419:. by administering teprotide to dogs and rats and observing that it inhibited the vasopressor response induced by angiotensin I. Teprotide was shown to be an effective antihyperension agent but it had limited use because of its expense and lack of oral activity. It was found that teprotide inhibits the enzyme that converts angiotensin I to angiotensin II. From this researchers conducted structure-activity studies which allowed them to identify the active binding site of the ACE which allowed for the development of antihypertension drugs to be developed.
205:
86:
238:
InChI=1S/C53H76N14O12/c1-3-29(2)43(51(77)66-25-9-16-39(66)50(76)67-26-10-17-40(67)52(78)79)63-45(71)34(18-20-41(54)68)60-46(72)37-14-7-23-64(37)48(74)35(13-6-22-57-53(55)56)61-47(73)38-15-8-24-65(38)49(75)36(62-44(70)33-19-21-42(69)59-33)27-30-28-58-32-12-5-4-11-31(30)32/h4-5,11-12,28-29,33-40,43,58H,3,6-10,13-27H2,1-2H3,(H2,54,68)(H,59,69)(H,60,72)(H,61,73)(H,62,70)(H,63,71)(H,78,79)(H4,55,56,57)/t29-,33-,34-,35-,36-,37-,38-,39-,40-,43-/m0/s1
228:
InChI=1S/C53H76N14O12/c1-3-29(2)43(51(77)66-25-9-16-39(66)50(76)67-26-10-17-40(67)52(78)79)63-45(71)34(18-20-41(54)68)60-46(72)37-14-7-23-64(37)48(74)35(13-6-22-57-53(55)56)61-47(73)38-15-8-24-65(38)49(75)36(62-44(70)33-19-21-42(69)59-33)27-30-28-58-32-12-5-4-11-31(30)32/h4-5,11-12,28-29,33-40,43,58H,3,6-10,13-27H2,1-2H3,(H2,54,68)(H,59,69)(H,60,72)(H,61,73)(H,62,70)(H,63,71)(H,78,79)(H4,55,56,57)/t29-,33-,34-,35-,36-,37-,38-,39-,40-,43-/m0/s1
24:
322:
528:
Ondetti, Miguel A.; Nina J. Williams; Emily F. Sabo; Josip
Pluscec; Eugene R. Weaver; Octavian Kocy (October 1971). "Angiotensin-converting enzyme inhibitors from the venom of bothrops jararaca. Isolation, elucidation of structure, and synthesis".
564:
Bianchi, A.; D.B. Evans; M. Cobb; M.T. Peschka; T.R. Schaeffer; R.J. Laffan (July 1973). "Inhibition by SQ 20881 of vasopressor response to angiotensin I in conscious animals".
335:
423:
was the first antihypertension drug developed by
Ondetti and Cushman. Many ACE inhibitors have been developed since this time but this was the start of them.
251:
367:(ACE inhibitor) which inhibits the conversion of angiotensin I to angiotensin II and may potentiate some of the pharmacological actions of
259:
O=C(O)7N(C(=O)6N(C(=O)(NC(=O)(NC(=O)5N(C(=O)(NC(=O)4N(C(=O)(NC(=O)1NC(=O)CC1)Cc3c2ccccc2c3)CCC4)CCC/N=C(\N)N)CCC5)CCC(=O)N)(C)CC)CCC6)CCC7
219:
395:
The antihypertensive effects of teprotide were first observed by Sergio
Ferreira in 1965 and it was first isolated by Ferreira
40:
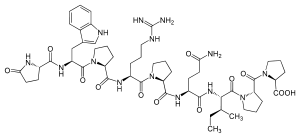
5-oxo-L-prolyl-L-tryptophyl-L-prolyl-N-(diaminomethylidene)-L-ornithyl-L-prolyl-L-glutaminyl-L-isoleucyl-L-prolyl-L-proline
342:
489:
Ferreira, Sergio H.; Diana C. Bartelt; Lewis J. Greene (June 1970). "Isolation of bradykinin-potentiating peptides from
162:
183:
651:
106:
646:
36:
200:
52:
622:
581:
546:
510:
471:
359:
612:
601:"History of the design of captopril and related inhibitors of angiotensin converting enzyme"
573:
538:
502:
461:
453:
274:
126:
171:
62:
204:
466:
457:
441:
399:
along with eight other peptides in 1970. Teprotide was synthesized in 1970 by
Ondetti
313:
640:
577:
364:
151:
442:"A bradykinin-potentiating factor (bpf) present in the venom of bothrops jararaca"
368:
301:
117:
617:
600:
420:
475:
626:
585:
550:
514:
403:
and from there its antihypertensive properties were studied more closely.
542:
506:
138:
23:
97:
312:
Except where otherwise noted, data are given for materials in their
85:
75:
188:
411:
Teprotide was chosen as a lead because of its long-lasting
387:
and has been investigated as an antihypertension agent.
330:
357:
is nonapeptide which has been isolated from the snake
150:
61:
8:
415:activity. This was demonstrated by Bianchi
599:Cushman, D.W.; M.A. Ondetti (April 1991).
203:
125:
15:
616:
465:
170:
432:
365:angiotensin converting enzyme inhibitor
256:
224:
199:
231:Key: UUUHXMGGBIUAPW-CSCXCSGISA-N
105:
7:
141:
458:10.1111/j.1476-5381.1965.tb02091.x
440:Ferreira, Sergio (February 1965).
14:
371:. It has a molecular formula of C
566:European Journal of Pharmacology
320:
22:
446:British Journal of Pharmacology
316:(at 25 °C , 100 kPa).
1:
578:10.1016/0014-2999(73)90248-3
668:
310:
267:
247:
215:
45:
35:
30:
21:
618:10.1161/01.hyp.17.4.589
391:Isolation and synthesis
306:1101.257
543:10.1021/bi00798a004
507:10.1021/bi00815a005
18:
343:Infobox references
16:
537:(22): 4033–4039.
501:(13): 2583–2593.
491:Bothrops jararaca
360:Bothrops jararaca
351:Chemical compound
349:
348:
184:CompTox Dashboard
87:Interactive image
659:
631:
630:
620:
596:
590:
589:
561:
555:
554:
525:
519:
518:
486:
480:
479:
469:
437:
333:
327:
324:
323:
275:Chemical formula
208:
207:
192:
190:
174:
154:
143:
129:
109:
89:
65:
26:
19:
667:
666:
662:
661:
660:
658:
657:
656:
637:
636:
635:
634:
598:
597:
593:
563:
562:
558:
527:
526:
522:
488:
487:
483:
439:
438:
434:
429:
409:
393:
386:
382:
378:
374:
352:
345:
340:
339:
338: ?)
329:
325:
321:
317:
295:
291:
287:
283:
277:
263:
260:
255:
254:
243:
240:
239:
233:
232:
229:
223:
222:
211:
201:DTXSID301029698
193:
186:
177:
157:
144:
132:
112:
92:
79:
68:
55:
41:
12:
11:
5:
665:
663:
655:
654:
652:ACE inhibitors
649:
639:
638:
633:
632:
611:(4): 589–592.
591:
556:
520:
481:
431:
430:
428:
425:
408:
405:
392:
389:
384:
380:
376:
372:
350:
347:
346:
341:
319:
318:
314:standard state
311:
308:
307:
304:
298:
297:
293:
289:
285:
281:
278:
273:
270:
269:
265:
264:
262:
261:
258:
250:
249:
248:
245:
244:
242:
241:
237:
236:
234:
230:
227:
226:
218:
217:
216:
213:
212:
210:
209:
196:
194:
182:
179:
178:
176:
175:
167:
165:
159:
158:
156:
155:
147:
145:
137:
134:
133:
131:
130:
122:
120:
114:
113:
111:
110:
102:
100:
94:
93:
91:
90:
82:
80:
73:
70:
69:
67:
66:
58:
56:
51:
48:
47:
43:
42:
39:
33:
32:
28:
27:
13:
10:
9:
6:
4:
3:
2:
664:
653:
650:
648:
645:
644:
642:
628:
624:
619:
614:
610:
606:
602:
595:
592:
587:
583:
579:
575:
571:
567:
560:
557:
552:
548:
544:
540:
536:
532:
524:
521:
516:
512:
508:
504:
500:
496:
492:
485:
482:
477:
473:
468:
463:
459:
455:
451:
447:
443:
436:
433:
426:
424:
422:
418:
414:
406:
404:
402:
398:
390:
388:
370:
366:
362:
361:
356:
344:
337:
332:
315:
309:
305:
303:
300:
299:
279:
276:
272:
271:
266:
257:
253:
246:
235:
225:
221:
214:
206:
202:
198:
197:
195:
185:
181:
180:
173:
169:
168:
166:
164:
161:
160:
153:
149:
148:
146:
140:
136:
135:
128:
124:
123:
121:
119:
116:
115:
108:
104:
103:
101:
99:
96:
95:
88:
84:
83:
81:
77:
72:
71:
64:
60:
59:
57:
54:
50:
49:
44:
38:
34:
29:
25:
20:
647:Nonapeptides
608:
605:Hypertension
604:
594:
572:(2): 90–96.
569:
565:
559:
534:
531:Biochemistry
530:
523:
498:
495:Biochemistry
494:
490:
484:
449:
445:
435:
416:
412:
410:
400:
396:
394:
363:. It is an
358:
354:
353:
107:ChEMBL408983
46:Identifiers
407:Medical use
268:Properties
641:Categories
452:(1): 169.
427:References
369:bradykinin
302:Molar mass
172:C3E5QBF1R6
118:ChemSpider
74:3D model (
63:35115-60-7
53:CAS Number
37:IUPAC name
17:Teprotide
421:Captopril
355:Teprotide
493:venom".
476:14302350
627:2013486
586:4354808
551:4334402
515:4317874
467:1704050
413:in vivo
336:what is
334: (
296:
139:PubChem
625:
584:
549:
513:
474:
464:
401:et al.
397:et al.
331:verify
328:
252:SMILES
152:443376
127:391608
98:ChEMBL
31:Names
417:et al
220:InChI
76:JSmol
623:PMID
582:PMID
547:PMID
511:PMID
472:PMID
163:UNII
613:doi
574:doi
539:doi
503:doi
462:PMC
454:doi
189:EPA
142:CID
643::
621:.
609:17
607:.
603:.
580:.
570:23
568:.
545:.
535:10
533:.
509:.
497:.
470:.
460:.
450:24
448:.
444:.
385:12
381:14
377:76
373:53
294:12
290:14
286:76
282:53
629:.
615::
588:.
576::
553:.
541::
517:.
505::
499:9
478:.
456::
383:O
379:N
375:H
326:Y
292:O
288:N
284:H
280:C
191:)
187:(
78:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.