364:
31:
232:
393:
613:β-Elimination, with loss of electrofuge and nucleofuge on vicinal carbon atoms, is by far the most common type of elimination. The ability to form a stable product containing a C=C or C=X bond, as well as orbital alignment considerations, strongly favors β-elimination over other elimination processes. However, other types are known, generally for systems where β-elimination cannot occur.
1034:
617:
1022:
643:, as a reactive intermediate. On the other hand, formic acid undergoes α-elimination to afford the stable products water and carbon monoxide under acidic conditions. α-Elimination may also occur on a metal center, one particularly common result of which is lowering of both the metal oxidation state and coordination number by 2 units in a process known as
658:
In certain special cases, γ- and higher eliminations to form three-membered or larger rings is also possible in both organic and organometallic processes. For instance, certain Pt(II) complexes undergo γ- and δ-elimination to give metallocycles. More recently, γ-silyl elimination of a silylcyclobutyl
801:
In rare cases in which β hydrogens are unavailable but substitution is disfavored, α-elimination to form a carbene can sometimes occur. In particular: (1) Trihalomethanes like chloroform can react with NaOH to form dihalocarbenes (substitution is electronically disfavored). (2) Allyl and benzyl
974:
Kelly, Christopher B.; Colthart, Allison M.; Constant, Brad D.; Corning, Sean R.; Dubois, Lily N. E.; Genovese, Jacqueline T.; Radziewicz, Julie L.; Sletten, Ellen M.; Whitaker, Katherine R. (2011-04-01). "Enabling the
Synthesis of Perfluoroalkyl Bicyclobutanes via 1,3 γ-Silyl Elimination".
70:. The numbers refer not to the number of steps in the mechanism, but rather to the kinetics of the reaction: E2 is bimolecular (second-order) while E1 is unimolecular (first-order). In cases where the molecule is able to stabilize an anion but possesses a poor
568:
2 substitution is hard to achieve when strong bases are used, as alkene products arising from elimination are almost always observed to some degree. On the other hand, clean E2 can be achieved by simply selecting a sterically hindered base (e.g., potassium
457:
being favored. Fluoride is not a good leaving group, so eliminations with fluoride as the leaving group have slower rates than other halogens . There is a certain level of competition between the elimination reaction and
542:> 11, e.g., hydroxide, alkoxide, acetylide), the result is generally elimination by E2, while weaker bases that are still good nucleophiles (e.g., acetate, azide, cyanide, iodide) will give primarily S
604:
2 reaction because in this reaction type the C-H bonds tighten in the transition state. The KIE's for the ethyl (0.99) and isopropyl (1.72) analogues suggest competition between the two reaction modes.
4678:
623:
The next most common type of elimination reaction is α-elimination. For a carbon center, the result of α-elimination is the formation of a carbene, which includes "stable carbenes" such as
939:
Moore, Stephen S.; DiCosimo, Robert; Sowinski, Allan F.; Whitesides, George M. (1981-02-01). "Ring strain in bis(triethylphosphine)-3,3-dimethylplatinacyclobutane is small".
577:
1 almost always result in a product mixture contaminated by some E1 product (again, with the exception of cases where the lack of β hydrogens makes elimination impossible).
3794:
837:
Stephanie M. Villano; Shuji Kato; Veronica M. Bierbaum (2006). "Deuterium
Kinetic Isotope Effects in Gas-Phase SN2 and E2 Reactions: Comparison of Experiment and Theory".
3739:
4507:
3849:
765:
Nash, J. J.; Leininger, M. A.; Keyes, K. (April 2008). "Pyrolysis of Aryl
Sulfonate Esters in the Absence of Solvent: E1 or E2? A Puzzle for the Organic Laboratory".
655:
refer to processes that result in formation of a metal-carbene complex. In these reactions, it is the carbon adjacent to the metal that undergoes α-elimination.)
3999:
2633:
4728:
1066:
4502:
3604:
2328:
802:
chloride can react with lithium tetramethylpiperide (LiTMP) to form vinylcarbene and phenylcarbene, respectively (substitution is sterically disfavored).
414:
Highly substituted carbocations are more stable than methyl or primary substituted cations. Such stability gives time for the two-step E1 mechanism to occur.
363:
3374:
1525:
564:
In general, with the exception of reactions in which E2 is impossible because β hydrogens are unavailable (e.g. methyl, allyl, and benzyl halides), clean S
4174:
2118:
525:
2 reaction) to essentially only 1° haloalkanes; 2° haloalkanes generally do not give synthetically useful yields, while 3° haloalkanes fail completely.
4269:
2373:
4249:
3744:
2911:
2792:
2348:
4339:
4094:
1561:
513:
For example, when a 3° haloalkane is reacts with an alkoxide, due to strong basic character of the alkoxide and unreactivity of 3° group towards S
183:
conformation with higher energy. The reaction mechanism involving staggered conformation is more favorable for E2 reactions (unlike E1 reactions).
2926:
4572:
4029:
528:
With strong base, 3° haloalkanes give elimination by E2. With weak bases, mixtures of elimination and substitution products form by competing S
4522:
4134:
4114:
4074:
2881:
4668:
4593:
4477:
3089:
2493:
1824:
411:
Highly substituted alkyl halides are bulky, limiting the room for the E2 one-step mechanism; therefore, the two-step E1 mechanism is favored.
4663:
4492:
4149:
4004:
3634:
3479:
2716:
326:
The reaction usually occurs in the complete absence of a base or the presence of only a weak base (acidic conditions and high temperature).
3839:
3329:
3004:
1278:
4743:
4527:
3549:
1155:
4104:
4738:
4452:
4314:
4069:
1112:
1038:
145:
It is typically undergone by primary substituted alkyl halides, but is possible with some secondary alkyl halides and other compounds.
4628:
4099:
4014:
3984:
3964:
3829:
3824:
3199:
3124:
2767:
2721:
2588:
1849:
1419:
4567:
1059:
915:
821:
742:
4733:
4693:
4643:
4119:
3869:
3799:
2288:
4329:
3934:
2827:
2548:
4319:
1859:
4487:
4244:
4194:
2984:
2916:
2807:
2383:
2138:
2063:
1844:
392:
231:
4199:
4009:
3484:
3394:
1518:
1026:
315:
is influenced only by the concentration of the alkyl halide because carbocation formation is the slowest step, as known as the
4773:
4557:
4497:
3899:
3874:
3784:
3364:
3244:
2278:
1774:
4827:
4822:
4658:
4144:
3939:
2208:
1327:
1322:
1132:
882:
4763:
4349:
3859:
3369:
3314:
3159:
3119:
2951:
2706:
2423:
2273:
1487:
4723:
4284:
4239:
3729:
3584:
1492:
767:
4758:
4673:
4532:
4447:
4344:
3419:
3074:
2742:
2153:
1714:
4832:
4648:
4623:
4608:
4304:
4169:
4124:
3889:
3434:
3284:
2498:
2178:
2123:
1052:
4653:
4598:
4129:
3544:
3259:
3254:
2747:
2563:
2553:
2268:
2128:
2078:
2073:
2048:
1954:
4708:
4309:
4229:
3844:
3809:
3654:
3079:
3039:
2936:
2711:
2463:
2408:
2008:
1719:
1709:
1684:
4683:
4384:
4189:
3624:
3189:
3164:
3104:
2696:
2403:
2053:
2238:
3974:
3509:
2961:
2183:
2148:
1744:
1679:
1511:
1457:
1147:
518:
4542:
4164:
3224:
3149:
2673:
2508:
2193:
1969:
1929:
1674:
4783:
4688:
4422:
4394:
4364:
4279:
4209:
4139:
4059:
3959:
3919:
3614:
3234:
2533:
2528:
1990:
1854:
1184:
1084:
680:
459:
75:
4748:
4618:
4482:
4324:
4184:
3704:
2678:
2228:
2188:
1939:
4638:
4234:
4204:
4079:
4034:
3864:
3774:
3589:
3579:
3409:
2966:
2906:
2871:
2658:
2618:
2393:
2263:
1779:
1769:
1699:
1414:
341:
213:
159:
Because the E2 mechanism results in the formation of a pi bond, the two leaving groups (often a hydrogen and a
4214:
3194:
2218:
1764:
1644:
4427:
4718:
4577:
4369:
4294:
4274:
3994:
3944:
3804:
3769:
3709:
3639:
2941:
2921:
2653:
2573:
2468:
2428:
2398:
2333:
2203:
2113:
2103:
1979:
1689:
1462:
1263:
668:
506:
546:
2. Finally, weakly nucleophilic species (e.g., water, alcohols, carboxylic acids) will give a mixture of S
62:
are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the
4457:
4179:
3929:
3909:
3884:
3834:
3749:
3724:
3679:
3649:
3629:
3599:
3564:
3519:
3494:
3469:
3354:
3279:
3059:
2752:
2688:
2488:
2213:
2133:
1819:
1794:
1571:
1566:
1217:
581:
561:
2 when the nucleophile is also unhindered. However, strongly basic and hindered nucleophiles favor E2.
386:
172:
4793:
3539:
2163:
517:
2, only alkene formation by E2 elimination is observed. Thus, elimination by E2 limits the scope of the
4379:
4334:
4049:
4019:
3989:
3924:
3904:
3819:
3814:
3779:
3734:
3719:
3714:
3694:
3684:
3619:
3609:
3489:
3009:
2812:
2388:
2343:
2173:
1909:
1629:
1591:
1447:
1379:
1237:
1227:
644:
316:
209:
194:
153:
1839:
1834:
4432:
4562:
4512:
4462:
4442:
4289:
4264:
3979:
3969:
3854:
3669:
3664:
3594:
3379:
3179:
3139:
3069:
3034:
2989:
2956:
2822:
2797:
2598:
2558:
2518:
2483:
2413:
2168:
2038:
2013:
1442:
776:
305:
4778:
4768:
4753:
4399:
4374:
4359:
4354:
4084:
4039:
4024:
3914:
3894:
3789:
3674:
3659:
3504:
3449:
3439:
3429:
3404:
3169:
3044:
3019:
2931:
2787:
2772:
2757:
2613:
2578:
2523:
2293:
2143:
2088:
1959:
1874:
1734:
1659:
1452:
1384:
1369:
1312:
1804:
30:
4517:
4467:
4437:
4299:
4089:
3879:
3764:
3699:
3689:
3454:
3384:
3349:
3344:
3324:
3319:
3264:
3174:
3024:
2886:
2876:
2782:
2568:
2513:
2443:
2363:
2258:
2158:
2093:
2018:
1864:
1729:
1664:
1477:
1247:
1076:
248:
127:
1649:
4254:
3574:
3459:
3424:
3389:
3334:
3289:
3249:
3204:
3184:
3134:
3129:
3099:
3084:
2994:
2901:
2837:
2802:
2628:
2503:
2378:
2303:
2283:
2198:
2033:
2028:
1974:
1884:
1789:
1749:
1704:
1586:
1581:
1546:
1472:
1467:
1429:
1374:
1293:
1273:
1209:
1000:
992:
956:
921:
911:
888:
878:
855:
839:
817:
748:
738:
732:
701:
260:
4788:
4633:
4603:
4547:
4472:
4404:
4159:
4109:
3954:
3759:
3534:
3529:
3474:
3464:
3239:
3049:
3029:
2999:
2896:
2832:
2817:
2648:
2603:
2593:
2583:
2478:
2458:
2453:
2438:
2433:
2313:
2308:
2248:
2233:
2223:
2068:
2058:
1924:
1914:
1904:
1814:
1809:
1784:
1724:
1576:
1535:
1404:
1353:
1307:
984:
948:
847:
784:
710:
589:
482:
187:
168:
139:
55:
271:
E1 is a model to explain a particular type of chemical elimination reaction. E1 stands for
4698:
4389:
4224:
4219:
3514:
3499:
3444:
3399:
3359:
3309:
3274:
3269:
3214:
3209:
3144:
3094:
3014:
2842:
2726:
2701:
2663:
2638:
2623:
2608:
2543:
2418:
2368:
2358:
2338:
2298:
2108:
2098:
2083:
1879:
1799:
1624:
1619:
1482:
1394:
1343:
636:
624:
593:
352:
1669:
1639:
780:
422:
1 and E1 pathways are competing, the E1 pathway can be favored by increasing the heat.
4703:
4613:
4552:
3644:
3554:
3524:
3299:
3154:
2891:
2668:
2538:
2353:
2323:
2023:
1919:
1694:
1556:
1189:
1178:
667:
Many of the concepts and terminology related to elimination reactions were proposed by
553:
For 1° haloalkanes with β-branching, E2 elimination is still generally preferred over S
164:
4816:
4713:
4414:
4259:
4154:
3949:
3339:
3304:
3294:
3229:
3219:
3109:
2946:
2762:
2473:
2448:
2318:
1964:
1949:
1934:
1829:
1759:
1739:
1654:
1437:
1409:
1317:
1268:
1242:
535:
The case of 2° haloalkanes is relatively complex. For strongly basic nucleophiles (p
442:
320:
312:
296:
176:
149:
71:
43:
27:
Reaction where 2 substituents are removed from a molecule in a 1 or 2 step mechanism
3754:
3114:
2866:
2643:
2243:
2043:
1894:
1889:
1754:
1609:
1389:
1195:
1102:
1092:
471:
463:
407:
E1 eliminations happen with highly substituted alkyl halides for two main reasons.
330:
220:
97:
35:
584:(KIE) was determined for the gas phase reaction of several alkyl halides with the
227:
mechanism if the base can also act as a nucleophile (true for many common bases).
2253:
1899:
1869:
1634:
1348:
1283:
728:
502:
491:
290:
59:
39:
4537:
4064:
3414:
1503:
628:
616:
286:
156:, because it's influenced by both the alkyl halide and the base (bimolecular).
996:
960:
925:
752:
734:
Advanced
Organic Chemistry: Reactions, Mechanisms, and Structure, 3rd edition
714:
635:) in the presence of strong base is a classic approach for the generation of
1399:
1044:
1033:
892:
348:
256:
82:
1004:
859:
17:
1021:
1944:
1614:
585:
404:
is the reaction of tert-butylbromide with potassium ethoxide in ethanol.
244:
180:
86:
952:
631:. For instance, α-elimination the elements of HCl from chloroform (CHCl
1604:
495:
454:
446:
356:
252:
160:
90:
988:
851:
788:
615:
557:
2 for strongly basic nucleophiles. Unhindered 1° haloalkanes favor S
450:
93:
600:
2 possible) on the other hand has a KIE of 0.85 consistent with a S
1302:
877:. Dougherty, Dennis A., 1952-. Sausalito, CA: University Science.
29:
376:
results from antiperiplanar elimination. The presence of product
308:
alkyl halides, but is possible with some secondary alkyl halides.
1988:
1507:
1048:
1122:
190:. It must be strong enough to remove a weakly acidic hydrogen.
659:
tosylate has been used to prepare strained bicyclic systems.
367:
E1 elimination Nash 2008, antiperiplanar relationship in blue
588:
ion. In accordance with an E2 elimination the reaction with
573:-butoxide). Similarly, attempts to effect substitution by S
391:
362:
230:
478:. Generally, elimination is favored over substitution when
647:. (Confusingly, in organometallic terminology, the terms
347:
There is no antiperiplanar requirement. An example is the
344:
of slightly larger than 1 (commonly 1 - 1.5) is observed.
96:
proceed through an "internal" elimination mechanism, the
699:
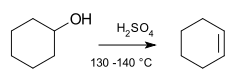
Coleman, G. H.; Johnstone, H. F. (1925). "Cyclohexene".
462:. More precisely, there are competitions between E2 and
337:
because they share a common carbocationic intermediate.
908:
The organometallic chemistry of the transition metals
4679:
Erlenmeyer–Plöchl azlactone and amino-acid synthesis
816:(5th ed.). New York: McGraw-Hill. p. 350.
4586:
4413:
4048:
3563:
3058:
2975:
2855:
2735:
2687:
1997:
1428:
1362:
1336:
1292:
1256:
1208:
1169:
1146:
1083:
380:
is an indication that an E1 mechanism is occurring.
3740:Divinylcyclopropane-cycloheptadiene rearrangement
432:Independent of concentration and basicity of base
138:E2 is a single step elimination, with a single
4000:Thermal rearrangement of aromatic hydrocarbons
2634:Thermal rearrangement of aromatic hydrocarbons
609:Elimination reactions other than β-elimination
216:much larger than 1 (commonly 2-6) is observed.
134:The specifics of the reaction are as follows:
4729:Lectka enantioselective beta-lactam synthesis
1519:
1060:
66:, and the two-step mechanism is known as the
8:
4508:Inverse electron-demand Diels–Alder reaction
2329:Heterogeneous metal catalyzed cross-coupling
193:In order for the pi bond to be created, the
3850:Lobry de Bruyn–Van Ekenstein transformation
289:: the carbon-halogen bond breaks to give a
4410:
2684:
1985:
1526:
1512:
1504:
1067:
1053:
1045:
4340:Petrenko-Kritschenko piperidone synthesis
3795:Fritsch–Buttenberg–Wiechell rearrangement
279:It is a two-step process of elimination:
116:, involves a one-step mechanism in which
4503:Intramolecular Diels–Alder cycloaddition
941:Journal of the American Chemical Society
691:
239:An example of this type of reaction in
4523:Metal-centered cycloaddition reactions
4175:Debus–Radziszewski imidazole synthesis
2119:Bodroux–Chichibabin aldehyde synthesis
1093:Unimolecular nucleophilic substitution
910:(5th ed.). Hoboken, N.J.: Wiley.
505:. Bases with steric bulk, (such as in
112:The E2 mechanism, where E2 stands for
4669:Diazoalkane 1,3-dipolar cycloaddition
4573:Vinylcyclopropane (5+2) cycloaddition
4478:Diazoalkane 1,3-dipolar cycloaddition
4250:Hurd–Mori 1,2,3-thiadiazole synthesis
3745:Dowd–Beckwith ring-expansion reaction
2912:Hurd–Mori 1,2,3-thiadiazole synthesis
1825:LFER solvent coefficients (data page)
1103:Bimolecular nucleophilic substitution
329:E1 reactions are in competition with
275:and has the following specifications
7:
3480:Sharpless asymmetric dihydroxylation
2717:Methoxymethylenetriphenylphosphorane
197:of carbons needs to be lowered from
3605:Allen–Millar–Trippett rearrangement
1156:Electrophilic aromatic substitution
445:is influenced by the reactivity of
385:It is accompanied by carbocationic
124:bonds break to form a double bond (
4744:Nitrone-olefin (3+2) cycloaddition
4739:Niementowski quinazoline synthesis
4528:Nitrone-olefin (3+2) cycloaddition
4453:Azide-alkyne Huisgen cycloaddition
4315:Niementowski quinazoline synthesis
4070:Azide-alkyne Huisgen cycloaddition
3375:Meerwein–Ponndorf–Verley reduction
2927:Leimgruber–Batcho indole synthesis
1123:Nucleophilic internal substitution
1113:Nucleophilic aromatic substitution
25:
4568:Trimethylenemethane cycloaddition
4270:Johnson–Corey–Chaykovsky reaction
4135:Cadogan–Sundberg indole synthesis
4115:Bohlmann–Rahtz pyridine synthesis
4075:Baeyer–Emmerling indole synthesis
2882:Cadogan–Sundberg indole synthesis
2374:Johnson–Corey–Chaykovsky reaction
875:Modern physical organic chemistry
4664:Cook–Heilbron thiazole synthesis
4493:Hexadehydro Diels–Alder reaction
4320:Niementowski quinoline synthesis
4150:Cook–Heilbron thiazole synthesis
4095:Bischler–Möhlau indole synthesis
4005:Tiffeneau–Demjanov rearrangement
3635:Baker–Venkataraman rearrangement
2793:Horner–Wadsworth–Emmons reaction
2464:Mizoroki-Heck vs. Reductive Heck
2349:Horner–Wadsworth–Emmons reaction
1860:Neighbouring group participation
1032:
1020:
208:The C-H bond is weakened in the
4200:Fiesselmann thiophene synthesis
4030:Westphalen–Lettré rearrangement
4010:Vinylcyclopropane rearrangement
3840:Kornblum–DeLaMare rearrangement
3485:Epoxidation of allylic alcohols
3395:Noyori asymmetric hydrogenation
3330:Kornblum–DeLaMare rearrangement
3005:Gallagher–Hollander degradation
1279:Lindemann–Hinshelwood mechanism
509:), are often poor nucleophiles.
396:Scheme 2. E1 reaction mechanism
235:Scheme 1: E2 reaction mechanism
4659:Chichibabin pyridine synthesis
4145:Chichibabin pyridine synthesis
4105:Blum–Ittah aziridine synthesis
3940:Ring expansion and contraction
2209:Cross dehydrogenative coupling
1328:Outer sphere electron transfer
1323:Inner sphere electron transfer
1133:Nucleophilic acyl substitution
485:around the α-carbon increases.
304:E1 typically takes place with
1:
4629:Bischler–Napieralski reaction
4587:Heterocycle forming reactions
4240:Hemetsberger indole synthesis
4100:Bischler–Napieralski reaction
4015:Wagner–Meerwein rearrangement
3985:Sommelet–Hauser rearrangement
3965:Seyferth–Gilbert homologation
3830:Ireland–Claisen rearrangement
3825:Hofmann–Martius rearrangement
3585:2,3-sigmatropic rearrangement
3200:Corey–Winter olefin synthesis
3125:Barton–McCombie deoxygenation
2768:Corey–Winter olefin synthesis
2722:Seyferth–Gilbert homologation
2589:Seyferth–Gilbert homologation
1493:Diffusion-controlled reaction
768:Journal of Chemical Education
592:results in a KIE of 2.3. The
179:transition state which is in
4734:Lehmstedt–Tanasescu reaction
4694:Gabriel–Colman rearrangement
4649:Bucherer carbazole synthesis
4644:Borsche–Drechsel cyclization
4624:Bernthsen acridine synthesis
4609:Bamberger triazine synthesis
4594:Algar–Flynn–Oyamada reaction
4305:Nazarov cyclization reaction
4170:De Kimpe aziridine synthesis
4125:Bucherer carbazole synthesis
4120:Borsche–Drechsel cyclization
3890:Nazarov cyclization reaction
3870:Meyer–Schuster rearrangement
3800:Gabriel–Colman rearrangement
3550:Wolffenstein–Böters reaction
3435:Reduction of nitro compounds
3285:Grundmann aldehyde synthesis
3090:Algar–Flynn–Oyamada reaction
2499:Olefin conversion technology
2494:Nozaki–Hiyama–Kishi reaction
2289:Gabriel–Colman rearrangement
2179:Claisen-Schmidt condensation
2124:Bouveault aldehyde synthesis
906:Crabtree, Robert H. (2009).
437:Competition among mechanisms
281:ionization and deprotonation
255:. The reaction products are
74:, a third type of reaction,
4709:Hantzsch pyridine synthesis
4488:Enone–alkene cycloadditions
4310:Nenitzescu indole synthesis
4230:Hantzsch pyridine synthesis
4195:Ferrario–Ackermann reaction
3845:Kowalski ester homologation
3810:Halogen dance rearrangement
3655:Benzilic acid rearrangement
3080:Akabori amino-acid reaction
3040:Von Braun amide degradation
2985:Barbier–Wieland degradation
2937:Nenitzescu indole synthesis
2917:Kharasch–Sosnovsky reaction
2808:Julia–Kocienski olefination
2712:Kowalski ester homologation
2409:Kowalski ester homologation
2384:Julia–Kocienski olefination
2139:Cadiot–Chodkiewicz coupling
2064:Aza-Baylis–Hillman reaction
2009:Acetoacetic ester synthesis
1720:Dynamic binding (chemistry)
1710:Conrotatory and disrotatory
1685:Charge remote fragmentation
1148:Electrophilic substitutions
186:E2 typically uses a strong
4849:
4774:Robinson–Gabriel synthesis
4724:Kröhnke pyridine synthesis
4558:Retro-Diels–Alder reaction
4498:Imine Diels–Alder reaction
4285:Kröhnke pyridine synthesis
3900:Newman–Kwart rearrangement
3875:Mislow–Evans rearrangement
3785:Fischer–Hepp rearrangement
3730:Di-π-methane rearrangement
3510:Stephen aldehyde synthesis
3245:Eschweiler–Clarke reaction
2962:Williamson ether synthesis
2279:Fujiwara–Moritani reaction
2184:Combes quinoline synthesis
2149:Carbonyl olefin metathesis
1850:More O'Ferrall–Jencks plot
1775:Grunwald–Winstein equation
1745:Electron-withdrawing group
1680:Catalytic resonance theory
1458:Energy profile (chemistry)
1420:More O'Ferrall–Jencks plot
1085:Nucleophilic substitutions
812:Carey, Francis A. (2003).
519:Williamson ether synthesis
425:Specific features :
4784:Urech hydantoin synthesis
4764:Pomeranz–Fritsch reaction
4689:Fischer oxazole synthesis
4423:1,3-Dipolar cycloaddition
4395:Urech hydantoin synthesis
4365:Reissert indole synthesis
4350:Pomeranz–Fritsch reaction
4280:Knorr quinoline synthesis
4210:Fischer oxazole synthesis
4140:Camps quinoline synthesis
4060:1,3-Dipolar cycloaddition
3960:Semipinacol rearrangement
3935:Ramberg–Bäcklund reaction
3920:Piancatelli rearrangement
3860:McFadyen–Stevens reaction
3615:Alpha-ketol rearrangement
3370:McFadyen–Stevens reaction
3315:Kiliani–Fischer synthesis
3235:Elbs persulfate oxidation
3160:Bouveault–Blanc reduction
3120:Baeyer–Villiger oxidation
2952:Schotten–Baumann reaction
2828:Ramberg–Bäcklund reaction
2707:Kiliani–Fischer synthesis
2549:Ramberg–Bäcklund reaction
2534:Pinacol coupling reaction
2529:Piancatelli rearrangement
2424:Liebeskind–Srogl coupling
2274:Fujimoto–Belleau reaction
1991:List of organic reactions
1855:Negative hyperconjugation
1600:
1542:
1488:Michaelis–Menten kinetics
681:E1cB-elimination reaction
460:nucleophilic substitution
175:with lower energy than a
81:, exists. Finally, the
4759:Pictet–Spengler reaction
4674:Einhorn–Brunner reaction
4639:Boger pyridine synthesis
4533:Oxo-Diels–Alder reaction
4448:Aza-Diels–Alder reaction
4345:Pictet–Spengler reaction
4245:Hofmann–Löffler reaction
4235:Hegedus indole synthesis
4205:Fischer indole synthesis
4080:Bartoli indole synthesis
4035:Willgerodt rearrangement
3865:McLafferty rearrangement
3775:Ferrier carbocyclization
3590:2,3-Wittig rearrangement
3580:1,2-Wittig rearrangement
3420:Parikh–Doering oxidation
3410:Oxygen rebound mechanism
3075:Adkins–Peterson reaction
2967:Yamaguchi esterification
2907:Hegedus indole synthesis
2872:Bartoli indole synthesis
2743:Bamford–Stevens reaction
2659:Weinreb ketone synthesis
2619:Stork enamine alkylation
2394:Knoevenagel condensation
2264:Ferrier carbocyclization
2154:Castro–Stephens coupling
1780:Hammett acidity function
1770:Free-energy relationship
1715:Curtin–Hammett principle
1700:Conformational isomerism
1415:Potential energy surface
1294:Electron/Proton transfer
1179:Unimolecular elimination
873:Anslyn, Eric V. (2006).
715:10.15227/orgsyn.005.0033
488:a stronger base is used.
470:and also between E1 and
342:deuterium isotope effect
273:unimolecular elimination
214:deuterium isotope effect
212:and therefore a primary
34:Elimination reaction of
4719:Knorr pyrrole synthesis
4654:Bucherer–Bergs reaction
4599:Allan–Robinson reaction
4578:Wagner-Jauregg reaction
4370:Ring-closing metathesis
4295:Larock indole synthesis
4275:Knorr pyrrole synthesis
4130:Bucherer–Bergs reaction
3995:Stieglitz rearrangement
3975:Skattebøl rearrangement
3945:Ring-closing metathesis
3805:Group transfer reaction
3770:Favorskii rearrangement
3710:Cornforth rearrangement
3640:Bamberger rearrangement
3545:Wolff–Kishner reduction
3365:Markó–Lam deoxygenation
3260:Fleming–Tamao oxidation
3255:Fischer–Tropsch process
2942:Oxymercuration reaction
2922:Knorr pyrrole synthesis
2748:Barton–Kellogg reaction
2654:Wagner-Jauregg reaction
2574:Ring-closing metathesis
2564:Reimer–Tiemann reaction
2554:Rauhut–Currier reaction
2469:Nef isocyanide reaction
2429:Malonic ester synthesis
2399:Knorr pyrrole synthesis
2334:High dilution principle
2269:Friedel–Crafts reaction
2204:Cross-coupling reaction
2129:Bucherer–Bergs reaction
2114:Blanc chloromethylation
2104:Blaise ketone synthesis
2079:Baylis–Hillman reaction
2074:Barton–Kellogg reaction
2049:Allan–Robinson reaction
1955:Woodward–Hoffmann rules
1690:Charge-transfer complex
1463:Transition state theory
1264:Intramolecular reaction
1190:Bimolecular elimination
669:Christopher Kelk Ingold
532:1 and E1 pathways.
507:potassium tert-butoxide
387:rearrangement reactions
114:bimolecular elimination
4684:Feist–Benary synthesis
4458:Bradsher cycloaddition
4428:4+4 Photocycloaddition
4385:Simmons–Smith reaction
4330:Paternò–Büchi reaction
4190:Feist–Benary synthesis
4180:Dieckmann condensation
3930:Pummerer rearrangement
3910:Oxy-Cope rearrangement
3885:Myers allene synthesis
3835:Jacobsen rearrangement
3750:Electrocyclic reaction
3725:Demjanov rearrangement
3680:Buchner ring expansion
3650:Beckmann rearrangement
3630:Aza-Cope rearrangement
3625:Arndt–Eistert reaction
3600:Alkyne zipper reaction
3520:Transfer hydrogenation
3495:Sharpless oxyamination
3470:Selenoxide elimination
3355:Lombardo methylenation
3280:Griesbaum coozonolysis
3190:Corey–Itsuno reduction
3165:Boyland–Sims oxidation
3105:Angeli–Rimini reaction
2753:Boord olefin synthesis
2697:Arndt–Eistert reaction
2689:Homologation reactions
2489:Nitro-Mannich reaction
2404:Kolbe–Schmitt reaction
2214:Cross-coupling partner
2134:Buchner ring expansion
2054:Arndt–Eistert reaction
1820:Kinetic isotope effect
1567:Rearrangement reaction
1257:Unimolecular reactions
1218:Electrophilic addition
1037:Quotations related to
620:
582:kinetic isotope effect
429:Rearrangement possible
397:
372:Only reaction product
368:
236:
173:staggered conformation
47:
4828:Olefination reactions
4823:Elimination reactions
4543:Pauson–Khand reaction
4380:Sharpless epoxidation
4335:Pechmann condensation
4215:Friedländer synthesis
4165:Davis–Beirut reaction
4020:Wallach rearrangement
3990:Stevens rearrangement
3925:Pinacol rearrangement
3905:Overman rearrangement
3820:Hofmann rearrangement
3815:Hayashi rearrangement
3780:Ferrier rearrangement
3735:Dimroth rearrangement
3720:Curtius rearrangement
3715:Criegee rearrangement
3695:Claisen rearrangement
3685:Carroll rearrangement
3620:Amadori rearrangement
3610:Allylic rearrangement
3490:Sharpless epoxidation
3225:Dess–Martin oxidation
3150:Bohn–Schmidt reaction
3010:Hofmann rearrangement
2813:Kauffmann olefination
2736:Olefination reactions
2674:Wurtz–Fittig reaction
2509:Palladium–NHC complex
2389:Kauffmann olefination
2344:Homologation reaction
2194:Corey–House synthesis
2174:Claisen rearrangement
1970:Yukawa–Tsuno equation
1930:Swain–Lupton equation
1910:Spherical aromaticity
1845:Möbius–Hückel concept
1630:Aromatic ring current
1592:Substitution reaction
1448:Rate-determining step
1380:Reactive intermediate
1238:Free-radical addition
1228:Nucleophilic addition
1171:Elimination reactions
1027:Elimination reactions
645:reductive elimination
619:
395:
366:
323:apply (unimolecular).
317:rate-determining step
234:
219:E2 competes with the
210:rate determining step
33:
4749:Paal–Knorr synthesis
4619:Barton–Zard reaction
4563:Staudinger synthesis
4513:Ketene cycloaddition
4483:Diels–Alder reaction
4463:Cheletropic reaction
4443:Alkyne trimerisation
4325:Paal–Knorr synthesis
4290:Kulinkovich reaction
4265:Jacobsen epoxidation
4185:Diels–Alder reaction
3980:Smiles rearrangement
3970:Sigmatropic reaction
3855:Lossen rearrangement
3705:Corey–Fuchs reaction
3670:Boekelheide reaction
3665:Bergmann degradation
3595:Achmatowicz reaction
3380:Methionine sulfoxide
3180:Clemmensen reduction
3140:Bergmann degradation
3070:Acyloin condensation
3035:Strecker degradation
2990:Bergmann degradation
2957:Ullmann condensation
2823:Peterson olefination
2798:Hydrazone iodination
2778:Elimination reaction
2679:Zincke–Suhl reaction
2599:Sonogashira coupling
2559:Reformatsky reaction
2519:Peterson olefination
2484:Nierenstein reaction
2414:Kulinkovich reaction
2229:Diels–Alder reaction
2189:Corey–Fuchs reaction
2169:Claisen condensation
2039:Alkyne trimerisation
2014:Acyloin condensation
1980:Σ-bishomoaromaticity
1940:Thorpe–Ingold effect
1552:Elimination reaction
1443:Equilibrium constant
1039:Elimination reaction
1029:at Wikimedia Commons
494:increases (increase
321:first-order kinetics
167:. An antiperiplanar
52:elimination reaction
4833:Reaction mechanisms
4769:Prilezhaev reaction
4754:Pellizzari reaction
4433:(4+3) cycloaddition
4400:Van Leusen reaction
4375:Robinson annulation
4360:Pschorr cyclization
4355:Prilezhaev reaction
4085:Bergman cyclization
4040:Wolff rearrangement
4025:Weerman degradation
3915:Pericyclic reaction
3895:Neber rearrangement
3790:Fries rearrangement
3675:Brook rearrangement
3660:Bergman cyclization
3505:Staudinger reaction
3450:Rosenmund reduction
3440:Reductive amination
3405:Oppenauer oxidation
3195:Corey–Kim oxidation
3170:Cannizzaro reaction
3045:Weerman degradation
3020:Isosaccharinic acid
2932:Mukaiyama hydration
2788:Hofmann elimination
2773:Dehydrohalogenation
2758:Chugaev elimination
2579:Robinson annulation
2524:Pfitzinger reaction
2294:Gattermann reaction
2239:Wulff–Dötz reaction
2219:Dakin–West reaction
2144:Carbonyl allylation
2089:Bergman cyclization
1875:Kennedy J. P. Orton
1795:Hammond's postulate
1765:Flippin–Lodge angle
1735:Electromeric effect
1660:Beta-silicon effect
1645:Baker–Nathan effect
1453:Reaction coordinate
1385:Radical (chemistry)
1370:Elementary reaction
1313:Grotthuss mechanism
1077:reaction mechanisms
953:10.1021/ja00394a043
781:2008JChEd..85..552N
737:, New York: Wiley,
501:the base is a poor
299:of the carbocation.
243:is the reaction of
4518:McCormack reaction
4468:Conia-ene reaction
4300:Madelung synthesis
4090:Biginelli reaction
3880:Mumm rearrangement
3765:Favorskii reaction
3700:Cope rearrangement
3690:Chan rearrangement
3455:Rubottom oxidation
3385:Miyaura borylation
3350:Lipid peroxidation
3345:Lindgren oxidation
3325:Kornblum oxidation
3320:Kolbe electrolysis
3265:Fukuyama reduction
3175:Carbonyl reduction
3025:Marker degradation
2887:Diazonium compound
2877:Boudouard reaction
2856:Carbon-heteroatom
2783:Grieco elimination
2569:Rieche formylation
2514:Passerini reaction
2444:Meerwein arylation
2364:Hydroxymethylation
2259:Favorskii reaction
2159:Chan rearrangement
2094:Biginelli reaction
2019:Aldol condensation
1865:2-Norbornyl cation
1840:Möbius aromaticity
1835:Markovnikov's rule
1730:Effective molarity
1675:Bürgi–Dunitz angle
1665:Bicycloaromaticity
1478:Arrhenius equation
1248:Oxidative addition
1210:Addition reactions
621:
398:
369:
249:potassium ethoxide
237:
48:
4810:
4809:
4806:
4805:
4802:
4801:
4794:Wohl–Aue reaction
4438:6+4 Cycloaddition
4255:Iodolactonization
3575:1,2-rearrangement
3540:Wohl–Aue reaction
3460:Sabatier reaction
3425:Pinnick oxidation
3390:Mozingo reduction
3335:Leuckart reaction
3290:Haloform reaction
3205:Criegee oxidation
3185:Collins oxidation
3135:Benkeser reaction
3130:Bechamp reduction
3100:Andrussow process
3085:Alcohol oxidation
2995:Edman degradation
2902:Haloform reaction
2851:
2850:
2838:Takai olefination
2803:Julia olefination
2629:Takai olefination
2504:Olefin metathesis
2379:Julia olefination
2304:Grignard reaction
2284:Fukuyama coupling
2199:Coupling reaction
2164:Chan–Lam coupling
2034:Alkyne metathesis
2029:Alkane metathesis
1885:Phosphaethynolate
1790:George S. Hammond
1750:Electronic effect
1705:Conjugated system
1587:Stereospecificity
1582:Stereoselectivity
1547:Addition reaction
1536:organic reactions
1501:
1500:
1473:Activated complex
1468:Activation energy
1430:Chemical kinetics
1375:Reaction dynamics
1274:Photodissociation
1025:Media related to
989:10.1021/ol200121f
852:10.1021/ja057491d
840:J. Am. Chem. Soc.
814:Organic Chemistry
789:10.1021/ed085p552
702:Organic Syntheses
580:In one study the
261:potassium bromide
16:(Redirected from
4840:
4789:Wenker synthesis
4779:Stollé synthesis
4634:Bobbitt reaction
4604:Auwers synthesis
4548:Povarov reaction
4473:Cyclopropanation
4411:
4405:Wenker synthesis
4160:Darzens reaction
4110:Bobbitt reaction
3955:Schmidt reaction
3760:Enyne metathesis
3535:Whiting reaction
3530:Wharton reaction
3475:Shapiro reaction
3465:Sarett oxidation
3430:Prévost reaction
3240:Emde degradation
3050:Wohl degradation
3030:Ruff degradation
3000:Emde degradation
2897:Grignard reagent
2833:Shapiro reaction
2818:McMurry reaction
2685:
2649:Ullmann reaction
2614:Stollé synthesis
2604:Stetter reaction
2594:Shapiro reaction
2584:Sakurai reaction
2479:Negishi coupling
2459:Minisci reaction
2454:Michael reaction
2439:McMurry reaction
2434:Mannich reaction
2314:Hammick reaction
2309:Grignard reagent
2249:Enyne metathesis
2234:Doebner reaction
2224:Darzens reaction
2069:Barbier reaction
2059:Auwers synthesis
1986:
1960:Woodward's rules
1925:Superaromaticity
1915:Spiroaromaticity
1815:Inductive effect
1810:Hyperconjugation
1785:Hammett equation
1725:Edwards equation
1577:Regioselectivity
1528:
1521:
1514:
1505:
1405:Collision theory
1354:Matrix isolation
1308:Harpoon reaction
1185:E1cB-elimination
1069:
1062:
1055:
1046:
1036:
1024:
1009:
1008:
983:(7): 1646–1649.
971:
965:
964:
936:
930:
929:
903:
897:
896:
870:
864:
863:
834:
828:
827:
809:
803:
799:
793:
792:
762:
756:
755:
725:
719:
718:
696:
596:reaction (only S
590:t-butyl chloride
483:steric hindrance
169:transition state
140:transition state
56:organic reaction
21:
4848:
4847:
4843:
4842:
4841:
4839:
4838:
4837:
4813:
4812:
4811:
4798:
4699:Gewald reaction
4582:
4409:
4390:Skraup reaction
4225:Graham reaction
4220:Gewald reaction
4051:
4044:
3566:
3559:
3515:Swern oxidation
3500:Stahl oxidation
3445:Riley oxidation
3400:Omega oxidation
3360:Luche reduction
3310:Jones oxidation
3275:Glycol cleavage
3270:Ganem oxidation
3215:Davis oxidation
3210:Dakin oxidation
3145:Birch reduction
3095:Amide reduction
3061:
3054:
3015:Hooker reaction
2977:
2971:
2859:
2857:
2847:
2843:Wittig reaction
2731:
2727:Wittig reaction
2702:Hooker reaction
2683:
2664:Wittig reaction
2639:Thorpe reaction
2624:Suzuki reaction
2609:Stille reaction
2544:Quelet reaction
2419:Kumada coupling
2369:Ivanov reaction
2359:Hydrovinylation
2339:Hiyama coupling
2299:Glaser coupling
2109:Blaise reaction
2099:Bingel reaction
2084:Benary reaction
2001:
1999:
1993:
1984:
1880:Passive binding
1800:Homoaromaticity
1650:Baldwin's rules
1625:Antiaromaticity
1620:Anomeric effect
1596:
1538:
1532:
1502:
1497:
1483:Eyring equation
1424:
1395:Stereochemistry
1358:
1344:Solvent effects
1332:
1288:
1252:
1233:
1223:
1204:
1199:
1165:
1161:
1142:
1138:
1128:
1118:
1108:
1098:
1079:
1073:
1017:
1012:
977:Organic Letters
973:
972:
968:
938:
937:
933:
918:
905:
904:
900:
885:
872:
871:
867:
836:
835:
831:
824:
811:
810:
806:
800:
796:
764:
763:
759:
745:
727:
726:
722:
698:
697:
693:
689:
677:
665:
642:
637:dichlorocarbene
634:
625:carbon monoxide
611:
603:
599:
594:methyl chloride
576:
567:
560:
556:
549:
545:
541:
531:
524:
516:
475:
467:
439:
421:
353:sulfonate ester
334:
269:
245:isobutylbromide
224:
118:carbon-hydrogen
110:
101:
79:
28:
23:
22:
15:
12:
11:
5:
4846:
4844:
4836:
4835:
4830:
4825:
4815:
4814:
4808:
4807:
4804:
4803:
4800:
4799:
4797:
4796:
4791:
4786:
4781:
4776:
4771:
4766:
4761:
4756:
4751:
4746:
4741:
4736:
4731:
4726:
4721:
4716:
4711:
4706:
4704:Hantzsch ester
4701:
4696:
4691:
4686:
4681:
4676:
4671:
4666:
4661:
4656:
4651:
4646:
4641:
4636:
4631:
4626:
4621:
4616:
4614:Banert cascade
4611:
4606:
4601:
4596:
4590:
4588:
4584:
4583:
4581:
4580:
4575:
4570:
4565:
4560:
4555:
4553:Prato reaction
4550:
4545:
4540:
4535:
4530:
4525:
4520:
4515:
4510:
4505:
4500:
4495:
4490:
4485:
4480:
4475:
4470:
4465:
4460:
4455:
4450:
4445:
4440:
4435:
4430:
4425:
4419:
4417:
4408:
4407:
4402:
4397:
4392:
4387:
4382:
4377:
4372:
4367:
4362:
4357:
4352:
4347:
4342:
4337:
4332:
4327:
4322:
4317:
4312:
4307:
4302:
4297:
4292:
4287:
4282:
4277:
4272:
4267:
4262:
4257:
4252:
4247:
4242:
4237:
4232:
4227:
4222:
4217:
4212:
4207:
4202:
4197:
4192:
4187:
4182:
4177:
4172:
4167:
4162:
4157:
4152:
4147:
4142:
4137:
4132:
4127:
4122:
4117:
4112:
4107:
4102:
4097:
4092:
4087:
4082:
4077:
4072:
4067:
4062:
4056:
4054:
4046:
4045:
4043:
4042:
4037:
4032:
4027:
4022:
4017:
4012:
4007:
4002:
3997:
3992:
3987:
3982:
3977:
3972:
3967:
3962:
3957:
3952:
3947:
3942:
3937:
3932:
3927:
3922:
3917:
3912:
3907:
3902:
3897:
3892:
3887:
3882:
3877:
3872:
3867:
3862:
3857:
3852:
3847:
3842:
3837:
3832:
3827:
3822:
3817:
3812:
3807:
3802:
3797:
3792:
3787:
3782:
3777:
3772:
3767:
3762:
3757:
3752:
3747:
3742:
3737:
3732:
3727:
3722:
3717:
3712:
3707:
3702:
3697:
3692:
3687:
3682:
3677:
3672:
3667:
3662:
3657:
3652:
3647:
3645:Banert cascade
3642:
3637:
3632:
3627:
3622:
3617:
3612:
3607:
3602:
3597:
3592:
3587:
3582:
3577:
3571:
3569:
3565:Rearrangement
3561:
3560:
3558:
3557:
3555:Zinin reaction
3552:
3547:
3542:
3537:
3532:
3527:
3525:Wacker process
3522:
3517:
3512:
3507:
3502:
3497:
3492:
3487:
3482:
3477:
3472:
3467:
3462:
3457:
3452:
3447:
3442:
3437:
3432:
3427:
3422:
3417:
3412:
3407:
3402:
3397:
3392:
3387:
3382:
3377:
3372:
3367:
3362:
3357:
3352:
3347:
3342:
3337:
3332:
3327:
3322:
3317:
3312:
3307:
3302:
3300:Hydrogenolysis
3297:
3292:
3287:
3282:
3277:
3272:
3267:
3262:
3257:
3252:
3250:Étard reaction
3247:
3242:
3237:
3232:
3227:
3222:
3217:
3212:
3207:
3202:
3197:
3192:
3187:
3182:
3177:
3172:
3167:
3162:
3157:
3155:Bosch reaction
3152:
3147:
3142:
3137:
3132:
3127:
3122:
3117:
3112:
3107:
3102:
3097:
3092:
3087:
3082:
3077:
3072:
3066:
3064:
3060:Organic redox
3056:
3055:
3053:
3052:
3047:
3042:
3037:
3032:
3027:
3022:
3017:
3012:
3007:
3002:
2997:
2992:
2987:
2981:
2979:
2973:
2972:
2970:
2969:
2964:
2959:
2954:
2949:
2944:
2939:
2934:
2929:
2924:
2919:
2914:
2909:
2904:
2899:
2894:
2892:Esterification
2889:
2884:
2879:
2874:
2869:
2863:
2861:
2853:
2852:
2849:
2848:
2846:
2845:
2840:
2835:
2830:
2825:
2820:
2815:
2810:
2805:
2800:
2795:
2790:
2785:
2780:
2775:
2770:
2765:
2760:
2755:
2750:
2745:
2739:
2737:
2733:
2732:
2730:
2729:
2724:
2719:
2714:
2709:
2704:
2699:
2693:
2691:
2682:
2681:
2676:
2671:
2669:Wurtz reaction
2666:
2661:
2656:
2651:
2646:
2641:
2636:
2631:
2626:
2621:
2616:
2611:
2606:
2601:
2596:
2591:
2586:
2581:
2576:
2571:
2566:
2561:
2556:
2551:
2546:
2541:
2539:Prins reaction
2536:
2531:
2526:
2521:
2516:
2511:
2506:
2501:
2496:
2491:
2486:
2481:
2476:
2471:
2466:
2461:
2456:
2451:
2446:
2441:
2436:
2431:
2426:
2421:
2416:
2411:
2406:
2401:
2396:
2391:
2386:
2381:
2376:
2371:
2366:
2361:
2356:
2354:Hydrocyanation
2351:
2346:
2341:
2336:
2331:
2326:
2324:Henry reaction
2321:
2316:
2311:
2306:
2301:
2296:
2291:
2286:
2281:
2276:
2271:
2266:
2261:
2256:
2251:
2246:
2241:
2236:
2231:
2226:
2221:
2216:
2211:
2206:
2201:
2196:
2191:
2186:
2181:
2176:
2171:
2166:
2161:
2156:
2151:
2146:
2141:
2136:
2131:
2126:
2121:
2116:
2111:
2106:
2101:
2096:
2091:
2086:
2081:
2076:
2071:
2066:
2061:
2056:
2051:
2046:
2041:
2036:
2031:
2026:
2024:Aldol reaction
2021:
2016:
2011:
2005:
2003:
1998:Carbon-carbon
1995:
1994:
1989:
1983:
1982:
1977:
1975:Zaitsev's rule
1972:
1967:
1962:
1957:
1952:
1947:
1942:
1937:
1932:
1927:
1922:
1920:Steric effects
1917:
1912:
1907:
1902:
1897:
1892:
1887:
1882:
1877:
1872:
1867:
1862:
1857:
1852:
1847:
1842:
1837:
1832:
1827:
1822:
1817:
1812:
1807:
1802:
1797:
1792:
1787:
1782:
1777:
1772:
1767:
1762:
1757:
1752:
1747:
1742:
1737:
1732:
1727:
1722:
1717:
1712:
1707:
1702:
1697:
1692:
1687:
1682:
1677:
1672:
1667:
1662:
1657:
1652:
1647:
1642:
1637:
1632:
1627:
1622:
1617:
1612:
1607:
1601:
1598:
1597:
1595:
1594:
1589:
1584:
1579:
1574:
1572:Redox reaction
1569:
1564:
1559:
1557:Polymerization
1554:
1549:
1543:
1540:
1539:
1533:
1531:
1530:
1523:
1516:
1508:
1499:
1498:
1496:
1495:
1490:
1485:
1480:
1475:
1470:
1465:
1460:
1455:
1450:
1445:
1440:
1434:
1432:
1426:
1425:
1423:
1422:
1417:
1412:
1407:
1402:
1397:
1392:
1387:
1382:
1377:
1372:
1366:
1364:
1363:Related topics
1360:
1359:
1357:
1356:
1351:
1346:
1340:
1338:
1337:Medium effects
1334:
1333:
1331:
1330:
1325:
1320:
1315:
1310:
1305:
1299:
1297:
1290:
1289:
1287:
1286:
1281:
1276:
1271:
1266:
1260:
1258:
1254:
1253:
1251:
1250:
1245:
1240:
1235:
1231:
1225:
1221:
1214:
1212:
1206:
1205:
1203:
1202:
1197:
1193:
1187:
1182:
1175:
1173:
1167:
1166:
1164:
1163:
1159:
1152:
1150:
1144:
1143:
1141:
1140:
1136:
1130:
1126:
1120:
1116:
1110:
1106:
1100:
1096:
1089:
1087:
1081:
1080:
1074:
1072:
1071:
1064:
1057:
1049:
1043:
1042:
1030:
1016:
1015:External links
1013:
1011:
1010:
966:
947:(4): 948–949.
931:
916:
898:
883:
865:
846:(3): 736–737.
829:
822:
804:
794:
757:
743:
720:
690:
688:
685:
684:
683:
676:
673:
671:in the 1920s.
664:
661:
640:
632:
610:
607:
601:
597:
574:
565:
558:
554:
547:
543:
539:
529:
522:
514:
511:
510:
499:
489:
486:
473:
465:
438:
435:
434:
433:
430:
419:
416:
415:
412:
400:An example in
390:
389:
382:
381:
361:
360:
345:
338:
332:
327:
324:
309:
302:
301:
300:
294:
268:
265:
259:, ethanol and
229:
228:
222:
217:
206:
191:
184:
165:antiperiplanar
157:
146:
143:
122:carbon-halogen
109:
106:
99:
77:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
4845:
4834:
4831:
4829:
4826:
4824:
4821:
4820:
4818:
4795:
4792:
4790:
4787:
4785:
4782:
4780:
4777:
4775:
4772:
4770:
4767:
4765:
4762:
4760:
4757:
4755:
4752:
4750:
4747:
4745:
4742:
4740:
4737:
4735:
4732:
4730:
4727:
4725:
4722:
4720:
4717:
4715:
4714:Herz reaction
4712:
4710:
4707:
4705:
4702:
4700:
4697:
4695:
4692:
4690:
4687:
4685:
4682:
4680:
4677:
4675:
4672:
4670:
4667:
4665:
4662:
4660:
4657:
4655:
4652:
4650:
4647:
4645:
4642:
4640:
4637:
4635:
4632:
4630:
4627:
4625:
4622:
4620:
4617:
4615:
4612:
4610:
4607:
4605:
4602:
4600:
4597:
4595:
4592:
4591:
4589:
4585:
4579:
4576:
4574:
4571:
4569:
4566:
4564:
4561:
4559:
4556:
4554:
4551:
4549:
4546:
4544:
4541:
4539:
4536:
4534:
4531:
4529:
4526:
4524:
4521:
4519:
4516:
4514:
4511:
4509:
4506:
4504:
4501:
4499:
4496:
4494:
4491:
4489:
4486:
4484:
4481:
4479:
4476:
4474:
4471:
4469:
4466:
4464:
4461:
4459:
4456:
4454:
4451:
4449:
4446:
4444:
4441:
4439:
4436:
4434:
4431:
4429:
4426:
4424:
4421:
4420:
4418:
4416:
4415:Cycloaddition
4412:
4406:
4403:
4401:
4398:
4396:
4393:
4391:
4388:
4386:
4383:
4381:
4378:
4376:
4373:
4371:
4368:
4366:
4363:
4361:
4358:
4356:
4353:
4351:
4348:
4346:
4343:
4341:
4338:
4336:
4333:
4331:
4328:
4326:
4323:
4321:
4318:
4316:
4313:
4311:
4308:
4306:
4303:
4301:
4298:
4296:
4293:
4291:
4288:
4286:
4283:
4281:
4278:
4276:
4273:
4271:
4268:
4266:
4263:
4261:
4260:Isay reaction
4258:
4256:
4253:
4251:
4248:
4246:
4243:
4241:
4238:
4236:
4233:
4231:
4228:
4226:
4223:
4221:
4218:
4216:
4213:
4211:
4208:
4206:
4203:
4201:
4198:
4196:
4193:
4191:
4188:
4186:
4183:
4181:
4178:
4176:
4173:
4171:
4168:
4166:
4163:
4161:
4158:
4156:
4155:Cycloaddition
4153:
4151:
4148:
4146:
4143:
4141:
4138:
4136:
4133:
4131:
4128:
4126:
4123:
4121:
4118:
4116:
4113:
4111:
4108:
4106:
4103:
4101:
4098:
4096:
4093:
4091:
4088:
4086:
4083:
4081:
4078:
4076:
4073:
4071:
4068:
4066:
4063:
4061:
4058:
4057:
4055:
4053:
4050:Ring forming
4047:
4041:
4038:
4036:
4033:
4031:
4028:
4026:
4023:
4021:
4018:
4016:
4013:
4011:
4008:
4006:
4003:
4001:
3998:
3996:
3993:
3991:
3988:
3986:
3983:
3981:
3978:
3976:
3973:
3971:
3968:
3966:
3963:
3961:
3958:
3956:
3953:
3951:
3950:Rupe reaction
3948:
3946:
3943:
3941:
3938:
3936:
3933:
3931:
3928:
3926:
3923:
3921:
3918:
3916:
3913:
3911:
3908:
3906:
3903:
3901:
3898:
3896:
3893:
3891:
3888:
3886:
3883:
3881:
3878:
3876:
3873:
3871:
3868:
3866:
3863:
3861:
3858:
3856:
3853:
3851:
3848:
3846:
3843:
3841:
3838:
3836:
3833:
3831:
3828:
3826:
3823:
3821:
3818:
3816:
3813:
3811:
3808:
3806:
3803:
3801:
3798:
3796:
3793:
3791:
3788:
3786:
3783:
3781:
3778:
3776:
3773:
3771:
3768:
3766:
3763:
3761:
3758:
3756:
3753:
3751:
3748:
3746:
3743:
3741:
3738:
3736:
3733:
3731:
3728:
3726:
3723:
3721:
3718:
3716:
3713:
3711:
3708:
3706:
3703:
3701:
3698:
3696:
3693:
3691:
3688:
3686:
3683:
3681:
3678:
3676:
3673:
3671:
3668:
3666:
3663:
3661:
3658:
3656:
3653:
3651:
3648:
3646:
3643:
3641:
3638:
3636:
3633:
3631:
3628:
3626:
3623:
3621:
3618:
3616:
3613:
3611:
3608:
3606:
3603:
3601:
3598:
3596:
3593:
3591:
3588:
3586:
3583:
3581:
3578:
3576:
3573:
3572:
3570:
3568:
3562:
3556:
3553:
3551:
3548:
3546:
3543:
3541:
3538:
3536:
3533:
3531:
3528:
3526:
3523:
3521:
3518:
3516:
3513:
3511:
3508:
3506:
3503:
3501:
3498:
3496:
3493:
3491:
3488:
3486:
3483:
3481:
3478:
3476:
3473:
3471:
3468:
3466:
3463:
3461:
3458:
3456:
3453:
3451:
3448:
3446:
3443:
3441:
3438:
3436:
3433:
3431:
3428:
3426:
3423:
3421:
3418:
3416:
3413:
3411:
3408:
3406:
3403:
3401:
3398:
3396:
3393:
3391:
3388:
3386:
3383:
3381:
3378:
3376:
3373:
3371:
3368:
3366:
3363:
3361:
3358:
3356:
3353:
3351:
3348:
3346:
3343:
3341:
3340:Ley oxidation
3338:
3336:
3333:
3331:
3328:
3326:
3323:
3321:
3318:
3316:
3313:
3311:
3308:
3306:
3305:Hydroxylation
3303:
3301:
3298:
3296:
3295:Hydrogenation
3293:
3291:
3288:
3286:
3283:
3281:
3278:
3276:
3273:
3271:
3268:
3266:
3263:
3261:
3258:
3256:
3253:
3251:
3248:
3246:
3243:
3241:
3238:
3236:
3233:
3231:
3230:DNA oxidation
3228:
3226:
3223:
3221:
3220:Deoxygenation
3218:
3216:
3213:
3211:
3208:
3206:
3203:
3201:
3198:
3196:
3193:
3191:
3188:
3186:
3183:
3181:
3178:
3176:
3173:
3171:
3168:
3166:
3163:
3161:
3158:
3156:
3153:
3151:
3148:
3146:
3143:
3141:
3138:
3136:
3133:
3131:
3128:
3126:
3123:
3121:
3118:
3116:
3113:
3111:
3110:Aromatization
3108:
3106:
3103:
3101:
3098:
3096:
3093:
3091:
3088:
3086:
3083:
3081:
3078:
3076:
3073:
3071:
3068:
3067:
3065:
3063:
3057:
3051:
3048:
3046:
3043:
3041:
3038:
3036:
3033:
3031:
3028:
3026:
3023:
3021:
3018:
3016:
3013:
3011:
3008:
3006:
3003:
3001:
2998:
2996:
2993:
2991:
2988:
2986:
2983:
2982:
2980:
2974:
2968:
2965:
2963:
2960:
2958:
2955:
2953:
2950:
2948:
2947:Reed reaction
2945:
2943:
2940:
2938:
2935:
2933:
2930:
2928:
2925:
2923:
2920:
2918:
2915:
2913:
2910:
2908:
2905:
2903:
2900:
2898:
2895:
2893:
2890:
2888:
2885:
2883:
2880:
2878:
2875:
2873:
2870:
2868:
2865:
2864:
2862:
2858:bond forming
2854:
2844:
2841:
2839:
2836:
2834:
2831:
2829:
2826:
2824:
2821:
2819:
2816:
2814:
2811:
2809:
2806:
2804:
2801:
2799:
2796:
2794:
2791:
2789:
2786:
2784:
2781:
2779:
2776:
2774:
2771:
2769:
2766:
2764:
2763:Cope reaction
2761:
2759:
2756:
2754:
2751:
2749:
2746:
2744:
2741:
2740:
2738:
2734:
2728:
2725:
2723:
2720:
2718:
2715:
2713:
2710:
2708:
2705:
2703:
2700:
2698:
2695:
2694:
2692:
2690:
2686:
2680:
2677:
2675:
2672:
2670:
2667:
2665:
2662:
2660:
2657:
2655:
2652:
2650:
2647:
2645:
2642:
2640:
2637:
2635:
2632:
2630:
2627:
2625:
2622:
2620:
2617:
2615:
2612:
2610:
2607:
2605:
2602:
2600:
2597:
2595:
2592:
2590:
2587:
2585:
2582:
2580:
2577:
2575:
2572:
2570:
2567:
2565:
2562:
2560:
2557:
2555:
2552:
2550:
2547:
2545:
2542:
2540:
2537:
2535:
2532:
2530:
2527:
2525:
2522:
2520:
2517:
2515:
2512:
2510:
2507:
2505:
2502:
2500:
2497:
2495:
2492:
2490:
2487:
2485:
2482:
2480:
2477:
2475:
2474:Nef synthesis
2472:
2470:
2467:
2465:
2462:
2460:
2457:
2455:
2452:
2450:
2449:Methylenation
2447:
2445:
2442:
2440:
2437:
2435:
2432:
2430:
2427:
2425:
2422:
2420:
2417:
2415:
2412:
2410:
2407:
2405:
2402:
2400:
2397:
2395:
2392:
2390:
2387:
2385:
2382:
2380:
2377:
2375:
2372:
2370:
2367:
2365:
2362:
2360:
2357:
2355:
2352:
2350:
2347:
2345:
2342:
2340:
2337:
2335:
2332:
2330:
2327:
2325:
2322:
2320:
2319:Heck reaction
2317:
2315:
2312:
2310:
2307:
2305:
2302:
2300:
2297:
2295:
2292:
2290:
2287:
2285:
2282:
2280:
2277:
2275:
2272:
2270:
2267:
2265:
2262:
2260:
2257:
2255:
2252:
2250:
2247:
2245:
2242:
2240:
2237:
2235:
2232:
2230:
2227:
2225:
2222:
2220:
2217:
2215:
2212:
2210:
2207:
2205:
2202:
2200:
2197:
2195:
2192:
2190:
2187:
2185:
2182:
2180:
2177:
2175:
2172:
2170:
2167:
2165:
2162:
2160:
2157:
2155:
2152:
2150:
2147:
2145:
2142:
2140:
2137:
2135:
2132:
2130:
2127:
2125:
2122:
2120:
2117:
2115:
2112:
2110:
2107:
2105:
2102:
2100:
2097:
2095:
2092:
2090:
2087:
2085:
2082:
2080:
2077:
2075:
2072:
2070:
2067:
2065:
2062:
2060:
2057:
2055:
2052:
2050:
2047:
2045:
2042:
2040:
2037:
2035:
2032:
2030:
2027:
2025:
2022:
2020:
2017:
2015:
2012:
2010:
2007:
2006:
2004:
2000:bond forming
1996:
1992:
1987:
1981:
1978:
1976:
1973:
1971:
1968:
1966:
1965:Y-aromaticity
1963:
1961:
1958:
1956:
1953:
1951:
1950:Walsh diagram
1948:
1946:
1943:
1941:
1938:
1936:
1935:Taft equation
1933:
1931:
1928:
1926:
1923:
1921:
1918:
1916:
1913:
1911:
1908:
1906:
1905:Σ-aromaticity
1903:
1901:
1898:
1896:
1893:
1891:
1888:
1886:
1883:
1881:
1878:
1876:
1873:
1871:
1868:
1866:
1863:
1861:
1858:
1856:
1853:
1851:
1848:
1846:
1843:
1841:
1838:
1836:
1833:
1831:
1830:Marcus theory
1828:
1826:
1823:
1821:
1818:
1816:
1813:
1811:
1808:
1806:
1805:Hückel's rule
1803:
1801:
1798:
1796:
1793:
1791:
1788:
1786:
1783:
1781:
1778:
1776:
1773:
1771:
1768:
1766:
1763:
1761:
1760:Evelyn effect
1758:
1756:
1753:
1751:
1748:
1746:
1743:
1741:
1740:Electron-rich
1738:
1736:
1733:
1731:
1728:
1726:
1723:
1721:
1718:
1716:
1713:
1711:
1708:
1706:
1703:
1701:
1698:
1696:
1693:
1691:
1688:
1686:
1683:
1681:
1678:
1676:
1673:
1671:
1668:
1666:
1663:
1661:
1658:
1656:
1655:Bema Hapothle
1653:
1651:
1648:
1646:
1643:
1641:
1638:
1636:
1633:
1631:
1628:
1626:
1623:
1621:
1618:
1616:
1613:
1611:
1608:
1606:
1603:
1602:
1599:
1593:
1590:
1588:
1585:
1583:
1580:
1578:
1575:
1573:
1570:
1568:
1565:
1563:
1560:
1558:
1555:
1553:
1550:
1548:
1545:
1544:
1541:
1537:
1529:
1524:
1522:
1517:
1515:
1510:
1509:
1506:
1494:
1491:
1489:
1486:
1484:
1481:
1479:
1476:
1474:
1471:
1469:
1466:
1464:
1461:
1459:
1456:
1454:
1451:
1449:
1446:
1444:
1441:
1439:
1438:Rate equation
1436:
1435:
1433:
1431:
1427:
1421:
1418:
1416:
1413:
1411:
1410:Arrow pushing
1408:
1406:
1403:
1401:
1398:
1396:
1393:
1391:
1388:
1386:
1383:
1381:
1378:
1376:
1373:
1371:
1368:
1367:
1365:
1361:
1355:
1352:
1350:
1347:
1345:
1342:
1341:
1339:
1335:
1329:
1326:
1324:
1321:
1319:
1318:Marcus theory
1316:
1314:
1311:
1309:
1306:
1304:
1301:
1300:
1298:
1295:
1291:
1285:
1282:
1280:
1277:
1275:
1272:
1270:
1269:Isomerization
1267:
1265:
1262:
1261:
1259:
1255:
1249:
1246:
1244:
1243:Cycloaddition
1241:
1239:
1236:
1229:
1226:
1219:
1216:
1215:
1213:
1211:
1207:
1201:
1194:
1191:
1188:
1186:
1183:
1180:
1177:
1176:
1174:
1172:
1168:
1157:
1154:
1153:
1151:
1149:
1145:
1134:
1131:
1124:
1121:
1114:
1111:
1104:
1101:
1094:
1091:
1090:
1088:
1086:
1082:
1078:
1070:
1065:
1063:
1058:
1056:
1051:
1050:
1047:
1040:
1035:
1031:
1028:
1023:
1019:
1018:
1014:
1006:
1002:
998:
994:
990:
986:
982:
978:
970:
967:
962:
958:
954:
950:
946:
942:
935:
932:
927:
923:
919:
917:9780470257623
913:
909:
902:
899:
894:
890:
886:
880:
876:
869:
866:
861:
857:
853:
849:
845:
842:
841:
833:
830:
825:
823:0-07-242458-3
819:
815:
808:
805:
798:
795:
790:
786:
782:
778:
774:
770:
769:
761:
758:
754:
750:
746:
744:9780471854722
740:
736:
735:
730:
724:
721:
716:
712:
708:
704:
703:
695:
692:
686:
682:
679:
678:
674:
672:
670:
662:
660:
656:
654:
653:α-abstraction
650:
649:α-elimination
646:
638:
630:
626:
618:
614:
608:
606:
595:
591:
587:
583:
578:
572:
562:
551:
538:
533:
526:
520:
508:
504:
500:
497:
493:
490:
487:
484:
481:
480:
479:
477:
469:
461:
456:
452:
448:
444:
443:reaction rate
436:
431:
428:
427:
426:
423:
413:
410:
409:
408:
405:
403:
394:
388:
384:
383:
379:
375:
371:
370:
365:
358:
354:
351:of a certain
350:
346:
343:
339:
336:
328:
325:
322:
319:. Therefore,
318:
314:
313:reaction rate
310:
307:
303:
298:
297:deprotonation
295:
293:intermediate.
292:
288:
285:
284:
282:
278:
277:
276:
274:
266:
264:
262:
258:
254:
250:
246:
242:
233:
226:
218:
215:
211:
207:
204:
200:
196:
195:hybridization
192:
189:
185:
182:
178:
177:synperiplanar
174:
170:
166:
163:) need to be
162:
158:
155:
151:
150:reaction rate
147:
144:
141:
137:
136:
135:
132:
130:
129:
123:
119:
115:
107:
105:
103:
95:
92:
88:
84:
80:
73:
72:leaving group
69:
65:
61:
58:in which two
57:
54:is a type of
53:
45:
44:sulfuric acid
41:
37:
32:
19:
3755:Ene reaction
3115:Autoxidation
2976:Degradation
2867:Azo coupling
2777:
2644:Ugi reaction
2244:Ene reaction
2044:Alkynylation
1895:Polyfluorene
1890:Polar effect
1755:Electrophile
1670:Bredt's rule
1640:Baird's rule
1610:Alpha effect
1551:
1390:Molecularity
1170:
1041:at Wikiquote
980:
976:
969:
944:
940:
934:
907:
901:
874:
868:
843:
838:
832:
813:
807:
797:
772:
766:
760:
733:
729:March, Jerry
723:
706:
700:
694:
666:
657:
652:
648:
622:
612:
579:
570:
563:
552:
536:
534:
527:
512:
440:
424:
417:
406:
401:
399:
377:
373:
340:A secondary
280:
272:
270:
267:E1 mechanism
240:
238:
202:
198:
154:second order
133:
125:
121:
117:
113:
111:
108:E2 mechanism
67:
63:
60:substituents
51:
49:
36:cyclohexanol
2254:Ethenolysis
1900:Ring strain
1870:Nucleophile
1695:Clar's rule
1635:Aromaticity
1349:Cage effect
1284:RRKM theory
1200:elimination
629:isocyanides
503:nucleophile
492:temperature
335:1 reactions
291:carbocation
68:E1 reaction
64:E2 reaction
40:cyclohexene
18:E1 reaction
4817:Categories
4538:Ozonolysis
4065:Annulation
3415:Ozonolysis
1534:Topics in
884:1891389319
775:(4): 552.
687:References
550:1 and E1.
287:Ionization
225:2 reaction
4052:reactions
3567:reactions
3062:reactions
2978:reactions
2860:reactions
2002:reactions
1400:Catalysis
1296:reactions
997:1523-7060
961:0002-7863
926:268790870
753:642506595
349:pyrolysis
257:isobutene
102:mechanism
83:pyrolysis
1945:Vinylogy
1615:Annulene
1562:Reagents
1005:21366262
893:55600610
860:16417360
731:(1985),
675:See also
586:chlorate
447:halogens
402:scheme 2
306:tertiary
241:scheme 1
181:eclipsed
87:xanthate
46:and heat
1605:A value
777:Bibcode
663:History
496:entropy
455:bromide
357:menthol
253:ethanol
161:halogen
128:Pi bond
91:acetate
1075:Basic
1003:
995:
959:
924:
914:
891:
881:
858:
820:
751:
741:
709:: 33.
639:, :CCl
451:iodide
94:esters
1303:Redox
1139:Acyl)
521:(an S
247:with
42:with
1192:(E2)
1181:(E1)
1001:PMID
993:ISSN
957:ISSN
922:OCLC
912:ISBN
889:OCLC
879:ISBN
856:PMID
818:ISBN
749:OCLC
739:ISBN
651:and
571:tert
453:and
441:The
418:If S
311:The
188:base
171:has
148:The
126:C=C
120:and
89:and
1162:Ar)
1119:Ar)
985:doi
949:doi
945:103
848:doi
844:128
785:doi
711:doi
627:or
355:of
251:in
201:to
152:is
131:).
85:of
50:An
38:to
4819::
1230:(A
1220:(A
1158:(S
1135:(S
1129:i)
1125:(S
1115:(S
1109:2)
1105:(S
1099:1)
1095:(S
999:.
991:.
981:13
979:.
955:.
943:.
920:.
887:.
854:.
783:.
773:85
771:.
747:,
705:.
540:aH
449:,
283:.
263:.
203:sp
199:sp
104:.
78:CB
76:E1
1527:e
1520:t
1513:v
1234:)
1232:N
1224:)
1222:E
1198:i
1196:E
1160:E
1137:N
1127:N
1117:N
1107:N
1097:N
1068:e
1061:t
1054:v
1007:.
987::
963:.
951::
928:.
895:.
862:.
850::
826:.
791:.
787::
779::
717:.
713::
707:5
641:2
633:3
602:N
598:N
575:N
566:N
559:N
555:N
548:N
544:N
537:K
530:N
523:N
515:N
498:)
476:1
474:N
472:S
468:2
466:N
464:S
420:N
378:B
374:A
359::
333:N
331:S
223:N
221:S
205:.
142:.
100:i
98:E
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.