136:
155:
86:
194:
78:
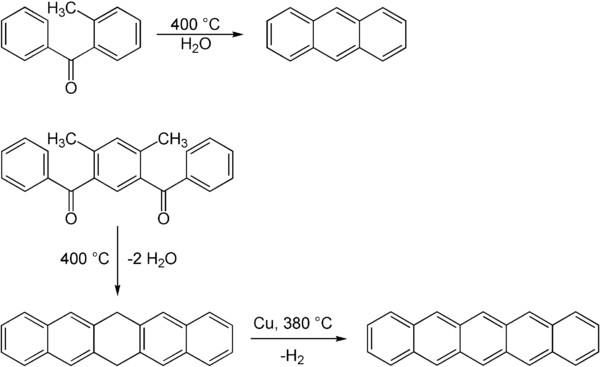
are also feasible. This reaction does not take place in a single step but leads first to dihydropentacene that is dehydrogenated in a second step with
321:
66:
The Elbs reaction enables the synthesis of condensed aromatic systems. As already demonstrated by Elbs in 1884 it is possible to obtain
54:. The reaction was published in 1884. Elbs however did not correctly interpret the reaction product due to a lack of knowledge about
354:
251:
371:
369:
G. M. Badger, B. J. Christie. (1956). "Polynuclear heterocyclic systems. Part X. The elbs reaction with heterocyclic ketones."
108:
The Elbs reaction is sometimes accompanied by elimination of substituents and can be unsuited for substituted polyaromatics.
85:
401:
116:
At least three plausible mechanisms for the Elbs reaction have been suggested. The first mechanism, suggested by
143:
Alternatively, in the second mechanism, due to Cook, the methylated aromatic compound instead first undergoes a
98:
135:
406:
154:
193:
411:
148:
175:
129:
125:
313:
218:
183:
32:
151:
to give the same intermediate, which then similarly undergoes a -hydride shift and dehydration.
350:
317:
308:
278:
102:
379:
342:
287:
260:
227:
24:
182:
derivative was published. The expected linear product was not obtained due to a change in
144:
51:
395:
309:
Organische Chemie: Grundlagen, Stoffklassen, Reaktionen, Konzepte, Molekülstrukturen
346:
187:
117:
43:
39:
291:
121:
71:
55:
231:
67:
264:
246:
213:
179:
163:
75:
47:
28:
249:. (1886) "Beiträge zur Kenntniss aromatischer Ketone. Erste Mittheilung."
383:
79:
35:
186:
after formation of the first intermediate which caused multiple
94:
97:
compounds required for this reaction can be obtained through a
46:. The reaction is named after its inventor, the German chemist
192:
153:
134:
84:
332:
330:
216:, Einar Larsen. (1884). "Ueber Paraxylylphenylketon."
178:
via the Elbs reaction. In 1956 an Elbs reaction of a
162:
A third mechanism has also been proposed, involving
339:Comprehensive Organic Name Reactions and Reagents
276:Fieser, Louis F. (1942). "The Elbs Reaction."
8:
306:Eberhard Breitmaier, Günther Jung (2005).
89:Elbs reaction to anthracite and pentacene
302:
300:
242:
240:
206:
7:
124:of the benzophenone, followed by a
174:It is also possible to synthesise
14:
132:then affords the polyaromatic.
74:. Larger aromatic systems like
347:10.1002/9780470638859.conrr213
1:
120:, begins with a heat-induced
292:10.1002/0471264180.or001.06
50:, also responsible for the
428:
341:. 2010. pp. 982–985.
312:(5th edition). Stuttgart:
197:Heterocyclic Elbs reaction
232:10.1002/cber.188401702247
128:to give the compound . A
265:10.1002/prac.18860330119
99:Friedel-Crafts acylation
219:Ber. Dtsch. Chem. Ges.
198:
176:heterocyclic compounds
159:
149:electrocyclic reaction
140:
90:
196:
157:
138:
88:
384:10.1039/JR9560003435
166:radical generation.
130:dehydration reaction
314:Georg Thieme Verlag
199:
184:reaction mechanism
160:
141:
139:Fieser's mechanism
91:
402:Organic reactions
337:"Elbs Reaction".
322:978-3-13-541505-5
103:aluminum chloride
16:Chemical reaction
419:
387:
367:
361:
360:
334:
325:
304:
295:
274:
268:
244:
235:
226:(2): 2847–2849,
211:
190:reaction steps.
158:Cook's mechanism
25:organic reaction
427:
426:
422:
421:
420:
418:
417:
416:
392:
391:
390:
368:
364:
357:
336:
335:
328:
305:
298:
275:
271:
252:J. Prakt. Chem.
245:
238:
212:
208:
204:
172:
147:followed by an
145:tautomerization
114:
82:as a catalyst.
64:
42:to a condensed
27:describing the
17:
12:
11:
5:
425:
423:
415:
414:
409:
407:Name reactions
404:
394:
393:
389:
388:
362:
355:
326:
296:
269:
259:(1): 180–188,
236:
205:
203:
200:
171:
168:
126:-hydride shift
113:
110:
63:
60:
52:Elbs oxidation
15:
13:
10:
9:
6:
4:
3:
2:
424:
413:
412:Benzophenones
410:
408:
405:
403:
400:
399:
397:
385:
381:
378:: 3435–3437,
377:
374:
373:
372:J. Chem. Soc.
366:
363:
358:
356:9780470638859
352:
348:
344:
340:
333:
331:
327:
323:
319:
315:
311:
310:
303:
301:
297:
293:
289:
285:
281:
280:
273:
270:
266:
262:
258:
255:(in German),
254:
253:
248:
243:
241:
237:
233:
229:
225:
222:(in German),
221:
220:
215:
210:
207:
201:
195:
191:
189:
185:
181:
177:
169:
167:
165:
156:
152:
150:
146:
137:
133:
131:
127:
123:
119:
111:
109:
106:
104:
100:
96:
87:
83:
81:
77:
73:
69:
61:
59:
57:
53:
49:
45:
41:
37:
34:
30:
26:
22:
21:Elbs reaction
375:
370:
365:
338:
307:
283:
277:
272:
256:
250:
223:
217:
209:
188:free radical
173:
161:
142:
115:
107:
92:
65:
44:polyaromatic
40:benzophenone
38:substituted
20:
18:
286:: 129-154,
279:Org. React.
122:cyclisation
72:dehydration
58:structure.
56:naphthalene
396:Categories
202:References
170:Variations
68:anthracene
214:Karl Elbs
180:thiophene
164:pyrolytic
112:Mechanism
76:pentacene
48:Karl Elbs
29:pyrolysis
70:through
247:K. Elbs
353:
320:
118:Fieser
80:copper
36:methyl
31:of an
23:is an
101:with
62:Scope
33:ortho
376:1956
351:ISBN
318:ISBN
95:acyl
93:The
19:The
380:doi
343:doi
288:doi
261:doi
228:doi
398::
349:.
329:^
316:,
299:^
282:,
257:33
239:^
224:17
105:.
386:.
382::
359:.
345::
324:.
294:.
290::
284:1
267:.
263::
234:.
230::
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.