488:) on one or more of the carbons of the ring. Anomers are named "alpha" or "axial" and "beta" or "equatorial" when substituting a cyclic ring structure that has single bonds between the carbon atoms of the ring for example, a hydroxyl group, a methyl hydroxyl group, a methoxy group or another pyranose or furanose group which are typical single bond substitutions but not limited to these. Axial geometric isomerism will be perpendicular (90 degrees) to a reference plane and equatorial will be 120 degrees away from the axial bond or deviate 30 degrees from the reference plane.
196:
403:
356:
294:, when looking at the source of light, the rotation of the plane of polarization may be either to the right (dextrorotary — d-rotary, represented by (+), clockwise), or to the left (levorotary — l-rotary, represented by (−), counter-clockwise) depending on which stereoisomer is dominant. For instance, sucrose and camphor are d-rotary whereas cholesterol is l-rotary.
31:
312:
Stereoisomerism about double bonds arises because rotation about the double bond is restricted, keeping the substituents fixed relative to each other. If the two substituents on at least one end of a double bond are the same, then there is no stereoisomer and the double bond is not a stereocenter,
451:
Conformational isomerism is a form of isomerism that describes the phenomenon of molecules with the same structural formula but with different shapes due to rotations about one or more bonds. Different conformations can have different energies, can usually interconvert, and are very rarely
418:)-2-fluoro-3-methylpent-2-ene because the highest-priority groups on each side of the double bond are on the same side of the double bond. Fluoro is the highest-priority group on the left side of the double bond, and ethyl is the highest-priority group on the right side of the molecule.
483:
is an identity for single bonded ring structures where "cis" or "Z" and "trans" or "E" (geometric isomerism) needs to name the substitutions on a carbon atom that also displays the identity of chirality; so anomers have carbon atoms that have geometric isomerism and optical isomerism
802:
287:. For instance, by definition, in a Fischer projection the penultimate carbon of D-sugars are depicted with hydrogen on the left and hydroxyl on the right. L-sugars will be shown with the hydrogen on the right and the hydroxyl on the left.
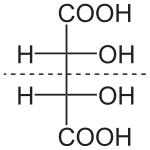
172:. Diastereomers seldom have the same physical properties. In the example shown below, the meso form of tartaric acid forms a diastereomeric pair with both levo- and dextro-tartaric acids, which form an enantiomeric pair.
107:
and how they interact with different enantiomers of other compounds. As a result, different enantiomers of a compound may have substantially different biological effects. Pure enantiomers also exhibit the phenomenon of
775:
471:
There are some molecules that can be isolated in several conformations, due to the large energy barriers between different conformations. 2,2',6,6'-Tetrasubstituted biphenyls can fit into this latter category.
468:
for this process, because there are lower-energy pathways. The conformational inversion of substituted cyclohexanes is a very rapid process at room temperature, with a half-life of 0.00001 seconds.
70:, which share the same molecular formula, but the bond connections or their order differs. By definition, molecules that are stereoisomers of each other represent the same structural isomer.
1352:
534:). This means that configurational isomers can be interconverted only by breaking covalent bonds to the stereocenter, for example, by inverting the configurations of some or all of the
103:
in one has the opposite configuration in the other. Two compounds that are enantiomers of each other have the same physical properties, except for the direction in which they rotate
1357:
371:-1,2-dichloroethene. Due to occasional ambiguity, IUPAC adopted a more rigorous system wherein the substituents at each end of the double bond are assigned priority based on their
383:, opposite). Since chlorine has a larger atomic number than hydrogen, it is the highest-priority group. Using this notation to name the above pictured molecules, molecule I is (
621:
1218:
464:, the boat conformation represents the energy maximum on a conformational itinerary between the two equivalent chair forms; however, it does not represent the
414:-2-fluoro-3-methylpent-2-ene because the alkyl groups that form the backbone chain (i.e., methyl and ethyl) reside across the double bond from each other, or (
460:
where four of the carbon atoms form the "seat" of the chair, one carbon atom is the "back" of the chair, and one carbon atom is the "foot rest"; and a
1462:
1093:
66:
and sequence of bonded atoms (constitution), but differ in the three-dimensional orientations of their atoms in space. This contrasts with
189:
181:
1211:
602:
1066:
828:
276:- labeling more commonly seen, explaining why these may appear reversed to those familiar with only the latter naming convention.
1390:
1013:
812:
785:
332:(Latin, across), in reference to the relative position of substituents on either side of a double bond. A simple example of
1375:
1204:
984:
1385:
149:
are stereoisomers not related through a reflection operation. They are not mirror images of each other. These include
1228:
1347:
554:
1452:
1273:
453:
446:
402:
303:
154:
756:
355:
597:
527:
1411:
1325:
1242:
935:
284:
113:
79:
960:
892:
526:
is a stereoisomer of a reference molecule that has the opposite configuration at a stereocenter (e.g.,
1038:
906:
1421:
1406:
1257:
868:
659:
1319:
1457:
1380:
1278:
1182:
998:
280:
100:
1367:
1151:
1062:
808:
781:
696:
672:
561:
195:
67:
63:
1078:
920:
731:
375:. If the high-priority substituents are on the same side of the bond, it is assigned Z (Ger.
307:
165:
99:
of each other that are non-superposable. Human hands are a macroscopic analog of this. Every
1416:
1342:
1252:
1141:
1133:
635:
465:
456:(which cyclohexane is an essential intermediate for the synthesis of nylon–6,6) including a
291:
109:
1426:
1314:
1268:
545:
is a diastereoisomer that has the opposite configuration at only one of the stereocenters.
169:
104:
47:
39:
1196:
843:
1146:
1121:
1446:
1309:
510:
506:
372:
150:
631:
1431:
1304:
1247:
535:
531:
141:
116:
agent. In nature, only one enantiomer of most chiral biological compounds, such as
96:
27:
When molecules have the same atoms and bond structure but differ in 3D orientation
513:
barrier to rotation is high enough to allow for the isolation of the conformers.
95:, are two stereoisomers that are related to each other by a reflection: they are
1137:
710:
626:
564:
atoms, there is a maximum of 2 different stereoisomers possible. As an example,
502:
497:
83:
429:
are also used to describe the relative position of two substituents on a ring;
124:, which is achiral), is present. An optically active compound shows two forms:
1299:
569:
485:
188:
180:
117:
630:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
639:
59:
17:
1155:
399:, are always interchangeable. Consider the following fluoromethylpentene:
1186:
1170:
121:
340:
isomerism is the 1,2-disubstituted ethenes, like the dichloroethene (C
30:
480:
35:
324:
Traditionally, double bond stereochemistry was described as either
711:"Geometric Isomers Definition And Examples | Chemistry Dictionary"
29:
1014:"The Big Damn Post Of Carbohydrate-Related Chemistry Definitions"
893:"Conformational Isomer - an overview | ScienceDirect Topics"
1200:
1061:
Morrison and Boyd
Organic Chemistry Sixth ed. pgs. 1170-1171
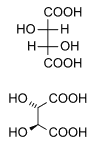
584:. Four of its six carbon atoms are stereogenic, which means
401:
354:
999:"Anomeric Effect - an overview | ScienceDirect Topics"
379:, together). If they are on opposite sides, it is E (Ger.
505:
are stereoisomers resulting from hindered rotation about
283:
can be used to differentiate between L- and D- molecules
697:"Diastereomer - an overview | ScienceDirect Topics"
242:
1079:"Atropisomer - an overview | ScienceDirect Topics"
921:"Cyclohexane - an overview | ScienceDirect Topics"
829:"Cis–trans isomerism | NAL Agricultural Thesaurus"
268:- labeling of the isomers above is not the same as the
907:"Isomerism - Conformational isomers | Britannica"
807:(second ed.). W. A. Benjamin, Inc. p. 19.6.
780:(second ed.). W. A. Benjamin, Inc. p. 19.7.
391:)-1,2-dichloroethene. It is not the case that Z and
1399:
1366:
1335:
1287:
1235:
452:isolatable. For example, there exists a variety of
42:
focuses on stereoisomers, red boxes in the picture.
985:"Anomer - an overview | ScienceDirect Topics"
321:where the two substituents at one end are both H.
1358:Ultraviolet–visible spectroscopy of stereoisomers
757:"Geometric Isomer Definition (Cis–Trans Isomers)"
588:-glucose is one of 2=16 possible stereoisomers.
174:
801:Roberts, John D.; Caserio, Marjorie C. (1977).
774:Roberts, John D.; Caserio, Marjorie C. (1977).
1212:
8:
410:The proper name for this molecule is either
235:
219:
202:
112:and can be separated only with the use of a
1219:
1205:
1197:
1126:Cold Spring Harbor Perspectives in Biology
650:Columbia Encyclopedia. "Stereoisomers" in
1145:
387:)-1,2-dichloroethene and molecule II is (
1122:"The Origin of Biological Homochirality"
961:"What do the α- and β- forms look like?"
193:
186:
177:
614:
367:-1,2-dichloroethene and molecule II is
844:"E–Z notation for geometric isomerism"
804:Basic Principles of Organic Chemistry
777:Basic Principles of Organic Chemistry
7:
732:"geometric (cis / trans) isomerism"
1092:Metrano, Anthony J. (2018-06-09).
869:"What are Conformational Isomers?"
627:Compendium of Chemical Terminology
603:Backbone-dependent rotamer library
25:
1353:NMR spectroscopy of stereoisomers
557:states that for a structure with
62:in which molecules have the same
1391:Diastereomeric recrystallization
1012:Ashenhurst, James (2022-08-03).
194:
187:
179:
433:if on the same side, otherwise
934:Reusch, William (2013-05-05).
1:
1463:Jacobus Henricus van 't Hoff
1386:Chiral column chromatography
1169:Brooks, Benjamin T. (1918).
842:Clark, Jim (November 2012).
730:Clark, Jim (February 2020).
671:Clark, Jim (November 2012).
524:configurational stereoisomer
1138:10.1101/cshperspect.a032540
298:Cis–trans and E–Z isomerism
1479:
1348:Chiral derivatizing agents
1229:enantioselective synthesis
1171:"The German Chemical Myth"
1018:masterorganicchemistry.com
495:
444:
301:
139:
77:
1175:The North American Review
1120:Blackmond, D. G. (2019).
755:Helmenstine, Anne Marie.
549:
454:Cyclohexane conformations
328:(Latin, on this side) or
1274:Supramolecular chirality
1101:knowleslab.princeton.edu
447:Conformational isomerism
640:10.1351/goldbook.S05983
352:) isomers shown below.
204:(natural) tartaric acid
168:, and non-enantiomeric
34:The different types of
598:Descriptor (chemistry)
555:Le Bel-van't Hoff rule
550:Le Bel-van't Hoff rule
407:
360:
359:Dichloroethene isomers
43:
1412:Chiral pool synthesis
1326:Diastereomeric excess
572:and has the formula C
405:
358:
285:Chirality (chemistry)
80:Chirality (chemistry)
33:
1422:Asymmetric catalysis
1407:Asymmetric induction
290:The other refers to
231:dextro-tartaric acid
1320:Enantiomeric excess
673:"Optical isomerism"
406:Fluoromethylpentene
304:Cis–trans isomerism
237:meso-tartaric acid
216:levo-tartaric acid
1417:Chiral auxiliaries
1381:Kinetic resolution
1279:Inherent chirality
1264:-symmetric ligands
458:chair conformation
408:
361:
281:Fischer projection
229:-(-)-tartaric acid
214:-(+)-tartaric acid
101:stereogenic center
68:structural isomers
44:
1440:
1439:
1376:Recrystallization
1368:Chiral resolution
1043:chemistry.msu.edu
1037:Reusch, William.
940:chemistry.msu.edu
587:
567:
562:asymmetric carbon
462:boat conformation
267:
263:
258:
257:
248:
228:
223:
213:
208:
131:
127:
64:molecular formula
56:spatial isomerism
16:(Redirected from
1470:
1343:Optical rotation
1288:Chiral molecules
1253:Planar chirality
1221:
1214:
1207:
1198:
1191:
1190:
1181:(756): 729–735.
1166:
1160:
1159:
1149:
1117:
1111:
1110:
1108:
1107:
1098:
1089:
1083:
1082:
1075:
1069:
1059:
1053:
1052:
1050:
1049:
1034:
1028:
1027:
1025:
1024:
1009:
1003:
1002:
995:
989:
988:
981:
975:
974:
972:
971:
965:chem.ucalgary.ca
956:
950:
949:
947:
946:
931:
925:
924:
917:
911:
910:
903:
897:
896:
889:
883:
882:
880:
879:
873:chem.ucalgary.ca
864:
858:
857:
855:
854:
839:
833:
832:
825:
819:
818:
798:
792:
791:
771:
765:
764:
752:
746:
745:
743:
742:
727:
721:
720:
718:
717:
707:
701:
700:
693:
687:
686:
684:
683:
668:
662:
652:Encyclopedia.com
648:
642:
619:
585:
565:
517:More definitions
466:transition state
313:e.g. propene, CH
292:Optical rotation
265:
261:
246:
226:
221:
211:
206:
198:
191:
183:
175:
129:
125:
110:optical activity
91:, also known as
21:
1478:
1477:
1473:
1472:
1471:
1469:
1468:
1467:
1453:Stereochemistry
1443:
1442:
1441:
1436:
1427:Organocatalysis
1395:
1362:
1331:
1315:Racemic mixture
1283:
1269:Axial chirality
1263:
1236:Chirality types
1231:
1225:
1195:
1194:
1168:
1167:
1163:
1119:
1118:
1114:
1105:
1103:
1096:
1091:
1090:
1086:
1077:
1076:
1072:
1060:
1056:
1047:
1045:
1039:"Stereoisomers"
1036:
1035:
1031:
1022:
1020:
1011:
1010:
1006:
997:
996:
992:
983:
982:
978:
969:
967:
958:
957:
953:
944:
942:
936:"Stereoisomers"
933:
932:
928:
919:
918:
914:
905:
904:
900:
891:
890:
886:
877:
875:
866:
865:
861:
852:
850:
848:chemguide.co.uk
841:
840:
836:
827:
826:
822:
815:
800:
799:
795:
788:
773:
772:
768:
754:
753:
749:
740:
738:
729:
728:
724:
715:
713:
709:
708:
704:
695:
694:
690:
681:
679:
677:chemguide.co.uk
670:
669:
665:
649:
645:
632:stereoisomerism
620:
616:
611:
594:
583:
579:
575:
568:-glucose is an
552:
519:
500:
494:
478:
449:
443:
351:
347:
343:
320:
316:
310:
302:Main articles:
300:
251:"racemic acid"
250:
245:
232:
230:
225:
215:
210:
205:
170:optical isomers
144:
138:
105:polarized light
93:optical isomers
86:
78:Main articles:
76:
58:, is a form of
52:stereoisomerism
48:stereochemistry
40:Stereochemistry
28:
23:
22:
15:
12:
11:
5:
1476:
1474:
1466:
1465:
1460:
1455:
1445:
1444:
1438:
1437:
1435:
1434:
1429:
1424:
1419:
1414:
1409:
1403:
1401:
1397:
1396:
1394:
1393:
1388:
1383:
1378:
1372:
1370:
1364:
1363:
1361:
1360:
1355:
1350:
1345:
1339:
1337:
1333:
1332:
1330:
1329:
1323:
1317:
1312:
1307:
1302:
1297:
1291:
1289:
1285:
1284:
1282:
1281:
1276:
1271:
1266:
1261:
1255:
1250:
1245:
1239:
1237:
1233:
1232:
1226:
1224:
1223:
1216:
1209:
1201:
1193:
1192:
1161:
1132:(3): a032540.
1112:
1094:"Atropisomers"
1084:
1070:
1054:
1029:
1004:
990:
976:
951:
926:
912:
898:
884:
859:
834:
820:
813:
793:
786:
766:
747:
722:
702:
688:
663:
643:
613:
612:
610:
607:
606:
605:
600:
593:
590:
581:
577:
573:
551:
548:
547:
546:
539:
538:in a compound.
518:
515:
496:Main article:
493:
490:
477:
474:
445:Main article:
442:
439:
363:Molecule I is
349:
345:
341:
318:
314:
299:
296:
256:
255:
253:
249:-tartaric acid
240:
239:
234:
224:-tartaric acid
218:
209:-tartaric acid
200:
199:
192:
185:
151:meso compounds
140:Main article:
137:
134:
128:-(+) form and
75:
72:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
1475:
1464:
1461:
1459:
1456:
1454:
1451:
1450:
1448:
1433:
1430:
1428:
1425:
1423:
1420:
1418:
1415:
1413:
1410:
1408:
1405:
1404:
1402:
1398:
1392:
1389:
1387:
1384:
1382:
1379:
1377:
1374:
1373:
1371:
1369:
1365:
1359:
1356:
1354:
1351:
1349:
1346:
1344:
1341:
1340:
1338:
1334:
1327:
1324:
1321:
1318:
1316:
1313:
1311:
1310:Meso compound
1308:
1306:
1303:
1301:
1298:
1296:
1293:
1292:
1290:
1286:
1280:
1277:
1275:
1272:
1270:
1267:
1265:
1260:
1256:
1254:
1251:
1249:
1246:
1244:
1241:
1240:
1238:
1234:
1230:
1222:
1217:
1215:
1210:
1208:
1203:
1202:
1199:
1188:
1184:
1180:
1176:
1172:
1165:
1162:
1157:
1153:
1148:
1143:
1139:
1135:
1131:
1127:
1123:
1116:
1113:
1102:
1095:
1088:
1085:
1080:
1074:
1071:
1068:
1067:0-13-643669-2
1064:
1058:
1055:
1044:
1040:
1033:
1030:
1019:
1015:
1008:
1005:
1000:
994:
991:
986:
980:
977:
966:
962:
955:
952:
941:
937:
930:
927:
922:
916:
913:
908:
902:
899:
894:
888:
885:
874:
870:
863:
860:
849:
845:
838:
835:
830:
824:
821:
816:
810:
806:
805:
797:
794:
789:
783:
779:
778:
770:
767:
762:
758:
751:
748:
737:
733:
726:
723:
712:
706:
703:
698:
692:
689:
678:
674:
667:
664:
661:
657:
653:
647:
644:
641:
637:
633:
629:
628:
623:
618:
615:
608:
604:
601:
599:
596:
595:
591:
589:
571:
563:
560:
556:
544:
540:
537:
536:stereocenters
533:
529:
525:
521:
520:
516:
514:
512:
511:steric strain
508:
504:
499:
491:
489:
487:
486:enantiomerism
482:
475:
473:
469:
467:
463:
459:
455:
448:
440:
438:
436:
432:
428:
424:
419:
417:
413:
404:
400:
398:
394:
390:
386:
382:
378:
374:
373:atomic number
370:
366:
357:
353:
339:
335:
331:
327:
322:
309:
305:
297:
295:
293:
288:
286:
282:
277:
275:
271:
254:
252:
241:
238:
233:
217:
201:
197:
190:
184:
182:
176:
173:
171:
167:
163:
161:
157:
152:
148:
147:Diastereomers
143:
136:Diastereomers
135:
133:
123:
119:
115:
111:
106:
102:
98:
97:mirror images
94:
90:
85:
81:
73:
71:
69:
65:
61:
57:
53:
49:
41:
37:
32:
19:
1432:Biocatalysis
1305:Diastereomer
1295:Stereoisomer
1294:
1258:
1248:Stereocenter
1227:Concepts in
1178:
1174:
1164:
1129:
1125:
1115:
1104:. Retrieved
1100:
1087:
1073:
1057:
1046:. Retrieved
1042:
1032:
1021:. Retrieved
1017:
1007:
993:
979:
968:. Retrieved
964:
954:
943:. Retrieved
939:
929:
915:
901:
887:
876:. Retrieved
872:
862:
851:. Retrieved
847:
837:
823:
803:
796:
776:
769:
760:
750:
739:. Retrieved
736:Chemguide.uk
735:
725:
714:. Retrieved
705:
691:
680:. Retrieved
676:
666:
655:
651:
646:
625:
617:
558:
553:
542:
523:
507:single bonds
503:Atropisomers
501:
492:Atropisomers
479:
470:
461:
457:
450:
434:
430:
426:
422:
420:
415:
411:
409:
396:
392:
388:
384:
380:
376:
368:
364:
362:
337:
333:
329:
325:
323:
311:
308:E–Z notation
289:
278:
273:
269:
259:
243:
236:
220:
203:
178:
159:
155:
146:
145:
142:Diastereomer
92:
88:
87:
55:
51:
45:
18:Stereoisomer
959:Hunt, Ian.
867:Hunt, Ian.
498:Atropisomer
395:, or E and
166:E-Z isomers
132:-(−) form.
118:amino acids
89:Enantiomers
84:Enantiomers
74:Enantiomers
1447:Categories
1300:Enantiomer
1106:2022-08-09
1048:2022-08-09
1023:2022-08-09
970:2022-08-09
945:2022-08-09
878:2022-08-09
853:2022-08-09
814:0805383298
787:0805383298
741:2022-08-09
716:2022-06-20
682:2022-08-09
609:References
570:aldohexose
509:where the
441:Conformers
421:The terms
1458:Isomerism
1400:Reactions
1243:Chirality
761:ThoughtCo
481:Anomerism
60:isomerism
1336:Analysis
1187:25151064
1156:30824575
654:, n.l.,
592:See also
532:E- vs Z-
528:R- vs S-
381:entgegen
377:zusammen
120:(except
1147:6396334
476:Anomers
162:isomers
122:glycine
36:isomers
1185:
1154:
1144:
1065:
811:
784:
543:epimer
272:- and
264:- and
114:chiral
1183:JSTOR
1097:(PDF)
622:IUPAC
435:trans
427:trans
412:trans
397:trans
369:trans
338:trans
330:trans
317:CH=CH
244:(1:1)
160:trans
54:, or
1328:(de)
1322:(ee)
1152:PMID
1063:ISBN
809:ISBN
782:ISBN
660:Link
656:2005
425:and
306:and
260:The
82:and
1179:208
1142:PMC
1134:doi
636:doi
634:".
541:An
530:or
431:cis
423:cis
393:cis
365:cis
334:cis
326:cis
156:cis
46:In
1449::
1177:.
1173:.
1150:.
1140:.
1130:11
1128:.
1124:.
1099:.
1041:.
1016:.
963:.
938:.
871:.
846:.
759:.
734:.
675:.
658:,
624:,
578:12
522:A
437:.
348:Cl
279:A
247:DL
164:,
153:,
50:,
38:.
1262:2
1259:C
1220:e
1213:t
1206:v
1189:.
1158:.
1136::
1109:.
1081:.
1051:.
1026:.
1001:.
987:.
973:.
948:.
923:.
909:.
895:.
881:.
856:.
831:.
817:.
790:.
763:.
744:.
719:.
699:.
685:.
638::
586:D
582:6
580:O
576:H
574:6
566:D
559:n
484:(
416:Z
389:E
385:Z
350:2
346:2
344:H
342:2
336:–
319:2
315:3
274:l
270:d
266:L
262:D
227:D
222:D
212:L
207:L
158:–
130:L
126:D
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.