438:
activity, reversing constipation, a non-motor symptom of
Parkinson’s disease. Squalamine also restored electrical signaling between the enteric nervous system and the brain ( the “gut-brain axis”). In addition the electrical signals induced by orally administered squalamine phenocopied those elicited by SSRI anti-depressant drugs suggesting that the compound could, via the gut-brain axis, elicit an anti-depressant effect. Based on these preclinical studies squalamine (as the phosphate salt (ENT-01)) was evaluated for the treatment of Parkinson’s disease associated constipation in two clinical trials: RASMET, an open label Phase 1b trial, and subsequently, KARMET, a Phase 2a placebo controlled randomized double blinded trial involving about 150 patients. Both trials, conducted by Enterin, Inc (Philadelphia) demonstrated that a 28 day course of orally administered ENT-03 effectively corrected constipation that had been previously intractable. In addition, positive efficacy signals were seen in circadian rhythm and sleep, dementia and hallucinations. ENT-01 is now (2024) positioned for Phase 3 clinical trials.
267:
422:. Squalamine was later identified in the white blood cells of the lamprey. Squalamine has broad spectrum microbicidal activity, and its use as a therapeutic has been studied preclinically. In the late 1990’s squalamine was discovered to exhibit antiangiogenic activity, and as a consequence was later studied in several early stage clinical trials for both cancer, age related macular degeneration, administered intravenously, and as an eye-drop in combination with intraocular ranibizumab.
31:
426:
cells. Once squalamine crosses the plasma membrane of an animal cell it binds to the cytoplasmic surface of the plasma membrane and displaces proteins that are bound electrostatically, a property that explains its inhibition of the sodium-hydrogen transporter type 3, neuronal synaptic AMPA receptors and its broad spectrum antiviral activity.
121:
425:
In aqueous solution at physiological pH squalamine exists as an amphipathic zwitterion with a net cationic charge. As a consequence the molecule is attracted by electrostatic forces to membranes that display negatively charged phospholipid headgroups, such as the intracellular membranes of animal
409:
was discovered in a search for anti-microbial compounds in the tissues of primitive vertebrates. The team speculated that animals with primitive immune systems, such as sharks and lampreys, might utilize antimicrobial compounds as a significant component of their immune repertoire. The dogfish
437:
where it forms toxic aggregates damaging or killing neurons, squalamine emerged as a potential therapeutic. Studies in mouse models of
Parkinson’s disease demonstrated that orally administered squalamine could restore the electrical activity of enteric neurons and thereby restore peristaltic
414:. In addition, large numbers of dogfish are harvested annually for consumption and could provide sufficient tissue for extraction during the early stages of compound isolation and characterization. The chemical synthesis was developed by William A. Kinney and colleagues,.
290:
InChI=1S/C34H65N3O5S/c1-23(2)31(42-43(39,40)41)12-9-24(3)27-10-11-28-32-29(14-16-34(27,28)5)33(4)15-13-26(21-25(33)22-30(32)38)37-20-8-19-36-18-7-6-17-35/h23-32,36-38H,6-22,35H2,1-5H3,(H,39,40,41)/t24-,25-,26+,27-,28+,29+,30-,31-,32+,33+,34-/m1/s1
300:
InChI=1/C34H65N3O5S/c1-23(2)31(42-43(39,40)41)12-9-24(3)27-10-11-28-32-29(14-16-34(27,28)5)33(4)15-13-26(21-25(33)22-30(32)38)37-20-8-19-36-18-7-6-17-35/h23-32,36-38H,6-22,35H2,1-5H3,(H,39,40,41)/t24-,25-,26+,27-,28+,29+,30-,31-,32+,33+,34-/m1/s1
417:
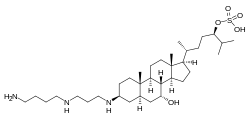
Squalamine consists of a spermidine coupled to a C-27 sulfated bile salt, a natural product with an unprecedented chemical structure. In addition 7 additional aminosterols were isolated from dogfish liver, including
316:
433:
Parkinson disease model. Since alpha synuclein accumulates within the enteric, peripheral and central nervous system of individuals suffering from
429:
In 2017, Perni et al reported that squalamine could displace alpha-synuclein from neuronal membranes both in vitro, in isolated cells, and in a
598:
A Phase I/II Trial of
Intravenous Squalamine Lactate for Treatment of Choroidal Neovascularization in Age Related Macular Degeneration (ARMD)
281:
718:
411:
394:
608:
224:
738:
245:
410:
shark (Squalus acanthias) was the first shark species studied since it was accessible for research purposes at the
743:
733:
434:
748:
148:
141:
57:
43:
723:
262:
728:
87:
339:
97:
233:
168:
463:
Squalus acanthias
Convention on the conservation of migratory species of wild animals 2008
266:
388:
712:
419:
213:
430:
376:
159:
17:
200:
30:
132:
387:
Except where otherwise noted, data are given for materials in their
188:
120:
110:
74:-cyclopentaphenanthren-1-yl]-2-methylheptan-3-yl hydrogen sulfate
51:)-3β-({3-propyl}amino)-7α-hydroxycholestan-24-yl hydrogen sulfate
179:
609:"Jason Slakter, MD: Squalamine Lactate Eye Drops in Wet AMD"
250:
324:
CC(C)(CC(C)1CC23(O)C4C(CC4(C)3CC12C)NCCCNCCCCN)OS(=O)(=O)O
70:)-6-propyl}amino)-4-hydroxy-9a,11a-dimethylhexadecahydro-1
212:
96:
648:doi: 10.1073/pnas.1610586114. Epub 2017 Jan 17
8:
265:
167:
22:
412:Mount Desert Marine Biological Laboratory
232:
447:
321:
286:
261:
702:doi: 10.7326/M22-1438. Epub 2022 Nov 8
293:Key: UIRKNQLZZXALBI-MSVGPLKSSA-N
147:
140:
7:
303:Key: UIRKNQLZZXALBI-MSVGPLKSBB
203:
187:
481:doi:/10.1016/S0040-4039(99)00896-5
14:
693:doi: 10.1016/j.prdoa.2019.06.001
630:doi:10.1016/j.neuron.2010.04.035
357:
351:
29:
684:doi: 10.1038/s41598-021-00615-w
639:doi.org/10.1073/pnas.1108558108
508:doi: 10.1194/jlr.M700294-JLR200
391:(at 25 °C , 100 kPa).
657:doi: 10.1007/s00702-002-0808-2
369:
363:
345:
1:
589:doi:10.1586/17469899.2.2.165
621:doi: 10.1038/emboj.2010.356
490:doi: 10.1073/pnas.90.4.1354
765:
454:doi:10.1073/pnas.90.4.1354
15:
385:
332:
312:
277:
80:
56:
42:
37:
28:
16:Not to be confused with
719:Angiogenesis inhibitors
675:doi: 10.3233/JPD-202076
666:doi: 10.3233/JPD-202076
544:doi: 10.1093/jac/dks230
381:628 g/mol
499:doi: 10.1021/np990514f
472:doi:10.1021/jo981344z
58:Systematic IUPAC name
611:. 13 November 2017.
435:Parkinson’s disease
25:
739:Secondary alcohols
395:Infobox references
23:
403:Chemical compound
401:
400:
246:CompTox Dashboard
122:Interactive image
756:
744:Secondary amines
703:
700:
694:
691:
685:
682:
676:
673:
667:
664:
658:
655:
649:
646:
640:
637:
631:
628:
622:
619:
613:
612:
605:
599:
596:
590:
587:
581:
578:
572:
569:
563:
560:
554:
551:
545:
542:
536:
533:
527:
524:
518:
515:
509:
506:
500:
497:
491:
488:
482:
479:
473:
470:
464:
461:
455:
452:
371:
365:
359:
353:
347:
340:Chemical formula
270:
269:
254:
252:
236:
216:
205:
191:
171:
151:
144:
124:
100:
33:
26:
764:
763:
759:
758:
757:
755:
754:
753:
734:Antiviral drugs
709:
708:
707:
706:
701:
697:
692:
688:
683:
679:
674:
670:
665:
661:
656:
652:
647:
643:
638:
634:
629:
625:
620:
616:
607:
606:
602:
597:
593:
588:
584:
579:
575:
570:
566:
561:
557:
552:
548:
543:
539:
534:
530:
525:
521:
516:
512:
507:
503:
498:
494:
489:
485:
480:
476:
471:
467:
462:
458:
453:
449:
444:
404:
397:
392:
368:
362:
356:
350:
342:
328:
325:
320:
319:
308:
305:
304:
301:
295:
294:
291:
285:
284:
273:
255:
248:
239:
219:
206:
194:
174:
154:
127:
114:
103:
90:
76:
75:
52:
21:
12:
11:
5:
762:
760:
752:
751:
749:Sulfate esters
746:
741:
736:
731:
726:
721:
711:
710:
705:
704:
695:
686:
677:
668:
659:
650:
641:
632:
623:
614:
600:
591:
582:
573:
564:
555:
546:
537:
528:
519:
510:
501:
492:
483:
474:
465:
456:
446:
445:
443:
440:
402:
399:
398:
393:
389:standard state
386:
383:
382:
379:
373:
372:
366:
360:
354:
348:
343:
338:
335:
334:
330:
329:
327:
326:
323:
315:
314:
313:
310:
309:
307:
306:
302:
299:
298:
296:
292:
289:
288:
280:
279:
278:
275:
274:
272:
271:
263:DTXSID40869971
258:
256:
244:
241:
240:
238:
237:
229:
227:
221:
220:
218:
217:
209:
207:
199:
196:
195:
193:
192:
184:
182:
176:
175:
173:
172:
164:
162:
156:
155:
153:
152:
145:
137:
135:
129:
128:
126:
125:
117:
115:
108:
105:
104:
102:
101:
93:
91:
86:
83:
82:
78:
77:
61:
60:
54:
53:
46:
40:
39:
35:
34:
13:
10:
9:
6:
4:
3:
2:
761:
750:
747:
745:
742:
740:
737:
735:
732:
730:
727:
725:
722:
720:
717:
716:
714:
699:
696:
690:
687:
681:
678:
672:
669:
663:
660:
654:
651:
645:
642:
636:
633:
627:
624:
618:
615:
610:
604:
601:
595:
592:
586:
583:
580:PMID 12855619
577:
574:
571:PMID 11751482
568:
565:
562:PMID 15128931
559:
556:
550:
547:
541:
538:
535:PMID 22998181
532:
529:
526:PMID 18648511
523:
520:
517:PMID 23735598
514:
511:
505:
502:
496:
493:
487:
484:
478:
475:
469:
466:
460:
457:
451:
448:
441:
439:
436:
432:
427:
423:
421:
420:Trodusquemine
415:
413:
408:
396:
390:
384:
380:
378:
375:
374:
344:
341:
337:
336:
331:
322:
318:
311:
297:
287:
283:
276:
268:
264:
260:
259:
257:
247:
243:
242:
235:
231:
230:
228:
226:
223:
222:
215:
211:
210:
208:
202:
198:
197:
190:
186:
185:
183:
181:
178:
177:
170:
166:
165:
163:
161:
158:
157:
150:
146:
143:
139:
138:
136:
134:
131:
130:
123:
119:
118:
116:
112:
107:
106:
99:
95:
94:
92:
89:
85:
84:
79:
73:
69:
65:
59:
55:
50:
45:
41:
36:
32:
27:
19:
698:
689:
680:
671:
662:
653:
644:
635:
626:
617:
603:
594:
585:
576:
567:
558:
553:PMID 9661892
549:
540:
531:
522:
513:
504:
495:
486:
477:
468:
459:
450:
428:
424:
416:
406:
405:
149:ChEMBL507931
142:ChEMBL444929
81:Identifiers
71:
67:
63:
48:
724:Cholestanes
333:Properties
98:148717-90-2
24:Squalamine
729:Polyamines
713:Categories
442:References
431:C. elegans
407:Squalamine
377:Molar mass
234:F8PO54Z4V7
160:ChemSpider
109:3D model (
88:CAS Number
44:IUPAC name
18:squalene
201:PubChem
317:SMILES
189:C16841
133:ChEMBL
38:Names
282:InChI
214:72495
169:65407
111:JSmol
225:UNII
180:KEGG
251:EPA
204:CID
47:(24
715::
355:65
349:34
66:,6
62:(3
370:S
367:5
364:O
361:3
358:N
352:H
346:C
253:)
249:(
113:)
72:H
68:R
64:R
49:R
20:.
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.