563:10.1016/B978-0-12-386547-2.00001-6
2538:10.1002/j.2050-0416.1907.tb02205.x
1917:equipment or a change of coolant.
1667:added to remove traces of ammonia.
1155:fractions, including at least one
1119:Batch or differential distillation
965:in 1961 with the hope of bringing
589:(Latin: Geber, ninth century) and
502:Ancient India and China (1–500 CE)
156:the distillate-cooling water bath.
25:
3910:Science and Civilisation in China
3396:A Century of Chemical Engineering
2557:. Academic Press. pp. 1–43.
2231:Chemistry in its beginnings used
1789:is the root of its common name –
1718:organic and inorganic components.
1592:Air-sensitive vacuum distillation
1448:Stirrer bar/anti-bumping granules
911:order to obtain a pure compound.
408:, was "never used in our sense".
92:mixture boiling-point thermometre
4392:
4049:
3232:. Harmondsworth: Penguin Books.
3162:(same content also available on
2295:
2275:
2260:
2244:
2224:
1402:are often used in perfumery and
693:(later translated into Latin as
683:'s (Latin: Abulcasis, 936–1013)
132:stirring mechanism speed control
3893:(4th ed.). Prentice Hall.
3003:. Routledge. pp. 147–148.
2833:Berthelot, M. P. E. M. (1893).
1356:system uses a spinning band of
1197:(usually laboratory scale only)
831:Simple liqueur distillation in
151:or mechanical stirring machine)
3913:. Cambridge University Press.
2170:Multi-stage flash distillation
1386:Steam distillation of various
561:dynasty (1st–2nd century CE).
441:Distilled water § History
112:the distillate-receiving flask
1:
3763:Energy Institute website page
3740:Separation Process Principles
3717:(1st ed.). McGraw-Hill.
3526:(6th ed.). McGraw-Hill.
3373:Traditional Alembic Pot Still
2176:Vapor compression evaporation
2012:or products having different
1232:is cooled back to the liquid
673:
662:
644:
617:
594:
480:
343:
32:Distillation (disambiguation)
27:Method of separating mixtures
3382:, accessed 16 November 2006.
3178:; Houdas, Octave V. (1893).
3127:; Levere, Trevor H. (eds.).
2334:Low-temperature distillation
1576:Distillate flask/distillate.
842:evolved into the science of
70:Laboratory model of a still.
3084:Moureau, Sébastien (2020).
2839:The Popular Science Monthly
2112:subject to process surges.
1928:Pressure-swing distillation
1890:liquids, such as water and
1244:pressure. Alternatively, a
686:
679:), and in the 28th book of
607:
437:Desalination § History
369:Greek and Roman terminology
302:into the component gases —
40:Distillery (disambiguation)
4610:
4429:Electrostatic precipitator
4112:Spinning band distillation
3624:Kravchenko, A. I. (2014).
3601:Kravchenko, A. I. (2014).
3578:Kravchenko, A. I. (2011).
3559:Spinning Band Distillation
2195:
1972:
1931:
1839:
1546:
1530:, where fluids are in the
1492:
1367:
1354:spinning band distillation
1320:
1207:Spinning band distillation
1147:
749:In 1500, German alchemist
574:
568:
434:
97:the condenser of the still
36:Distiller (disambiguation)
29:
4469:Rotary vacuum-drum filter
4390:
4047:
3817:10.1016/j.cep.2006.05.003
3711:Kister, Henry Z. (1992).
3477:Science History Institute
3070:10.1017/S0269889701000096
2820:10.1080/00033794500201451
2153:Multi-effect distillation
1807:where the product is the
1509:decomposition temperature
973:of distillation columns.
854:and retorts are forms of
823:Old Ukrainian vodka still
795:Science History Institute
769:The Green Tree Distillery
632:De anima in arte alkimiae
122:the receiver of the still
117:vacuum pump and gas inlet
4512:Aqueous two-phase system
4334:Liquid–liquid extraction
4041:Vapor–liquid equilibrium
3907:Needham, Joseph (1980).
3661:10.1021/acs.oprd.9b00368
3632:(in Russian) (7): 68–72.
3378:21 November 2006 at the
3313:. London: Richard Cotes.
3255:Braunschweig, Hieronymus
3199:The Origins of Chemistry
3169:29 December 2015 at the
2997:"Wine, women and poison"
2995:Haw, Stephen G. (2012).
2948:20 December 2019 at the
2924::3:289-314 (Jul 1, 1993)
2905:20 December 2019 at the
2877:13 December 2022 at the
2745:(2nd ed.). Leiden:
1878:water of crystallization
1819:to increase ethanol and
1767:destructive distillation
1104:data is by measurement.
1102:vapor–liquid equilibrium
789:Hieronymus Brunschwig's
729:(10th–13th century) and
488:Alexander of Aphrodisias
211:destructive distillation
4409:API oil–water separator
4279:Dissolved air flotation
4071:Continuous distillation
3768:12 October 2007 at the
3630:Perspectivnye Materialy
3547:Fractional Distillation
3436:4 February 2017 at the
3310:The Art of Distillation
2723:. 3 vol., Paris, p. 161
2634:Forbes, R. J. (1948) .
2586:Forbes, R. J. (1948) .
1975:Continuous distillation
1866:azeotropic distillation
1842:Azeotropic distillation
1687:Extractive distillation
1604:apparatus can be used.
1549:Short-path distillation
1543:Short path distillation
1323:Fractional distillation
1317:Fractional distillation
1150:Continuous distillation
1144:Continuous distillation
1054:vapor–liquid equilibria
1014:continuous distillation
987:industrial distillation
780:The Art of Distillation
602:fractional distillation
514:and receivers found at
476:in the 1st century CE.
281:Fractional distillation
271:Crude oil stabilisation
179:of the mixture and the
58:Distillate (motor fuel)
4374:Solid-phase extraction
3564:25 August 2006 at the
3180:La Chimie au Moyen Âge
2496:Levey, Martin (1959).
2129:structured sheet metal
2120:
2083:
2028:Industrial towers use
2025:
1994:natural gas processing
1970:
1671:Catalytic distillation
1580:
1520:Molecular distillation
1515:Molecular distillation
1485:
1437:
1273:
1260:, into the apparatus.
1128:
1064:. This law applies to
941:With the emergence of
835:
824:
816:
808:
797:
756:
704:
696:
631:
625:
461:
425:
390:
381:
217:, breaking down large
165:classical distillation
157:
4579:Laboratory techniques
4494:Vacuum ceramic filter
4489:Sublimation apparatus
4294:Electrochromatography
4254:Cross-flow filtration
3946:(searchable database)
3201:. London: Oldbourne.
3184:vol. I, pp. 141, 143.
2483:16 April 2021 at the
2464:14 April 2021 at the
2354:Random column packing
2118:
2081:
2023:
1968:
1932:Further information:
1656:reactive distillation
1556:
1443:
1424:
1271:
1214:Laboratory procedures
1126:
1098:computational methods
830:
822:
814:
803:
788:
751:Hieronymus Brunschwig
741:(13th–14th century).
544:Frank Raymond Allchin
448:
245:, is a distillery of
87:the head of the still
68:
4574:Separation processes
4569:Alchemical processes
4444:Fractionating column
4239:Acid–base extraction
4220:Separation processes
4076:Fractionating column
4059:Industrial processes
4026:McCabe–Thiele method
3932:Alcohol distillation
3176:Berthelot, Marcellin
3164:the author's website
2617:Aristotle. (1952) .
2520:Fairley, T. (1907).
2329:Fragrance extraction
2237:laboratory equipment
2088:McCabe–Thiele method
2000:petroleum products.
1982:petroleum refineries
1722:The unit process of
1712:Dean-Stark apparatus
1454:Fractionating column
1428:usually boils at 189
1394:as well as a watery
1337:fractionating column
1178:fractionating column
1172:General improvements
1038:atmospheric pressure
1034:normal boiling point
971:computer simulations
951:McCabe–Thiele method
943:chemical engineering
653:The distillation of
626:Liber de septuaginta
498:in the 3rd century.
496:Zosimus of Panopolis
451:Zosimos of Panopolis
431:Alexandrian chemists
167:, is the process of
4264:Cyclonic separation
3844:10.1021/ie00069a021
3809:2006CEPPI..45.1011S
3742:. New York: Wiley.
3714:Distillation Design
3362:on 4 November 2012.
3352:"Sealing Technique"
3338:10.1021/ie50318a015
3224:Holmyard, Eric John
3195:Multhauf, Robert P.
3154:al-Hassan, Ahmad Y.
3125:Holmes, Frederic L.
3001:Marco Polo in China
2739:(1 December 1970).
2715:Berthelot, Marcelin
1874:potassium carbonate
1797:Freeze distillation
1751:Kugelrohr apparatus
1586:Kugelrohr apparatus
1532:free molecular flow
1495:Vacuum distillation
1489:Vacuum distillation
1376:vacuum distillation
1311:ground glass joints
1264:Simple distillation
1113:chemical separation
793:(Strassburg, 1512)
508:Indian subcontinent
364:Classical antiquity
295:for livestock feed.
243:distilled beverages
224:relative volatility
183:of the vapors in a
4594:Ancient inventions
4324:Gravity separation
4090:Laboratory methods
4066:Batch distillation
3700:. 2021/0178287 Al.
3285:. 28 December 2018
3267:] (in German).
3117:Newman, William R.
3058:Science in Context
2737:Forbes, Robert J.
2305:round-bottom flask
2198:Distilled beverage
2187:In food processing
2142:theoretical stages
2121:
2084:
2063:. Distillation of
2059:, and high purity
2039:Chemical engineers
2035:theoretical plates
2026:
1971:
1961:Industrial process
1836:Azeotropic process
1732:rotary evaporation
1581:
1534:regime, i.e., the
1486:
1438:
1426:Dimethyl sulfoxide
1370:Steam distillation
1364:Steam distillation
1304:3, then through a
1274:
1238:distillation types
1129:
1007:batch distillation
836:
825:
817:
809:
798:
623:) under the title
565:Islamic Golden Age
462:
322:Chemical synthesis
158:
56:, and
4584:Phase transitions
4546:
4545:
4464:Rapid sand filter
4359:Recrystallization
4339:Electroextraction
4299:Electrofiltration
4186:
4185:
4107:Rotary evaporator
4031:Theoretical plate
3900:978-0-13-101367-4
3749:978-0-471-58626-5
3724:978-0-07-034909-4
3533:978-0-07-049479-4
3239:978-0-486-26298-7
3010:978-1-134-27542-7
2971:Pearson Education
2808:Annals of Science
2756:978-90-04-00617-1
2701:978-0-618-22123-3
2668:The Volatile Oils
2651:978-90-04-00617-1
2603:978-90-04-00617-1
2572:978-0-12-386547-2
2392:978-0-632-02016-4
2284:rotary evaporator
2159:energy efficiency
2101:equilibrium stage
1899:hydrochloric acid
1805:recrystallization
1771:chemical reaction
1697:Flash evaporation
1631:Zone distillation
1567:Cooling water out
1502:rotary evaporator
1466:Cooling water out
1396:herbal distillate
1347:theoretical plate
1342:theoretical plate
991:herbal distillate
846:, vessels called
666: 801–873 CE
614:Gerard of Cremona
459:Parisinus graces.
239:chemical reaction
235:unit of operation
215:chemical cracking
16:(Redirected from
4601:
4589:Gas technologies
4396:
4213:
4206:
4199:
4190:
4053:
4036:Partial pressure
3985:
3978:
3971:
3962:
3957:
3955:
3953:
3947:
3904:
3885:
3848:
3847:
3827:
3821:
3820:
3792:
3786:
3779:
3773:
3760:
3754:
3753:
3735:
3729:
3728:
3708:
3702:
3701:
3689:
3683:
3682:
3672:
3640:
3634:
3633:
3621:
3615:
3614:
3598:
3592:
3591:
3575:
3569:
3556:
3550:
3544:
3538:
3537:
3517:
3504:
3495:
3489:
3488:
3486:
3484:
3469:
3463:
3462:
3461:
3457:
3446:
3440:
3428:
3427:
3423:
3412:
3406:
3392:
3383:
3370:
3364:
3363:
3358:. Archived from
3348:
3342:
3341:
3332:(6): 677. 1936.
3324:"Distillation".
3321:
3315:
3314:
3301:
3295:
3294:
3292:
3290:
3275:
3269:
3268:
3251:
3245:
3243:
3220:
3214:
3212:
3191:
3185:
3183:
3161:
3150:
3144:
3142:
3113:
3107:
3105:
3081:
3064:(1–2): 249–288.
3053:
3024:
3018:
3017:
2992:
2981:
2958:
2952:
2941::1:55-63 (1979)
2931:
2925:
2915:
2909:
2898::1:55-63 (1979)
2888:
2882:
2857:
2851:
2850:
2830:
2824:
2823:
2803:
2797:
2796:
2747:Brill Publishers
2733:
2724:
2712:
2706:
2705:
2689:
2679:
2673:
2672:
2662:
2656:
2655:
2631:
2625:
2624:
2614:
2608:
2607:
2583:
2577:
2576:
2548:
2542:
2541:
2517:
2511:
2510:
2493:
2487:
2474:
2468:
2455:
2449:
2446:
2440:
2439:
2436:Internet Archive
2433:
2431:
2384:
2374:
2299:
2279:
2264:
2248:
2228:
1882:Molecular sieves
1763:Dry distillation
1573:Vacuum/gas inlet
1570:cooling water in
1475:Vacuum/gas inlet
1469:Cooling water in
1435:
1431:
1306:Liebig condenser
1294:magnetic stirrer
1109:partial pressure
1052:and assume that
983:laboratory scale
761:
715:Taddeo Alderotti
707:
699:
697:Liber servatoris
691:
678:
675:
667:
664:
649:
648: 1220–1292
646:
634:
628:
622:
621: 1114–1187
619:
610:
599:
596:
593:(Latin: Rhazes,
591:Abū Bakr al-Rāzī
587:Jābir ibn Ḥayyān
485:
482:
394:Robert J. Forbes
351:
345:
316:industrial gases
207:dry distillation
191:Dry distillation
21:
4609:
4608:
4604:
4603:
4602:
4600:
4599:
4598:
4564:Unit operations
4549:
4548:
4547:
4542:
4526:
4504:
4498:
4459:Protein skimmer
4397:
4388:
4384:Ultrafiltration
4364:Reverse osmosis
4344:Microfiltration
4319:Froth flotation
4259:Crystallization
4222:
4217:
4187:
4182:
4121:
4085:
4054:
4045:
4021:Fenske equation
3994:
3989:
3951:
3949:
3945:
3941:
3928:
3901:
3888:
3874:10.2307/2801640
3859:
3856:
3854:Further reading
3851:
3829:
3828:
3824:
3794:
3793:
3789:
3780:
3776:
3770:Wayback Machine
3761:
3757:
3750:
3737:
3736:
3732:
3725:
3710:
3709:
3705:
3691:
3690:
3686:
3642:
3641:
3637:
3623:
3622:
3618:
3600:
3599:
3595:
3577:
3576:
3572:
3566:Wayback Machine
3557:
3553:
3545:
3541:
3534:
3519:
3518:
3507:
3496:
3492:
3482:
3480:
3479:. 19 March 2019
3471:
3470:
3466:
3459:
3448:
3447:
3443:
3438:Wayback Machine
3425:
3414:
3413:
3409:
3393:
3386:
3380:Wayback Machine
3371:
3367:
3350:
3349:
3345:
3323:
3322:
3318:
3303:
3302:
3298:
3288:
3286:
3277:
3276:
3272:
3253:
3252:
3248:
3240:
3222:
3221:
3217:
3209:
3193:
3192:
3188:
3174:
3171:Wayback Machine
3152:
3151:
3147:
3139:
3115:
3114:
3110:
3083:
3055:
3042:
3026:
3025:
3021:
3011:
2994:
2993:
2984:
2959:
2955:
2950:Wayback Machine
2932:
2928:
2916:
2912:
2907:Wayback Machine
2889:
2885:
2879:Wayback Machine
2858:
2854:
2832:
2831:
2827:
2805:
2804:
2800:
2757:
2735:
2734:
2727:
2713:
2709:
2702:
2681:
2680:
2676:
2664:
2663:
2659:
2652:
2633:
2632:
2628:
2616:
2615:
2611:
2604:
2585:
2584:
2580:
2573:
2550:
2549:
2545:
2519:
2518:
2514:
2495:
2494:
2490:
2485:Wayback Machine
2475:
2471:
2466:Wayback Machine
2456:
2452:
2447:
2443:
2429:
2427:
2393:
2376:
2375:
2366:
2362:
2339:Microdistillery
2315:
2308:
2300:
2291:
2280:
2271:
2265:
2256:
2249:
2240:
2229:
2220:
2200:
2194:
2189:
2163:ultrapure water
2155:
2092:Fenske equation
2057:liquid nitrogen
1990:chemical plants
1977:
1963:
1936:
1930:
1907:
1857:example, 95.6%
1844:
1838:
1740:water aspirator
1654:The process of
1651:
1642:
1633:
1602:Perkin triangle
1594:
1579:
1551:
1545:
1517:
1497:
1491:
1484:
1433:
1429:
1412:food processing
1380:heating element
1372:
1366:
1325:
1319:
1266:
1216:
1174:
1152:
1146:
1121:
1066:ideal solutions
1022:
1020:Idealized model
995:food processing
979:
963:Freeport, Texas
959:Fenske equation
771:, was founded.
747:
723:
688:Kitāb al-Taṣrīf
676:
665:
647:
620:
608:Kitāb al-Sabʿīn
597:
583:Muslim chemists
579:
573:
567:
504:
492:Byzantine Egypt
483:
455:Byzantine Greek
443:
433:
414:
406:Pliny the Elder
371:
366:
346:
336:
331:
152:
143:
138:
133:
128:
123:
118:
113:
108:
103:
98:
93:
88:
83:
78:
73:
72:
61:
28:
23:
22:
15:
12:
11:
5:
4607:
4605:
4597:
4596:
4591:
4586:
4581:
4576:
4571:
4566:
4561:
4551:
4550:
4544:
4543:
4541:
4540:
4538:Unit operation
4534:
4532:
4528:
4527:
4525:
4524:
4519:
4514:
4508:
4506:
4500:
4499:
4497:
4496:
4491:
4486:
4481:
4476:
4471:
4466:
4461:
4456:
4451:
4446:
4441:
4436:
4431:
4426:
4421:
4416:
4411:
4405:
4403:
4399:
4398:
4391:
4389:
4387:
4386:
4381:
4376:
4371:
4366:
4361:
4356:
4351:
4346:
4341:
4336:
4331:
4326:
4321:
4316:
4311:
4306:
4301:
4296:
4291:
4286:
4281:
4276:
4271:
4266:
4261:
4256:
4251:
4249:Chromatography
4246:
4241:
4236:
4230:
4228:
4224:
4223:
4218:
4216:
4215:
4208:
4201:
4193:
4184:
4183:
4181:
4180:
4175:
4170:
4165:
4160:
4155:
4150:
4145:
4140:
4135:
4129:
4127:
4123:
4122:
4120:
4119:
4114:
4109:
4104:
4099:
4093:
4091:
4087:
4086:
4084:
4083:
4078:
4073:
4068:
4062:
4060:
4056:
4055:
4048:
4046:
4044:
4043:
4038:
4033:
4028:
4023:
4018:
4013:
4008:
4002:
4000:
3996:
3995:
3990:
3988:
3987:
3980:
3973:
3965:
3959:
3958:
3939:
3934:
3927:
3926:External links
3924:
3923:
3922:
3905:
3899:
3886:
3855:
3852:
3850:
3849:
3822:
3787:
3774:
3755:
3748:
3730:
3723:
3703:
3684:
3635:
3616:
3593:
3586:(in Russian).
3570:
3551:
3539:
3532:
3505:
3490:
3464:
3454:Solvay, Ernest
3441:
3407:
3384:
3365:
3356:copper-alembic
3343:
3316:
3296:
3270:
3246:
3238:
3215:
3207:
3186:
3145:
3137:
3108:
3040:
3019:
3009:
2982:
2953:
2926:
2910:
2883:
2852:
2825:
2798:
2755:
2725:
2707:
2700:
2674:
2657:
2650:
2644:. p. 14.
2626:
2609:
2602:
2596:. p. 15.
2578:
2571:
2543:
2532:(6): 559–582.
2512:
2506:. p. 36.
2488:
2469:
2450:
2441:
2391:
2363:
2361:
2358:
2357:
2356:
2351:
2346:
2341:
2336:
2331:
2326:
2321:
2314:
2311:
2310:
2309:
2301:
2294:
2292:
2281:
2274:
2272:
2269:oil refineries
2266:
2259:
2257:
2250:
2243:
2241:
2230:
2223:
2219:
2216:
2196:Main article:
2193:
2190:
2188:
2185:
2180:
2179:
2173:
2154:
2151:
2049:air separation
2014:boiling points
1973:Main article:
1962:
1959:
1929:
1926:
1906:
1903:
1840:Main article:
1837:
1834:
1833:
1832:
1828:
1794:
1756:
1755:
1747:
1720:
1719:
1715:
1708:
1694:
1684:
1674:
1668:
1650:
1647:
1641:
1638:
1632:
1629:
1593:
1590:
1578:
1577:
1574:
1571:
1568:
1565:
1562:
1558:
1547:Main article:
1544:
1541:
1536:mean free path
1516:
1513:
1493:Main article:
1490:
1487:
1483:
1482:
1481:Still receiver
1479:
1476:
1473:
1470:
1467:
1464:
1461:
1458:
1455:
1452:
1449:
1445:
1400:essential oils
1368:Main article:
1365:
1362:
1321:Main article:
1318:
1315:
1302:Vigreux column
1265:
1262:
1215:
1212:
1211:
1210:
1204:
1198:
1195:Vigreux column
1192:
1173:
1170:
1148:Main article:
1145:
1142:
1120:
1117:
1056:are attained.
1030:vapor pressure
1021:
1018:
978:
975:
967:water security
905:essential oils
746:
743:
722:
721:Medieval China
719:
677: 872–950
598: 865–925
569:Main article:
566:
563:
520:Shaikhan Dheri
503:
500:
432:
429:
413:
410:
370:
367:
365:
362:
342:tablets dated
335:
332:
330:
327:
326:
325:
319:
300:Air separation
296:
287:for producing
278:
268:
262:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
4606:
4595:
4592:
4590:
4587:
4585:
4582:
4580:
4577:
4575:
4572:
4570:
4567:
4565:
4562:
4560:
4557:
4556:
4554:
4539:
4536:
4535:
4533:
4529:
4523:
4520:
4518:
4515:
4513:
4510:
4509:
4507:
4501:
4495:
4492:
4490:
4487:
4485:
4482:
4480:
4479:Spinning cone
4477:
4475:
4472:
4470:
4467:
4465:
4462:
4460:
4457:
4455:
4454:Mixer-settler
4452:
4450:
4447:
4445:
4442:
4440:
4437:
4435:
4432:
4430:
4427:
4425:
4422:
4420:
4417:
4415:
4412:
4410:
4407:
4406:
4404:
4400:
4395:
4385:
4382:
4380:
4377:
4375:
4372:
4370:
4369:Sedimentation
4367:
4365:
4362:
4360:
4357:
4355:
4354:Precipitation
4352:
4350:
4347:
4345:
4342:
4340:
4337:
4335:
4332:
4330:
4327:
4325:
4322:
4320:
4317:
4315:
4312:
4310:
4307:
4305:
4302:
4300:
4297:
4295:
4292:
4290:
4287:
4285:
4282:
4280:
4277:
4275:
4272:
4270:
4267:
4265:
4262:
4260:
4257:
4255:
4252:
4250:
4247:
4245:
4242:
4240:
4237:
4235:
4232:
4231:
4229:
4225:
4221:
4214:
4209:
4207:
4202:
4200:
4195:
4194:
4191:
4179:
4176:
4174:
4171:
4169:
4166:
4164:
4161:
4159:
4156:
4154:
4151:
4149:
4146:
4144:
4141:
4139:
4136:
4134:
4131:
4130:
4128:
4124:
4118:
4115:
4113:
4110:
4108:
4105:
4103:
4100:
4098:
4095:
4094:
4092:
4088:
4082:
4081:Spinning cone
4079:
4077:
4074:
4072:
4069:
4067:
4064:
4063:
4061:
4057:
4052:
4042:
4039:
4037:
4034:
4032:
4029:
4027:
4024:
4022:
4019:
4017:
4014:
4012:
4009:
4007:
4004:
4003:
4001:
3997:
3993:
3986:
3981:
3979:
3974:
3972:
3967:
3966:
3963:
3944:
3940:
3938:
3935:
3933:
3930:
3929:
3925:
3920:
3919:0-521-08573-X
3916:
3912:
3911:
3906:
3902:
3896:
3892:
3887:
3883:
3879:
3875:
3871:
3867:
3863:
3858:
3857:
3853:
3845:
3841:
3837:
3833:
3826:
3823:
3818:
3814:
3810:
3806:
3802:
3798:
3791:
3788:
3784:
3778:
3775:
3771:
3767:
3764:
3759:
3756:
3751:
3745:
3741:
3734:
3731:
3726:
3720:
3716:
3715:
3707:
3704:
3699:
3695:
3688:
3685:
3680:
3676:
3671:
3666:
3662:
3658:
3654:
3650:
3646:
3639:
3636:
3631:
3627:
3620:
3617:
3612:
3608:
3604:
3597:
3594:
3589:
3585:
3581:
3574:
3571:
3567:
3563:
3560:
3555:
3552:
3548:
3543:
3540:
3535:
3529:
3525:
3524:
3516:
3514:
3512:
3510:
3506:
3503:
3499:
3494:
3491:
3478:
3474:
3468:
3465:
3455:
3451:
3445:
3442:
3439:
3435:
3432:
3421:
3417:
3411:
3408:
3405:
3404:0-306-40895-3
3401:
3397:
3391:
3389:
3385:
3381:
3377:
3374:
3369:
3366:
3361:
3357:
3353:
3347:
3344:
3339:
3335:
3331:
3327:
3320:
3317:
3312:
3311:
3306:
3300:
3297:
3284:
3280:
3274:
3271:
3266:
3262:
3261:
3256:
3250:
3247:
3241:
3235:
3231:
3230:
3225:
3219:
3216:
3210:
3208:9782881245947
3204:
3200:
3196:
3190:
3187:
3181:
3177:
3172:
3168:
3165:
3159:
3155:
3149:
3146:
3140:
3138:9780262082822
3134:
3130:
3126:
3122:
3118:
3112:
3109:
3106:pp. 106, 111.
3103:
3102:2078.1/211340
3099:
3095:
3091:
3087:
3079:
3075:
3071:
3067:
3063:
3059:
3051:
3047:
3043:
3041:9783487091150
3037:
3033:
3030:(1942–1943).
3029:
3023:
3020:
3016:
3012:
3006:
3002:
2998:
2991:
2989:
2987:
2983:
2980:
2979:9788131727911
2976:
2972:
2968:
2967:
2962:
2957:
2954:
2951:
2947:
2944:
2940:
2937:, New Series
2936:
2930:
2927:
2923:
2920:
2914:
2911:
2908:
2904:
2901:
2897:
2894:, New Series
2893:
2887:
2884:
2880:
2876:
2873:
2869:
2865:
2861:
2860:John Marshall
2856:
2853:
2848:
2844:
2840:
2836:
2829:
2826:
2821:
2817:
2813:
2809:
2802:
2799:
2794:
2790:
2786:
2782:
2778:
2774:
2770:
2766:
2762:
2758:
2752:
2748:
2744:
2743:
2738:
2732:
2730:
2726:
2722:
2721:
2716:
2711:
2708:
2703:
2697:
2693:
2688:
2687:
2678:
2675:
2670:
2669:
2661:
2658:
2653:
2647:
2643:
2639:
2638:
2630:
2627:
2622:
2621:
2620:Meteorologica
2613:
2610:
2605:
2599:
2595:
2591:
2590:
2582:
2579:
2574:
2568:
2564:
2560:
2556:
2555:
2547:
2544:
2539:
2535:
2531:
2527:
2523:
2516:
2513:
2509:
2505:
2501:
2500:
2492:
2489:
2486:
2482:
2479:
2473:
2470:
2467:
2463:
2460:
2454:
2451:
2445:
2442:
2437:
2426:
2422:
2418:
2414:
2410:
2406:
2402:
2398:
2394:
2388:
2383:
2382:
2373:
2371:
2369:
2365:
2359:
2355:
2352:
2350:
2347:
2345:
2342:
2340:
2337:
2335:
2332:
2330:
2327:
2325:
2322:
2320:
2317:
2316:
2312:
2306:
2298:
2293:
2289:
2285:
2278:
2273:
2270:
2263:
2258:
2254:
2247:
2242:
2238:
2234:
2227:
2222:
2217:
2215:
2212:
2208:
2204:
2199:
2191:
2186:
2184:
2177:
2174:
2171:
2168:
2167:
2166:
2164:
2160:
2152:
2150:
2148:
2143:
2137:
2134:
2133:mass transfer
2130:
2126:
2125:Raschig rings
2117:
2113:
2110:
2109:plate columns
2104:
2102:
2097:
2093:
2089:
2080:
2076:
2074:
2073:semiconductor
2071:for use as a
2070:
2066:
2065:chlorosilanes
2062:
2058:
2054:
2053:liquid oxygen
2050:
2047:
2042:
2040:
2036:
2031:
2022:
2018:
2015:
2011:
2007:
2001:
1997:
1995:
1991:
1987:
1986:petrochemical
1983:
1976:
1967:
1960:
1958:
1956:
1952:
1951:ethyl acetate
1947:
1943:
1941:
1935:
1927:
1925:
1922:
1918:
1914:
1911:
1904:
1902:
1900:
1895:
1893:
1889:
1885:
1883:
1879:
1875:
1871:
1867:
1862:
1860:
1855:
1854:
1849:
1843:
1835:
1829:
1826:
1822:
1818:
1814:
1810:
1809:mother liquor
1806:
1802:
1798:
1795:
1792:
1788:
1784:
1780:
1776:
1772:
1768:
1764:
1761:
1760:
1759:
1752:
1748:
1745:
1744:membrane pump
1741:
1737:
1733:
1729:
1728:
1727:
1725:
1716:
1713:
1709:
1706:
1702:
1698:
1695:
1692:
1688:
1685:
1682:
1678:
1677:Pervaporation
1675:
1672:
1669:
1666:
1665:sulfuric acid
1662:
1657:
1653:
1652:
1648:
1646:
1639:
1637:
1630:
1628:
1626:
1622:
1618:
1614:
1610:
1605:
1603:
1599:
1598:air sensitive
1591:
1589:
1587:
1575:
1572:
1569:
1566:
1563:
1560:
1559:
1555:
1550:
1542:
1540:
1537:
1533:
1529:
1525:
1521:
1514:
1512:
1510:
1505:
1503:
1496:
1488:
1480:
1477:
1474:
1471:
1468:
1465:
1462:
1459:
1456:
1453:
1450:
1447:
1446:
1442:
1427:
1423:
1419:
1417:
1413:
1409:
1405:
1401:
1397:
1393:
1392:essential oil
1389:
1384:
1381:
1377:
1371:
1363:
1361:
1359:
1355:
1350:
1348:
1344:
1343:
1338:
1333:
1330:
1324:
1316:
1314:
1312:
1307:
1303:
1299:
1295:
1291:
1286:
1282:
1280:
1270:
1263:
1261:
1259:
1255:
1251:
1247:
1241:
1239:
1235:
1231:
1227:
1223:
1222:
1213:
1208:
1205:
1202:
1201:Packed column
1199:
1196:
1193:
1191:
1190:Air condenser
1188:
1187:
1186:
1182:
1179:
1171:
1169:
1166:
1161:
1158:
1151:
1143:
1141:
1140:component B.
1137:
1133:
1125:
1118:
1116:
1114:
1110:
1105:
1103:
1099:
1095:
1091:
1087:
1082:
1078:
1075:
1069:
1067:
1063:
1062:mole fraction
1057:
1055:
1051:
1047:
1041:
1039:
1035:
1031:
1027:
1026:boiling point
1019:
1017:
1015:
1010:
1008:
1002:
1000:
996:
992:
988:
984:
976:
974:
972:
968:
964:
960:
956:
955:Ernest Thiele
952:
948:
944:
939:
937:
933:
932:Ernest Solvay
929:
925:
924:Aeneas Coffey
921:
917:
912:
908:
906:
902:
898:
894:
890:
886:
882:
881:Irish whiskey
878:
877:Scotch whisky
874:
870:
865:
861:
857:
853:
849:
845:
841:
834:
829:
821:
813:
807:
802:
796:
792:
787:
783:
781:
777:
772:
770:
766:
762:
760:
759:
752:
744:
742:
740:
736:
732:
728:
727:Southern Song
720:
718:
716:
712:
708:
706:
700:
698:
692:
690:
689:
682:
671:
660:
656:
651:
642:
638:
633:
627:
615:
611:
609:
603:
592:
588:
584:
578:
572:
564:
562:
560:
555:
553:
549:
545:
541:
537:
533:
529:
525:
521:
517:
513:
509:
501:
499:
497:
493:
489:
484: 200 CE
477:
475:
471:
467:
460:
456:
452:
447:
442:
438:
430:
428:
424:
420:
418:
411:
409:
407:
403:
399:
395:
389:
387:
380:
375:
368:
363:
361:
359:
355:
350:
341:
333:
328:
323:
320:
317:
314:— for use as
313:
309:
305:
301:
297:
294:
293:raw materials
291:and chemical
290:
286:
282:
279:
276:
272:
269:
266:
263:
260:
259:ethyl alcohol
256:
252:
251:
250:
248:
244:
240:
236:
233:is used as a
232:
227:
225:
220:
216:
212:
208:
204:
200:
196:
192:
188:
186:
182:
178:
174:
170:
166:
162:
155:
150:
149:boiling chips
146:
141:
136:
131:
126:
121:
116:
111:
106:
101:
96:
91:
86:
81:
76:
71:
67:
63:
59:
55:
53:
48:
46:
41:
37:
33:
19:
4559:Distillation
4439:Filter press
4424:Depth filter
4314:Flocculation
4284:Distillation
4283:
4178:Vacuum-based
4011:Dalton's law
4006:Raoult's law
3992:Distillation
3991:
3950:. Retrieved
3909:
3890:
3868:(1): 55–63.
3865:
3861:
3835:
3831:
3825:
3803:(11): 1011.
3800:
3796:
3790:
3782:
3777:
3758:
3739:
3733:
3713:
3706:
3697:
3687:
3655:(1): 25–31.
3652:
3648:
3638:
3629:
3619:
3613:(20): 64–65.
3610:
3606:
3596:
3590:(19): 24–26.
3587:
3583:
3573:
3554:
3542:
3522:
3493:
3481:. Retrieved
3476:
3467:
3444:
3410:
3395:
3368:
3360:the original
3355:
3346:
3329:
3325:
3319:
3309:
3305:French, John
3299:
3287:. Retrieved
3282:
3273:
3264:
3259:
3249:
3228:
3218:
3213:pp. 204–206.
3198:
3189:
3179:
3157:
3148:
3128:
3111:
3093:
3089:
3061:
3057:
3031:
3022:
3014:
3000:
2965:
2961:Habib, Irfan
2956:
2938:
2934:
2929:
2921:
2918:
2913:
2895:
2891:
2886:
2867:
2863:
2855:
2842:
2838:
2828:
2811:
2807:
2801:
2793:Google Books
2791:– via
2741:
2719:
2717:(1887–1888)
2710:
2685:
2677:
2667:
2660:
2636:
2629:
2619:
2612:
2588:
2581:
2553:
2546:
2529:
2525:
2515:
2507:
2498:
2491:
2472:
2453:
2444:
2434:– via
2428:. Retrieved
2380:
2203:Carbohydrate
2201:
2181:
2156:
2138:
2122:
2105:
2085:
2051:, producing
2043:
2027:
2009:
2002:
1998:
1978:
1948:
1944:
1937:
1923:
1919:
1915:
1912:
1908:
1896:
1886:
1865:
1863:
1851:
1848:Raoult's law
1845:
1791:wood alcohol
1790:
1758:Other uses:
1757:
1721:
1701:vaporization
1643:
1634:
1606:
1595:
1582:
1518:
1506:
1498:
1478:Teflon tap 3
1472:Teflon tap 2
1460:Teflon tap 1
1408:aromatherapy
1404:aromatherapy
1385:
1373:
1351:
1346:
1340:
1334:
1329:Raoult's law
1326:
1298:silicone oil
1287:
1283:
1279:Raoult's law
1275:
1250:drying tubes
1242:
1225:
1219:
1217:
1183:
1175:
1165:steady state
1162:
1153:
1138:
1134:
1130:
1106:
1083:
1079:
1070:
1058:
1050:Dalton's law
1046:Raoult's law
1042:
1023:
1011:
1003:
980:
977:Applications
940:
920:patent still
913:
909:
837:
815:Distillation
790:
779:
773:
764:
754:
748:
739:Yuan dynasty
724:
702:
694:
684:
652:
580:
556:
505:
478:
463:
458:
426:
422:
415:
397:
391:
386:distillation
385:
383:
377:
372:
347: 1200
337:
285:oil refinery
275:hydrocarbons
265:Desalination
231:distillation
230:
228:
206:
189:
181:condensation
164:
161:Distillation
160:
159:
153:
144:
139:
134:
129:
124:
119:
114:
109:
104:
99:
94:
89:
84:
79:
74:
69:
62:
51:
44:
4414:Belt filter
4379:Sublimation
4269:Decantation
4173:Steam-based
4168:Salt-effect
4143:Destructive
3838:(9): 1845.
3549:. fandm.edu
3500:, DIDAC by
3283:Bored Panda
3028:Kraus, Paul
2349:Dixon rings
2344:Sublimation
1724:evaporation
1649:Other types
1528:high vacuum
1463:Cold finger
1246:vacuum pump
922:. In 1830,
901:bootleggers
895:, and some
776:John French
705:aqua ardens
641:Roger Bacon
559:Eastern Han
474:Roman Egypt
468:working in
457:manuscript
453:, from the
396:, the word
358:Mesopotamia
356:of ancient
354:Babylonians
253:Distilling
219:hydrocarbon
195:thermolysis
4553:Categories
4503:Multiphase
4434:Evaporator
4419:Centrifuge
4309:Filtration
4304:Extraction
4244:Adsorption
4234:Absorption
4158:Fractional
4153:Extractive
4133:Azeotropic
4126:Techniques
3999:Principles
3420:Coffey, A.
3244:pp. 51–52.
3096:: 87–141.
3090:Micrologus
2814:(3): 185.
2789:Q107312970
2773:1060799375
2425:Q107313989
2360:References
2096:simulation
1888:Immiscible
1872:, such as
869:pot stills
860:condensers
833:East Timor
778:published
753:published
745:Modern era
681:al-Zahrāwī
575:See also:
540:Common Era
532:Rang Mahal
470:Alexandria
466:alchemists
435:See also:
398:distillare
298:Cryogenic
169:separating
4517:Azeotrope
4227:Processes
4138:Catalytic
4102:Kugelrohr
3698:US Patent
3483:27 August
3450:US 198699
3078:143006568
3050:468740510
2973:. p. 55.
2943:full text
2900:full text
2845:: 85–94.
2781:13686623M
2192:Beverages
2046:cryogenic
2010:fractions
2006:crude oil
1870:desiccant
1853:azeotrope
1825:applejack
1775:pyrolysis
1773:known as
1691:azeotrope
1451:Still pot
1416:skin care
1296:13 via a
1094:azeotrope
999:volatiles
947:petroleum
928:archetype
856:glassware
844:chemistry
774:In 1651,
670:al-Fārābī
668:) and to
581:Medieval
524:Charsadda
417:Aristotle
412:Aristotle
255:fermented
199:pyrolysis
54:(journal)
4531:Concepts
4522:Eutectic
4474:Scrubber
4449:Leachate
4329:Leaching
4274:Dialysis
4163:Reactive
3766:Archived
3679:34092997
3562:Archived
3434:Archived
3376:Archived
3307:(1651).
3257:(1500).
3226:(1957).
3197:(1966).
3167:Archived
3119:(2000).
3082:p. 280;
2963:(2011),
2946:Archived
2903:Archived
2875:Archived
2847:Archived
2785:Wikidata
2765:71879886
2504:Elsevier
2481:Archived
2462:Archived
2421:Wikidata
2417:2032038M
2409:18816225
2401:88007680
2313:See also
1996:plants.
1817:ice wine
1813:ice beer
1801:freezing
1787:methanol
1785:to give
1779:reducing
1736:solvents
1681:membrane
1621:nitrogen
1388:aromatic
1290:hotplate
1258:nitrogen
1254:bubblers
1157:volatile
957:and the
864:condense
852:alembics
659:al-Kindī
637:Avicenna
585:such as
548:Gandhara
528:Pakistan
340:Akkadian
334:Iron Age
308:nitrogen
4505:systems
4402:Devices
4349:Osmosis
4097:Alembic
3882:2801640
3805:Bibcode
3670:8174813
3416:GB 5974
3289:28 July
3229:Alchemy
3173:); cf.
2430:22 June
2324:Clyssus
2253:toluene
2233:retorts
2218:Gallery
2207:whiskey
2090:or the
2069:silicon
1955:ethanol
1892:toluene
1859:ethanol
1209:system.
1090:toluene
1086:benzene
1074:boiling
993:), and
936:ammonia
893:cachaça
885:tequila
848:retorts
840:alchemy
512:retorts
486:, when
379:manner.
329:History
247:alcohol
213:and of
177:boiling
173:mixture
163:, also
52:Distill
47:(album)
45:Distill
4289:Drying
4016:Reflux
3917:
3897:
3880:
3746:
3721:
3677:
3667:
3530:
3460:
3452:,
3426:
3418:,
3402:
3236:
3205:
3143:p. 44.
3135:
3076:
3048:
3038:
3007:
2977:
2881:, 1951
2864:Taxila
2787:
2779:
2771:
2763:
2753:
2698:
2648:
2600:
2569:
2423:
2415:
2407:
2399:
2389:
2288:vacuum
2030:reflux
1617:reflux
1609:Teflon
1434:
1430:
1398:. The
1358:Teflon
916:reflux
897:vodkas
873:cognac
806:retort
552:liquor
522:, and
516:Taxila
494:under
439:, and
402:Seneca
310:, and
304:oxygen
49:,
42:,
38:,
34:,
4484:Still
4117:Still
3952:5 May
3878:JSTOR
3502:IUPAC
3431:image
3263:[
3123:. In
3074:S2CID
2843:XLIII
2642:BRILL
2594:BRILL
2061:argon
1821:sugar
1749:In a
1742:or a
1705:valve
1625:argon
1613:still
1374:Like
1234:state
1230:vapor
1221:still
735:Hebei
536:India
312:argon
289:fuels
203:fluid
185:still
3954:2007
3915:ISBN
3895:ISBN
3744:ISBN
3719:ISBN
3675:PMID
3528:ISBN
3485:2019
3400:ISBN
3291:2024
3234:ISBN
3203:ISBN
3133:ISBN
3046:OCLC
3036:ISBN
3005:ISBN
2975:ISBN
2769:OCLC
2761:LCCN
2751:ISBN
2696:ISBN
2646:ISBN
2598:ISBN
2567:ISBN
2432:2021
2405:OCLC
2397:LCCN
2387:ISBN
2209:and
1992:and
1988:and
1815:and
1783:wood
1661:lime
1524:torr
1414:and
1292:and
1088:and
1048:and
1024:The
711:salt
655:wine
530:and
404:and
197:and
4148:Dry
3870:doi
3862:Man
3840:doi
3813:doi
3665:PMC
3657:doi
3334:doi
3098:hdl
3066:doi
2935:Man
2892:Man
2872:420
2816:doi
2559:doi
2534:doi
2235:as
2211:rum
2127:or
1765:or
1730:In
1623:or
1436:°C.
1240:).
1226:pot
1012:In
953:by
889:rum
862:to
838:As
731:Jin
650:).
534:in
526:in
472:in
349:BCE
154:16:
145:15:
140:14:
135:13:
130:12:
125:11:
120:10:
4555::
3876:.
3866:14
3864:.
3836:26
3834:.
3811:.
3801:45
3799:.
3696:.
3673:.
3663:.
3653:24
3651:.
3647:.
3609:.
3605:.
3508:^
3475:.
3429:;
3398:.
3387:^
3354:.
3330:28
3328:.
3281:.
3094:28
3092:.
3088:.
3072:.
3062:14
3060:.
3044:.
3013:.
2999:.
2985:^
2969:.
2939:14
2922:41
2896:14
2866:,
2862:,
2841:.
2837:.
2810:.
2783:.
2777:OL
2775:.
2767:.
2759:.
2749:.
2728:^
2694:.
2692:88
2640:.
2592:.
2565:.
2530:13
2528:.
2524:.
2502:.
2419:.
2413:OL
2411:.
2403:.
2395:.
2367:^
2282:A
2075:.
2055:,
1984:,
1957:.
1942:.
1880:.
1504:.
1418:.
1410:,
1313:.
1281:.
1040:.
985:,
907:.
891:,
887:,
883:,
879:,
875:,
804:A
674:c.
663:c.
645:c.
618:c.
595:c.
542:.
518:,
481:c.
360:.
344:c.
306:,
187:.
115:9:
110:8:
105:7:
100:6:
95:5:
90:4:
85:3:
80:2:
75:1:
4212:e
4205:t
4198:v
3984:e
3977:t
3970:v
3956:.
3921:.
3903:.
3884:.
3872::
3846:.
3842::
3819:.
3815::
3807::
3752:.
3727:.
3681:.
3659::
3611:1
3588:6
3536:.
3487:.
3340:.
3336::
3293:.
3242:.
3211:.
3141:.
3104:.
3100::
3080:.
3068::
3052:.
2870::
2868:2
2822:.
2818::
2812:5
2795:.
2704:.
2654:.
2606:.
2575:.
2561::
2540:.
2536::
2438:.
2290:.
2255:.
1793:.
1746:.
1683:.
763:(
672:(
661:(
643:(
616:(
318:.
277:.
261:.
193:(
60:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.