82:
653:
694:
254:
45:
444:
65:
843:
858:
430:
818:
272:
701:
By varying the apparatus and catalyst loading, Shirakawa and coworkers were able to synthesize polyacetylene as thin films, rather than insoluble black powders. They obtained these films by coating the walls of a reaction flask under inert conditions with a solution of the
Ziegler–Natta catalyst and
1160:
Polyacetylene has no commercial applications, although the discovery of polyacetylene as a conductive organic polymer led to many developments in materials science. Conducting polymers are of interest for solution-processing for film-forming conductive polymers. Therefore, attention has shifted to
387:
in 2000. Early work in the field of polyacetylene research was aimed at using doped polymers as easily processable and lightweight "plastic metals". Despite the promise of this polymer in the field of conductive polymers, many of its properties such as instability to air and difficulty with
1295:
Gorman, C. B.; Ginsburg, E. J.; Grubbs, R. H. (1993). "Soluble, Highly
Conjugated Derivatives of Polyacetylene from the Ring-Opening Metathesis Polymerization of Monosubstituted Cyclooctratetraenes: Synthesis and the Relationship between Polymer Structure and Physical Properties".
1109:
1084:
The synthesis and processing of polyacetylene films affects the properties. Increasing the catalyst ratio creates thicker films with a greater draw ratio, allowing them to be stretched further. Lower catalyst loadings leads to the formation of dark red
830:
Polyacetylene can also be synthesized from other polymers. This method enables modification and processing of the polymer before conversion into the highly insoluble polyacetylene. Short, irregular segments of polyacetylene can be obtained by
690:. This method allows control over the structure and properties of the final polymer by varying temperature and catalyst loading. Mechanistic studies suggest that this polymerization involves metal insertion into the triple bond of acetylene.
849:
More efficient methos for synthesizing long polyacetylene chains exist and include the Durham precursor route in which precusor polymers are prepared by ring-opening metathesis polymerization, and a subsequent heat-induced reverse
234:
1011:-polyacetylene has a lower conductivity of 1.7×10 Ωcm. Doping with bromine causes an increase in conductivity to 0.5 Ωcm, while a higher conductivity of 38 Ωcm is obtained through doping with iodine. Doping of either
524:
in 1958. The resulting polyacetylene was linear, of high molecular weight, displayed high crystallinity, and had a regular structure. X-ray diffraction studies demonstrated that the resulting polyacetylene was
536:’s group who were able to prepare silvery films of polyacetylene. They discovered that the polymerization of polyacetylene could be achieved at the surface of a concentrated solution of the catalyst system of
619:
further increased the conductivities, bringing them close to that of copper. Furthermore, it was found that heat treatment of the catalyst used for polymerization led to films with higher conductivities.
529:-polyacetylene. After this first reported synthesis, few chemists were interested in polyacetylene because the product of Natta's preparation was an insoluble, air sensitive, and infusible black powder.
519:
One of the earliest reported acetylene polymers was named
Cuprene. Its highly cross-linked nature led to no further studies in the field for quite some time. Linear polyacetylene was first prepared by
1097:, then freezing and subliming the benzene. Polyacetylene has a bulk density of 0.4 g/cm, while density of the foam is significantly lower, at 0.02–0.04 g/cm. The morphology consists of
1124:-polyacetylene show high thermal stability, exposure to air causes a large decrease in the flexibility and conductivity. When polyacetylene is exposed to air, oxidation of the backbone by O
503:
nature of the polyacetylene backbone, not all of the carbon–carbon bonds in the material are equal: a distinct single/double alternation exists. Each hydrogen atom can be replaced by a
587:. These materials exhibited the largest room temperature conductivity observed for a covalent organic polymer, and this seminal report was key in furthering the development of organic
507:. Substituted polyacetylenes tend to be more rigid than saturated polymers. Furthermore, placing different functional groups as substituents on the polymer backbone leads to a twisted
1205:
Perego, Giovanni; Lugli, Gabriele; Pedretti, Ugo; Cesari, Marco (1988). "X-ray investigation on highly oriented polyacetylene, 1. Crystal structure of cis- and trans-polyacetylene".
1743:
Ito, T.; Shirakawa, H.; Ikeda, S. (1974). "Simultaneous
Polymerization and Formation of Polyacetylene Film on the Surface of Concentrated Soluble Ziegler-Type Catalyst Solution".
1553:
Shirakawa, H.; Louis, E.J.; MacDiarmid, A.G.; Chiang, C.K.; Heeger, A.J. (1977). "Synthesis of
Electrically Conducting Organic Polymers: Halogen Derivatives of Polyacetylene, (CH)
285:
739:
to produce usable polymer. Gas-phase polymerization typically produces irregular cuprene, whereas liquid-phase polymerization, conducted at −78 °C produces linear
1875:
MacDiarmid, Alan Graham; Mammone, R. J.; Kaner, R. B.; Porter, Lord; Pethig, R.; Heeger, A. J.; Rosseinsky, D. R.; Gillespie, Ronald James; Day, Peter (1985-05-30).
702:
adding gaseous acetylene resulting in immediate formation of a film. Enkelmann and coworkers further improved polyacetylene synthesis by changing the catalyst to a
1116:
For applications, polyacetylenes suffer from many drawbacks. They are insoluble in solvents, making it essentially impossible to process the material. While both
1105:
between chains. The insolubility of polyacetylene makes it difficult to characterize this material and to determine the extent of cross-linking in the material.
81:
1089:, which can be converted to films by cutting and pressing between glass plates. A foam-like material can be obtained from the gel by displacing the
756:
660:
A variety of methods have been developed to synthesize polyacetylene. One of the most common methods is via passing acetylene gas over a
632:
770:. This synthetic route also provides a facile method for adding solubilizing groups to the polymer while maintaining the conjugation.
560:, a related but inorganic polymer. Polythiazyl caught Heeger's interest as a chain-like metallic material, and he collaborated with
1716:
Clarke, T. C.; Yannoni, T. S.; Katz, T. J. (1983). "Mechanism of
Ziegler–Natta Polymerization of Acetylene: A Nutation NMR Study".
1383:
913:
604:
352:
2106:
1497:
2096:
1959:
292:
1043:, and found that the structure depends on synthetic conditions. When the synthesis is performed below −78 °C, the
30:
This article is about polymers with alternating double and single bonds. For compounds with multiple triple bonds, see
661:
1207:
1778:
Klavetter, Floyd L.; Grubbs, Robert H. (1988). "Polycyclooctatetraene (Polyacetylene): Synthesis and
Properties".
2101:
1805:
851:
1829:
Chiang, C.K.; Gau, S.C.; Fincher, C.R.; Park, Y.W.; MacDiarmid, A.G.; Heeger, A.J. (1978). "Polyacetylene, (CH)
508:
384:
44:
469:
568:
at low temperatures. Shirakawa, Heeger, and MacDiarmid collaborated on further development of polyacetylene.
253:
968:
925:
917:
878:
652:
693:
624:
572:
356:
64:
1174:
1129:
1036:
665:
636:
380:
246:
105:
1881:
Philosophical
Transactions of the Royal Society of London. Series A, Mathematical and Physical Sciences
1019:-polyacetylene leads to an increase in their conductivities by at least six orders of magnitude. Doped
2048:
1888:
1842:
1752:
1630:
1466:
Saxon, A.M.; Liepins, F.; Aldissi, M. (1985). "Polyacetylene: Its
Synthesis, Doping, and Structure".
1246:
991:
987:
980:
964:
952:
941:
882:
871:
836:
87:
2081:
1101:, with an average width of 200 Å. These fibrils form an irregular, web-like network, with some
491:-polyacetylene, can be achieved by changing the temperature at which the reaction is conducted. The
1235:"Nobel Lecture: Semiconducting and metallic polymers: The fourth generation of polymeric materials"
1077:
1067:
1049:
832:
588:
129:
735:
irradiation have all been used. This method avoid the use of catalysts and solvents, but requires
391:
Compounds called polyacetylenes also occur in nature, although in this context the term refers to
1904:
1620:
1162:
1040:
714:
703:
564:
who had previous experience with this material. By the early 1970s, this polymer was known to be
360:
1876:
351:
groups. This compound is conceptually important, as the discovery of polyacetylene and its high
1580:
Shirakawa, Hideki (1995). "Synthesis and characterization of highly conducting polyacetylene".
743:-polyacetylene, and solid-phase polymerization, conducted at still lower temperature, produces
2111:
2076:
1664:
1646:
1516:
1354:
937:
795:
760:
676:
579:, the conductivity increased seven orders of magnitude. Similar results were achieved using Cl
537:
500:
2056:
2019:
1986:
1951:
1896:
1850:
1787:
1760:
1725:
1698:
1654:
1638:
1589:
1562:
1508:
1475:
1421:
1344:
1336:
1305:
1274:
1254:
1215:
1108:
533:
504:
376:
364:
200:
172:
50:
139:
807:
774:
and coworkers synthesized a variety of polyacetylene derivatives with linear and branched
728:
565:
561:
553:
372:
316:
2052:
1892:
1846:
1756:
1634:
1250:
814:) unit directly connected to the polymer reduces steric crowding and prevents twisting.
1659:
1608:
1349:
1324:
1023:-polyacetylene films usually have conductivities two or three times greater than doped
936:. The "hole" on the polymer backbone is weakly associated with the anionic acceptor by
842:
724:
443:
340:
320:
263:
2090:
1955:
1702:
1593:
1479:
1425:
1166:
771:
1908:
1682:
Feast, W. J.; Tsibouklis, J.; Pouwer, K. L.; Groenendaal, L.; Meijer, E. W. (1996).
928:
occurs from the polymer to the acceptor compound; the polyacetylene chain acts as a
1524:
1145:
521:
400:
1877:"The concept of 'doping' of conducting polymers: the role of reduction potentials"
1683:
1406:
1340:
857:
1405:
Feast, W.J.; Tsibouklis, J.; Pouwer, K.L.; Groenendaal, L.; Meijer, E.W. (1996).
1053:
form is favored. At room temperature, the polymerization yields a ratio of 60:40
1609:"Observation of the topological soliton state in the Su–Schrieffer–Heeger model"
1219:
1170:
870:
When polyacetylene films are exposed to vapors of electron-accepting compounds (
732:
557:
549:
461:
368:
2060:
1932:
1259:
1234:
1102:
817:
736:
150:
17:
1650:
1149:
948:
retain their high conductivity even after exposure to air for several days.
881:
of the material increases by orders of magnitude over the undoped material.
764:
375:
for this polymer led to intense interest in the use of organic compounds in
344:
221:
1900:
1668:
1520:
1358:
1003:
The conductivity of polyacetylene depends on structure and doping. Undoped
548:
in an inert solvent such as toluene. In parallel with
Shirakawa's studies,
1764:
2024:
2007:
1991:
1974:
1566:
1325:"Biosynthesis and function of polyacetylenes and allied natural products"
1141:
1133:
976:
909:
723:
Polyacetylene can also be produced by photopolymerization of acetylene.
465:
1791:
1729:
1642:
1309:
1152:
temporarily, while coating with glass increases stability indefinitely.
697:
Mechanism of polyacetylene synthesis from acetylene and a metal catalyst
429:
1186:
1137:
1094:
1090:
994:
972:
945:
921:
767:
628:
532:
The next major development of polyacetylene polymerization was made by
392:
31:
1933:"Organic Metals and Semiconductors: The Chemistry of Polyacetylene (CH
1684:"Synthesis, processing and material properties of conjugated polymers"
1407:"Synthesis, processing and material properties of conjugated polymers"
2039:
Will, F.G.; D.W. McKee (1983). "Thermal Oxidation of Polyacetylene".
1512:
1098:
1075:-polyacetylene are very flexible and can be readily stretched, while
983:
960:
956:
929:
905:
885:
874:
861:
Durham precursor polymer (Reverse Diels–Alder) route to polyacetylene
799:
480:
457:
348:
1854:
262:
Except where otherwise noted, data are given for materials in their
1625:
1177:
could also be a potential application of conductive polyacetylene.
1027:-polyacetylene even though the parent film has lower conductivity.
90:
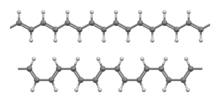
of the transoidal (top) and cisoidal (bottom) conformations of the
1107:
963:
for polyacetylene include lithium, sodium, and potassium. As with
933:
856:
816:
791:
783:
775:
692:
651:
623:
To account for such an increase in conductivity in polyacetylene,
308:
395:, compounds containing multiple acetylene groups ("poly" meaning
1035:
The structure of polyacetylene films have been examined by both
916:
of these polymers is believed to be a result of the creation of
786:
had high conductivity but low solubility, while highly branched
627:
and Heeger considered the existence of topologically protected
1926:
1924:
1922:
1920:
1918:
1086:
1007:-polyacetylene films have a conductivity of 4.4×10 Ωcm, while
986:
is not as significant as those achieved upon treatment with a
495:
form of the polymer is thermodynamically less stable than the
854:
yields the final polymer, as well as volatile side products.
802:
crowding. They obtained soluble and conductive polymers with
388:
processing have led to avoidance in commercial applications.
2008:"Raman Scattering and Electronic Spectra of Poly(acetylene)"
959:
can also be used to create conductive polyacetylene. n-Type
635:, which has served as model in other contexts to understand
1607:
Meier, Eric J.; An, Fangzhao Alex; Gadway, Bryce (2016).
1384:"The Nobel Prize in Chemistry, 2000: Conductive Polymers"
1559:
Journal of the Chemical Society, Chemical Communications
280:
979:. The increase in conductivity upon treatment with an
339:. The name refers to its conceptual construction from
2082:
The Nobel Prize in Chemistry 2000 presentation speech
1000:Polyacetylene can also be doped electrochemically.
720:system, which was stable to both oxygen and water.
511:of the polymer chain to interrupt the conjugation.
591:. Further studies led to improved control of the
363:. The high electrical conductivity discovered by
2006:Shirakawa, H. S.; Ito, T. S.; Ikeda, S. (1973).
1973:Shirakawa, H. S.; Ito, T. S.; Ikeda, S. (1971).
1496:Hall, N; McDiarmid, Alan; Heeger, Alan (2003).
1047:form predominates, while above 150 °C the
138:
1833:: n-type and p-type doping and compensation".
997:are extremely sensitive to air and moisture.
8:
1548:
1546:
1544:
778:chains. Polymers with linear groups such as
971:are created, where the polymer backbone is
27:Organic polymer made of the repeating unit
1975:"Infrared Spectroscopy of Poly(acetylene)"
1498:"Twenty-five years of conducting polymers"
794:groups increased solubility but decreased
456:Polyacetylene consists of a long chain of
36:
2023:
1990:
1824:
1822:
1658:
1624:
1461:
1459:
1457:
1455:
1348:
1258:
806:-butyl and neopentyl groups, because the
631:defects, their model is now known as the
556:were studying the metallic properties of
1962:from the original on September 24, 2017.
1780:Journal of the American Chemical Society
1718:Journal of the American Chemical Society
1453:
1451:
1449:
1447:
1445:
1443:
1441:
1439:
1437:
1435:
1377:
1375:
1373:
1371:
1369:
1298:Journal of the American Chemical Society
1290:
1288:
1286:
1284:
990:dopant. Polyacetylene chains doped with
841:
383:). This discovery was recognized by the
333:
329:
325:
188:
184:
180:
1400:
1398:
1396:
1197:
763:, a material easier to handle than the
468:atom. The double bonds can have either
1491:
1489:
1112:Products of oxidation of polyacetylene
757:ring-opening metathesis polymerisation
751:Ring-opening metathesis polymerization
359:helped to launch the field of organic
1870:
1868:
1866:
1864:
1081:-polyacetylene is much more brittle.
7:
755:Polyacetylene can be synthesized by
603:-polyacetylene doping led to higher
1275:"The Nobel Prize in Chemistry 2000"
1165:for application purposes including
599:isomer ratio and demonstrated that
479:. The controlled synthesis of each
1931:MacDiarmid, A; Heeger, A. (1979).
460:atoms with alternating single and
25:
1745:Journal of Polymer Science Part A
798:due to polymer twisting to avoid
442:
428:
270:
252:
80:
63:
43:
1065:form appear coppery, while the
347:to give a chain with repeating
266:(at 25 °C , 100 kPa).
1:
1341:10.1016/j.plipres.2008.02.002
821:Grubbs route to polyacetylene
1956:10.1016/0379-6779(80)90002-8
1703:10.1016/0032-3861(96)00439-9
1594:10.1016/0379-6779(94)02340-5
1480:10.1016/0079-6700(85)90008-5
1426:10.1016/0032-3861(96)00439-9
940:. Polyacetylene doped with (
912:from the polymer chain. The
464:between them, each with one
399:), rather than to chains of
1220:10.1002/macp.1988.021891113
2128:
2061:10.1002/pol.1983.170211210
2041:Journal of Polymer Science
1382:Norden, B; Krutmeijer, E.
1329:Progress in Lipid Research
1071:form is silvery. Films of
633:Su–Schrieffer–Heeger model
29:
1323:Minto, Robert E. (2008).
1260:10.1103/RevModPhys.73.681
1239:Reviews of Modern Physics
969:charge-transfer complexes
918:charge-transfer complexes
260:
233:
228:
210:
165:
122:
114:
104:
99:
79:
62:
42:
920:between the polymer and
385:Nobel Prize in Chemistry
1061:. Films containing the
932:and the acceptor as an
879:electrical conductivity
826:From precursor polymers
615:-polyacetylene with AsF
611:-polyacetylene. Doping
403:groups ("poly" meaning
315:) usually refers to an
2107:Organic semiconductors
1937:) and its Derivatives"
1901:10.1098/rsta.1985.0004
1113:
908:act by abstracting an
862:
846:
822:
698:
662:Ziegler–Natta catalyst
657:
637:topological insulators
381:organic semiconductors
2097:Molecular electronics
1806:"Conducting Polymers"
1765:10.1002/pola.1996.854
1613:Nature Communications
1233:Heeger, Alan (2001).
1175:Molecular electronics
1130:Infrared spectroscopy
1111:
1037:infrared spectroscopy
860:
845:
820:
696:
655:
88:Ball-and-stick models
2025:10.1295/polymj.4.460
1992:10.1295/polymj.2.231
1567:10.1039/C39770000578
1148:or wax can slow the
852:Diels–Alder reaction
837:poly(vinyl chloride)
656:Ziegler–Natta scheme
575:polyacetylene with I
499:isomer. Despite the
449:Ball-and-stick model
70:Skeletal formula of
2053:1983JPoSA..21.3479W
1893:1985RSPTA.314....3M
1847:1978ApPhL..33...18C
1792:10.1021/ja00231a036
1757:1996JPoSA..34.2533I
1730:10.1021/ja00364a076
1643:10.1038/ncomms13986
1635:2016NatCo...713986M
1310:10.1021/ja00057a024
1251:2001RvMP...73..681H
1163:conductive polymers
1132:shows formation of
951:Electron-donating (
833:dehydrohalogenation
589:conductive polymers
361:conductive polymers
201:Solubility in water
39:
1114:
1041:Raman spectroscopy
863:
847:
823:
699:
658:
487:-polyacetylene or
435:Structural diagram
293:Infobox references
211:Related compounds
117:Polyacetylene, PAc
37:
2047:(12): 3479–3492.
1786:(23): 7807–7813.
1724:(26): 7787–7789.
1697:(22): 5017–5047.
1214:(11): 2657–2669.
975:and the donor is
938:Coulomb potential
761:cyclooctatetraene
405:polymerization of
301:Chemical compound
299:
298:
217:Related compounds
16:(Redirected from
2119:
2102:Organic polymers
2065:
2064:
2036:
2030:
2029:
2027:
2003:
1997:
1996:
1994:
1970:
1964:
1963:
1950:(101–118): 101.
1944:Synthetic Metals
1941:
1928:
1913:
1912:
1872:
1859:
1858:
1835:Appl. Phys. Lett
1826:
1817:
1816:
1810:
1802:
1796:
1795:
1775:
1769:
1768:
1740:
1734:
1733:
1713:
1707:
1706:
1688:
1679:
1673:
1672:
1662:
1628:
1604:
1598:
1597:
1582:Synthetic Metals
1577:
1571:
1570:
1550:
1539:
1538:
1536:
1535:
1529:
1523:. Archived from
1513:10.1039/B210718J
1502:
1493:
1484:
1483:
1468:Prog. Polym. Sci
1463:
1430:
1429:
1411:
1402:
1391:
1390:
1388:
1379:
1364:
1362:
1352:
1320:
1314:
1313:
1304:(4): 1397–1409.
1292:
1279:
1278:
1271:
1265:
1264:
1262:
1230:
1224:
1223:
1202:
747:-polyacetylene.
625:J. R. Schrieffer
534:Hideki Shirakawa
505:functional group
483:of the polymer,
446:
432:
377:microelectronics
365:Hideki Shirakawa
338:
283:
277:
274:
273:
256:
193:
173:Chemical formula
142:
84:
67:
51:Skeletal formula
47:
40:
21:
2127:
2126:
2122:
2121:
2120:
2118:
2117:
2116:
2087:
2086:
2073:
2068:
2038:
2037:
2033:
2005:
2004:
2000:
1972:
1971:
1967:
1939:
1936:
1930:
1929:
1916:
1874:
1873:
1862:
1855:10.1063/1.90166
1832:
1828:
1827:
1820:
1808:
1804:
1803:
1799:
1777:
1776:
1772:
1742:
1741:
1737:
1715:
1714:
1710:
1686:
1681:
1680:
1676:
1606:
1605:
1601:
1579:
1578:
1574:
1561:(16): 578–580.
1556:
1552:
1551:
1542:
1533:
1531:
1527:
1500:
1495:
1494:
1487:
1465:
1464:
1433:
1409:
1404:
1403:
1394:
1386:
1381:
1380:
1367:
1322:
1321:
1317:
1294:
1293:
1282:
1273:
1272:
1268:
1232:
1231:
1227:
1208:Makromol. Chem.
1204:
1203:
1199:
1195:
1183:
1158:
1144:. Coating with
1127:
1033:
926:Charge transfer
903:
899:
895:
891:
868:
828:
813:
753:
718:
711:
707:
688:
684:
680:
673:
650:
645:
618:
607:than doping of
586:
582:
578:
566:superconductive
562:Alan MacDiarmid
554:Alan MacDiarmid
547:
541:
517:
454:
453:
452:
451:
450:
447:
438:
437:
436:
433:
424:
423:
413:
373:Alan MacDiarmid
337:
331:
327:
323:
317:organic polymer
302:
295:
290:
289:
288: ?)
279:
275:
271:
267:
249:
218:
203:
192:
186:
182:
178:
175:
161:
145:
132:
118:
110:
95:
85:
75:
68:
58:
48:
35:
28:
23:
22:
15:
12:
11:
5:
2125:
2123:
2115:
2114:
2109:
2104:
2099:
2089:
2088:
2085:
2084:
2079:
2072:
2071:External links
2069:
2067:
2066:
2031:
2018:(4): 460–462.
1998:
1985:(2): 231–244.
1965:
1934:
1914:
1887:(1528): 3–15.
1860:
1830:
1818:
1797:
1770:
1735:
1708:
1674:
1599:
1572:
1554:
1540:
1485:
1431:
1392:
1365:
1335:(4): 233–306.
1315:
1280:
1266:
1245:(3): 681–700.
1225:
1196:
1194:
1191:
1190:
1189:
1182:
1179:
1157:
1154:
1125:
1032:
1029:
901:
897:
893:
889:
867:
864:
827:
824:
811:
752:
749:
725:Glow-discharge
716:
709:
705:
686:
682:
678:
671:
649:
648:From acetylene
646:
644:
641:
616:
584:
580:
576:
545:
539:
516:
513:
448:
441:
440:
439:
434:
427:
426:
425:
422:-polyacetylene
417:
416:
415:
414:
412:
409:
341:polymerization
321:repeating unit
300:
297:
296:
291:
269:
268:
264:standard state
261:
258:
257:
250:
245:
242:
241:
231:
230:
226:
225:
224:gas (monomer)
219:
216:
213:
212:
208:
207:
204:
199:
196:
195:
176:
171:
168:
167:
163:
162:
160:
159:
155:
153:
147:
146:
144:
143:
135:
133:
128:
125:
124:
120:
119:
116:
112:
111:
108:
102:
101:
97:
96:
86:
77:
76:
74:-polyacetylene
69:
60:
59:
57:-polyacetylene
49:
38:Polyacetylene
26:
24:
18:Polyacetylenes
14:
13:
10:
9:
6:
4:
3:
2:
2124:
2113:
2110:
2108:
2105:
2103:
2100:
2098:
2095:
2094:
2092:
2083:
2080:
2078:
2077:Polyacetylene
2075:
2074:
2070:
2062:
2058:
2054:
2050:
2046:
2042:
2035:
2032:
2026:
2021:
2017:
2013:
2009:
2002:
1999:
1993:
1988:
1984:
1980:
1976:
1969:
1966:
1961:
1957:
1953:
1949:
1945:
1938:
1927:
1925:
1923:
1921:
1919:
1915:
1910:
1906:
1902:
1898:
1894:
1890:
1886:
1882:
1878:
1871:
1869:
1867:
1865:
1861:
1856:
1852:
1848:
1844:
1840:
1836:
1825:
1823:
1819:
1814:
1807:
1801:
1798:
1793:
1789:
1785:
1781:
1774:
1771:
1766:
1762:
1758:
1754:
1751:(13): 11–20.
1750:
1746:
1739:
1736:
1731:
1727:
1723:
1719:
1712:
1709:
1704:
1700:
1696:
1692:
1685:
1678:
1675:
1670:
1666:
1661:
1656:
1652:
1648:
1644:
1640:
1636:
1632:
1627:
1622:
1618:
1614:
1610:
1603:
1600:
1595:
1591:
1587:
1583:
1576:
1573:
1568:
1564:
1560:
1549:
1547:
1545:
1541:
1530:on 2016-03-04
1526:
1522:
1518:
1514:
1510:
1506:
1499:
1492:
1490:
1486:
1481:
1477:
1473:
1469:
1462:
1460:
1458:
1456:
1454:
1452:
1450:
1448:
1446:
1444:
1442:
1440:
1438:
1436:
1432:
1427:
1423:
1419:
1415:
1408:
1401:
1399:
1397:
1393:
1385:
1378:
1376:
1374:
1372:
1370:
1366:
1360:
1356:
1351:
1346:
1342:
1338:
1334:
1330:
1326:
1319:
1316:
1311:
1307:
1303:
1299:
1291:
1289:
1287:
1285:
1281:
1276:
1270:
1267:
1261:
1256:
1252:
1248:
1244:
1240:
1236:
1229:
1226:
1221:
1217:
1213:
1210:
1209:
1201:
1198:
1192:
1188:
1185:
1184:
1180:
1178:
1176:
1172:
1168:
1167:polythiophene
1164:
1155:
1153:
1151:
1147:
1143:
1139:
1135:
1131:
1123:
1119:
1110:
1106:
1104:
1103:cross-linking
1100:
1096:
1092:
1088:
1082:
1080:
1079:
1074:
1070:
1069:
1064:
1060:
1056:
1052:
1051:
1046:
1042:
1038:
1030:
1028:
1026:
1022:
1018:
1014:
1010:
1006:
1001:
998:
996:
993:
989:
985:
982:
978:
974:
970:
966:
962:
958:
954:
949:
947:
943:
939:
935:
931:
927:
923:
919:
915:
911:
907:
887:
884:
880:
876:
873:
865:
859:
855:
853:
844:
840:
838:
834:
825:
819:
815:
809:
805:
801:
797:
793:
789:
785:
781:
777:
773:
772:Robert Grubbs
769:
766:
762:
758:
750:
748:
746:
742:
738:
734:
730:
726:
721:
719:
712:
695:
691:
689:
674:
669:
663:
654:
647:
642:
640:
638:
634:
630:
626:
621:
614:
610:
606:
602:
598:
594:
590:
574:
569:
567:
563:
559:
555:
551:
543:
535:
530:
528:
523:
514:
512:
510:
506:
502:
498:
494:
490:
486:
482:
478:
476:
472:
467:
463:
459:
445:
431:
421:
418:A segment of
410:
408:
406:
402:
398:
394:
389:
386:
382:
378:
374:
370:
366:
362:
358:
354:
350:
346:
342:
336:
322:
318:
314:
310:
306:
305:Polyacetylene
294:
287:
282:
265:
259:
255:
251:
248:
244:
243:
239:
237:
232:
227:
223:
220:
215:
214:
209:
205:
202:
198:
197:
191:
177:
174:
170:
169:
164:
157:
156:
154:
152:
149:
148:
141:
137:
136:
134:
131:
127:
126:
121:
113:
107:
103:
98:
93:
89:
83:
78:
73:
66:
61:
56:
52:
46:
41:
33:
19:
2044:
2040:
2034:
2015:
2011:
2001:
1982:
1978:
1968:
1947:
1943:
1884:
1880:
1838:
1834:
1812:
1800:
1783:
1779:
1773:
1748:
1744:
1738:
1721:
1717:
1711:
1694:
1690:
1677:
1619:(1): 13986.
1616:
1612:
1602:
1585:
1581:
1575:
1558:
1532:. Retrieved
1525:the original
1504:
1471:
1467:
1420:(22): 5017.
1417:
1413:
1332:
1328:
1318:
1301:
1297:
1269:
1242:
1238:
1228:
1211:
1206:
1200:
1159:
1156:Applications
1146:polyethylene
1121:
1117:
1115:
1083:
1076:
1072:
1066:
1062:
1058:
1054:
1048:
1044:
1034:
1024:
1020:
1016:
1012:
1008:
1004:
1002:
999:
950:
914:conductivity
869:
848:
829:
803:
787:
779:
759:(ROMP) from
754:
744:
740:
722:
700:
667:
659:
622:
612:
608:
605:conductivity
600:
596:
592:
570:
531:
526:
522:Giulio Natta
518:
509:conformation
496:
492:
488:
484:
474:
470:
462:double bonds
455:
419:
404:
396:
390:
353:conductivity
334:
312:
304:
303:
235:
189:
123:Identifiers
115:Other names
91:
71:
54:
1813:ch.ic.ac.uk
1505:Chem. Comm.
1474:(1–2): 57.
1171:polyaniline
796:conjugation
733:ultraviolet
558:polythiazyl
550:Alan Heeger
544:and Ti(OBu)
369:Alan Heeger
166:Properties
2091:Categories
1626:1607.02811
1588:(1–3): 3.
1534:2014-03-14
1507:(1): 1–4.
1193:References
1031:Properties
888:include Br
737:cryogenics
664:, such as
501:conjugated
313:polyethyne
247:Pictograms
206:insoluble
151:ChemSpider
140:25067-58-7
130:CAS Number
109:Polyethyne
106:IUPAC name
1841:(1): 18.
1651:2041-1723
1150:oxidation
1142:peroxides
967:dopants,
900:, and AsF
808:methylene
765:acetylene
643:Synthesis
629:solitonic
411:Structure
345:acetylene
319:with the
238:labelling
222:Acetylene
2112:Polyenes
2012:Polym. J
1979:Polym. J
1960:Archived
1909:91941666
1669:28008924
1521:12610942
1359:18387369
1181:See also
1138:epoxides
1136:groups,
1134:carbonyl
1128:occurs.
977:cationic
910:electron
904:. These
477:geometry
466:hydrogen
393:polyynes
229:Hazards
2049:Bibcode
1889:Bibcode
1843:Bibcode
1753:Bibcode
1691:Polymer
1660:5196433
1631:Bibcode
1414:Polymer
1350:2515280
1247:Bibcode
1187:Polyene
1099:fibrils
1095:benzene
1091:solvent
995:dopants
973:anionic
961:dopants
957:dopants
946:dopants
922:halogen
906:dopants
886:dopants
877:), the
875:dopants
768:monomer
515:History
286:what is
284: (
194:
32:polyyne
1907:
1667:
1657:
1649:
1519:
1357:
1347:
1161:other
1140:, and
992:n-type
988:p-type
984:dopant
981:n-type
965:p-type
953:n-type
942:p-type
930:cation
883:p-Type
872:p-type
866:Doping
800:steric
731:, and
583:and Br
573:doping
481:isomer
458:carbon
401:olefin
371:, and
357:doping
349:olefin
324:[C
311:name:
281:verify
278:
179:[C
100:Names
94:isomer
1940:(PDF)
1905:S2CID
1809:(PDF)
1687:(PDF)
1621:arXiv
1528:(PDF)
1501:(PDF)
1410:(PDF)
1387:(PDF)
1122:trans
1093:with
1078:trans
1068:trans
1059:trans
1050:trans
1025:trans
1017:trans
1015:- or
1005:trans
934:anion
792:butyl
784:octyl
776:alkyl
745:trans
729:gamma
704:Co(NO
609:trans
597:trans
571:Upon
527:trans
497:trans
489:trans
475:trans
420:trans
355:upon
309:IUPAC
92:trans
55:trans
1665:PMID
1647:ISSN
1517:PMID
1355:PMID
1169:and
1120:and
1087:gels
1039:and
896:, Cl
788:tert
715:NaBH
677:Al(C
666:Ti(O
552:and
397:many
158:none
2057:doi
2020:doi
1987:doi
1952:doi
1897:doi
1885:314
1851:doi
1788:doi
1784:110
1761:doi
1726:doi
1722:105
1699:doi
1655:PMC
1639:doi
1590:doi
1563:doi
1557:".
1509:doi
1476:doi
1422:doi
1345:PMC
1337:doi
1306:doi
1302:115
1255:doi
1216:doi
1212:189
1118:cis
1073:cis
1063:cis
1055:cis
1045:cis
1021:cis
1013:cis
1009:cis
892:, I
835:of
810:(CH
804:sec
741:cis
670:Pr)
613:cis
601:cis
593:cis
493:cis
485:cis
473:or
471:cis
407:).
343:of
236:GHS
72:cis
53:of
2093::
2055:.
2045:21
2043:.
2014:.
2010:.
1981:.
1977:.
1958:.
1946:.
1942:.
1917:^
1903:.
1895:.
1883:.
1879:.
1863:^
1849:.
1839:33
1837:.
1821:^
1811:.
1782:.
1759:.
1749:12
1747:.
1720:.
1695:37
1693:.
1689:.
1663:.
1653:.
1645:.
1637:.
1629:.
1615:.
1611:.
1586:69
1584:.
1543:^
1515:.
1503:.
1488:^
1472:11
1470:.
1434:^
1418:37
1416:.
1412:.
1395:^
1368:^
1353:.
1343:.
1333:47
1331:.
1327:.
1300:.
1283:^
1253:.
1243:73
1241:.
1237:.
1173:.
955:)
944:)
924:.
839::
727:,
639:.
542:Al
538:Et
367:,
240::
2063:.
2059::
2051::
2028:.
2022::
2016:4
1995:.
1989::
1983:2
1954::
1948:1
1935:x
1911:.
1899::
1891::
1857:.
1853::
1845::
1831:x
1815:.
1794:.
1790::
1767:.
1763::
1755::
1732:.
1728::
1705:.
1701::
1671:.
1641::
1633::
1623::
1617:7
1596:.
1592::
1569:.
1565::
1555:x
1537:.
1511::
1482:.
1478::
1428:.
1424::
1389:.
1363:.
1361:.
1339::
1312:.
1308::
1277:.
1263:.
1257::
1249::
1222:.
1218::
1126:2
1057::
902:5
898:2
894:2
890:2
812:2
790:-
782:-
780:n
717:4
713:/
710:2
708:)
706:3
687:3
685:)
683:5
681:H
679:2
675:/
672:4
668:i
617:5
595:/
585:2
581:2
577:2
546:4
540:3
379:(
335:n
332:]
330:2
328:H
326:2
307:(
276:N
190:n
187:]
185:2
183:H
181:2
34:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.