869:
molecule cannot be represented by one structure, but rather a resonance hybrid of different structures, such as with the two resonance structures of benzene. These molecules cannot be found in either one of these representations, with the longer single bonds in one location and the shorter double bond in another (See Theory below). Rather, the molecule exhibits bond lengths in between those of single and double bonds. This commonly seen model of aromatic rings, namely the idea that benzene was formed from a six-membered carbon ring with alternating single and double bonds (cyclohexatriene), was developed by August Kekulé (see
History section below). The model for benzene consists of two resonance forms, which corresponds to the double and single bonds superimposing to produce six one-and-a-half bonds. Benzene is a more stable molecule than would be expected without accounting for charge delocalization.
1408:
114:
1453:
1553:
1270:
1206:
1183:
1484:
88:
149:
1255:
1315:
1389:
1225:
1431:
1519:
1285:
1353:
1338:
1500:
680:
899:
1164:
1240:
1538:
561:, because of the interactions depicted by the arcs shown). Medium rings (8-11 atoms) are the most strained, with between 9-13 (kcal/mol) strain energy, and analysis of factors important in the conformations of larger macrocycles can be modeled using medium ring conformations. Conformational analysis of odd-membered rings suggests they tend to reside in less symmetrical forms with smaller energy differences between stable conformations.
565:
1300:
1452:
113:
1407:
865:
property and the olfactory properties of such compounds (how they smell), although in 1855, before the structure of benzene or organic compounds was understood, chemists like
Hofmann were beginning to understand that odiferous molecules from plants, such as terpenes, had chemical properties we recognize today are similar to unsaturated petroleum hydrocarbons like benzene.
1552:
549:). Heterocycles with carbon in the rings may have limited non-carbon atoms in their rings (e.g., in lactones and lactams whose rings are rich in carbon but have limited number of non-carbon atoms), or be rich in non-carbon atoms and displaying significant symmetry (e.g., in the case of chelating macrocycles). Macrocycles can access a number of stable
792:
861:
benzene derivatives, and this is how it was first defined. Nevertheless, many non-benzene aromatic compounds exist. In living organisms, for example, the most common aromatic rings are the double-ringed bases in RNA and DNA. A functional group or other substituent that is aromatic is called an aryl group.
868:
In terms of the electronic nature of the molecule, aromaticity describes a conjugated system often made of alternating single and double bonds in a ring. This configuration allows for the electrons in the molecule's pi system to be delocalized around the ring, increasing the molecule's stability. The
856:
is non-aromatic. In organic chemistry, the term aromaticity is used to describe a cyclic (ring-shaped), planar (flat) molecule that exhibits unusual stability as compared to other geometric or connective arrangements of the same set of atoms. As a result of their stability, it is very difficult to
864:
The earliest use of the term “aromatic” was in an article by August
Wilhelm Hofmann in 1855. Hofmann used the term for a class of benzene compounds, many of which do have odors (aromas), unlike pure saturated hydrocarbons. Today, there is no general relationship between aromaticity as a chemical
860:
Since one of the most commonly encountered aromatic systems of compounds in organic chemistry is based on derivatives of the prototypical aromatic compound benzene (an aromatic hydrocarbon common in petroleum and its distillates), the word “aromatic” is occasionally used to refer informally to
148:
87:
523:
777:
stereochemistry projects both of these groups toward the same side of the ring. Hence, if forced into the higher energy boat form, these methyl groups are in steric contact, repel one another, and drive the equilibrium toward the chair conformation.
1182:
857:
cause aromatic molecules to break apart and to react with other substances. Organic compounds that are not aromatic are classified as aliphatic compounds—they might be cyclic, but only aromatic rings have especial stability (low reactivity).
1352:
1388:
1314:
194:
is displayed. Indeed, the development of this important chemical concept arose historically in reference to cyclic compounds. Finally, cyclic compounds, because of the unique shapes, reactivities, properties, and
1483:
1518:
2046:
László Kürti & Barbara Czakó, 2005, "Strategic
Applications of Named Reactions in Organic Synthesis: Background and Detailed Mechanisms, Amsterdam, NH, NLD:Elsevier Academic Press, 2005ISBN 0124297854, see
749:
chair conformations predominate in cyclohexanes bearing one or more substituents depends on the substituents, and where they are located on the ring; generally, "bulky" substituents—those groups with large
910:
organic reactions that historically have been crucial in the development, first, of understanding the concepts of ring chemistry, and second, of reliable procedures for preparing ring structures in high
1499:
1269:
1430:
1205:
215:
in which at least some its atoms are connected to form a ring. Rings vary in size from three to many tens or even hundreds of atoms. Examples of ring compounds readily include cases where:
2004:
Francis A. Carey & Richard J. Sundberg, 2006, "Title
Advanced Organic Chemistry: Part A: Structure and Mechanisms," 4th Edn., New York, NY, USA:Springer Science & Business Media,
636:
is formally a polycyclic compound, but is more specifically named as a bicyclic compound. Several examples of macrocyclic and polycyclic structures are given in the final gallery below.
1254:
1224:
1163:
280:); As well, depending on ring size, the three-dimensional shapes of particular cyclic structures — typically rings of five atoms and larger — can vary and interconvert such that
190:). As well, depending on ring size, the three-dimensional shapes of particular cyclic structures – typically rings of five atoms and larger – can vary and interconvert such that
819:
1465:
with a tricyclic core: with a heterocyclic, 4-membered D ring, fused to further 6- and 8-membered carbocyclic (A/C and B) rings (non-aromatic), and with three further pendant
906:
There are a variety of specialized reactions whose use is solely the formation of rings, and these will be discussed below. In addition to those, there are a wide variety of
1284:
617:
1337:
832:
for details of when such copying and pasting is acceptable and when it is not, and how to correctly attribute using links in the edit summaries. You can also read the "
74:
of common atoms and their ability to form rings, the number of possible cyclic structures, even of small size (e.g., < 17 total atoms) numbers in the many billions.
1990:
Michael B. Smith & Jerry March, 2007, "March's
Advanced Organic Chemistry: Reactions, Mechanisms, and Structure," 6th Ed., New York, NY, USA:Wiley & Sons,
1537:
710:
Depending on ring size, the three-dimensional shapes of particular cyclic structures—typically rings of 5-atoms and larger—can vary and interconvert such that
1239:
1084:
A variety of further synthetic procedures are particularly useful in opening carbocyclic and other rings, generally which contain a double bound or other
1753:
885:, and in the man-made molecules (e.g., drugs, herbicides, etc.) through which man attempts to exert control over nature and biological systems.
1096:
296:, and of these, a significant and conceptually important portion are composed of rings made only of carbon atoms (i.e., they are carbocycles).
257:
possible in cyclic structures, the number of possible cyclic structures, even of small size (e.g., <17 atoms) numbers in the many billions.
714:
is displayed. Indeed, the development of this important chemical concept arose, historically, in reference to cyclic compounds. For instance,
514:
in the rings). Hantzsch–Widman nomenclature is recommended by the IUPAC for naming heterocycles, but many common names remain in regular use.
1299:
142:
shorthand is used where hydrogen atoms are inferred as present to fill the carbon's valence of 4 (rather than their being shown explicitly).
1859:
Anet, F.A.L.; St. Jacques, M.; Henrichs, P.M.; Cheng, A.K.; Krane, J.; Wong, L. (1974). "Conformational analysis of medium-ring ketones".
161:, also a natural product, displaying a complex multi-ring structure including 4-, 6-, and 8-membered rings (carbocyclic and heterocyclic,
1903:
1113:
881:
that they engender, cyclic compounds are the largest majority of all molecules involved in the biochemistry, structure, and function of
533:
is used for compounds having a rings of 8 or more atoms. Macrocycles may be fully carbocyclic (rings containing only carbon atoms, e.g.
70:
to having varying numbers of multiple bonds between the ring atoms. Because of the tremendous diversity allowed, in combination, by the
67:
948:
In organic chemistry, a variety of synthetic procures are particularly useful in closing carbocyclic and other rings; these are termed
722:
with no double bonds, to which various substituents might be attached, see image—display an equilibrium between two conformations, the
1976:
Jürgen-Hinrich
Fuhrhop & Gustav Penzlin, 1986, "Organic synthesis: concepts, methods, starting materials," Weinheim, BW, DEU:VCH,
1897:
1631:
620:
nomenclature has extensive rules to cover the naming of cyclic structures, both as core structures, and as substituents appended to
62:
of the individual links between ring atoms, and their arrangements within the rings, carbocyclic and heterocyclic compounds may be
557:
nonbonded interactions within the ring (e.g., with the chair and chair-boat being more stable than the boat-boat conformation for
1424:
possible for "fusing" the two rings together, and how this impacts the shapes available to this bicyclic compound (non-aromatic).
829:
2037:
2023:
2009:
1995:
1981:
1127:
2032:
Jonathan
Clayden, Nick Greeves & Stuart Warren, 2012, "Organic Chemistry," Oxford, Oxon, GBR:Oxford University Press,
773:-1,4-dimethylcyclohexane. In this molecule, the two methyl groups are in opposing positions of the ring (1,4-), and their
1021:
993:
241:) form varying numbers of bonds, and many common atoms readily form rings. In addition, depending on the ring size, the
107:
that follows, which displays a complex ring structure including 3-, 5-, and 7-membered non-aromatic, carbocyclic rings.
2095:
1100:
1014:
758:
199:
that they engender, are the majority of all molecules involved in the biochemistry, structure, and function of living
54:), none of the atoms are carbon (inorganic cyclic compounds), or where both carbon and non-carbon atoms are present (
1260:
50:. Rings may vary in size from three to many atoms, and include examples where all the atoms are carbon (i.e., are
2090:
1379:
The following are examples of cyclic compounds exhibiting more complex ring systems and stereochemical features:
568:
Chelating macrocyclic structures of interest in inorganic and supramolecular chemistry, an example array. A, the
974:
2074:
2064:
1953:
711:
550:
334:
281:
191:
187:
1744:
1154:
The following are examples of simple and aromatic carbocycles, inorganic cyclic compounds, and heterocycles:
245:
of the individual links between ring atoms, and their arrangements within the rings, cyclic compounds may be
1158:
Simple mono-cyclic compounds: Carbocyclic, inorganic, and heterocyclic (aromatic and non-aromatic) examples.
1131:
1010:
254:
170:
Adding to their complexity and number, closing of atoms into rings may lock particular atoms with distinct
1230:
936:
761:—prefer to occupy an equatorial location. An example of interactions within a molecule that would lead to
546:
324:
1797:
Still, W.Clark; Galynker, Igor (1981). "Chemical consequences of conformation in macrocyclic compounds".
1126:. Ring expansions and contractions can involve the insertion of a functional group such as the case with
545:
containing rings of 8 or more atoms), or non-carbon (containing only non-carbon atoms in the rings, e.g.
1566:
1275:
370:
253:
to having varying numbers of multiple bonds. As a consequence of the constitutional variability that is
55:
1134:
956:
923:
2018:
Michael B. Smith, 2011, "Organic
Chemistry: An Acid—Base Approach," Boca Raton, FL, USA:CRC Press,
1598:
1123:
1004:
963:
679:
629:
486:
as members of its ring(s). Cyclic compounds that have both carbon and non-carbon atoms present are
238:
196:
71:
276:
being associated with the compound, including some manifestations that are unique to rings (e.g.,
2070:
1671:
1588:
815:
250:
51:
2060:
898:
2033:
2019:
2005:
1991:
1977:
1945:
1893:
1887:
1712:
1637:
1627:
1621:
1138:
1119:
665:
597:
415:
313:
277:
249:
or non-aromatic; in the case of non-aromatic cyclic compounds, they may vary from being fully
246:
212:
139:
63:
39:
1937:
1868:
1806:
1702:
1663:
1245:
1085:
1051:
929:
752:
738:
483:
352:
293:
261:
175:
47:
1573:. The R group at right refers to one of several possible four-carbon side chains (see main
765:, leading to a shift in equilibrium from boat to chair, is the interaction between the two
1777:
1562:
1462:
1443:
1421:
1328:
1173:
916:
882:
825:
657:
605:
522:
407:
269:
179:
100:
79:
1397:, technically a polycyclic, more specifically a bicyclic compound, with circles showing
564:
1524:
1398:
997:
912:
733:
The chair conformation is the favored configuration, because in this conformation, the
459:
186:
of the compound results, including some manifestations that are unique to rings (e.g.,
1872:
1810:
482:
A heterocyclic compound is a cyclic compound that has atoms of at least two different
2084:
1069:
1055:
986:
833:
762:
734:
463:
388:
1675:
1169:
766:
742:
554:
490:
carbon compounds, and the name refers to inorganic cyclic compounds as well (e.g.,
487:
230:
131:
123:
58:
with rings containing both carbon and non-carbon). Depending on the ring size, the
2048:
2041:
2027:
2013:
1999:
1985:
628:
is used when a ring-containing compound has a ring of 12 or more atoms. The term
1617:
1528:
1505:
1436:
1417:
1394:
1366:
1343:
1196:
1065:
878:
853:
845:
810:
715:
686:
653:
633:
569:
558:
534:
471:
467:
455:
451:
265:
171:
162:
135:
127:
119:
664:
of the compound, including some manifestations that are unique to rings (e.g.,
1574:
1558:
1490:
1458:
1215:
719:
625:
530:
366:
242:
220:
154:
104:
59:
1641:
1623:
Advanced
Organic Chemistry: Reactions, Mechanisms, and Structure, 3rd edition
1320:
1305:
1059:
814:
due to copying and pasting between articles. This may be a violation of the
661:
621:
581:
273:
183:
46:
in which one or more series of atoms in the compound is connected to form a
43:
1949:
1941:
1716:
1546:, a complex, highly symmetric, man-made polycyclic compound (non-aromatic).
17:
822:
if proper attribution was not made in an edit summary or on the talk page.
1570:
1543:
1358:
1324:
1290:
1073:
1029:
1025:
967:
573:
511:
503:
491:
437:
309:
200:
1780:
652:
The closing of atoms into rings may lock particular atoms with distinct
639:
The atoms that are part of the ring structure are called annular atoms.
1667:
1593:
1509:
1440:
1413:
1362:
1192:
1188:
849:
601:
585:
538:
495:
450:). When carbon in benzene is "replaced" by other elements, e.g., as in
348:
158:
97:
93:
1707:
1690:
1569:, and a toxic component associated with the organisms responsible for
791:
1466:
982:
577:
542:
499:
384:
305:
1925:
78:
Cyclic compound examples: All-carbon (carbocyclic) and more complex
66:
or non-aromatic; in the latter case, they may vary from being fully
1654:
Halduc, I. (1961). "Classification of inorganic cyclic compounds".
1130:
of cyclic ketones, rearrangements of cyclic carbocycles as seen in
1013:
reactions, which also can be used to accomplish a specific type of
915:, and with defined orientation of ring substituents (i.e., defined
226:
none of the atoms are carbon (inorganic cyclic compounds), or where
978:
897:
697:
at right. Axial and equatorial hydrogen atoms are denoted with an
593:
589:
563:
537:), heterocyclic containing both carbon and non-carbon atoms (e.g.
507:
433:
411:
1293:, a 6-membered inorganic heterocyclic compound (may be aromatic).
604:/imine Jäger macrocycle, and F, the tetracarboxylate-derivative
304:
Inorganic atoms form cyclic compounds as well. Examples include
1233:, an 8-membered inorganic heterocyclic compound (non-aromatic).
785:
632:
is used when more than one ring appears in a single molecule.
260:
Moreover, the closing of atoms into rings may lock particular
1383:
Complex cyclic compounds: Macrocyclic and polycyclic examples
1346:, a 7-membered heterocyclic organic compound (non-aromatic).
1263:, a 6-membered organic heterocyclic compound (non-aromatic).
877:
Because of the unique shapes, reactivities, properties, and
790:
521:
233:
compounds with rings containing both carbon and non-carbon).
203:, and in man-made molecules such as drugs, pesticides, etc.
553:, with preference to reside in conformations that minimize
1416:(decahydronaphthalene), the fully saturated derivative of
1278:, a 6-membered inorganic heterocyclic compound (aromatic).
1099:, which can also be used to accomplish a specific type of
1827:
J. D. Dunitz (1968). J. D. Dunitz and J. A. Ibers (ed.).
1218:, an 8-membered inorganic cyclic compound (non-aromatic).
1527:, a pentacyclic compound both natural and man-made, and
1469:-rings on its "tail", and attached to C-2 (abbrev. Ph, C
1446:, and an example of a tricyclic molecule (non-aromatic).
1248:, a 5-membered inorganic cyclic compound (non-aromatic).
745:
that are otherwise possible are minimized. Which of the
1118:
Ring expansion and contraction reactions are common in
803:
1926:"Refined structure of αβ-tubulin at 3.5 Å resolution"
1508:, another terpene natural product, in particular, a
1489:
A representative three-dimensional shape adopted by
1308:, a 5-membered inorganic cyclic compound (aromatic).
852:
is an example of an aromatic cyclic compound, while
1889:
Academic Press Dictionary of Science and Technology
1088:"handle" to facilitate chemistry; these are termed
836:" guideline for an overview of the issues involved.
1512:, a class of tetracyclic molecules (non-aromatic).
808:Some of the article text's edit history exists at
1924:Löwe, J; Li, H; Downing, K.H; Nogales, E (2001).
1331:hydrogen atoms implied, not shown (non-aromatic).
1886:Morris, Christopher G.; Press, Academic (1992).
757:or groups that are otherwise repulsive in their
1822:
1820:
1531:π-electrons shown as discrete bonds (aromatic).
1369:π-electrons shown as discrete bonds (aromatic).
1831:. Vol. 2. New York: Wiley. pp. 1–70.
229:both carbon and non-carbon atoms are present (
1892:. Gulf Professional Publishing. p. 120.
1493:, as a result of its unique cyclic structure.
1191:, a 6-membered carbocyclic organic compound.
1108:Ring expansion and ring contraction reactions
894:Important general reactions for forming rings
656:by functional groups such that the result is
8:
1792:
1790:
1172:, a simple 7-membered carbocyclic compound,
1841:Eliel, E.L., Wilen, S.H. and Mander, L.S. (
1738:
1736:
1734:
1732:
1730:
1728:
1726:
1195:hydrogens shown, and 6 electrons shown as
292:The vast majority of cyclic compounds are
2073:at the U.S. National Library of Medicine
2063:at the U.S. National Library of Medicine
1706:
1042:
1035:
996:, originally being the cyclization of a
844:Cyclic compounds may or may not exhibit
446:
442:
428:
424:
420:
402:
379:
375:
361:
357:
343:
339:
329:
318:
211:A cyclic compound or ring compound is a
1773:
1771:
1609:
1381:
1365:hydrogen atoms implied, not shown, and
1156:
572:, 18-crown-6; B, the simple tetra-aza
474:are also known and well-characterized.
237:Common atoms can (as a result of their
76:
1361:, a 6 membered heterocyclic compound,
122:, the simplest carbocycles, including
1847:Stereochemistry of Organic Compounds,
1748:Virtual Textbook of Organic Chemistry
1199:through drawing of circle (aromatic).
7:
1849:John Wiley and Sons, Inc., New York.
1829:Perspectives in Structural Chemistry
1122:, and are frequently encountered in
919:). These general reactions include:
219:all the atoms are carbon (i.e., are
27:Molecule with a ring of bonded atoms
1565:with ten rings, all fused, and all
1114:Ring expansion and ring contraction
472:aromatic inorganic cyclic compounds
1756:from the original on 10 March 2015
1137:, or collapse or rearrangement of
470:, aromaticity is retained, and so
103:, related to but simpler than the
25:
157:, another complex, plant-derived
1551:
1536:
1517:
1498:
1482:
1451:
1429:
1406:
1387:
1351:
1336:
1313:
1298:
1283:
1268:
1253:
1238:
1223:
1204:
1181:
1162:
830:Knowledge:How to break up a page
678:
147:
112:
86:
1956:from the original on 2021-01-22
1906:from the original on 2021-04-13
1656:Journal of Structural Chemistry
1375:Complex and polycyclic examples
1176:hydrogens shown (non-aromatic).
902:Dieckmann ring-closing reaction
268:atoms into place, resulting in
1:
1873:10.1016/S0040-4020(01)90685-4
1811:10.1016/S0040-4020(01)93273-9
1752:. Michigan State University.
1695:Accounts of Chemical Research
1327:(aza) heterocyclic compound,
939:as applied to ring formation.
1930:Journal of Molecular Biology
1691:"The Chemical Space Project"
1689:Reymond, Jean-Louis (2015).
1150:Simple, mono-cyclic examples
1072:, as in the biosynthesis of
1064:other reactions, such as an
1022:Ruzicka large ring synthesis
994:Nazarov cyclization reaction
436:), boron and nitrogen (e.g.
207:Structure and classification
2112:
2029:, accessed 19 June 2015.
1746:"Stereoisomers Part I" in
1401:of π-electrons (aromatic).
1261:Hexamethylcyclotrisiloxane
1111:
300:Inorganic cyclic compounds
1128:Baeyer–Villiger oxidation
1028:groups combine to form a
2075:Medical Subject Headings
2065:Medical Subject Headings
2050:, accessed 19 June 2015.
2043:, accessed 19 June 2015.
2015:, accessed 19 June 2015.
2001:, accessed 19 June 2015.
1987:, accessed 19 June 2015.
834:copying within Knowledge
712:conformational isomerism
672:Conformational isomerism
335:tetrasulfur tetranitride
282:conformational isomerism
192:conformational isomerism
1743:William Reusch (2010).
1577:article; non-aromatic).
1097:ring opening metathesis
1011:ring-closing metathesis
977:, between a conjugated
730:as shown in the image.
666:configurational isomers
278:configurational isomers
188:configurational isomers
1942:10.1006/jmbi.2001.5077
1782:heterocyclic compounds
1231:Diselenium hexasulfide
1090:ring-opening reactions
1080:Ring-opening reactions
950:ring-closing reactions
944:Ring-closing reactions
937:Dieckmann condensation
903:
795:
609:
547:diselenium hexasulfide
526:
478:Heterocyclic compounds
325:trithiazyl trichloride
56:heterocyclic compounds
2071:Macrocyclic+Compounds
1276:Hexachlorophosphazene
1141:as several examples.
1135:Diels-Alder reactions
901:
794:
624:structures. The term
567:
525:
506:, which contain only
494:, which contain only
371:hexachlorophosphazene
138:. Note, elsewhere an
2061:Polycyclic+Compounds
1124:pericyclic reactions
1092:. Examples include:
1005:radical cyclizations
957:alkyne trimerisation
952:. Examples include:
924:Acyloin condensation
584:, the unsubstituted
369:and nitrogen (e.g.,
1626:, New York: Wiley,
1599:Open-chain compound
1032:group with loss of
964:Bergman cyclization
889:Synthetic reactions
2096:Molecular geometry
1668:10.1007/BF01141802
1589:Effective molarity
1420:, showing the two
1139:bicyclic compounds
1054:converting a beta
981:and a substituted
904:
796:
685:Two conformers of
610:
600:; E, the related
527:
502:in the rings, and
414:and oxygen (e.g.,
383:), phosphorus and
165:and non-aromatic).
38:) is a term for a
1708:10.1021/ar500432k
1422:stereochemistries
1120:organic synthesis
930:Anodic oxidations
842:
841:
693:at left, and the
598:Curtis macrocycle
580:; C, an example
416:sodium metaborate
406:and other cyclic
314:heptasulfur imide
255:thermodynamically
176:functional groups
140:organic chemistry
16:(Redirected from
2103:
2091:Cyclic compounds
1965:
1964:
1962:
1961:
1921:
1915:
1914:
1912:
1911:
1883:
1877:
1876:
1856:
1850:
1839:
1833:
1832:
1824:
1815:
1814:
1794:
1785:
1775:
1766:
1765:
1763:
1761:
1740:
1721:
1720:
1710:
1686:
1680:
1679:
1651:
1645:
1644:
1614:
1555:
1540:
1521:
1502:
1486:
1455:
1433:
1410:
1391:
1355:
1340:
1317:
1302:
1287:
1272:
1257:
1246:Cyclopentasilane
1242:
1227:
1208:
1185:
1166:
1086:functional group
1068:reacting with a
1052:Wenker synthesis
1046:
1038:
883:living organisms
837:
806:is not complete.
786:
739:eclipsing strain
682:
449:
431:
405:
400:
399:
396:
382:
364:
353:cyclopentasilane
346:
332:
322:
262:functional group
151:
116:
90:
82:cyclic compounds
42:in the field of
21:
2111:
2110:
2106:
2105:
2104:
2102:
2101:
2100:
2081:
2080:
2057:
1973:
1971:Further reading
1968:
1959:
1957:
1923:
1922:
1918:
1909:
1907:
1900:
1885:
1884:
1880:
1867:(12): 1629–37.
1858:
1857:
1853:
1840:
1836:
1826:
1825:
1818:
1805:(23): 3981–96.
1796:
1795:
1788:
1778:IUPAC Gold Book
1776:
1769:
1759:
1757:
1742:
1741:
1724:
1688:
1687:
1683:
1653:
1652:
1648:
1634:
1616:
1615:
1611:
1607:
1585:
1578:
1563:natural product
1556:
1547:
1541:
1532:
1522:
1513:
1503:
1494:
1487:
1478:
1476:
1472:
1463:natural product
1461:, a polycyclic
1456:
1447:
1444:natural product
1434:
1425:
1411:
1402:
1392:
1377:
1370:
1356:
1347:
1341:
1332:
1323:, a 4-membered
1318:
1309:
1303:
1294:
1288:
1279:
1273:
1264:
1258:
1249:
1243:
1234:
1228:
1219:
1209:
1200:
1186:
1177:
1167:
1152:
1147:
1116:
1110:
1082:
1044:
1040:
1037:
1033:
1024:, in which two
946:
917:stereochemistry
896:
891:
875:
838:
826:Knowledge:Merge
823:
802:This article's
797:
784:
708:
707:
706:
705:, respectively.
683:
674:
658:stereochemistry
650:
648:Stereochemistry
645:
615:
520:
480:
448:
444:
440:
430:
426:
422:
418:
408:phosphoric acid
404:
397:
394:
393:
391:
381:
377:
373:
363:
359:
355:
345:
341:
337:
331:
327:
320:
316:
302:
290:
270:stereochemistry
209:
180:stereochemistry
166:
152:
143:
117:
108:
101:natural product
91:
32:cyclic compound
28:
23:
22:
15:
12:
11:
5:
2109:
2107:
2099:
2098:
2093:
2083:
2082:
2079:
2078:
2068:
2056:
2055:External links
2053:
2052:
2051:
2044:
2030:
2016:
2002:
1988:
1972:
1969:
1967:
1966:
1936:(5): 1045–57.
1916:
1898:
1878:
1851:
1834:
1816:
1786:
1767:
1722:
1681:
1646:
1632:
1608:
1606:
1603:
1602:
1601:
1596:
1591:
1584:
1581:
1580:
1579:
1557:
1550:
1548:
1542:
1535:
1533:
1523:
1516:
1514:
1504:
1497:
1495:
1488:
1481:
1479:
1474:
1470:
1457:
1450:
1448:
1435:
1428:
1426:
1412:
1405:
1403:
1399:delocalization
1393:
1386:
1384:
1376:
1373:
1372:
1371:
1357:
1350:
1348:
1342:
1335:
1333:
1319:
1312:
1310:
1304:
1297:
1295:
1289:
1282:
1280:
1274:
1267:
1265:
1259:
1252:
1250:
1244:
1237:
1235:
1229:
1222:
1220:
1210:
1203:
1201:
1187:
1180:
1178:
1168:
1161:
1159:
1151:
1148:
1146:
1143:
1132:intramolecular
1112:Main article:
1109:
1106:
1105:
1104:
1101:polymerization
1081:
1078:
1077:
1076:
1062:
1048:
1018:
1015:polymerization
1008:
1001:
998:divinyl ketone
990:
971:
960:
945:
942:
941:
940:
933:
927:
895:
892:
890:
887:
874:
873:Principal uses
871:
840:
839:
800:
798:
789:
783:
780:
718:—six membered
684:
677:
676:
675:
673:
670:
649:
646:
644:
641:
614:
611:
588:; D, a mixed
519:
516:
479:
476:
460:germanabenzene
410:derivatives),
389:metaphosphates
301:
298:
289:
286:
284:is displayed.
235:
234:
227:
224:
208:
205:
168:
167:
153:
146:
144:
118:
111:
109:
92:
85:
83:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
2108:
2097:
2094:
2092:
2089:
2088:
2086:
2076:
2072:
2069:
2066:
2062:
2059:
2058:
2054:
2049:
2045:
2042:
2039:
2035:
2031:
2028:
2025:
2021:
2017:
2014:
2011:
2007:
2003:
2000:
1997:
1993:
1989:
1986:
1983:
1979:
1975:
1974:
1970:
1955:
1951:
1947:
1943:
1939:
1935:
1931:
1927:
1920:
1917:
1905:
1901:
1899:9780122004001
1895:
1891:
1890:
1882:
1879:
1874:
1870:
1866:
1862:
1855:
1852:
1848:
1844:
1838:
1835:
1830:
1823:
1821:
1817:
1812:
1808:
1804:
1800:
1793:
1791:
1787:
1784:
1783:
1779:
1774:
1772:
1768:
1755:
1751:
1750:
1747:
1739:
1737:
1735:
1733:
1731:
1729:
1727:
1723:
1718:
1714:
1709:
1704:
1701:(3): 722–30.
1700:
1696:
1692:
1685:
1682:
1677:
1673:
1669:
1665:
1661:
1657:
1650:
1647:
1643:
1639:
1635:
1633:9780471854722
1629:
1625:
1624:
1619:
1613:
1610:
1604:
1600:
1597:
1595:
1592:
1590:
1587:
1586:
1582:
1576:
1572:
1568:
1564:
1560:
1554:
1549:
1545:
1539:
1534:
1530:
1526:
1520:
1515:
1511:
1507:
1501:
1496:
1492:
1485:
1480:
1477:; aromatics).
1468:
1464:
1460:
1454:
1449:
1445:
1442:
1438:
1432:
1427:
1423:
1419:
1415:
1409:
1404:
1400:
1396:
1390:
1385:
1382:
1380:
1374:
1368:
1364:
1360:
1354:
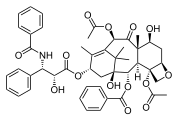
1349:
1345:
1339:
1334:
1330:
1326:
1322:
1316:
1311:
1307:
1301:
1296:
1292:
1286:
1281:
1277:
1271:
1266:
1262:
1256:
1251:
1247:
1241:
1236:
1232:
1226:
1221:
1217:
1213:
1207:
1202:
1198:
1194:
1190:
1184:
1179:
1175:
1171:
1165:
1160:
1157:
1155:
1149:
1144:
1142:
1140:
1136:
1133:
1129:
1125:
1121:
1115:
1107:
1102:
1098:
1095:
1094:
1093:
1091:
1087:
1079:
1075:
1071:
1070:hydroxy group
1067:
1063:
1061:
1057:
1056:amino alcohol
1053:
1049:
1031:
1027:
1023:
1019:
1016:
1012:
1009:
1006:
1002:
999:
995:
991:
988:
987:cycloaddition
984:
980:
976:
972:
969:
965:
961:
958:
955:
954:
953:
951:
943:
938:
934:
931:
928:
925:
922:
921:
920:
918:
914:
909:
900:
893:
888:
886:
884:
880:
879:bioactivities
872:
870:
866:
862:
858:
855:
851:
847:
835:
831:
827:
821:
817:
813:
812:
807:
805:
799:
793:
788:
787:
781:
779:
776:
772:
768:
767:methyl groups
764:
763:steric strain
760:
756:
754:
748:
744:
740:
736:
735:steric strain
731:
729:
725:
721:
717:
713:
704:
700:
696:
692:
688:
681:
671:
669:
667:
663:
659:
655:
647:
642:
640:
637:
635:
631:
627:
623:
619:
612:
607:
603:
599:
595:
591:
587:
583:
579:
575:
571:
566:
562:
560:
556:
552:
551:conformations
548:
544:
540:
536:
532:
524:
517:
515:
513:
509:
505:
501:
497:
493:
489:
485:
477:
475:
473:
469:
465:
464:stannabenzene
461:
457:
453:
439:
435:
417:
413:
409:
390:
386:
372:
368:
354:
350:
336:
326:
315:
311:
307:
299:
297:
295:
287:
285:
283:
279:
275:
271:
267:
263:
258:
256:
252:
248:
244:
240:
232:
228:
225:
222:
218:
217:
216:
214:
206:
204:
202:
198:
197:bioactivities
193:
189:
185:
181:
177:
173:
164:
160:
156:
150:
145:
141:
137:
133:
129:
125:
121:
115:
110:
106:
102:
99:
96:, a complex,
95:
89:
84:
81:
77:
75:
73:
69:
65:
61:
57:
53:
49:
45:
41:
37:
36:ring compound
33:
19:
1958:. Retrieved
1933:
1929:
1919:
1908:. Retrieved
1888:
1881:
1864:
1860:
1854:
1846:
1842:
1837:
1828:
1802:
1798:
1781:
1758:. Retrieved
1749:
1745:
1698:
1694:
1684:
1662:(3): 350–8.
1659:
1655:
1649:
1622:
1618:March, Jerry
1612:
1567:heterocyclic
1559:Brevetoxin A
1378:
1211:
1170:Cycloheptane
1153:
1117:
1089:
1083:
985:, and other
949:
947:
907:
905:
876:
867:
863:
859:
843:
809:
804:edit history
801:
774:
770:
759:interactions
751:
746:
743:angle strain
732:
727:
723:
716:cyclohexanes
709:
702:
698:
694:
690:
654:substitution
651:
638:
616:
613:Nomenclature
555:transannular
528:
488:heterocyclic
481:
303:
291:
259:
236:
231:heterocyclic
210:
178:) such that
172:substitution
169:
132:cyclopentane
124:cyclopropane
120:Cycloalkanes
35:
31:
29:
1861:Tetrahedron
1799:Tetrahedron
1529:delocalized
1525:Benzopyrene
1506:Cholesterol
1437:Longifolene
1418:naphthalene
1395:Naphthalene
1367:delocalized
1344:Caprolactam
1197:delocalized
1066:amino group
975:Diels–Alder
854:cyclohexane
846:aromaticity
824:Please see
811:Aromaticity
782:Aromaticity
720:carbocycles
687:cyclohexane
634:Naphthalene
608:macrocycle.
570:crown ether
559:cyclooctane
535:cyclooctane
518:Macrocycles
468:phosphorine
456:silabenzene
452:borabenzene
288:Carbocycles
266:substituted
221:carbocycles
136:cyclohexane
128:cyclobutane
52:carbocycles
18:Ring pucker
2085:Categories
2038:0199270295
2024:1420079212
2010:0306468565
1996:0470084944
1982:0895732467
1960:2020-09-14
1910:2020-09-14
1605:References
1575:Brevetoxin
1491:paclitaxel
1459:Paclitaxel
1216:octasulfur
989:reactions;
630:polycyclic
626:macrocycle
531:macrocycle
367:phosphorus
243:bond order
155:Paclitaxel
105:paclitaxel
60:bond order
1642:642506595
1571:red tides
1329:methylene
1321:Azetidine
1306:Pentazole
1174:methylene
1060:aziridine
662:chirality
643:Isomerism
622:alicyclic
582:porphyrin
529:The term
504:borazines
492:siloxanes
274:chirality
251:saturated
201:organisms
184:chirality
159:terpenoid
98:terpenoid
68:saturated
44:chemistry
1954:Archived
1950:11700061
1904:Archived
1754:Archived
1717:25687211
1676:93804259
1620:(1985),
1583:See also
1544:Pagodane
1359:Pyridine
1325:nitrogen
1291:Borazine
1145:Examples
1074:solanine
1030:carbonyl
1026:carboxyl
1003:various
968:enediyne
816:CC BY-SA
747:possible
726:and the
586:porphine
574:chelator
539:lactones
512:nitrogen
484:elements
438:borazine
310:nitrogen
247:aromatic
239:valences
213:compound
163:aromatic
72:valences
64:aromatic
40:compound
1760:7 April
1594:Lactone
1510:steroid
1441:terpene
1414:Decalin
1363:methine
1193:methine
1189:Benzene
908:general
850:benzene
818:and/or
753:volumes
602:enamine
543:lactams
496:silicon
387:(e.g.,
351:(e.g.,
349:silicon
294:organic
94:Ingenol
80:natural
2077:(MeSH)
2067:(MeSH)
2040:, see
2036:
2026:, see
2022:
2012:, see
2008:
1998:, see
1994:
1984:, see
1980:
1948:
1896:
1715:
1674:
1640:
1630:
1467:phenyl
1058:to an
983:alkene
966:of an
741:, and
689:, the
596:, the
578:cyclam
500:oxygen
466:, and
385:oxygen
328:(NSCl)
312:(e.g.
306:sulfur
134:, and
1672:S2CID
1212:Cyclo
979:diene
932:; and
913:yield
728:boat,
724:chair
691:chair
618:IUPAC
594:imine
590:amine
508:boron
434:borax
412:boron
374:(NPCl
2034:ISBN
2020:ISBN
2006:ISBN
1992:ISBN
1978:ISBN
1946:PMID
1894:ISBN
1843:1994
1762:2015
1713:PMID
1638:OCLC
1628:ISBN
1561:, a
1439:, a
1050:the
1039:and
1020:the
992:the
973:the
962:the
935:the
828:and
820:GFDL
701:and
695:boat
660:and
606:DOTA
541:and
510:and
498:and
441:(BN)
356:(SiH
308:and
272:and
182:and
174:(by
48:ring
34:(or
1938:doi
1934:313
1869:doi
1807:doi
1703:doi
1664:doi
775:cis
771:cis
769:in
668:).
423:(BO
392:(PO
365:),
347:),
2087::
1952:.
1944:.
1932:.
1928:.
1902:.
1865:30
1863:.
1845:)
1819:^
1803:37
1801:.
1789:^
1770:^
1725:^
1711:.
1699:48
1697:.
1693:.
1670:.
1658:.
1636:,
1034:CO
848:;
737:,
576:,
462:,
458:,
454:,
432:,
419:Na
333:,
323:,
321:NH
223:),
130:,
126:,
30:A
1963:.
1940::
1913:.
1875:.
1871::
1813:.
1809::
1764:.
1719:.
1705::
1678:.
1666::
1660:2
1475:5
1473:H
1471:6
1214:-
1103:.
1047:;
1045:O
1043:2
1041:H
1036:2
1017:;
1007:;
1000:;
970:;
959:;
926:;
755:,
703:e
699:a
592:/
447:6
445:H
443:3
429:3
427:)
425:2
421:3
403:3
401:)
398:3
395:−
380:3
378:)
376:2
362:5
360:)
358:2
344:4
342:N
340:4
338:S
330:3
319:7
317:S
264:–
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.