402:
357:
387:
372:
342:
312:
38:
327:
237:(cyclobutadiene, annulene). Cyclobutadiene is the only annulene with considerable antiaromaticity, since planarity is unavoidable. With annulene, the molecule takes on a tub shape that allows it to avoid conjugation of double bonds. Annulene is of the wrong size to achieve a planar structure: in a planar conformation,
256:
When the annulene is large enough, annulene for example, there is enough room internally to accommodate hydrogen atoms without significant distortion of bond angles. Annulene possesses several properties that qualify it as aromatic. However, none of the larger annulenes are as stable as benzene, as
114:= 3 to 6) can also be informally referred to as annulenes. Using this form of nomenclature 1,3,5,7-cyclooctatetraene is annulene and benzene is annulene (and occasionally referred to as just 'annulene').
447:
100:
accepts the use of 'annulene nomenclature' in naming carbocyclic ring systems with 7 or more carbon atoms, using the name 'annulene' for the mancude hydrocarbon with
117:
The discovery that annulene possesses a number of key properties associated with other aromatic molecules was an important development in the understanding of
549:
Oth, Jean F. M.; Bünzli, Jean-Claude; De Julien De Zélicourt, Yves (1974-11-06). "The
Stabilization Energy of Annulene. A thermochemical determination".
756:
621:
497:
401:
356:
593:
371:
341:
110:
in its ring, though in certain contexts (e.g., discussions of aromaticity for different ring sizes), smaller rings (
386:
311:
614:
42:
672:
481:
377:
684:
678:
392:
751:
666:
607:
587:
362:
97:
716:
661:
656:
566:
531:
347:
326:
711:
558:
523:
461:
242:
257:
their reactivity more closely resembles a conjugated polyene than an aromatic hydrocarbon.
706:
501:
233:
53:
494:
644:
317:
745:
17:
457:
37:
701:
276:
3) are aromatic, provided a planar conformation can be achieved. For instance, C
452:
238:
129:
118:
56:
59:
that contain the maximum number of non-cumulated or conjugated double bonds ('
570:
562:
535:
456:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
465:
423:
60:
732:
527:
515:
428:
418:
223:
125:
650:
332:
31:
253:) is unavoidable. Thus, it does not exhibit appreciable aromaticity.
104:
36:
107:
603:
599:
516:"[16]Annulene: the crystal and molecular structure"
514:
Johnson, Suzanne M.; Paul, Iain C.; King, G. S. D. (1970).
249:) or bond angle distortion (when the double bonds are all
260:
In general, charged annulene species of the form (
694:
637:
520:
Journal of the
Chemical Society B: Physical Organic
245:of internal hydrogens (when some double bonds are
615:
27:Completely conjugated monocyclic hydrocarbons
8:
622:
608:
600:
478:Organic Chemistry:Structure and Reactivity
139:
440:
307:
45:image of a hexadehydrotribenzoannulene
7:
128:, one double bond is replaced by a
63:'). They have the general formula C
453:Compendium of Chemical Terminology
227:(benzene, annulene and annulene),
141:Aromaticity of the even annulenes
25:
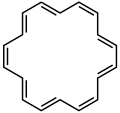
407:Cyclodocosahendecaene (-annulene)
400:
385:
370:
355:
340:
325:
310:
300:are all known aromatic species.
1:
773:
757:Physical organic chemistry
29:
725:
563:10.1002/hlca.19740570745
121:as a chemical concept.
30:Not to be confused with
492:Dublin City University
466:10.1351/goldbook.A00368
96:is an odd number). The
79:is an even number) or C
673:Cyclotetradecaheptaene
588:NIST Chemistry WebBook
551:Helvetica Chimica Acta
500:April 7, 2005, at the
482:D.C. Heath and Company
378:Cyclotetradecaheptaene
46:
40:
18:Cyclodocosahendecaene
685:Cyclooctadecanonaene
679:Cyclohexadecaoctaene
528:10.1039/j29700000643
393:Cyclooctadecanonaene
231:( and annulene), or
191:weakly antiaromatic
142:
667:Cyclododecahexaene
363:Cyclododecahexaene
140:
47:
739:
738:
717:Cyclononatetraene
662:Cyclodecapentaene
657:Cyclooctatetraene
348:Cyclooctatetraene
221:Annulenes may be
219:
218:
16:(Redirected from
764:
712:Cycloheptatriene
624:
617:
610:
601:
596:of and annulene
575:
574:
557:(7): 2276–2288.
546:
540:
539:
511:
505:
490:
484:
474:
468:
445:
404:
389:
374:
359:
344:
329:
314:
264:= 0, 1, 2, ...,
243:steric hindrance
199:weakly aromatic
143:
21:
772:
771:
767:
766:
765:
763:
762:
761:
742:
741:
740:
735:
721:
707:Cyclopentadiene
690:
633:
628:
584:
579:
578:
548:
547:
543:
513:
512:
508:
502:Wayback Machine
491:
487:
476:Ege, S. (1994)
475:
471:
446:
442:
437:
415:
408:
405:
396:
390:
381:
375:
366:
360:
351:
345:
336:
330:
321:
315:
306:
299:
295:
291:
287:
283:
279:
138:
124:In the related
91:
84:
74:
68:
35:
28:
23:
22:
15:
12:
11:
5:
770:
768:
760:
759:
754:
744:
743:
737:
736:
726:
723:
722:
720:
719:
714:
709:
704:
698:
696:
692:
691:
689:
688:
681:
676:
669:
664:
659:
654:
647:
645:Cyclobutadiene
641:
639:
635:
634:
629:
627:
626:
619:
612:
604:
598:
597:
591:
583:
582:External links
580:
577:
576:
541:
506:
485:
469:
439:
438:
436:
433:
432:
431:
426:
421:
414:
411:
410:
409:
406:
399:
397:
391:
384:
382:
376:
369:
367:
361:
354:
352:
346:
339:
337:
331:
324:
322:
318:Cyclobutadiene
316:
309:
305:
302:
297:
293:
289:
285:
281:
277:
268:= 0, ±1, ±2, 4
241:due to either
217:
216:
213:
209:
208:
205:
201:
200:
197:
193:
192:
189:
185:
184:
181:
177:
176:
173:
169:
168:
165:
161:
160:
157:
153:
152:
149:
137:
134:
86:
80:
70:
64:
41:Structure and
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
769:
758:
755:
753:
750:
749:
747:
734:
730:
727:Compounds in
724:
718:
715:
713:
710:
708:
705:
703:
700:
699:
697:
693:
687:
686:
682:
680:
677:
675:
674:
670:
668:
665:
663:
660:
658:
655:
653:
652:
648:
646:
643:
642:
640:
638:Even–numbered
636:
632:
625:
620:
618:
613:
611:
606:
605:
602:
595:
592:
589:
586:
585:
581:
572:
568:
564:
560:
556:
552:
545:
542:
537:
533:
529:
525:
521:
517:
510:
507:
504:
503:
499:
496:
489:
486:
483:
479:
473:
470:
467:
463:
459:
455:
454:
449:
444:
441:
434:
430:
427:
425:
422:
420:
417:
416:
412:
403:
398:
394:
388:
383:
379:
373:
368:
364:
358:
353:
349:
343:
338:
334:
328:
323:
319:
313:
308:
303:
301:
275:
271:
267:
263:
258:
254:
252:
248:
244:
240:
236:
235:
234:anti-aromatic
230:
226:
225:
214:
211:
210:
206:
203:
202:
198:
195:
194:
190:
187:
186:
182:
179:
178:
174:
171:
170:
166:
163:
162:
159:antiaromatic
158:
155:
154:
150:
148:
145:
144:
135:
133:
131:
127:
122:
120:
115:
113:
109:
106:
103:
99:
95:
89:
83:
78:
73:
67:
62:
58:
55:
51:
44:
39:
33:
19:
728:
702:Cyclopropene
695:Odd–numbered
683:
671:
649:
630:
554:
550:
544:
519:
509:
493:
488:
477:
472:
451:
443:
273:
269:
265:
261:
259:
255:
250:
246:
232:
229:non-aromatic
228:
222:
220:
207:nonaromatic
183:nonaromatic
175:nonaromatic
151:aromaticity
146:
123:
116:
111:
101:
93:
87:
81:
76:
71:
65:
57:hydrocarbons
49:
48:
522:: 643–649.
239:ring strain
136:Aromaticity
130:triple bond
119:aromaticity
746:Categories
590:- annulene
435:References
395:(annulene)
380:(annulene)
365:(annulene)
350:(annulene)
335:(annulene)
320:(annulene)
54:monocyclic
752:Annulenes
631:Annulenes
594:Structure
571:0018-019X
536:0045-6470
495:Annulenes
424:Circulene
215:aromatic
167:aromatic
126:annulynes
50:Annulenes
733:aromatic
498:Archived
480:3rd ed.
458:annulene
429:Fulvenes
419:Annulyne
413:See also
224:aromatic
729:italics
651:Benzene
333:Benzene
304:Gallery
292:, and C
61:mancude
32:aniline
569:
534:
272:+ 2 +
105:carbon
92:(when
75:(when
448:IUPAC
247:trans
108:atoms
98:IUPAC
731:are
567:ISSN
532:ISSN
52:are
559:doi
524:doi
462:doi
460:".
284:, C
274:q ≥
251:cis
212:18
204:16
196:14
188:12
180:10
43:AFM
748::
565:.
555:57
553:.
530:.
518:.
450:,
172:8
164:6
156:4
132:.
90:+1
623:e
616:t
609:v
573:.
561::
538:.
526::
464::
298:8
296:H
294:8
290:3
288:H
286:3
282:5
280:H
278:5
270:n
266:q
262:n
147:n
112:n
102:n
94:n
88:n
85:H
82:n
77:n
72:n
69:H
66:n
34:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.