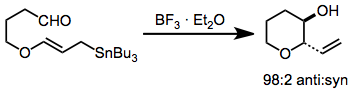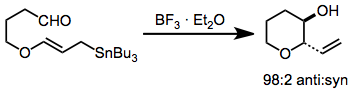244:
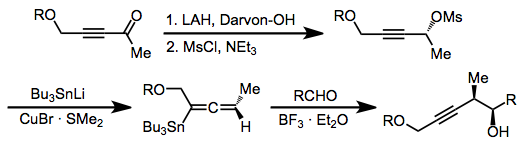
The possibility of incorporating oxygen-containing substituents into allyl- and allenylstannanes expands their scope and utility substantially over methods relying on more reactive organometallics. These compounds are usually prepared by enantioselective reduction with a chiral reducing agent such as
224:
Thermal additions of stannanes are limited (because of the high temperatures and pressures required) to only simple aldehyde substrates. Lewis acid promoted and transmetalation reactions are much milder and have achieved synthetic utility. Intramolecular addition gives five- or six-membered rings
146:
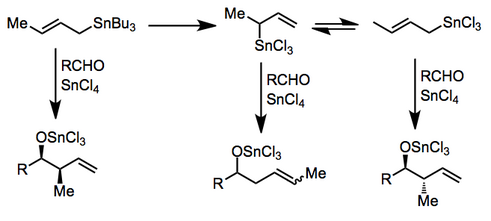
In the presence of certain Lewis acids, transmetalation may occur before addition. Complex reaction mixtures may result if transmetalation is not complete or if an equilibrium between allylic isomers exists. Tin(IV) chloride and indium(III) chloride have been employed for useful reactions in this
63:
Three modes allow the addition of allylstannanes to carbonyls: thermal addition, Lewis-acid-promoted addition, and addition involving prior transmetalation. Each of these modes invokes a unique model for stereocontrol, but in all cases, a distinction is made between reagent and substrate control.
83:
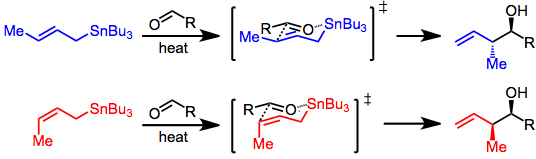
With the allylstannane and aldehyde in high-temperature conditions, addition proceeds through a six-membered, cyclic transition state, with the tin center serving as an organizing element. The configuration of the double bond in the allylstannane controls the sense of diastereoselectivity of the
29:
addition to carbonyl groups constitutes one of the most common and efficient methods for the construction of contiguous, oxygen-containing stereocenters in organic molecules. As many molecules containing this motif—polypropionates and polyacetates, for instance—are desired by natural products
49:
Organostannanes are known for their stability, ease of handling, and selective reactivity. Chiral allylstannanes often react with good stereoselectivity to give single diastereomers. Models explaining the sense of selectivity are reliable. In terms of disadvantages, stoichiometric amounts of
287:
Nucleophilic addition to propargyl mesylates or tosylates is used to form allenylstannanes. These compounds react similarly to allylstannanes to afford homopropargyl alcohols, and any of the three reaction modes described above can be used with this class of reagents as well.
362:
Repeated use of the allylic stannane addition in an intramolecular sense was used in the synthesis of hemibrevetoxin B (one example is shown below). The pseudoequatorial positions of both "appendages" in the starting material lead to the observed stereoisomer.
377:
161:
30:
chemists, the title reaction has become important synthetically and has been heavily studied over the years. Substituted allylstannanes may create one or two new stereocenters, often with a very high degree of stereocontrol.
44:
343:
diastereomer as a single stereoisomer. A subsequent sigmatropic rearrangement increased the distance between the stereocenters even further. This step was carried out en route to (±)-patulolide C.
307:
Imines are less reactive than the corresponding aldehydes, but palladium catalysis can be used to facilitate addition into imines. The use of iminium ions as electrophiles has also been reported.
19:
reactions comprise the nucleophilic addition of an allyl-, allenyl-, or propargylstannane to an aldehyde, imine, or, in rare cases, a ketone. The reaction is widely used for
264:
The use of chiral electrophiles is common and can provide "double diastereoselection" if the stannane is also chiral. Chelation control using Lewis acids such as
115:
product predominantly (Type II). The origin of this selectivity has been debated, and depends on the relative energies of a number of acyclic transition states. (
321:
302:
282:
245:
BINAL-H. In the presence of a Lewis acid, isomerization of α-alkoxy allylstannanes to the corresponding γ-alkoxy isomers takes place.
213:
98:
259:
239:
357:
141:
50:
metal-containing byproducts are generated. Additions to sterically encumbered pi bonds, such as those of ketones, are uncommon.
754:
172:
393:
204:
20:
388:
265:
80:' mechanism involving concerted dissociation of tin and C-C bond formation at the γ position.
425:
64:
Substrate-controlled additions typically involve chiral aldehydes or imines and invoke the
65:
748:
175:, non-racemic Lewis acids are known. The chiral (acyloxy)borane or "CAB" catalyst
429:
413:
103:
This is not the case in Lewis-acid-promoted reactions, in which either the (
26:
320:
301:
281:
258:
238:
212:
97:
376:
268:
can lead to high stereoselectivities for reactions of α-alkoxy aldehydes.
336:
180:
356:
192:
160:
140:
68:. When all reagents are achiral, only simple diastereoselectivity (
196:
184:
43:
76:, see above) must be considered. Addition takes place via an S
500:
Keck, G. E.; Savin, K. A.; Cressman, E. N. K.; Abbott, D. E.
207:
via the Lewis-acid-promoted mechanism described above.
171:
A wide variety of enantioselective additions employing
576:Marshall, J. A.; Jablonowski, J. A.; Jiang, H.
8:
520:Keck, G. E.; Dougherty, S. M.; Savin, K. A.
460:Keck, G. E.; Dougherty, S. M.; Savin, K. A.
225:under Lewis acidic or thermal conditions.
404:
672:Nakamura, H.; Iwama, H.; Yamamoto, Y.
7:
123:selectivity than the corresponding (
636:Hara, O.; Hamada, Y.; Shioiri, T.
616:Marshall, J. A.; Yashunsky, D. V.
203:provide addition products in high
14:
375:
355:
319:
300:
280:
257:
237:
211:
159:
139:
96:
42:
556:Marshall, J. A.; Hinkle, K. W.
540:McNeill, A. H.; Thomas, E. J.
1:
596:Marshall, J. A.; Gung, W. Y.
440:Denmark, S. E.; Weber, E. J.
54:Mechanism and stereochemistry
708:Dorling, E.K.; Thomas, E.J.
694:J. Chem. Soc., Chem. Commun.
652:Ruitenberg, K.; Vermeer, P.
331:The chiral allylic stannane
480:Denmark, S.E.; Weber, E.J.
430:10.1002/0471264180.or064.01
119:)-Stannanes exhibit higher
771:
728:Kadota, I.; Yamamoto, Y.
692:Yamamoto, Y.; Schmid, M.
167:Enantioselective variants
111:)-stannane affords the
17:Organostannane addition
327:Synthetic applications
220:Scope and limitations
59:Prevailing mechanism
394:Carbonyl allylation
21:carbonyl allylation
389:Krische allylation
755:Organic reactions
710:Tetrahedron Lett.
674:J. Am. Chem. Soc.
654:Tetrahedron Lett.
598:Tetrahedron Lett.
522:J. Am. Chem. Soc.
462:J. Am. Chem. Soc.
442:J. Am. Chem. Soc.
339:to yield the 1,5-
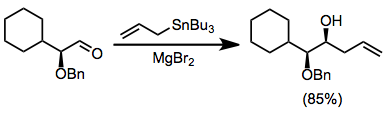
266:magnesium bromide
762:
740:
726:
720:
706:
700:
690:
684:
670:
664:
650:
644:
634:
628:
614:
608:
594:
588:
574:
568:
554:
548:
538:
532:
518:
512:
498:
492:
482:Helv. Chim. Acta
478:
472:
458:
452:
438:
432:
409:
383:Related articles
379:
372:
359:
352:
323:
316:
304:
297:
284:
277:
261:
254:
241:
234:
215:
163:
156:
143:
136:
100:
93:
66:Felkin-Anh model
46:
39:
770:
769:
765:
764:
763:
761:
760:
759:
745:
744:
743:
727:
723:
707:
703:
691:
687:
671:
667:
651:
647:
635:
631:
615:
611:
595:
591:
575:
571:
555:
551:
539:
535:
519:
515:
499:
495:
479:
475:
459:
455:
439:
435:
410:
406:
402:
385:
380:
366:
360:
346:
329:
324:
310:
305:
291:
285:
271:
262:
248:
242:
228:
222:
169:
164:
150:
144:
130:
101:
87:
79:
61:
56:
47:
33:
12:
11:
5:
768:
766:
758:
757:
747:
746:
742:
741:
721:
701:
685:
665:
645:
629:
609:
589:
569:
549:
533:
513:
493:
473:
453:
433:
403:
401:
398:
397:
396:
391:
384:
381:
374:
354:
328:
325:
318:
299:
279:
256:
236:
221:
218:
217:
216:
168:
165:
158:
138:
95:
77:
60:
57:
55:
52:
41:
27:Organostannane
13:
10:
9:
6:
4:
3:
2:
767:
756:
753:
752:
750:
738:
734:
731:
730:J. Org. Chem.
725:
722:
718:
714:
711:
705:
702:
698:
695:
689:
686:
682:
678:
675:
669:
666:
662:
658:
655:
649:
646:
642:
639:
633:
630:
626:
622:
619:
618:J. Org. Chem.
613:
610:
606:
602:
599:
593:
590:
586:
582:
579:
578:J. Org. Chem.
573:
570:
566:
562:
559:
558:J. Org. Chem.
553:
550:
546:
543:
537:
534:
530:
526:
523:
517:
514:
510:
506:
503:
502:J. Org. Chem.
497:
494:
490:
486:
483:
477:
474:
470:
466:
463:
457:
454:
450:
446:
443:
437:
434:
431:
427:
423:
419:
416:
415:
408:
405:
399:
395:
392:
390:
387:
386:
382:
378:
373:
371:
370:
364:
358:
353:
351:
350:
344:
342:
338:
334:
326:
322:
317:
315:
314:
308:
303:
298:
296:
295:
289:
283:
278:
276:
275:
269:
267:
260:
255:
253:
252:
246:
240:
235:
233:
232:
226:
219:
214:
210:
209:
208:
206:
202:
198:
194:
190:
186:
182:
178:
174:
166:
162:
157:
155:
154:
148:
142:
137:
135:
134:
128:
127:)-stannanes.
126:
122:
118:
114:
110:
106:
99:
94:
92:
91:
85:
81:
75:
71:
67:
58:
53:
51:
45:
40:
38:
37:
31:
28:
24:
22:
18:
736:
732:
729:
724:
716:
712:
709:
704:
696:
693:
688:
680:
676:
673:
668:
660:
656:
653:
648:
640:
637:
632:
624:
620:
617:
612:
604:
600:
597:
592:
584:
580:
577:
572:
564:
560:
557:
552:
544:
541:
536:
528:
524:
521:
516:
508:
504:
501:
496:
488:
484:
481:
476:
468:
464:
461:
456:
448:
444:
441:
436:
421:
417:
412:
411:Gung, B. W.
407:
368:
367:
365:
361:
348:
347:
345:
340:
332:
330:
312:
311:
309:
306:
293:
292:
290:
286:
273:
272:
270:
263:
250:
249:
247:
243:
230:
229:
227:
223:
200:
188:
176:
170:
152:
151:
149:
145:
132:
131:
129:
124:
120:
116:
112:
108:
104:
102:
89:
88:
86:
82:
73:
69:
62:
48:
35:
34:
32:
25:
16:
15:
414:Org. React.
400:References
84:reaction.
542:Synthesis
424:, 1-112.
749:Category
337:acrolein
335:adds to
181:titanium
739:, 6597.
699:, 1310.
683:, 6641.
663:, 3019.
638:Synlett
627:, 5493.
607:, 7349.
587:, 2152.
567:, 1920.
531:, 6210.
511:, 7889.
491:, 1655.
471:, 6210.
451:, 7970.
199:system
187:system
107:)- or (
72:versus
719:, 471.
643:, 283.
547:, 322.
193:silver
191:, and
173:chiral
147:mode.
197:BINAP
185:BINOL
733:1998
713:1999
697:1989
677:1996
657:1984
641:1991
621:1991
601:1989
581:1999
561:1995
545:1994
525:1995
505:1994
485:1983
465:1995
445:1984
418:2004
369:(12)
349:(11)
313:(10)
74:anti
681:118
529:117
469:117
449:106
426:doi
341:syn
294:(9)
274:(8)
251:(7)
231:(6)
153:(4)
133:(3)
121:syn
113:syn
90:(2)
70:syn
36:(1)
751::
737:63
735:,
717:40
715:,
679:,
661:25
659:,
625:56
623:,
605:30
603:,
585:64
583:,
565:60
563:,
527:,
509:59
507:,
489:66
487:,
467:,
447:,
422:64
420:,
205:ee
179:,
23:.
428::
333:1
201:3
195:-
189:2
183:-
177:1
125:Z
117:E
109:E
105:Z
78:E
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.