162:
145:
43:
693:
1372:
805:
A strong electrolyte is a solute that exists in solution completely or nearly completely as ions. Again, the strength of an electrolyte is defined as the percentage of solute that is ions, rather than molecules. The higher the percentage, the stronger the electrolyte. Thus, even if a substance is not
430:
is the fraction of original solute molecules that have dissociated. It is usually indicated by the Greek symbol α. More accurately, degree of dissociation refers to the amount of solute dissociated into ions or radicals per mole. In case of very strong acids and bases, degree of dissociation will be
1582:
constant. In fact, increasing the pressure of the equilibrium favours a shift to the left favouring the formation of dinitrogen tetroxide (as on this side of the equilibrium there is less pressure since pressure is proportional to number of moles) hence decreasing the extent of dissociation
753:
A weak electrolyte is a substance whose solute exists in solution mostly in the form of molecules (which are said to be "undissociated"), with only a small fraction in the form of ions. Simply because a substance does not readily dissolve does not make it a weak electrolyte.
696:
700:
699:
695:
694:
701:
1549:
1146:
782:) are good examples. Acetic acid is extremely soluble in water, but most of the compound dissolves into molecules, rendering it a weak electrolyte. Weak bases and weak acids are generally weak electrolytes. In an aqueous solution there will be some
698:
1116:
806:
very soluble, but does dissociate completely into ions, the substance is defined as a strong electrolyte. Similar logic applies to a weak electrolyte. Strong acids and bases are good examples, such as HCl and
1401:
1929:
2035:
1783:
615:
1687:
978:
397:
697:
313:
524:
1367:{\displaystyle K_{p}={\frac {p_{T}^{2}{\bigl (}x\,{\ce {NO2}}{\bigr )}^{2}}{p_{T}\cdot x\,{\ce {N2O4}}}}={\frac {p_{T}{\bigl (}x\,{\ce {NO2}}{\bigr )}^{2}}{x\,{\ce {N2O4}}}}}
676:
1012:
2081:
428:
644:
474:
453:
750:
medium. Most of the solute does not dissociate in a weak electrolyte, whereas in a strong electrolyte a higher ratio of solute dissociates to form free ions.
1789:
1696:
COOH. The double arrow means that this is an equilibrium process, with dissociation and recombination occurring at the same time. This implies that the
60:
431:
close to 1. Less powerful acids and bases will have lesser degree of dissociation. There is a simple relationship between this parameter and the
2316:
2274:
2244:
107:
126:
79:
881:
86:
2339:
64:
31:
1817:
1544:{\displaystyle K_{p}={\frac {p_{T}(4\alpha ^{2})}{(1+\alpha )(1-\alpha )}}={\frac {p_{T}(4\alpha ^{2})}{1-\alpha ^{2}}}}
93:
2334:
1554:
1944:
1705:
1697:
1596:
535:
75:
2132:
1607:
334:
2196:
247:
2170:
2151:
2136:
747:
53:
2084:
687:
1564:
will remain constant with temperature. The addition of pressure to the system will increase the value of
2213:
2140:
319:
233:
432:
482:
1006:
853:
718:
239:
189:
2173:
of the ligand to the receptor. The higher the affinity of the ligand for the receptor the lower the
100:
209:
193:
649:
1938:
or formation of ions (for the case when HA has no net charge). The equilibrium constant is then
151:
161:
2312:
2288:
2280:
2270:
2240:
2201:
205:
2043:
413:
1125:
868:
722:
185:
144:
2083:
is not included because in dilute solution the solvent is essentially a pure liquid with a
837:
refers to the percentage of gas molecules which dissociate. Various relationships between
706:
168:
623:
459:
438:
2328:
2305:
2263:
1140:
849:
201:
1111:{\displaystyle K_{p}={\frac {p{\bigl (}{\ce {NO2}}{\bigr )}^{2}}{p\,{\ce {N2O4}}}}}
983:
755:
742:
734:
42:
2110:. It serves as an indicator of the acid strength: stronger acids have a higher
2207:
1601:
The reaction of an acid in water solvent is often described as a dissociation
2292:
1801:
714:
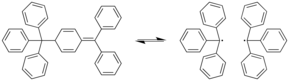
181:
27:
Separation of molecules or ionic compounds into smaller constituent entities
17:
767:
402:
where the brackets denote the equilibrium concentrations of the species.
2155:
2261:
Petrucci, Ralph H.; Harwood, William S.; Herring, F. Geoffrey (2002).
1394:. Thus, substituting the mole fractions with actual values in term of
2159:
730:
746:
refers to a substance that contains free ions and can be used as an
184:
is a general process in which molecules (or ionic compounds such as
30:"Dissociate" and "Dissociating" redirect here. For other uses, see
2284:
2204:, dissociation of molecules by photons (light, gamma rays, x-rays)
987:
726:
691:
197:
982:
If the initial concentration of dinitrogen tetroxide is 1
1128:. Hence, through the definition of partial pressure and using
826:
213:
192:) separate or split into other things such as atoms, ions, or
36:
2062:
1978:
1894:
1838:
1357:
1344:
1309:
1264:
1251:
1203:
1101:
1088:
1053:
966:
909:
896:
2269:(8th ed.). Upper Saddle River, N.J: Prentice Hall.
196:, usually in a reversible manner. For instance, when an
1788:
However a more explicit description is provided by the
1865:
1636:
933:
564:
328:
is the ratio of dissociated to undissociated compound
276:
2265:
General chemistry: principles and modern applications
2046:
1947:
1820:
1796:
H+ does not exist as such in solution but is instead
1708:
1610:
1404:
1149:
1015:
884:
817:. These will all exist as ions in an aqueous medium.
652:
626:
538:
485:
462:
441:
416:
337:
250:
1924:{\displaystyle {\ce {HA + H2O <=> H3O+ + A-}}}
67:. Unsourced material may be challenged and removed.
2262:
2075:
2029:
1923:
1777:
1681:
1543:
1366:
1110:
972:
670:
638:
609:
518:
468:
447:
422:
391:
307:
2210:, dissociation of molecules by ionizing radiation
1873:
1872:
1855:
1854:
1692:where HA is a proton acid such as acetic acid, CH
1644:
1643:
1626:
1625:
941:
940:
923:
922:
572:
571:
554:
553:
284:
283:
266:
265:
1009:(in terms of pressure) is given by the equation
2309:Physical Chemistry with Biological Applications
2030:{\displaystyle K_{\ce {a}}={\ce {\frac {}{}}}}
1778:{\displaystyle K_{\ce {a}}={\ce {\frac {}{}}}}
610:{\displaystyle {\ce {KCl <=> K+ + Cl-}}}
2135:of a molecule can take place by a process of
1316:
1289:
1210:
1183:
1060:
1037:
709:crystals dissolving and dissociating in water
529:For instance, for the following dissociation
8:
1800:by (bonded to) a water molecule to form the
1682:{\displaystyle {\ce {HA <=> H+ + A-}}}
1376:The total number of moles at equilibrium is
973:{\displaystyle {\ce {N2O4 <=> 2NO2}}}
456:. If the solute substance dissociates into
392:{\displaystyle K_{d}=\mathrm {\frac {}{}} }
308:{\displaystyle {\ce {AB <=> A + B}}}
212:, which gives a proton (H) and a negative
2061:
2056:
2048:
2047:
2045:
2014:
2003:
1995:
1985:
1977:
1972:
1964:
1961:
1952:
1946:
1914:
1901:
1893:
1888:
1874:
1867:
1866:
1864:
1856:
1849:
1847:
1846:
1844:
1837:
1832:
1821:
1819:
1811:The reaction can therefore be written as
1762:
1751:
1743:
1733:
1725:
1722:
1713:
1707:
1672:
1659:
1645:
1638:
1637:
1635:
1627:
1620:
1618:
1617:
1615:
1611:
1609:
1532:
1511:
1495:
1488:
1441:
1425:
1418:
1409:
1403:
1356:
1351:
1343:
1338:
1333:
1332:
1321:
1315:
1314:
1308:
1303:
1298:
1297:
1288:
1287:
1281:
1274:
1263:
1258:
1250:
1245:
1240:
1239:
1227:
1215:
1209:
1208:
1202:
1197:
1192:
1191:
1182:
1181:
1175:
1170:
1163:
1154:
1148:
1100:
1095:
1087:
1082:
1077:
1076:
1065:
1059:
1058:
1052:
1047:
1042:
1036:
1035:
1029:
1020:
1014:
994:at equilibrium giving, by stoichiometry,
965:
960:
955:
942:
935:
934:
932:
924:
917:
915:
914:
912:
908:
903:
895:
890:
885:
883:
651:
625:
600:
587:
573:
566:
565:
563:
555:
548:
546:
545:
543:
539:
537:
484:
461:
440:
415:
351:
342:
336:
285:
278:
277:
275:
267:
260:
258:
257:
255:
251:
249:
127:Learn how and when to remove this message
2231:
2229:
1001:
874:
863:
859:
813:
809:
793:
785:
761:
2225:
1848:
1619:
916:
547:
259:
208:atom and a hydrogen atom is broken by
7:
2256:
2254:
2252:
1553:This equation is in accordance with
1135:to represent the total pressure and
65:adding citations to reliable sources
380:
377:
366:
357:
238:For reversible dissociations in a
216:. Dissociation is the opposite of
25:
2239:(8th ed. W.H.Freeman 2006) p.763
2311:(Benjamin/Cummings) 1978, p.307
852:of the equation. The example of
519:{\displaystyle i=1+\alpha (n-1)}
160:
143:
41:
1790:Brønsted–Lowry acid–base theory
733:. The salt can be recovered by
52:needs additional citations for
2068:
2049:
2021:
2015:
2009:
1996:
1991:
1965:
1875:
1850:
1769:
1763:
1757:
1744:
1739:
1726:
1646:
1621:
1517:
1501:
1479:
1467:
1464:
1452:
1447:
1431:
943:
918:
825:The degree of dissociation in
725:, means the separation of the
574:
549:
513:
501:
383:
374:
369:
363:
360:
354:
286:
261:
1:
713:The dissociation of salts by
76:"Dissociation" chemistry
2169:is used as indicator of the
2162:. The dissociation constant
2180:value (and the higher the p
1934:and better described as an
1792:, which specifies that the
671:{\displaystyle i=1+\alpha }
2356:
2235:Atkins P. and de Paula J.
1698:acid dissociation constant
1597:Acid dissociation constant
1594:
685:
231:
29:
1591:Acids in aqueous solution
1387:, which is equivalent to
829:is denoted by the symbol
2197:Bond-dissociation energy
2100:acid ionization constant
2076:{\displaystyle {\ce {}}}
1555:Le Chatelier's principle
990:, this will decrease by
410:The dissociation degree
167:Dissociation diagram of
848:exist depending on the
748:electrically conductive
423:{\displaystyle \alpha }
2085:thermodynamic activity
2077:
2031:
1925:
1779:
1683:
1575:must decrease to keep
1545:
1368:
1112:
974:
710:
688:Solubility equilibrium
672:
640:
611:
520:
470:
449:
424:
393:
309:
200:dissolves in water, a
2340:Equilibrium chemistry
2214:Thermal decomposition
2096:dissociation constant
2094:is variously named a
2078:
2032:
1926:
1780:
1684:
1546:
1369:
1113:
975:
704:
673:
646:, we would have that
641:
612:
521:
471:
450:
425:
394:
320:dissociation constant
310:
234:Dissociation constant
228:Dissociation constant
2117:value (and a lower p
2044:
1945:
1818:
1706:
1608:
1402:
1147:
1013:
1007:equilibrium constant
882:
854:dinitrogen tetroxide
650:
624:
536:
483:
460:
439:
414:
335:
248:
240:chemical equilibrium
61:improve this article
2108:ionization constant
2064:
1980:
1896:
1861:
1840:
1632:
1359:
1346:
1311:
1266:
1253:
1205:
1180:
1103:
1090:
1055:
968:
929:
911:
898:
639:{\displaystyle n=2}
560:
406:Dissociation degree
272:
210:heterolytic fission
2335:Chemical processes
2237:Physical Chemistry
2073:
2052:
2027:
1968:
1921:
1884:
1880:
1828:
1775:
1679:
1651:
1541:
1364:
1347:
1334:
1299:
1254:
1241:
1193:
1166:
1108:
1091:
1078:
1043:
970:
956:
948:
899:
886:
867:) dissociating to
711:
668:
636:
607:
579:
516:
466:
445:
433:van 't Hoff factor
420:
389:
305:
291:
2317:978-0-8053-5680-9
2276:978-0-13-014329-7
2245:978-0-7167-8759-4
2202:Photodissociation
2067:
2055:
2025:
2020:
2002:
1984:
1971:
1955:
1913:
1900:
1887:
1882:
1843:
1831:
1824:
1773:
1768:
1750:
1732:
1716:
1671:
1658:
1653:
1614:
1539:
1483:
1398:and simplifying;
1362:
1350:
1337:
1302:
1269:
1257:
1244:
1196:
1139:to represent the
1106:
1094:
1081:
1046:
959:
950:
902:
889:
878:) will be taken.
702:
599:
586:
581:
542:
469:{\displaystyle n}
448:{\displaystyle i}
387:
303:
297:
293:
254:
137:
136:
129:
111:
16:(Redirected from
2347:
2319:
2303:
2297:
2296:
2268:
2258:
2247:
2233:
2158:that bind small
2104:acidity constant
2082:
2080:
2079:
2074:
2072:
2071:
2065:
2063:
2060:
2053:
2036:
2034:
2033:
2028:
2026:
2024:
2018:
2013:
2012:
2008:
2007:
2000:
1994:
1990:
1989:
1982:
1979:
1976:
1969:
1962:
1957:
1956:
1953:
1930:
1928:
1927:
1922:
1920:
1919:
1918:
1911:
1906:
1905:
1898:
1895:
1892:
1885:
1883:
1881:
1879:
1878:
1871:
1863:
1862:
1860:
1853:
1845:
1841:
1839:
1836:
1829:
1822:
1784:
1782:
1781:
1776:
1774:
1772:
1766:
1761:
1760:
1756:
1755:
1748:
1742:
1738:
1737:
1730:
1723:
1718:
1717:
1714:
1688:
1686:
1685:
1680:
1678:
1677:
1676:
1669:
1664:
1663:
1656:
1654:
1652:
1650:
1649:
1642:
1634:
1633:
1631:
1624:
1616:
1612:
1586:
1581:
1574:
1570:
1563:
1550:
1548:
1547:
1542:
1540:
1538:
1537:
1536:
1520:
1516:
1515:
1500:
1499:
1489:
1484:
1482:
1450:
1446:
1445:
1430:
1429:
1419:
1414:
1413:
1397:
1393:
1386:
1373:
1371:
1370:
1365:
1363:
1361:
1360:
1358:
1355:
1348:
1345:
1342:
1335:
1327:
1326:
1325:
1320:
1319:
1312:
1310:
1307:
1300:
1293:
1292:
1286:
1285:
1275:
1270:
1268:
1267:
1265:
1262:
1255:
1252:
1249:
1242:
1232:
1231:
1221:
1220:
1219:
1214:
1213:
1206:
1204:
1201:
1194:
1187:
1186:
1179:
1174:
1164:
1159:
1158:
1138:
1134:
1126:partial pressure
1123:
1117:
1115:
1114:
1109:
1107:
1105:
1104:
1102:
1099:
1092:
1089:
1086:
1079:
1071:
1070:
1069:
1064:
1063:
1056:
1054:
1051:
1044:
1041:
1040:
1030:
1025:
1024:
1004:
997:
993:
979:
977:
976:
971:
969:
967:
964:
957:
951:
949:
947:
946:
939:
931:
930:
928:
921:
913:
910:
907:
900:
897:
894:
887:
877:
869:nitrogen dioxide
866:
847:
843:
836:
832:
816:
801:
797:
789:
781:
780:
779:
776:
765:
737:of the solvent.
703:
677:
675:
674:
669:
645:
643:
642:
637:
616:
614:
613:
608:
606:
605:
604:
597:
592:
591:
584:
582:
580:
578:
577:
570:
562:
561:
559:
552:
544:
540:
525:
523:
522:
517:
475:
473:
472:
467:
454:
452:
451:
446:
429:
427:
426:
421:
398:
396:
395:
390:
388:
386:
372:
352:
347:
346:
314:
312:
311:
306:
304:
301:
295:
294:
292:
290:
289:
282:
274:
273:
271:
264:
256:
252:
164:
150:Dissociation of
147:
132:
125:
121:
118:
112:
110:
69:
45:
37:
21:
2355:
2354:
2350:
2349:
2348:
2346:
2345:
2344:
2325:
2324:
2323:
2322:
2304:
2300:
2277:
2260:
2259:
2250:
2234:
2227:
2222:
2193:
2186:
2179:
2168:
2149:
2130:
2123:
2116:
2093:
2042:
2041:
1999:
1981:
1963:
1948:
1943:
1942:
1910:
1897:
1816:
1815:
1807:
1747:
1729:
1724:
1709:
1704:
1703:
1695:
1668:
1655:
1606:
1605:
1599:
1593:
1584:
1580:
1576:
1572:
1569:
1565:
1562:
1558:
1528:
1521:
1507:
1491:
1490:
1451:
1437:
1421:
1420:
1405:
1400:
1399:
1395:
1388:
1377:
1328:
1313:
1277:
1276:
1223:
1222:
1207:
1165:
1150:
1145:
1144:
1136:
1133:
1129:
1124:represents the
1121:
1072:
1057:
1031:
1016:
1011:
1010:
1003:
999:
995:
991:
880:
879:
876:
872:
865:
861:
857:
845:
842:
838:
834:
830:
823:
815:
811:
807:
799:
795:
791:
787:
783:
777:
774:
773:
771:
763:
759:
707:sodium chloride
692:
690:
684:
648:
647:
622:
621:
596:
583:
534:
533:
481:
480:
458:
457:
437:
436:
412:
411:
408:
373:
353:
338:
333:
332:
327:
246:
245:
236:
230:
206:electronegative
175:
174:
173:
172:
171:
169:phosphoric acid
165:
156:
155:
154:
152:Gomberg's dimer
148:
133:
122:
116:
113:
70:
68:
58:
46:
35:
28:
23:
22:
15:
12:
11:
5:
2353:
2351:
2343:
2342:
2337:
2327:
2326:
2321:
2320:
2298:
2275:
2248:
2224:
2223:
2221:
2218:
2217:
2216:
2211:
2205:
2199:
2192:
2189:
2184:
2177:
2166:
2148:
2145:
2129:
2126:
2121:
2114:
2091:
2070:
2059:
2051:
2038:
2037:
2023:
2017:
2011:
2006:
1998:
1993:
1988:
1975:
1967:
1960:
1951:
1932:
1931:
1917:
1909:
1904:
1891:
1877:
1870:
1859:
1852:
1835:
1827:
1805:
1786:
1785:
1771:
1765:
1759:
1754:
1746:
1741:
1736:
1728:
1721:
1712:
1693:
1690:
1689:
1675:
1667:
1662:
1648:
1641:
1630:
1623:
1595:Main article:
1592:
1589:
1578:
1567:
1560:
1535:
1531:
1527:
1524:
1519:
1514:
1510:
1506:
1503:
1498:
1494:
1487:
1481:
1478:
1475:
1472:
1469:
1466:
1463:
1460:
1457:
1454:
1449:
1444:
1440:
1436:
1433:
1428:
1424:
1417:
1412:
1408:
1354:
1341:
1331:
1324:
1318:
1306:
1296:
1291:
1284:
1280:
1273:
1261:
1248:
1238:
1235:
1230:
1226:
1218:
1212:
1200:
1190:
1185:
1178:
1173:
1169:
1162:
1157:
1153:
1131:
1098:
1085:
1075:
1068:
1062:
1050:
1039:
1034:
1028:
1023:
1019:
963:
954:
945:
938:
927:
920:
906:
893:
840:
822:
819:
683:
680:
667:
664:
661:
658:
655:
635:
632:
629:
618:
617:
603:
595:
590:
576:
569:
558:
551:
527:
526:
515:
512:
509:
506:
503:
500:
497:
494:
491:
488:
465:
444:
419:
407:
404:
400:
399:
385:
382:
379:
376:
371:
368:
365:
362:
359:
356:
350:
345:
341:
325:
316:
315:
300:
288:
281:
270:
263:
232:Main article:
229:
226:
166:
159:
158:
157:
149:
142:
141:
140:
139:
138:
135:
134:
49:
47:
40:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
2352:
2341:
2338:
2336:
2333:
2332:
2330:
2318:
2314:
2310:
2307:
2302:
2299:
2294:
2290:
2286:
2282:
2278:
2272:
2267:
2266:
2257:
2255:
2253:
2249:
2246:
2242:
2238:
2232:
2230:
2226:
2219:
2215:
2212:
2209:
2206:
2203:
2200:
2198:
2195:
2194:
2190:
2188:
2183:
2176:
2172:
2165:
2161:
2157:
2153:
2146:
2144:
2142:
2138:
2134:
2133:Fragmentation
2128:Fragmentation
2127:
2125:
2120:
2113:
2109:
2105:
2101:
2097:
2088:
2086:
2057:
2004:
1986:
1973:
1958:
1949:
1941:
1940:
1939:
1937:
1915:
1907:
1902:
1889:
1868:
1857:
1833:
1825:
1814:
1813:
1812:
1809:
1803:
1799:
1795:
1791:
1752:
1734:
1719:
1710:
1702:
1701:
1700:
1699:
1673:
1665:
1660:
1639:
1628:
1604:
1603:
1602:
1598:
1590:
1588:
1556:
1551:
1533:
1529:
1525:
1522:
1512:
1508:
1504:
1496:
1492:
1485:
1476:
1473:
1470:
1461:
1458:
1455:
1442:
1438:
1434:
1426:
1422:
1415:
1410:
1406:
1392:
1385:
1381:
1374:
1352:
1339:
1329:
1322:
1304:
1294:
1282:
1278:
1271:
1259:
1246:
1236:
1233:
1228:
1224:
1216:
1198:
1188:
1176:
1171:
1167:
1160:
1155:
1151:
1142:
1141:mole fraction
1127:
1118:
1096:
1083:
1073:
1066:
1048:
1032:
1026:
1021:
1017:
1008:
989:
985:
980:
961:
952:
936:
925:
904:
891:
870:
855:
851:
850:stoichiometry
828:
820:
818:
803:
769:
757:
751:
749:
745:
744:
738:
736:
732:
728:
724:
720:
716:
708:
689:
681:
679:
665:
662:
659:
656:
653:
633:
630:
627:
601:
593:
588:
567:
556:
532:
531:
530:
510:
507:
504:
498:
495:
492:
489:
486:
479:
478:
477:
463:
455:
442:
434:
417:
405:
403:
348:
343:
339:
331:
330:
329:
324:
321:
298:
279:
268:
244:
243:
242:
241:
235:
227:
225:
223:
222:recombination
219:
215:
211:
207:
203:
202:covalent bond
199:
195:
191:
187:
183:
179:
170:
163:
153:
146:
131:
128:
120:
109:
106:
102:
99:
95:
92:
88:
85:
81:
78: –
77:
73:
72:Find sources:
66:
62:
56:
55:
50:This article
48:
44:
39:
38:
33:
19:
2308:
2306:Laidler K.J.
2301:
2264:
2236:
2181:
2174:
2163:
2150:
2131:
2118:
2111:
2107:
2103:
2099:
2095:
2089:
2039:
1935:
1933:
1810:
1797:
1793:
1787:
1691:
1600:
1552:
1390:
1383:
1379:
1375:
1119:
981:
824:
804:
752:
741:
739:
712:
619:
528:
435:
409:
401:
322:
317:
237:
221:
217:
178:Dissociation
177:
176:
123:
114:
104:
97:
90:
83:
71:
59:Please help
54:verification
51:
32:Dissociation
2137:heterolysis
756:Acetic acid
743:electrolyte
735:evaporation
705:A video of
476:ions, then
218:association
204:between an
2329:Categories
2285:2001032331
2220:References
2208:Radiolysis
1936:ionization
721:, such as
686:See also:
87:newspapers
18:Dissociate
2152:Receptors
2147:Receptors
2141:homolysis
2005:−
1916:−
1876:⇀
1869:−
1858:−
1851:↽
1802:hydronium
1753:−
1674:−
1647:⇀
1640:−
1629:−
1622:↽
1530:α
1526:−
1509:α
1477:α
1474:−
1462:α
1439:α
1234:⋅
998:moles of
944:⇀
937:−
926:−
919:↽
790:and some
715:solvation
666:α
602:−
575:⇀
568:−
557:−
550:↽
508:−
499:α
418:α
287:⇀
280:−
269:−
262:↽
190:complexes
182:chemistry
117:June 2014
2293:46872308
2191:See also
2187:value).
2171:affinity
2156:proteins
2124:value).
2087:of one.
1798:accepted
833:, where
768:ammonium
719:solution
194:radicals
2160:ligands
731:cations
101:scholar
2315:
2291:
2283:
2273:
2243:
2106:or an
2040:where
1794:proton
1120:where
1005:. The
766:) and
727:anions
103:
96:
89:
82:
74:
2102:, an
2098:, an
1804:ion H
1571:, so
1382:) + 2
1378:(1 –
988:litre
827:gases
821:Gases
723:water
717:in a
682:Salts
188:, or
186:salts
108:JSTOR
94:books
2313:ISBN
2289:OCLC
2281:LCCN
2271:ISBN
2241:ISBN
2154:are
1389:1 +
986:per
984:mole
844:and
798:and
788:COOH
764:COOH
729:and
318:the
198:acid
80:news
2139:or
1808:O.
796:COO
740:An
620:As
541:KCl
220:or
214:ion
180:in
63:by
2331::
2287:.
2279:.
2251:^
2228:^
2143:.
2019:HA
1823:HA
1767:HA
1613:HA
1587:.
1557:.
1301:NO
1195:NO
1143:;
1045:NO
1000:NO
958:NO
873:NO
812:SO
802:.
792:CH
784:CH
772:NH
760:CH
678:.
598:Cl
253:AB
224:.
2295:.
2185:d
2182:K
2178:d
2175:K
2167:d
2164:K
2122:a
2119:K
2115:a
2112:K
2092:a
2090:K
2069:]
2066:O
2058:2
2054:H
2050:[
2022:]
2016:[
2010:]
2001:A
1997:[
1992:]
1987:+
1983:O
1974:3
1970:H
1966:[
1959:=
1954:a
1950:K
1912:A
1908:+
1903:+
1899:O
1890:3
1886:H
1842:O
1834:2
1830:H
1826:+
1806:3
1770:]
1764:[
1758:]
1749:A
1745:[
1740:]
1735:+
1731:H
1727:[
1720:=
1715:a
1711:K
1694:3
1670:A
1666:+
1661:+
1657:H
1585:α
1579:p
1577:K
1573:α
1568:T
1566:p
1561:p
1559:K
1534:2
1523:1
1518:)
1513:2
1505:4
1502:(
1497:T
1493:p
1486:=
1480:)
1471:1
1468:(
1465:)
1459:+
1456:1
1453:(
1448:)
1443:2
1435:4
1432:(
1427:T
1423:p
1416:=
1411:p
1407:K
1396:α
1391:α
1384:α
1380:α
1353:4
1349:O
1340:2
1336:N
1330:x
1323:2
1317:)
1305:2
1295:x
1290:(
1283:T
1279:p
1272:=
1260:4
1256:O
1247:2
1243:N
1237:x
1229:T
1225:p
1217:2
1211:)
1199:2
1189:x
1184:(
1177:2
1172:T
1168:p
1161:=
1156:p
1152:K
1137:x
1132:T
1130:p
1122:p
1097:4
1093:O
1084:2
1080:N
1074:p
1067:2
1061:)
1049:2
1038:(
1033:p
1027:=
1022:p
1018:K
1002:2
996:α
992:α
962:2
953:2
905:4
901:O
892:2
888:N
875:2
871:(
864:4
862:O
860:2
858:N
856:(
846:α
841:p
839:K
835:α
831:α
814:4
810:2
808:H
800:H
794:3
786:3
778:4
775:+
770:(
762:3
758:(
663:+
660:1
657:=
654:i
634:2
631:=
628:n
594:+
589:+
585:K
514:)
511:1
505:n
502:(
496:+
493:1
490:=
487:i
464:n
443:i
384:]
381:B
378:A
375:[
370:]
367:B
364:[
361:]
358:A
355:[
349:=
344:d
340:K
326:d
323:K
302:B
299:+
296:A
130:)
124:(
119:)
115:(
105:·
98:·
91:·
84:·
57:.
34:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.