1595:
1670:
333:
310:
207:
232:
1882:
these disorders have been found to be in vivo and in vitro substrates of tTG. Although tTG is up regulated in the areas of the brain affected by
Huntington's disease, a recent study showed that increasing levels of tTG do not affect the onset and/or progression of the disease in mice. Recent studies show that tTG may not be involved in AD as studies show it is associated with erythrocyte lysis and is a consequence of the disease rather than a cause.
2018:
591:
584:
598:
339:
238:
4343:
1653:-1 has been shown to activate extracellular tTG by reducing the disulfide bond. Another disuplhide bond can form in tTG, between the residues Cys-230 and Cys-370. While this bond does not exist in the enzyme's native state, it appears when the enzyme is inactivated via oxidation. The presence of calcium protects against the formation of both disulfide bonds, thus making the enzyme more resistant to oxidation.
5046:
2077:
cultured human umbilical vein endothelial cells (HUVECs). ERp57 oxidized TG2 with a rate constant that was 400-2000-fold higher than those of the aforementioned small molecule oxidants. Moreover, its specificity for TG2 was also markedly higher than those of other secreted redox proteins, including protein disulfide isomerase (PDI), ERp72, TRX, and quiescin sulfhydryl oxidase 1 (QSOX1).
1685:
1657:
1450:, GTP-binding/hydrolyzing, and isopeptidase activities. Unlike other members of the transglutaminase family, tTG can be found both in the intracellular and the extracellular spaces of various types of tissues and is found in many different organs including the heart, the liver, and the small intestine. Intracellular tTG is abundant in the
1740:. Evidence shows that intracellular tTG crosslinks itself to myosin. It is also believed that tTG may stabilize the structure of the dying cells during apoptosis by polymerizing the components of the cytoskeleton, therefore preventing the leakage of the cellular contents into the extracellular space.
1827:
and tumor biology. tTG expression is elevated in multiple cancer cell types and is implicated in drug resistance and metastasis due to its ability to promote mesenchymal transition and stem cell like properties. In its GTP bound form, tTG contributes to cancer cell survival and appears to be a cancer
1747:
activity: In the presence of GTP, it suggested to function as a G protein participating in signaling processes. Besides its transglutaminase activity, tTG is proposed to also act as kinase, and protein disulfide isomerase, and deamidase. This latter activity is important in the deamidation of gliadin
1648:
residues, namely Cys 370 and Cys 371. When this disulfide bond forms, the enzyme remains in an open confirmation but becomes catalytically inactive. The, oxidation/reduction of the disulfide bond serves as a third allosteric regulatory mechanism (along with GTP/GDP and Ca) for the activation of tTG.
2076:
Endoplasmic reticulum (ER)-resident protein 57 (ERp57), a protein in the ER that promotes folding of nascent proteins and is also present in the extracellular environment, has the cellular and biochemical characteristics for inactivating TG2. We found that ERp57 colocalizes with extracellular TG2 in
1622:
Crosslinking activity by tTG requires the binding of Ca ions. Multiple Ca can bind to a single tTG molecule. Specifically, tTG binds up to 6 calcium ions at 5 different binding sites. Mutations to these binding sites causing lower calcium affinity, decrease the enzyme's transglutaminase activity. In
1881:
diseases by affecting transcription, differentiation and migration and adhesion . Such neurological diseases are characterized in part by the abnormal aggregation of proteins due to the increased activity of protein crosslinking in the affected brain. Additionally, specific proteins associated with
1665:
Recent studies have suggested that interferon-γ may serve as an activator of extracellular tTG in the small intestine; these studies have a direct implication to the pathogenesis of celiac disease. Activation of tTG has been shown to be accompanied by large conformational changes, switching from a
1771:
tTG is the most comprehensively studied transglutaminase and has been associated with many diseases. However, none of these diseases are related to an enzyme deficiency. Indeed, thus far no disease has been attributed to the lack of tTG activity and this has been attested through the study of tTG
1976:
of TG2 are known to affect TG2 activity, which enables it to subsequently execute diverse biological functions in the cell. However, the importance of non-enzymatic interactions in regulating TG2 activities is yet to be revealed. Recent studies indicate that non-enzymatic interactions play
1546:
have demonstrated that these forms of TG2 adopt a "closed" conformation, whereas TG2 with the active site occupied by an inhibitory gluten peptide mimic or other similar inhibitors adopts an "open" conformation. In the open conformation the four domains of TG2 are arranged in an extended
1631:
inhibits the crosslinking activity of the enzyme. Therefore, intracellular tTG is mostly inactive due to the relatively high concentration of GTP/GDP and the low levels of calcium inside the cell. Although extracellular tTG is expected to be active due to the low concentration of
1660:
Figure 2: Cystein residues relevant in tTG activity. The disulfide bond between Cys 370 and Cys 371 has formed, therefore the enzyme is in an active conformation. The distance between Cys 370 and Cys 230 is 11.3 Å. Cys 277 is the cystein located within the active site of the
62:
36:
1591:(i.e. deamidation). The deamidation of glutamine residues catalyzed by tTG is thought to be linked to the pathological immune response to gluten in celiac disease. A schematic for the crosslinking and the deamidation reactions is provided in Figure 1.
3623:
Rossin F, Villella VR, D'Eletto M, Farrace MG, Esposito S, Ferrari E, Monzani R, Occhigrossi L, Pagliarini V, Sette C, Cozza G, Barlev NA, Falasca L, Fimia GM, Kroemer G, Raia V, Maiuri L, Piacentini M (July 2018).
4077:
Sblattero D, Berti I, Trevisiol C, Marzari R, Tommasini A, Bradbury A, Fasano A, Ventura A, Not T (May 2000). "Human recombinant tissue transglutaminase ELISA: an innovative diagnostic assay for celiac disease".
3984:
Martin A, Giuliano A, Collaro D, De Vivo G, Sedia C, Serretiello E, Gentile V (January 2013). "Possible involvement of transglutaminase-catalyzed reactions in the physiopathology of neurodegenerative diseases".
1860:
have showed promise in using tTG inhibitors as anti-cancer therapeutic agents. However, other studies have noted that tTG transamidation activity could be linked to the inhibition of tumor cell invasiveness.
1639:
and the high levels of calcium in the extracellular space, evidence has shown that extracellular tTG is mostly inactive. Recent studies suggest that extracellular tTG is kept inactive by the formation of a
1681:, TG2 is "turned off", due primarily to the oxidizing activity of endoplasmic reticulum protein 57 (ERp57). Thus, tTG is allosterically regulated by two separate proteins, Erp57 and TRX-1. (See Figure 4).
1518:
which catalyze the crosslinking of proteins by epsilon-(gamma-glutamyl)lysine isopeptide bonds. Similarly to other transglutaminases, tTG consists of a GTP/ GDP binding site, a
5103:
1587:
bond between the two substrates (i.e. crosslinking). Alternatively, the thioester intermediate can be hydrolyzed, resulting in the net conversion of the glutamine residue to
1856:, lower survival rates and generally poor prognosis. Cancer cells can be killed by increasing calcium levels through the activation of tTG transamidation activity.
1763:(Heat Shock Factor 1) and thus the body's response to heat shock. In the absence of tTG, the response to heat shock is impaired since the necessary trimer is not formed.
3727:
Dieterich W, Ehnis T, Bauer M, Donner P, Volta U, Riecken EO, Schuppan D (July 1997). "Identification of tissue transglutaminase as the autoantigen of celiac disease".
346:
245:
1360:
2025:
These Mutant ES Cells can be studied directly or used to generate mice with this gene knocked out. Study of these mice can shed light on the function of Tgm2: see
2746:
1379:
2017:
4582:
4577:
2388:"Transglutaminase-2 interaction with heparin: identification of a heparin binding site that regulates cell adhesion to fibronectin-transglutaminase-2 matrix"
1470:. Extracellular tTG has been linked to cell adhesion, ECM stabilization, wound healing, receptor signaling, cellular proliferation, and cellular motility.
1489:
atrophy. It has also been implicated in the pathophysiology of many other diseases, including such as many different cancers and neurogenerative diseases.
4383:
4224:"Physiological, pathological, and structural implications of non-enzymatic protein-protein interactions of the multifunctional human transglutaminase 2"
2870:
Han BG, Cho JW, Cho YD, Jeong KC, Kim SY, Lee BI (August 2010). "Crystal structure of human transglutaminase 2 in complex with adenosine triphosphate".
906:
3429:"Tissue transglutaminase has intrinsic kinase activity: identification of transglutaminase 2 as an insulin-like growth factor-binding protein-3 kinase"
887:
168:
1697:
tTG is expressed ubiquitously and is present in various cellular compartments, such as the cytosol, the nucleus, and the plasma membrane. It requires
4565:
1594:
2605:
Di
Sabatino A, Vanoli A, Giuffrida P, Luinetti O, Solcia E, Corazza GR (August 2012). "The function of tissue transglutaminase in celiac disease".
1784:. It was first associated with celiac disease in 1997 when the enzyme was found to be the antigen recognized by the antibodies specific to celiac.
1736:
tTG is thought to be involved in the regulation of the cytoskeleton by crosslinking various cytoskeletal proteins including myosin, actin, and
5078:
4572:
5326:
4322:
2153:
2135:
4560:
3878:"Presence of tissue transglutaminase in granular endoplasmic reticulum is characteristic of melanized neurons in Parkinson's disease brain"
1755:
tTG also presents PDI (Protein
Disulfide Isomerase) activity. Based on its PDI activity, tTG plays an important role in the regulation of
3235:"Cytosolic guanine nucledotide binding deficient form of transglutaminase 2 (R580a) potentiates cell death in oxygen glucose deprivation"
4732:
4342:
2649:"Functional significance of five noncanonical Ca2+-binding sites of human transglutaminase 2 characterized by site-directed mutagenesis"
2555:"Interferon-γ activates transglutaminase 2 via a phosphatidylinositol-3-kinase-dependent pathway: implications for celiac sprue therapy"
1611:. Once synthesized, most of the protein is found in the cytoplasm, plasma membrane and ECM, but a small fraction is translocated to the
2813:"Structural basis for the guanine nucleotide-binding activity of tissue transglutaminase and its regulation of transamidation activity"
1688:
Figure 4: The proteins that allosterically regulate tTG. On the left Erp57 which oxidizes tTG and on the right TRX-1 which reduces tTG.
332:
4700:
1145:
1138:
797:
positive regulation of cytosolic calcium ion concentration involved in phospholipase C-activating G protein-coupled signaling pathway
4765:
4516:
4454:
3358:
2722:
1803:
responses that eventually result in the production of anti-transglutaminase antibodies IgA and IgG. tTG specifically deamidates the
1948:
diseases. This indicates that tTG inhibitors could also serve as a tool to mitigate the progression of tTG brain related diseases.
309:
5260:
4511:
4506:
1785:
1372:
4449:
2122:
2101:
4123:"Cardiac fibrosis can be attenuated by blocking the activity of transglutaminase 2 using a selective small-molecule inhibitor"
4030:"Tissue transglutaminase overexpression does not modify the disease phenotype of the R6/2 mouse model of Huntington's disease"
2058:
Transglutaminase 2 (TG2) is a ubiquitously expressed (intracellular as well as extracellular) protein, with multiple modes of
4464:
4376:
1323:
1299:
231:
206:
4921:
4284:
2647:
Király R, Csosz E, Kurtán T, Antus S, Szigeti K, Simon-Vecsei Z, Korponay-Szabó IR, Keresztessy Z, Fésüs L (December 2009).
2118:
1579:
intermediate. The thioester intermediate can then be attacked by the surface amine of a second substrate (typically from a
59:
5131:
4629:
4609:
4523:
2059:
2097:
1994:
4550:
4444:
4439:
2386:
Lortat-Jacob H, Burhan I, Scarpellini A, Thomas A, Imberty A, Vivès RR, Johnson T, Gutierrez A, Verderio EA (May 2012).
148:
3821:"Transglutaminase 2 takes center stage as a cancer cell survival factor and therapy target: Transglutaminase in cancer"
1466:. In the extracellular space, tTG binds to proteins of the extracellular matrix (ECM), binding particularly tightly to
4712:
4486:
2176:"Protein transamidation by transglutaminase 2 in cells: a disputed Ca2+-dependent action of a multifunctional protein"
1957:
1924:(>90%) for identifying celiac disease. Modern anti-tTG assays rely on a human recombinant protein as an antigen.
345:
244:
5108:
5071:
5036:
4722:
4555:
4429:
4906:
5022:
5009:
4996:
4983:
4970:
4957:
4944:
4670:
4646:
4597:
4481:
4434:
4409:
4369:
4315:
338:
237:
4916:
1317:
4870:
4813:
4675:
4400:
1897:. Specifically, in kidney fibrosis, tTG contributes to the stabilization and accumulation of the ECM affecting
1718:
1412:
1210:
951:
156:
20:
1446:(protein degradation). Aside from its crosslinking function, tTG catalyzes other types of reactions including
1304:
4818:
4717:
1932:
It's still experimental to use tTG as a form of surgical glue. It is also being studied as an attenuator of
1916:
has superseded older serological tests (anti-endomysium, anti-gliadin, and anti-reticulin) and has a strong
1878:
1853:
932:
5113:
4685:
4680:
4545:
2963:"Structural Basis for Antigen Recognition by Transglutaminase 2-specific Autoantibodies in Celiac Disease"
2342:
2001:
Mutant Mouse
Embryonic Stem Cell Clones. These are the known targeted mutations for this gene in a mouse.
1874:
1669:
1624:
1543:
1539:
1485:
causes a pathological immune response resulting in the inflammation of the small intestine and subsequent
1384:
5336:
5295:
5064:
4839:
4758:
4665:
4660:
3392:
Fesus L, Piacentini M (October 2002). "Transglutaminase 2: an enigmatic enzyme with diverse functions".
2063:
2052:
1973:
1628:
1535:
1292:
220:
4911:
2961:
Chen X, Hnida K, Graewert MA, Andersen JT, Iversen R, Tuukkanen A, Svergun D, Sollid LM (August 2015).
1725:. It has been noted that tTG may have very different activity in different cell types. For example, in
1227:
4727:
4614:
4601:
4501:
4308:
3246:
2824:
2048:
1714:
1678:
1616:
135:
3876:
Wilhelmus MM, Verhaar R, Andringa G, Bol JG, Cras P, Shan L, Hoozemans JJ, Drukarch B (March 2011).
3133:"Endoplasmic reticulum-resident protein 57 (ERp57) oxidatively inactivates human transglutaminase 2"
2347:
4875:
4459:
2040:
1921:
1917:
1608:
1320:
1222:
1092:
1088:
1084:
1037:
1033:
1029:
1567:
group from a Cys residue in the active site of tTG. The thiol group attacks the carboxamide of a
1244:
5234:
4808:
4261:
4103:
4028:
Kumar A, Kneynsberg A, Tucholski J, Perry G, van Groen T, Detloff PJ, Lesort M (September 2012).
4010:
3915:
3858:
3752:
3709:
2740:
2678:
2368:
2205:
1789:
1698:
1531:
180:
3468:
Hasegawa G, Suwa M, Ichikawa Y, Ohtsuka T, Kumagai S, Kikuchi M, Sato Y, Saito Y (August 2003).
1121:
1100:
1096:
1058:
1025:
1021:
5331:
4690:
4419:
4253:
4204:
4152:
4095:
4059:
4002:
3966:
3907:
3850:
3801:
3744:
3701:
3655:
3602:
3548:
3499:
3450:
3409:
3374:
3354:
3323:
3274:
3215:
3164:
3104:
3044:
2994:
2943:
2887:
2852:
2793:
2728:
2718:
2670:
2622:
2584:
2524:
2468:
2419:
2360:
2333:
McConkey DJ, Orrenius S (October 1997). "The role of calcium in the regulation of apoptosis".
2315:
2269:
2197:
1945:
1944:. tTG inhibitors have also been shown to inhibit the formation of toxic inclusions related to
1857:
1841:
1793:
1515:
1486:
1311:
128:
52:
1598:
Figure 1: Transamidation (crosslinking) and deamidation mechanisms of tissue transglutaminase
5270:
5123:
4854:
4849:
4823:
4751:
4655:
4619:
4243:
4235:
4194:
4186:
4142:
4134:
4087:
4049:
4041:
3994:
3956:
3946:
3897:
3889:
3840:
3832:
3791:
3783:
3736:
3691:
3645:
3637:
3592:
3582:
3538:
3530:
3489:
3481:
3440:
3401:
3364:
3346:
3313:
3305:
3264:
3254:
3205:
3195:
3154:
3144:
3094:
3084:
3036:
3028:
2984:
2974:
2933:
2923:
2879:
2842:
2832:
2783:
2773:
2660:
2614:
2574:
2566:
2514:
2506:
2458:
2450:
2409:
2399:
2352:
2305:
2259:
2251:
2187:
1941:
1937:
1419:
590:
583:
425:
356:
300:
255:
3013:
1547:
configuration, allowing for catalytic activity, whereas in the closed conformation the two
1280:
5244:
5239:
5229:
5224:
5219:
5214:
5209:
5204:
4901:
4885:
4798:
4695:
4396:
4361:
4288:
3014:"Spotlight on the transglutaminase 2 gene: a focus on genomic and transcriptional aspects"
1960:. Enzymatic interactions are formed between TG2 and its substrate proteins containing the
1845:
1837:
1749:
597:
400:
176:
3571:"The Role of Tissue Transglutaminase in Cancer Cell Initiation, Survival and Progression"
2715:
Transglutaminases : multiple functional modifiers and targets for new drug discovery
1256:
4248:
4223:
3250:
2828:
1215:
35:
5194:
5189:
5184:
5179:
5174:
5169:
5164:
5159:
5154:
5050:
4939:
4880:
4199:
4174:
4147:
4122:
4054:
4029:
3961:
3934:
3902:
3877:
3845:
3820:
3796:
3771:
3650:
3625:
3597:
3570:
3543:
3518:
3494:
3469:
3369:
3350:
3318:
3293:
3269:
3234:
3210:
3183:
3159:
3132:
3099:
3072:
2989:
2962:
2938:
2911:
2788:
2761:
2579:
2554:
2519:
2494:
2463:
2438:
2414:
2387:
2264:
2239:
2026:
1828:
driver. tTG is upregulated in cancer cells and tissues in many cancer types, including
1781:
1615:, where it participates in the control of its own expression through the regulation of
1478:
1355:
3626:"TG2 regulates the heat-shock response by the post-translational modification of HSF1"
3405:
2847:
2812:
1335:
821:
816:
811:
806:
801:
796:
791:
786:
781:
776:
771:
766:
761:
756:
751:
746:
730:
725:
720:
715:
710:
705:
700:
695:
690:
685:
669:
664:
659:
654:
649:
644:
639:
5320:
5305:
5095:
4844:
4803:
4091:
3893:
3534:
2665:
2648:
2192:
2175:
2004:
1833:
1722:
1706:
1702:
1607:
The expression of tTG is regulated at the transcriptional level depending on complex
1588:
1527:
1503:
1330:
626:
4265:
4107:
4045:
4014:
3862:
3756:
3713:
3345:. International Review of Cell and Molecular Biology. Vol. 294. pp. 1–97.
2682:
2439:"Tissue transglutaminase is an integrin-binding adhesion coreceptor for fibronectin"
2372:
2209:
5290:
5087:
4793:
2883:
1977:
physiological roles and enable diverse TG2 functions in a context-specific manner.
1824:
1756:
1612:
1551:
are folded in on the catalytic core domain which includes the residue Cys-277. The
1474:
1459:
1455:
418:
197:
3919:
3294:"Transglutaminase 2: Friend or foe? The discordant role in neurons and astrocytes"
1988:
Marker Symbol for Mouse Gene. This symbol is assigned to the genomic locus by the
160:
4281:
4190:
3787:
3259:
3200:
2778:
2618:
184:
139:, G-ALPHA-h, GNAH, HEL-S-45, TG2, TGC, TG(C), transglutaminase 2, G(h), hTG2, tTG
5275:
5146:
5017:
4952:
4788:
4634:
4392:
4293:
A collection of substrates and interaction partners of TG2 is accessible in the
3470:"A novel function of tissue-type transglutaminase: protein disulphide isomerase"
2158:
National Center for
Biotechnology Information, U.S. National Library of Medicine
2140:
National Center for
Biotechnology Information, U.S. National Library of Medicine
1936:
in certain tumors. tTG shows promise as a potential therapeutic target to treat
1650:
1636:
1523:
1519:
1467:
1447:
1443:
1435:
1339:
5045:
3569:
Tabolacci C, De
Martino A, Mischiati C, Feriotto G, Beninati S (January 2019).
3071:
Jin X, Stamnaes J, Klöck C, DiRaimondo TR, Sollid LM, Khosla C (October 2011).
2817:
Proceedings of the
National Academy of Sciences of the United States of America
1869:
tTG is believed to contribute to several neurodegenerative disorders including
1442:
residue, creating an inter- or intramolecular bond that is highly resistant to
501:
5280:
4294:
4239:
4138:
3998:
2255:
1933:
1898:
1849:
1584:
1552:
1548:
317:
214:
164:
4121:
Wang Z, Stuckey DJ, Murdoch CE, Camelliti P, Lip GY, Griffin M (April 2018).
3149:
2732:
1555:
only shows minor structural changes between the two different conformations.
5285:
4991:
4965:
4175:"New insight into transglutaminase 2 and link to neurodegenerative diseases"
3641:
3184:"Transglutaminase 2 undergoes a large conformational change upon activation"
3089:
2979:
2928:
2762:"Transglutaminase 2 undergoes a large conformational change upon activation"
2570:
2404:
2066:
1961:
1870:
1804:
1799:
that are crosslinked to tTG are able to stimulate transglutaminase specific
1730:
1710:
1641:
1576:
1568:
1463:
1439:
851:
561:
439:
384:
371:
283:
270:
172:
4257:
4208:
4156:
4099:
4063:
4006:
3970:
3951:
3911:
3854:
3805:
3705:
3659:
3606:
3552:
3503:
3454:
3445:
3428:
3413:
3378:
3327:
3278:
3219:
3168:
3108:
3048:
2998:
2947:
2891:
2856:
2837:
2797:
2674:
2626:
2588:
2528:
2472:
2423:
2356:
2319:
2273:
2201:
3748:
3696:
3679:
3587:
3519:"A role for tissue transglutaminase in alpha-gliadin peptide cytotoxicity"
2910:
Stamnaes J, Pinkas DM, Fleckenstein B, Khosla C, Sollid LM (August 2010).
2454:
2364:
1185:
1180:
5300:
4424:
3040:
2696:
1913:
1909:
1886:
1829:
1737:
1733:
knocking out the gene expression for tTG is beneficial to cell survival.
1701:
as a cofactor for transamidation activity. Transcription is increased by
1645:
1169:
996:
977:
3032:
1666:
compact (inactive) to an extended (active) conformation. (see Figure 3)
1415:
1268:
3740:
3485:
2510:
2044:
1969:
1812:
1684:
1673:
Figure 3: Compact (inactive) and extended (active) conformations of tTG
1633:
1572:
1451:
1423:
1287:
963:
918:
752:
phospholipase C-activating G protein-coupled receptor signaling pathway
3309:
2011:
Example structure of targeted conditional mutant allele for this gene
762:
negative regulation of endoplasmic reticulum calcium ion concentration
105:
101:
97:
93:
89:
85:
81:
5004:
4774:
4705:
4533:
4528:
4469:
3836:
2294:"The role of transglutaminase-2 and its substrates in human diseases"
2070:
1965:
1894:
1808:
1807:
residues creating epitopes that increase the binding affinity of the
1800:
1744:
1726:
1580:
1482:
1431:
1408:
1367:
1263:
1251:
1239:
1153:
873:
1729:, tTG supports the survival of cells subjected to injury whereas in
1656:
1571:
residue on the surface of a protein or peptide substrate, releasing
1418:) of the protein-glutamine γ-glutamyltransferases family (or simply
3935:"Physio-pathological roles of transglutaminase-catalyzed reactions"
3233:
Colak G, Keillor JW, Johnson GV (January 2011). Polymenis M (ed.).
1563:
The catalytic mechanism for crosslinking in human tTG involves the
5265:
5199:
4978:
4587:
4474:
1705:. Among its many supposed functions, it appears to play a role in
1683:
1668:
1655:
1593:
1564:
1427:
2310:
2293:
1848:. Higher tTG expression also correlates with higher instances of
836:
832:
4491:
1890:
1760:
1275:
152:
5060:
5056:
4747:
4365:
4304:
3073:"Activation of extracellular transglutaminase 2 by thioredoxin"
2437:
Akimov SS, Krylov D, Fleischman LF, Belkin AM (February 2000).
606:
4300:
767:
positive regulation of mitochondrial calcium ion concentration
3680:"Transglutaminase diseases: from biochemistry to the bedside"
1989:
1462:. Intracellular tTG is thought to play an important role in
2240:"Role of transglutaminase 2 in celiac disease pathogenesis"
4743:
3182:
Pinkas DM, Strop P, Brunger AT, Khosla C (December 2007).
2760:
Pinkas DM, Strop P, Brunger AT, Khosla C (December 2007).
1748:
peptides, thus playing important role in the pathology of
792:
positive regulation of I-kappaB kinase/NF-kappaB signaling
3517:
Sakly W, Thomas V, Quash G, El Alaoui S (December 2006).
3131:
Yi MC, Melkonian AV, Ousey JA, Khosla C (February 2018).
2559:
The
Journal of Pharmacology and Experimental Therapeutics
1481:, a lifelong illness in which the consumption of dietary
802:
isopeptide cross-linking via N6-(L-isoglutamyl)-L-lysine
408:
817:
positive regulation of smooth muscle cell proliferation
3933:
Ricotta M, Iannuzzi M, Vivo GD, Gentile V (May 2010).
1583:
residue). The end product of the reaction is a stable
5034:
4297:, an interactive transglutaminase substrate database.
1956:
TG2 participates in both enzymatic and non-enzymatic
1823:
Recent studies suggest that tTG also plays a role in
1422:
family). Like other transglutaminases, it crosslinks
573:
5104:
Branched-chain alpha-keto acid dehydrogenase complex
2493:
Griffin M, Casadio R, Bergamini CM (December 2002).
650:
protein-glutamine gamma-glutamyltransferase activity
5253:
5145:
5122:
5094:
4930:
4894:
4863:
4832:
4781:
4645:
4596:
4408:
2335:
2292:Facchiano F, Facchiano A, Facchiano AM (May 2006).
1378:
1366:
1354:
1349:
1329:
1310:
1298:
1286:
1274:
1262:
1250:
1238:
1233:
1221:
1209:
1204:
1199:
1114:
1077:
1051:
1014:
3292:Quinn BR, Yunes-Medina L, Johnson GV (July 2018).
2872:International Journal of Biological Macromolecules
2114:
2112:
2110:
2093:
2091:
2089:
782:branching involved in salivary gland morphogenesis
4351:: HUMAN TISSUE TRANSGLUTAMINASE IN GDP BOUND FORM
3564:
3562:
2912:"Redox regulation of transglutaminase 2 activity"
1940:, through the activity of a highly selective tTG
355:
254:
3770:Murray JA, Frey MR, Oliva-Hemker M (June 2018).
3012:Bianchi N, Beninati S, Bergamini CM (May 2018).
2905:
2903:
2901:
1885:tTG has also been linked to the pathogenesis of
1514:TG2 is a multifunctional enzyme that belongs to
19:"TGM2" redirects here. For the Tetris game, see
4222:Kanchan K, Fuxreiter M, Fésüs L (August 2015).
2553:Diraimondo TR, Klöck C, Khosla C (April 2012).
2039:Endoplasmic reticulum protein 57 (Erp57), is a
16:Protein-coding gene in the species Homo sapiens
3673:
3671:
3669:
3618:
3616:
2708:
2706:
2642:
2640:
2638:
2636:
2495:"Transglutaminases: nature's biological glues"
2238:Klöck C, Diraimondo TR, Khosla C (July 2012).
2119:GRCm38: Ensembl release 89: ENSMUSG00000037820
2069:bond between Cys-370-Cys-371 that renders the
5072:
4759:
4377:
4316:
3343:Cellular functions of tissue transglutaminase
2548:
2546:
2544:
2542:
2540:
2538:
2174:Király R, Demény M, Fésüs L (December 2011).
1454:but smaller amounts can also be found in the
8:
4583:2-acylglycerol-3-phosphate O-acyltransferase
4578:1-acylglycerol-3-phosphate O-acyltransferase
787:positive regulation of inflammatory response
4168:
4166:
2600:
2598:
2287:
2285:
2283:
2098:GRCh38: Ensembl release 89: ENSG00000198959
1200:Protein-glutamine gamma-glutamyltransferase
5079:
5065:
5057:
4766:
4752:
4744:
4384:
4370:
4362:
4323:
4309:
4301:
3126:
3124:
3122:
3120:
3118:
2811:Liu S, Cerione RA, Clardy J (March 2002).
2745:: CS1 maint: location missing publisher (
2488:
2486:
2484:
2482:
1815:, initiating an adaptive immune response.
1346:
847:
622:
396:
295:
192:
70:
4247:
4198:
4146:
4053:
3960:
3950:
3901:
3844:
3795:
3695:
3649:
3596:
3586:
3542:
3493:
3444:
3368:
3317:
3268:
3258:
3209:
3199:
3158:
3148:
3098:
3088:
2988:
2978:
2937:
2927:
2846:
2836:
2787:
2777:
2664:
2578:
2518:
2462:
2413:
2403:
2346:
2309:
2263:
2191:
1623:contrast, the binding of one molecule of
4080:The American Journal of Gastroenterology
2169:
2167:
1979:
807:positive regulation of apoptotic process
731:collagen-containing extracellular matrix
5041:
4338:
2233:
2231:
2229:
2227:
2225:
2223:
2221:
2219:
2085:
4573:Glycerol-3-phosphate O-acyltransferase
3066:
3064:
3062:
3060:
3058:
2738:
2697:"Entrez Gene: TGM2 transglutaminase 2"
2073:inactive in the extracellular matrix.
1196:
691:intrinsic component of plasma membrane
25:
4566:Lecithin—cholesterol acyltransferase
3939:World Journal of Biological Chemistry
1759:, by catalyzing the trimerization of
1502:The human tTG gene is located on the
360:
321:
316:
259:
218:
213:
7:
4561:Glyceronephosphate O-acyltransferase
4228:Cellular and Molecular Life Sciences
3678:Lorand L, Iismaa SE (January 2019).
3523:Clinical and Experimental Immunology
2713:Hitomi K, Kojima S, Fesus L (2015).
1780:tTG is best known for its link with
777:positive regulation of cell adhesion
4733:Sulfoacetaldehyde acetyltransferase
4425:Acetyl-Coenzyme A acetyltransferase
3433:The Journal of Biological Chemistry
3137:The Journal of Biological Chemistry
3077:The Journal of Biological Chemistry
2967:The Journal of Biological Chemistry
2916:The Journal of Biological Chemistry
2392:The Journal of Biological Chemistry
4649:: converted into alkyl on transfer
3351:10.1016/B978-0-12-394305-7.00001-X
3341:Nurminskaya MV, Belkin AM (2012).
1811:peptide to the antigen presenting
1111:
1074:
1048:
1011:
987:
968:
942:
923:
897:
878:
578:
496:
434:
413:
14:
4455:Chloramphenicol acetyltransferase
3819:Eckert, Richard L. (2019-01-29).
3427:Mishra S, Murphy LJ (June 2004).
1407:) is a 78-kDa, calcium-dependent
5044:
4507:Carnitine O-palmitoyltransferase
4341:
4173:Min B, Chung KC (January 2018).
4092:10.1111/j.1572-0241.2000.02018.x
3894:10.1111/j.1750-3639.2010.00429.x
3535:10.1111/j.1365-2249.2006.03236.x
3298:Journal of Neuroscience Research
2666:10.1111/j.1742-4658.2009.07420.x
2193:10.1111/j.1742-4658.2011.08345.x
2016:
1968:donor groups in the presence of
1889:in various organs including the
1792:in which a cellular response to
1786:Anti-transglutaminase antibodies
596:
589:
582:
344:
337:
331:
308:
243:
236:
230:
205:
34:
4450:Beta-galactoside transacetylase
4046:10.1016/j.expneurol.2012.05.015
670:protein domain specific binding
4465:Serotonin N-acetyl transferase
4412:: other than amino-acyl groups
3394:Trends in Biochemical Sciences
2884:10.1016/j.ijbiomac.2010.04.023
1983:Mouse Mutant Alleles for Tgm2
607:More reference expression data
562:More reference expression data
1:
4630:Keratinocyte transglutaminase
4610:Gamma-glutamyl transpeptidase
4524:Serine C-palmitoyltransferase
3406:10.1016/S0968-0004(02)02182-5
2060:Post-translational regulation
2043:molecule involved in loading
520:tunica media of zone of aorta
329:
228:
5327:Genes on human chromosome 20
4551:Aminolevulinic acid synthase
4445:Acetyl-CoA C-acyltransferase
4440:Dihydrolipoyl transacetylase
4191:10.5483/BMBRep.2018.51.1.227
3788:10.1053/j.gastro.2017.12.026
3260:10.1371/journal.pone.0016665
3201:10.1371/journal.pbio.0050327
2779:10.1371/journal.pbio.0050327
2619:10.1016/j.autrev.2012.01.007
4713:2-hydroxyglutarate synthase
2443:The Journal of Cell Biology
2244:Seminars in Immunopathology
812:protein homooligomerization
5353:
5132:Epidermal transglutaminase
5109:Oxoglutarate dehydrogenase
4723:2-isopropylmalate synthase
4556:Beta-ketoacyl-ACP synthase
4430:N-Acetylglutamate synthase
18:
4922:Michaelis–Menten kinetics
4671:Decylhomocitrate synthase
4482:Histone acetyltransferase
4435:Choline acetyltransferase
4336:
4240:10.1007/s00018-015-1909-z
4139:10.1038/s41419-018-0573-2
3999:10.1007/s00726-011-1081-1
2256:10.1007/s00281-012-0305-0
2154:"Mouse PubMed Reference:"
2136:"Human PubMed Reference:"
2024:
2015:
2010:
1982:
1644:bond between two vicinal
1345:
1184:
1179:
1175:
1168:
1152:
1146:Chr 2: 157.96 – 157.99 Mb
1133:
1118:
1081:
1070:
1055:
1018:
1007:
994:
990:
975:
971:
962:
949:
945:
930:
926:
917:
904:
900:
885:
881:
872:
857:
850:
846:
830:
757:salivary gland cavitation
625:
621:
604:
581:
572:
559:
508:
499:
446:
437:
407:
399:
395:
378:
365:
328:
307:
298:
294:
277:
264:
227:
204:
195:
191:
146:
143:
133:
126:
121:
78:
73:
56:
51:
46:
42:
33:
28:
5286:Filaggrin (Citrullinate)
4814:Diffusion-limited enzyme
4676:2-methylcitrate synthase
4127:Cell Death & Disease
3825:Molecular Carcinogenesis
3150:10.1074/jbc.RA117.001382
1139:Chr 20: 38.13 – 38.17 Mb
747:apoptotic cell clearance
645:acyltransferase activity
21:Tetris: The Grand Master
5137:Tissue transglutaminase
4718:3-propylmalate synthase
4625:Tissue transglutaminase
3642:10.15252/embr.201745067
3474:The Biochemical Journal
3090:10.1074/jbc.M111.287490
3021:The Biochemical Journal
2980:10.1074/jbc.M115.669895
2929:10.1074/jbc.M109.097162
2571:10.1124/jpet.111.187385
2499:The Biochemical Journal
2405:10.1074/jbc.M111.337089
2298:Frontiers in Bioscience
1854:chemotherapy resistance
1717:development as well as
1397:Tissue transglutaminase
822:blood vessel remodeling
548:external carotid artery
482:upper lobe of left lung
5261:Acetylcholine receptor
5114:Pyruvate dehydrogenase
4686:3-ethylmalate synthase
4681:2-ethylmalate synthase
4546:Acyltransferase like 2
4034:Experimental Neurology
3952:10.4331/wjbc.v1.i5.181
3446:10.1074/jbc.M311919200
2838:10.1073/pnas.042454899
2357:10.1006/bbrc.1997.7409
1689:
1674:
1662:
1599:
536:mesenteric lymph nodes
5296:Sp100 nuclear antigen
4907:Eadie–Hofstee diagram
4840:Allosteric regulation
4666:Citrate (Re)-synthase
4661:Decylcitrate synthase
4602:Aminoacyltransferases
4502:palmitoyltransferases
4282:Endomysial antibodies
3697:10.1096/fj.201801544R
3588:10.3390/medsci7020019
2455:10.1083/jcb.148.4.825
2053:endoplasmic reticulum
2049:MHC class I molecules
1767:Clinical significance
1687:
1672:
1659:
1617:transcription factors
1597:
772:peptide cross-linking
716:endoplasmic reticulum
701:extracellular exosome
454:right coronary artery
221:Chromosome 20 (human)
4917:Lineweaver–Burk plot
4728:Homocitrate synthase
4615:Peptidyl transferase
2607:Autoimmunity Reviews
1788:result in a form of
1715:extracellular matrix
1679:extracellular matrix
726:extracellular matrix
640:transferase activity
458:left coronary artery
362:2 H1|2 78.72 cM
323:Chromosome 2 (mouse)
74:List of PDB id codes
47:Available structures
4460:N-acetyltransferase
3251:2011PLoSO...616665C
3033:10.1042/BCJ20170601
2829:2002PNAS...99.2743L
512:conjunctival fornix
462:canal of the cervix
4876:Enzyme superfamily
4809:Enzyme promiscuity
4420:acetyltransferases
4287:2021-05-12 at the
3741:10.1038/nm0797-797
3486:10.1042/BJ20021084
2511:10.1042/BJ20021234
1858:Preclinical trials
1790:gluten sensitivity
1690:
1675:
1663:
1600:
1575:, and producing a
1549:C-terminal domains
1534:of TG2 with bound
1532:Crystal structures
952:ENSMUSG00000037820
740:Biological process
679:Cellular component
633:Molecular function
478:right uterine tube
5314:
5313:
5032:
5031:
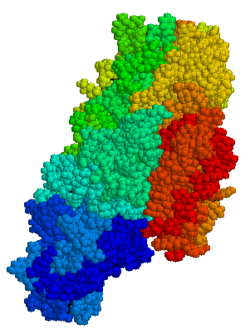
4741:
4740:
4691:ATP citrate lyase
4359:
4358:
3480:(Pt 3): 793–803.
3310:10.1002/jnr.24239
2032:
2031:
1946:neurodegenerative
1842:pancreatic cancer
1553:N-terminal domain
1516:transglutaminases
1394:
1393:
1390:
1389:
1293:metabolic pathway
1195:
1194:
1191:
1190:
1164:
1163:
1129:
1128:
1108:
1107:
1066:
1065:
1045:
1044:
1003:
1002:
984:
983:
958:
957:
939:
938:
913:
912:
894:
893:
842:
841:
660:metal ion binding
617:
616:
613:
612:
568:
567:
555:
554:
493:
492:
391:
390:
290:
289:
117:
116:
113:
112:
57:Ortholog search:
5344:
5271:Apolipoprotein H
5124:Transglutaminase
5081:
5074:
5067:
5058:
5049:
5048:
5040:
4912:Hanes–Woolf plot
4855:Enzyme activator
4850:Enzyme inhibitor
4824:Enzyme catalysis
4768:
4761:
4754:
4745:
4701:HMG-CoA synthase
4656:Citrate synthase
4620:Transglutaminase
4397:acyltransferases
4386:
4379:
4372:
4363:
4345:
4325:
4318:
4311:
4302:
4270:
4269:
4251:
4219:
4213:
4212:
4202:
4170:
4161:
4160:
4150:
4118:
4112:
4111:
4074:
4068:
4067:
4057:
4025:
4019:
4018:
3981:
3975:
3974:
3964:
3954:
3930:
3924:
3923:
3905:
3873:
3867:
3866:
3848:
3837:10.1002/mc.22986
3816:
3810:
3809:
3799:
3782:(8): 2005–2008.
3776:Gastroenterology
3772:"Celiac Disease"
3767:
3761:
3760:
3724:
3718:
3717:
3699:
3675:
3664:
3663:
3653:
3620:
3611:
3610:
3600:
3590:
3575:Medical Sciences
3566:
3557:
3556:
3546:
3514:
3508:
3507:
3497:
3465:
3459:
3458:
3448:
3424:
3418:
3417:
3389:
3383:
3382:
3372:
3338:
3332:
3331:
3321:
3304:(7): 1150–1158.
3289:
3283:
3282:
3272:
3262:
3230:
3224:
3223:
3213:
3203:
3179:
3173:
3172:
3162:
3152:
3143:(8): 2640–2649.
3128:
3113:
3112:
3102:
3092:
3083:(43): 37866–73.
3068:
3053:
3052:
3027:(9): 1643–1667.
3018:
3009:
3003:
3002:
2992:
2982:
2973:(35): 21365–75.
2958:
2952:
2951:
2941:
2931:
2907:
2896:
2895:
2867:
2861:
2860:
2850:
2840:
2808:
2802:
2801:
2791:
2781:
2757:
2751:
2750:
2744:
2736:
2710:
2701:
2700:
2693:
2687:
2686:
2668:
2653:The FEBS Journal
2644:
2631:
2630:
2602:
2593:
2592:
2582:
2550:
2533:
2532:
2522:
2505:(Pt 2): 377–96.
2490:
2477:
2476:
2466:
2434:
2428:
2427:
2417:
2407:
2398:(22): 18005–17.
2383:
2377:
2376:
2350:
2330:
2324:
2323:
2313:
2289:
2278:
2277:
2267:
2235:
2214:
2213:
2195:
2180:The FEBS Journal
2171:
2162:
2161:
2150:
2144:
2143:
2132:
2126:
2116:
2105:
2095:
2020:
1980:
1938:cardiac fibrosis
1520:catalytic domain
1434:residue and a γ-
1420:transglutaminase
1399:(abbreviated as
1347:
1197:
1177:
1176:
1148:
1141:
1124:
1112:
1103:
1075:
1071:RefSeq (protein)
1061:
1049:
1040:
1012:
988:
969:
943:
924:
898:
879:
848:
623:
609:
600:
593:
586:
579:
564:
540:efferent ductule
504:
502:Top expressed in
497:
466:popliteal artery
442:
440:Top expressed in
435:
414:
397:
387:
374:
363:
348:
341:
335:
324:
312:
296:
286:
273:
262:
247:
240:
234:
223:
209:
193:
187:
185:TGM2 - orthologs
138:
131:
108:
71:
65:
44:
43:
38:
26:
5352:
5351:
5347:
5346:
5345:
5343:
5342:
5341:
5317:
5316:
5315:
5310:
5249:
5141:
5118:
5090:
5085:
5055:
5043:
5035:
5033:
5028:
4940:Oxidoreductases
4926:
4902:Enzyme kinetics
4890:
4886:List of enzymes
4859:
4828:
4799:Catalytic triad
4777:
4772:
4742:
4737:
4696:Malate synthase
4641:
4592:
4404:
4390:
4360:
4355:
4352:
4346:
4332:
4329:
4289:Wayback Machine
4278:
4273:
4234:(16): 3009–35.
4221:
4220:
4216:
4172:
4171:
4164:
4120:
4119:
4115:
4076:
4075:
4071:
4027:
4026:
4022:
3983:
3982:
3978:
3932:
3931:
3927:
3882:Brain Pathology
3875:
3874:
3870:
3818:
3817:
3813:
3769:
3768:
3764:
3729:Nature Medicine
3726:
3725:
3721:
3677:
3676:
3667:
3622:
3621:
3614:
3568:
3567:
3560:
3516:
3515:
3511:
3467:
3466:
3462:
3439:(23): 23863–8.
3426:
3425:
3421:
3391:
3390:
3386:
3361:
3340:
3339:
3335:
3291:
3290:
3286:
3232:
3231:
3227:
3181:
3180:
3176:
3130:
3129:
3116:
3070:
3069:
3056:
3016:
3011:
3010:
3006:
2960:
2959:
2955:
2922:(33): 25402–9.
2909:
2908:
2899:
2869:
2868:
2864:
2810:
2809:
2805:
2759:
2758:
2754:
2737:
2725:
2712:
2711:
2704:
2695:
2694:
2690:
2659:(23): 7083–96.
2646:
2645:
2634:
2604:
2603:
2596:
2552:
2551:
2536:
2492:
2491:
2480:
2436:
2435:
2431:
2385:
2384:
2380:
2348:10.1.1.483.2738
2332:
2331:
2327:
2291:
2290:
2281:
2237:
2236:
2217:
2186:(24): 4717–39.
2173:
2172:
2165:
2152:
2151:
2147:
2134:
2133:
2129:
2117:
2108:
2096:
2087:
2083:
2062:, including an
2037:
1954:
1930:
1907:
1867:
1846:cervical cancer
1838:prostate cancer
1821:
1778:
1772:knockout mice.
1769:
1750:coeliac disease
1719:differentiation
1695:
1609:signal cascades
1605:
1561:
1512:
1506:(20q11.2-q12).
1504:20th chromosome
1500:
1495:
1186:View/Edit Mouse
1181:View/Edit Human
1144:
1137:
1134:Location (UCSC)
1120:
1099:
1095:
1091:
1087:
1083:
1057:
1036:
1032:
1028:
1024:
1020:
933:ENSG00000198959
826:
735:
721:plasma membrane
674:
655:protein binding
605:
595:
594:
588:
587:
560:
551:
546:
542:
538:
534:
530:
528:right lung lobe
526:
524:ascending aorta
522:
518:
514:
500:
489:
484:
480:
476:
472:
470:tibial arteries
468:
464:
460:
456:
452:
438:
382:
369:
361:
351:
350:
349:
342:
322:
299:Gene location (
281:
268:
260:
250:
249:
248:
241:
219:
196:Gene location (
147:
134:
127:
80:
58:
24:
17:
12:
11:
5:
5350:
5348:
5340:
5339:
5334:
5329:
5319:
5318:
5312:
5311:
5309:
5308:
5303:
5298:
5293:
5288:
5283:
5278:
5273:
5268:
5263:
5257:
5255:
5251:
5250:
5248:
5247:
5242:
5237:
5232:
5227:
5222:
5217:
5212:
5207:
5202:
5197:
5192:
5187:
5182:
5177:
5172:
5167:
5162:
5157:
5151:
5149:
5143:
5142:
5140:
5139:
5134:
5128:
5126:
5120:
5119:
5117:
5116:
5111:
5106:
5100:
5098:
5092:
5091:
5086:
5084:
5083:
5076:
5069:
5061:
5054:
5053:
5030:
5029:
5027:
5026:
5013:
5000:
4987:
4974:
4961:
4948:
4934:
4932:
4928:
4927:
4925:
4924:
4919:
4914:
4909:
4904:
4898:
4896:
4892:
4891:
4889:
4888:
4883:
4878:
4873:
4867:
4865:
4864:Classification
4861:
4860:
4858:
4857:
4852:
4847:
4842:
4836:
4834:
4830:
4829:
4827:
4826:
4821:
4816:
4811:
4806:
4801:
4796:
4791:
4785:
4783:
4779:
4778:
4773:
4771:
4770:
4763:
4756:
4748:
4739:
4738:
4736:
4735:
4730:
4725:
4720:
4715:
4710:
4709:
4708:
4698:
4693:
4688:
4683:
4678:
4673:
4668:
4663:
4658:
4652:
4650:
4643:
4642:
4640:
4639:
4638:
4637:
4632:
4627:
4617:
4612:
4606:
4604:
4594:
4593:
4591:
4590:
4585:
4580:
4575:
4569:
4568:
4563:
4558:
4553:
4548:
4539:
4538:
4537:
4536:
4531:
4521:
4520:
4519:
4514:
4497:
4496:
4495:
4494:
4489:
4479:
4478:
4477:
4472:
4467:
4457:
4452:
4447:
4442:
4437:
4432:
4427:
4415:
4413:
4406:
4405:
4391:
4389:
4388:
4381:
4374:
4366:
4357:
4356:
4354:
4353:
4347:
4340:
4337:
4334:
4333:
4330:
4328:
4327:
4320:
4313:
4305:
4299:
4298:
4291:
4277:
4276:External links
4274:
4272:
4271:
4214:
4162:
4113:
4069:
4020:
3976:
3925:
3868:
3831:(6): 837–853.
3811:
3762:
3735:(7): 797–801.
3719:
3665:
3612:
3558:
3509:
3460:
3419:
3384:
3359:
3333:
3284:
3225:
3174:
3114:
3054:
3004:
2953:
2897:
2862:
2803:
2752:
2723:
2702:
2688:
2632:
2613:(10): 746–53.
2594:
2534:
2478:
2429:
2378:
2325:
2279:
2215:
2163:
2145:
2127:
2106:
2084:
2082:
2079:
2036:
2033:
2030:
2029:
2027:Knockout mouse
2022:
2021:
2013:
2012:
2008:
2007:
2002:
1998:
1997:
1992:
1985:
1984:
1953:
1950:
1929:
1926:
1906:
1903:
1866:
1865:Other Diseases
1863:
1820:
1817:
1782:celiac disease
1777:
1776:Celiac Disease
1774:
1768:
1765:
1694:
1691:
1604:
1601:
1560:
1557:
1511:
1508:
1499:
1496:
1494:
1491:
1479:celiac disease
1392:
1391:
1388:
1387:
1382:
1376:
1375:
1370:
1364:
1363:
1358:
1352:
1351:
1343:
1342:
1333:
1327:
1326:
1315:
1308:
1307:
1302:
1296:
1295:
1290:
1284:
1283:
1278:
1272:
1271:
1266:
1260:
1259:
1254:
1248:
1247:
1242:
1236:
1235:
1231:
1230:
1225:
1219:
1218:
1213:
1207:
1206:
1202:
1201:
1193:
1192:
1189:
1188:
1183:
1173:
1172:
1166:
1165:
1162:
1161:
1159:
1157:
1150:
1149:
1142:
1135:
1131:
1130:
1127:
1126:
1116:
1115:
1109:
1106:
1105:
1079:
1078:
1072:
1068:
1067:
1064:
1063:
1053:
1052:
1046:
1043:
1042:
1016:
1015:
1009:
1005:
1004:
1001:
1000:
992:
991:
985:
982:
981:
973:
972:
966:
960:
959:
956:
955:
947:
946:
940:
937:
936:
928:
927:
921:
915:
914:
911:
910:
902:
901:
895:
892:
891:
883:
882:
876:
870:
869:
864:
859:
855:
854:
844:
843:
840:
839:
828:
827:
825:
824:
819:
814:
809:
804:
799:
794:
789:
784:
779:
774:
769:
764:
759:
754:
749:
743:
741:
737:
736:
734:
733:
728:
723:
718:
713:
708:
706:focal adhesion
703:
698:
693:
688:
682:
680:
676:
675:
673:
672:
667:
662:
657:
652:
647:
642:
636:
634:
630:
629:
619:
618:
615:
614:
611:
610:
602:
601:
576:
570:
569:
566:
565:
557:
556:
553:
552:
550:
549:
545:
541:
537:
533:
529:
525:
521:
517:
513:
509:
506:
505:
494:
491:
490:
488:
487:
483:
479:
475:
471:
467:
463:
459:
455:
451:
447:
444:
443:
431:
430:
422:
411:
405:
404:
401:RNA expression
393:
392:
389:
388:
380:
376:
375:
367:
364:
359:
353:
352:
343:
336:
330:
326:
325:
320:
314:
313:
305:
304:
292:
291:
288:
287:
279:
275:
274:
266:
263:
258:
252:
251:
242:
235:
229:
225:
224:
217:
211:
210:
202:
201:
189:
188:
145:
141:
140:
132:
124:
123:
119:
118:
115:
114:
111:
110:
76:
75:
67:
66:
55:
49:
48:
40:
39:
31:
30:
15:
13:
10:
9:
6:
4:
3:
2:
5349:
5338:
5335:
5333:
5330:
5328:
5325:
5324:
5322:
5307:
5306:Topoisomerase
5304:
5302:
5299:
5297:
5294:
5292:
5289:
5287:
5284:
5282:
5279:
5277:
5274:
5272:
5269:
5267:
5264:
5262:
5259:
5258:
5256:
5252:
5246:
5243:
5241:
5238:
5236:
5233:
5231:
5228:
5226:
5223:
5221:
5218:
5216:
5213:
5211:
5208:
5206:
5203:
5201:
5198:
5196:
5193:
5191:
5188:
5186:
5183:
5181:
5178:
5176:
5173:
5171:
5168:
5166:
5163:
5161:
5158:
5156:
5153:
5152:
5150:
5148:
5144:
5138:
5135:
5133:
5130:
5129:
5127:
5125:
5121:
5115:
5112:
5110:
5107:
5105:
5102:
5101:
5099:
5097:
5096:Dehydrogenase
5093:
5089:
5082:
5077:
5075:
5070:
5068:
5063:
5062:
5059:
5052:
5047:
5042:
5038:
5024:
5020:
5019:
5014:
5011:
5007:
5006:
5001:
4998:
4994:
4993:
4988:
4985:
4981:
4980:
4975:
4972:
4968:
4967:
4962:
4959:
4955:
4954:
4949:
4946:
4942:
4941:
4936:
4935:
4933:
4929:
4923:
4920:
4918:
4915:
4913:
4910:
4908:
4905:
4903:
4900:
4899:
4897:
4893:
4887:
4884:
4882:
4881:Enzyme family
4879:
4877:
4874:
4872:
4869:
4868:
4866:
4862:
4856:
4853:
4851:
4848:
4846:
4845:Cooperativity
4843:
4841:
4838:
4837:
4835:
4831:
4825:
4822:
4820:
4817:
4815:
4812:
4810:
4807:
4805:
4804:Oxyanion hole
4802:
4800:
4797:
4795:
4792:
4790:
4787:
4786:
4784:
4780:
4776:
4769:
4764:
4762:
4757:
4755:
4750:
4749:
4746:
4734:
4731:
4729:
4726:
4724:
4721:
4719:
4716:
4714:
4711:
4707:
4704:
4703:
4702:
4699:
4697:
4694:
4692:
4689:
4687:
4684:
4682:
4679:
4677:
4674:
4672:
4669:
4667:
4664:
4662:
4659:
4657:
4654:
4653:
4651:
4648:
4644:
4636:
4633:
4631:
4628:
4626:
4623:
4622:
4621:
4618:
4616:
4613:
4611:
4608:
4607:
4605:
4603:
4599:
4595:
4589:
4586:
4584:
4581:
4579:
4576:
4574:
4571:
4570:
4567:
4564:
4562:
4559:
4557:
4554:
4552:
4549:
4547:
4544:
4541:
4540:
4535:
4532:
4530:
4527:
4526:
4525:
4522:
4518:
4515:
4513:
4510:
4509:
4508:
4505:
4503:
4499:
4498:
4493:
4490:
4488:
4485:
4484:
4483:
4480:
4476:
4473:
4471:
4468:
4466:
4463:
4462:
4461:
4458:
4456:
4453:
4451:
4448:
4446:
4443:
4441:
4438:
4436:
4433:
4431:
4428:
4426:
4423:
4421:
4417:
4416:
4414:
4411:
4407:
4402:
4398:
4394:
4387:
4382:
4380:
4375:
4373:
4368:
4367:
4364:
4350:
4344:
4339:
4335:
4326:
4321:
4319:
4314:
4312:
4307:
4306:
4303:
4296:
4292:
4290:
4286:
4283:
4280:
4279:
4275:
4267:
4263:
4259:
4255:
4250:
4245:
4241:
4237:
4233:
4229:
4225:
4218:
4215:
4210:
4206:
4201:
4196:
4192:
4188:
4184:
4180:
4176:
4169:
4167:
4163:
4158:
4154:
4149:
4144:
4140:
4136:
4132:
4128:
4124:
4117:
4114:
4109:
4105:
4101:
4097:
4093:
4089:
4086:(5): 1253–7.
4085:
4081:
4073:
4070:
4065:
4061:
4056:
4051:
4047:
4043:
4039:
4035:
4031:
4024:
4021:
4016:
4012:
4008:
4004:
4000:
3996:
3992:
3988:
3980:
3977:
3972:
3968:
3963:
3958:
3953:
3948:
3944:
3940:
3936:
3929:
3926:
3921:
3917:
3913:
3909:
3904:
3899:
3895:
3891:
3887:
3883:
3879:
3872:
3869:
3864:
3860:
3856:
3852:
3847:
3842:
3838:
3834:
3830:
3826:
3822:
3815:
3812:
3807:
3803:
3798:
3793:
3789:
3785:
3781:
3777:
3773:
3766:
3763:
3758:
3754:
3750:
3746:
3742:
3738:
3734:
3730:
3723:
3720:
3715:
3711:
3707:
3703:
3698:
3693:
3689:
3685:
3684:FASEB Journal
3681:
3674:
3672:
3670:
3666:
3661:
3657:
3652:
3647:
3643:
3639:
3636:(7): e45067.
3635:
3631:
3627:
3619:
3617:
3613:
3608:
3604:
3599:
3594:
3589:
3584:
3580:
3576:
3572:
3565:
3563:
3559:
3554:
3550:
3545:
3540:
3536:
3532:
3528:
3524:
3520:
3513:
3510:
3505:
3501:
3496:
3491:
3487:
3483:
3479:
3475:
3471:
3464:
3461:
3456:
3452:
3447:
3442:
3438:
3434:
3430:
3423:
3420:
3415:
3411:
3407:
3403:
3400:(10): 534–9.
3399:
3395:
3388:
3385:
3380:
3376:
3371:
3366:
3362:
3360:9780123943057
3356:
3352:
3348:
3344:
3337:
3334:
3329:
3325:
3320:
3315:
3311:
3307:
3303:
3299:
3295:
3288:
3285:
3280:
3276:
3271:
3266:
3261:
3256:
3252:
3248:
3245:(1): e16665.
3244:
3240:
3236:
3229:
3226:
3221:
3217:
3212:
3207:
3202:
3197:
3193:
3189:
3185:
3178:
3175:
3170:
3166:
3161:
3156:
3151:
3146:
3142:
3138:
3134:
3127:
3125:
3123:
3121:
3119:
3115:
3110:
3106:
3101:
3096:
3091:
3086:
3082:
3078:
3074:
3067:
3065:
3063:
3061:
3059:
3055:
3050:
3046:
3042:
3041:11392/2388638
3038:
3034:
3030:
3026:
3022:
3015:
3008:
3005:
3000:
2996:
2991:
2986:
2981:
2976:
2972:
2968:
2964:
2957:
2954:
2949:
2945:
2940:
2935:
2930:
2925:
2921:
2917:
2913:
2906:
2904:
2902:
2898:
2893:
2889:
2885:
2881:
2877:
2873:
2866:
2863:
2858:
2854:
2849:
2844:
2839:
2834:
2830:
2826:
2823:(5): 2743–7.
2822:
2818:
2814:
2807:
2804:
2799:
2795:
2790:
2785:
2780:
2775:
2771:
2767:
2763:
2756:
2753:
2748:
2742:
2734:
2730:
2726:
2724:9784431558255
2720:
2716:
2709:
2707:
2703:
2698:
2692:
2689:
2684:
2680:
2676:
2672:
2667:
2662:
2658:
2654:
2650:
2643:
2641:
2639:
2637:
2633:
2628:
2624:
2620:
2616:
2612:
2608:
2601:
2599:
2595:
2590:
2586:
2581:
2576:
2572:
2568:
2565:(1): 104–14.
2564:
2560:
2556:
2549:
2547:
2545:
2543:
2541:
2539:
2535:
2530:
2526:
2521:
2516:
2512:
2508:
2504:
2500:
2496:
2489:
2487:
2485:
2483:
2479:
2474:
2470:
2465:
2460:
2456:
2452:
2449:(4): 825–38.
2448:
2444:
2440:
2433:
2430:
2425:
2421:
2416:
2411:
2406:
2401:
2397:
2393:
2389:
2382:
2379:
2374:
2370:
2366:
2362:
2358:
2354:
2349:
2344:
2341:(2): 357–66.
2340:
2336:
2329:
2326:
2321:
2317:
2312:
2307:
2303:
2299:
2295:
2288:
2286:
2284:
2280:
2275:
2271:
2266:
2261:
2257:
2253:
2250:(4): 513–22.
2249:
2245:
2241:
2234:
2232:
2230:
2228:
2226:
2224:
2222:
2220:
2216:
2211:
2207:
2203:
2199:
2194:
2189:
2185:
2181:
2177:
2170:
2168:
2164:
2159:
2155:
2149:
2146:
2141:
2137:
2131:
2128:
2124:
2120:
2115:
2113:
2111:
2107:
2103:
2099:
2094:
2092:
2090:
2086:
2080:
2078:
2074:
2072:
2068:
2065:
2061:
2056:
2054:
2050:
2046:
2042:
2034:
2028:
2023:
2019:
2014:
2009:
2006:
2003:
2000:
1999:
1996:
1993:
1991:
1987:
1986:
1981:
1978:
1975:
1971:
1967:
1963:
1959:
1951:
1949:
1947:
1943:
1939:
1935:
1927:
1925:
1923:
1919:
1915:
1912:for anti-tTG
1911:
1904:
1902:
1900:
1896:
1892:
1888:
1883:
1880:
1876:
1872:
1864:
1862:
1859:
1855:
1851:
1847:
1843:
1839:
1835:
1834:breast cancer
1831:
1826:
1818:
1816:
1814:
1810:
1806:
1802:
1798:
1796:
1791:
1787:
1783:
1775:
1773:
1766:
1764:
1762:
1758:
1753:
1751:
1746:
1743:tTG also has
1741:
1739:
1734:
1732:
1728:
1724:
1723:cell adhesion
1720:
1716:
1712:
1708:
1707:wound healing
1704:
1703:retinoic acid
1700:
1692:
1686:
1682:
1680:
1671:
1667:
1658:
1654:
1652:
1647:
1643:
1638:
1635:
1630:
1626:
1620:
1618:
1614:
1610:
1602:
1596:
1592:
1590:
1589:glutamic acid
1586:
1582:
1578:
1574:
1570:
1566:
1558:
1556:
1554:
1550:
1545:
1541:
1537:
1533:
1529:
1528:beta-sandwich
1525:
1521:
1517:
1509:
1507:
1505:
1497:
1492:
1490:
1488:
1484:
1480:
1476:
1471:
1469:
1465:
1461:
1457:
1453:
1449:
1445:
1441:
1437:
1433:
1429:
1426:between an ε-
1425:
1421:
1417:
1414:
1410:
1406:
1402:
1398:
1386:
1383:
1381:
1377:
1374:
1371:
1369:
1365:
1362:
1359:
1357:
1353:
1348:
1344:
1341:
1337:
1334:
1332:
1331:Gene Ontology
1328:
1325:
1322:
1319:
1316:
1313:
1309:
1306:
1303:
1301:
1297:
1294:
1291:
1289:
1285:
1282:
1279:
1277:
1273:
1270:
1269:NiceZyme view
1267:
1265:
1261:
1258:
1255:
1253:
1249:
1246:
1243:
1241:
1237:
1232:
1229:
1226:
1224:
1220:
1217:
1214:
1212:
1208:
1203:
1198:
1187:
1182:
1178:
1174:
1171:
1167:
1160:
1158:
1155:
1151:
1147:
1143:
1140:
1136:
1132:
1125:
1123:
1117:
1113:
1110:
1104:
1102:
1098:
1094:
1090:
1086:
1080:
1076:
1073:
1069:
1062:
1060:
1054:
1050:
1047:
1041:
1039:
1035:
1031:
1027:
1023:
1017:
1013:
1010:
1008:RefSeq (mRNA)
1006:
999:
998:
993:
989:
986:
980:
979:
974:
970:
967:
965:
961:
954:
953:
948:
944:
941:
935:
934:
929:
925:
922:
920:
916:
909:
908:
903:
899:
896:
890:
889:
884:
880:
877:
875:
871:
868:
865:
863:
860:
856:
853:
849:
845:
838:
834:
829:
823:
820:
818:
815:
813:
810:
808:
805:
803:
800:
798:
795:
793:
790:
788:
785:
783:
780:
778:
775:
773:
770:
768:
765:
763:
760:
758:
755:
753:
750:
748:
745:
744:
742:
739:
738:
732:
729:
727:
724:
722:
719:
717:
714:
712:
711:mitochondrion
709:
707:
704:
702:
699:
697:
694:
692:
689:
687:
684:
683:
681:
678:
677:
671:
668:
666:
663:
661:
658:
656:
653:
651:
648:
646:
643:
641:
638:
637:
635:
632:
631:
628:
627:Gene ontology
624:
620:
608:
603:
599:
592:
585:
580:
577:
575:
571:
563:
558:
547:
543:
539:
535:
531:
527:
523:
519:
515:
511:
510:
507:
503:
498:
495:
485:
481:
477:
474:right auricle
473:
469:
465:
461:
457:
453:
449:
448:
445:
441:
436:
433:
432:
429:
427:
423:
421:
420:
416:
415:
412:
410:
406:
402:
398:
394:
386:
381:
377:
373:
368:
358:
354:
347:
340:
334:
327:
319:
315:
311:
306:
302:
297:
293:
285:
280:
276:
272:
267:
257:
253:
246:
239:
233:
226:
222:
216:
212:
208:
203:
199:
194:
190:
186:
182:
178:
174:
170:
166:
162:
158:
154:
150:
142:
137:
130:
125:
120:
109:
107:
103:
99:
95:
91:
87:
83:
77:
72:
69:
68:
64:
61:
54:
50:
45:
41:
37:
32:
27:
22:
5337:Autoantigens
5291:Gangliosides
5147:Nucleoporins
5136:
5088:Autoantigens
5018:Translocases
5015:
5002:
4989:
4976:
4963:
4953:Transferases
4950:
4937:
4794:Binding site
4624:
4542:
4500:
4418:
4393:Transferases
4348:
4231:
4227:
4217:
4182:
4178:
4130:
4126:
4116:
4083:
4079:
4072:
4040:(1): 78–89.
4037:
4033:
4023:
3993:(1): 111–8.
3990:
3986:
3979:
3945:(5): 181–7.
3942:
3938:
3928:
3888:(2): 130–9.
3885:
3881:
3871:
3828:
3824:
3814:
3779:
3775:
3765:
3732:
3728:
3722:
3687:
3683:
3633:
3630:EMBO Reports
3629:
3578:
3574:
3529:(3): 550–8.
3526:
3522:
3512:
3477:
3473:
3463:
3436:
3432:
3422:
3397:
3393:
3387:
3342:
3336:
3301:
3297:
3287:
3242:
3238:
3228:
3194:(12): e327.
3191:
3188:PLOS Biology
3187:
3177:
3140:
3136:
3080:
3076:
3024:
3020:
3007:
2970:
2966:
2956:
2919:
2915:
2878:(2): 190–5.
2875:
2871:
2865:
2820:
2816:
2806:
2772:(12): e327.
2769:
2766:PLOS Biology
2765:
2755:
2714:
2691:
2656:
2652:
2610:
2606:
2562:
2558:
2502:
2498:
2446:
2442:
2432:
2395:
2391:
2381:
2338:
2334:
2328:
2311:10.2741/1921
2301:
2297:
2247:
2243:
2183:
2179:
2157:
2148:
2139:
2130:
2075:
2057:
2038:
1958:interactions
1955:
1952:Interactions
1931:
1908:
1884:
1868:
1825:inflammation
1822:
1794:
1779:
1770:
1757:proteostasis
1754:
1742:
1735:
1696:
1676:
1664:
1621:
1606:
1562:
1513:
1501:
1472:
1460:mitochondria
1404:
1400:
1396:
1395:
1257:BRENDA entry
1119:
1093:NP_001310247
1089:NP_001310246
1085:NP_001310245
1082:
1056:
1038:NM_001323318
1034:NM_001323317
1030:NM_001323316
1019:
995:
976:
950:
931:
905:
886:
866:
861:
544:carotid body
532:ciliary body
424:
417:
383:157,988,356
370:157,958,322
144:External IDs
79:
5276:Cardiolipin
4789:Active site
4635:Factor XIII
4331:PDB gallery
4185:(1): 5–13.
4179:BMB Reports
3987:Amino Acids
3690:(1): 3–12.
2304:: 1758–73.
1928:Therapeutic
1922:specificity
1918:sensitivity
1651:Thioredoxin
1637:nucleotides
1524:beta barrel
1475:autoantigen
1473:tTG is the
1468:fibronectin
1448:deamidation
1444:proteolysis
1436:carboxamide
1430:group of a
1245:IntEnz view
1228:80146-85-6
1205:Identifiers
665:GTP binding
516:Paneth cell
282:38,166,578
269:38,127,385
122:Identifiers
5321:Categories
5281:Centromere
4992:Isomerases
4966:Hydrolases
4833:Regulation
4133:(6): 613.
2125:, May 2017
2104:, May 2017
2081:References
2064:allosteric
1974:Substrates
1964:donor and
1934:metastasis
1920:(99%) and
1914:antibodies
1905:Diagnostic
1901:activity.
1879:Huntington
1850:metastasis
1731:astrocytes
1603:Regulation
1585:isopeptide
1314:structures
1281:KEGG entry
428:(ortholog)
165:HomoloGene
4871:EC number
3581:(2): 19.
2741:cite book
2733:937392418
2717:. Tokyo.
2343:CiteSeerX
2067:disulfide
2041:chaperone
1962:glutamine
1942:inhibitor
1875:Parkinson
1871:Alzheimer
1805:glutamine
1795:Triticeae
1711:apoptosis
1642:disulfide
1577:thioester
1569:glutamine
1559:Mechanism
1493:Structure
1464:apoptosis
1440:glutamine
1438:group of
1234:Databases
1122:NP_033399
1101:NP_945189
1097:NP_004604
1059:NM_009373
1026:NM_198951
1022:NM_004613
852:Orthologs
686:cytoplasm
450:beta cell
173:GeneCards
5332:EC 2.3.2
5301:Thrombin
4895:Kinetics
4819:Cofactor
4782:Activity
4487:P300/CBP
4295:TRANSDAB
4285:Archived
4266:14849506
4258:25943306
4249:11113818
4209:29187283
4157:29795262
4108:11018740
4100:10811336
4064:22698685
4015:16143202
4007:21938398
3971:21541002
3912:20731657
3863:59341070
3855:30693974
3806:29550590
3757:20033968
3714:58551851
3706:30593123
3660:29752334
3607:30691081
3553:17100777
3504:12737632
3455:15069073
3414:12368090
3379:22364871
3328:29570839
3279:21304968
3239:PLOS ONE
3220:18092889
3169:29305423
3109:21908620
3049:29764956
2999:26160175
2948:20547769
2892:20450932
2857:11867708
2798:18092889
2683:21883387
2675:19878304
2627:22326684
2589:22228808
2529:12366374
2473:10684262
2424:22442151
2373:11242870
2320:16368554
2274:22437759
2210:19217277
2202:21902809
2121:–
2100:–
1910:Serology
1899:TGF beta
1893:and the
1887:fibrosis
1830:leukemia
1738:spectrin
1693:Function
1646:cysteine
1458:and the
1424:proteins
1416:2.3.2.13
1385:proteins
1373:articles
1361:articles
1318:RCSB PDB
1216:2.3.2.13
1170:Wikidata
831:Sources:
261:20q11.23
5051:Biology
5005:Ligases
4775:Enzymes
4200:5796628
4148:5966415
4055:3418489
3962:3083958
3903:8094245
3846:7754084
3797:6203336
3749:9212111
3651:6030705
3598:6409630
3544:1810403
3495:1223550
3370:3746560
3319:5980740
3270:3031627
3247:Bibcode
3211:2140088
3160:5827427
3100:3199528
2990:4571865
2939:2919103
2825:Bibcode
2789:2140088
2580:3310700
2520:1223021
2464:2169362
2415:3365763
2365:9344835
2265:3712867
2123:Ensembl
2102:Ensembl
2051:in the
2045:peptide
1970:calcium
1813:T cells
1797:glutens
1727:neurons
1699:calcium
1677:In the
1661:enzyme.
1634:guanine
1613:nucleus
1573:ammonia
1510:Protein
1487:villous
1456:nucleus
1452:cytosol
1340:QuickGO
1305:profile
1288:MetaCyc
1223:CAS no.
964:UniProt
919:Ensembl
858:Species
837:QuickGO
696:cytosol
486:decidua
403:pattern
129:Aliases
5245:NUP214
5240:NUP205
5235:NUP210
5230:NUP188
5225:NUP160
5220:NUP155
5215:NUP153
5210:NUP133
5205:NUP107
5037:Portal
4979:Lyases
4706:HMGCS2
4543:other:
4534:SPTLC2
4529:SPTLC1
4470:HGSNAT
4264:
4256:
4246:
4207:
4197:
4155:
4145:
4106:
4098:
4062:
4052:
4013:
4005:
3969:
3959:
3920:586174
3918:
3910:
3900:
3861:
3853:
3843:
3804:
3794:
3755:
3747:
3712:
3704:
3658:
3648:
3605:
3595:
3551:
3541:
3502:
3492:
3453:
3412:
3377:
3367:
3357:
3326:
3316:
3277:
3267:
3218:
3208:
3167:
3157:
3107:
3097:
3047:
2997:
2987:
2946:
2936:
2890:
2855:
2848:122418
2845:
2796:
2786:
2731:
2721:
2681:
2673:
2625:
2587:
2577:
2527:
2517:
2471:
2461:
2422:
2412:
2371:
2363:
2345:
2318:
2272:
2262:
2208:
2200:
2071:enzyme
1966:lysine
1895:kidney
1819:Cancer
1809:gluten
1801:B-cell
1745:GTPase
1713:, and
1581:lysine
1526:and a
1522:, two
1483:gluten
1432:lysine
1409:enzyme
1368:PubMed
1350:Search
1336:AmiGO
1324:PDBsum
1264:ExPASy
1252:BRENDA
1240:IntEnz
1211:EC no.
1156:search
1154:PubMed
997:P21981
978:P21980
874:Entrez
574:BioGPS
153:190196
5266:Actin
5254:Other
5200:NUP98
5195:NUP93
5190:NUP88
5185:NUP85
5180:NUP62
5175:NUP54
5170:NUP50
5165:NUP43
5160:NUP37
5155:NUP35
4931:Types
4647:2.3.3
4598:2.3.2
4588:ABHD5
4475:ARD1A
4410:2.3.1
4262:S2CID
4104:S2CID
4011:S2CID
3916:S2CID
3859:S2CID
3753:S2CID
3710:S2CID
3017:(PDF)
2679:S2CID
2369:S2CID
2206:S2CID
2047:onto
2035:Erp57
1565:thiol
1542:, or
1428:amino
1300:PRIAM
907:21817
867:Mouse
862:Human
833:Amigo
426:Mouse
419:Human
366:Start
301:Mouse
265:Start
198:Human
161:98731
5023:list
5016:EC7
5010:list
5003:EC6
4997:list
4990:EC5
4984:list
4977:EC4
4971:list
4964:EC3
4958:list
4951:EC2
4945:list
4938:EC1
4517:CPT2
4512:CPT1
4492:NAT2
4403:2.3)
4349:1kv3
4254:PMID
4205:PMID
4153:PMID
4096:PMID
4060:PMID
4003:PMID
3967:PMID
3908:PMID
3851:PMID
3802:PMID
3745:PMID
3702:PMID
3656:PMID
3603:PMID
3549:PMID
3500:PMID
3451:PMID
3410:PMID
3375:PMID
3355:ISBN
3324:PMID
3275:PMID
3216:PMID
3165:PMID
3105:PMID
3045:PMID
2995:PMID
2944:PMID
2888:PMID
2853:PMID
2794:PMID
2747:link
2729:OCLC
2719:ISBN
2671:PMID
2623:PMID
2585:PMID
2525:PMID
2469:PMID
2420:PMID
2361:PMID
2316:PMID
2270:PMID
2198:PMID
2005:Tgm2
1995:Tgm2
1891:lung
1877:and
1844:and
1761:HSF1
1721:and
1498:Gene
1380:NCBI
1321:PDBe
1276:KEGG
888:7052
409:Bgee
357:Band
318:Chr.
256:Band
215:Chr.
177:TGM2
169:3391
149:OMIM
136:TGM2
106:3S3S
102:3S3P
98:3S3J
94:3LY6
90:2Q3Z
86:1KV3
82:4PYG
63:RCSB
60:PDBe
29:TGM2
4244:PMC
4236:doi
4195:PMC
4187:doi
4143:PMC
4135:doi
4088:doi
4050:PMC
4042:doi
4038:237
3995:doi
3957:PMC
3947:doi
3898:PMC
3890:doi
3841:PMC
3833:doi
3792:PMC
3784:doi
3780:154
3737:doi
3692:doi
3646:PMC
3638:doi
3593:PMC
3583:doi
3539:PMC
3531:doi
3527:146
3490:PMC
3482:doi
3478:373
3441:doi
3437:279
3402:doi
3365:PMC
3347:doi
3314:PMC
3306:doi
3265:PMC
3255:doi
3206:PMC
3196:doi
3155:PMC
3145:doi
3141:293
3095:PMC
3085:doi
3081:286
3037:hdl
3029:doi
3025:475
2985:PMC
2975:doi
2971:290
2934:PMC
2924:doi
2920:285
2880:doi
2843:PMC
2833:doi
2784:PMC
2774:doi
2661:doi
2657:276
2615:doi
2575:PMC
2567:doi
2563:341
2515:PMC
2507:doi
2503:368
2459:PMC
2451:doi
2447:148
2410:PMC
2400:doi
2396:287
2353:doi
2339:239
2306:doi
2260:PMC
2252:doi
2188:doi
2184:278
1990:MGI
1629:GDP
1627:or
1625:GTP
1544:ATP
1540:GTP
1536:GDP
1477:in
1405:TG2
1403:or
1401:tTG
1356:PMC
1312:PDB
379:End
278:End
181:OMA
157:MGI
53:PDB
5323::
4600::
4401:EC
4395::
4260:.
4252:.
4242:.
4232:72
4230:.
4226:.
4203:.
4193:.
4183:51
4181:.
4177:.
4165:^
4151:.
4141:.
4129:.
4125:.
4102:.
4094:.
4084:95
4082:.
4058:.
4048:.
4036:.
4032:.
4009:.
4001:.
3991:44
3989:.
3965:.
3955:.
3941:.
3937:.
3914:.
3906:.
3896:.
3886:21
3884:.
3880:.
3857:.
3849:.
3839:.
3829:58
3827:.
3823:.
3800:.
3790:.
3778:.
3774:.
3751:.
3743:.
3731:.
3708:.
3700:.
3688:33
3686:.
3682:.
3668:^
3654:.
3644:.
3634:19
3632:.
3628:.
3615:^
3601:.
3591:.
3577:.
3573:.
3561:^
3547:.
3537:.
3525:.
3521:.
3498:.
3488:.
3476:.
3472:.
3449:.
3435:.
3431:.
3408:.
3398:27
3396:.
3373:.
3363:.
3353:.
3322:.
3312:.
3302:96
3300:.
3296:.
3273:.
3263:.
3253:.
3241:.
3237:.
3214:.
3204:.
3190:.
3186:.
3163:.
3153:.
3139:.
3135:.
3117:^
3103:.
3093:.
3079:.
3075:.
3057:^
3043:.
3035:.
3023:.
3019:.
2993:.
2983:.
2969:.
2965:.
2942:.
2932:.
2918:.
2914:.
2900:^
2886:.
2876:47
2874:.
2851:.
2841:.
2831:.
2821:99
2819:.
2815:.
2792:.
2782:.
2768:.
2764:.
2743:}}
2739:{{
2727:.
2705:^
2677:.
2669:.
2655:.
2651:.
2635:^
2621:.
2611:11
2609:.
2597:^
2583:.
2573:.
2561:.
2557:.
2537:^
2523:.
2513:.
2501:.
2497:.
2481:^
2467:.
2457:.
2445:.
2441:.
2418:.
2408:.
2394:.
2390:.
2367:.
2359:.
2351:.
2337:.
2314:.
2302:11
2300:.
2296:.
2282:^
2268:.
2258:.
2248:34
2246:.
2242:.
2218:^
2204:.
2196:.
2182:.
2178:.
2166:^
2156:.
2138:.
2109:^
2088:^
2055:.
1972:.
1873:,
1852:,
1840:,
1836:,
1832:,
1752:.
1709:,
1619:.
1538:,
1530:.
1413:EC
1338:/
835:/
385:bp
372:bp
284:bp
271:bp
179:;
175::
171:;
167::
163:;
159::
155:;
151::
104:,
100:,
96:,
92:,
88:,
84:,
5080:e
5073:t
5066:v
5039::
5025:)
5021:(
5012:)
5008:(
4999:)
4995:(
4986:)
4982:(
4973:)
4969:(
4960:)
4956:(
4947:)
4943:(
4767:e
4760:t
4753:v
4504::
4422::
4399:(
4385:e
4378:t
4371:v
4324:e
4317:t
4310:v
4268:.
4238::
4211:.
4189::
4159:.
4137::
4131:9
4110:.
4090::
4066:.
4044::
4017:.
3997::
3973:.
3949::
3943:1
3922:.
3892::
3865:.
3835::
3808:.
3786::
3759:.
3739::
3733:3
3716:.
3694::
3662:.
3640::
3609:.
3585::
3579:7
3555:.
3533::
3506:.
3484::
3457:.
3443::
3416:.
3404::
3381:.
3349::
3330:.
3308::
3281:.
3257::
3249::
3243:6
3222:.
3198::
3192:5
3171:.
3147::
3111:.
3087::
3051:.
3039::
3031::
3001:.
2977::
2950:.
2926::
2894:.
2882::
2859:.
2835::
2827::
2800:.
2776::
2770:5
2749:)
2735:.
2699:.
2685:.
2663::
2629:.
2617::
2591:.
2569::
2531:.
2509::
2475:.
2453::
2426:.
2402::
2375:.
2355::
2322:.
2308::
2276:.
2254::
2212:.
2190::
2160:.
2142:.
1411:(
303:)
200:)
183::
23:.
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.