291:
134:. At the same time, it consists of primary carbon particles and boasts a high degree of aggregation. Carbon black's grouping facilitates the formation of a conductive structure in plastics, rubbers and other composites. These characteristics predetermine electroconductive carbon black's primary area of application, i.e. electrical conductivity modification of nearly all types of plastic materials by adding a relatively low volume of carbon black. Such modifications can be used for numerous purposes, from establishing antistatic properties to adjusting polymer conductivity. Another valuable property of electroconductive carbon black is its excellent ability to absorb
341:
carbon black structure consists of branched chains with many secondarily created spaces in the aggregate. On the other hand, aggregate structure that is developed only a little represents smaller clusters of the spherical particles and thus also smaller spaces inside of the aggregate. The aggregate structure does not depend on the size of the particles. It has been established that particles of the same size can have aggregates with completely different structures. Generally speaking, carbon black that has a highly developed and complex structure is easier to disperse, has lower wettability, higher electric conductivity and higher viscosity.
368:
328:
307:
299:
190:
production principle lies in isolating carbon from the water using granulation petrol, where intensive homogenization causes the carbon to transform from its aqueous to an organic phase, i.e. transformation of the water-carbon suspension to petrol-carbon suspension in the form of carbonaceous granules. The carbonaceous granules are subsequently processed into a finished product – carbonaceous substrate. The two mediums’ proportionality primarily depends on carbon content and physical and chemical properties of the carbon black water and granulation petrol.
287:(aggregate) into chains or clusters. These aggregates then form the smallest carbon black units. They define what is known as the primary structure. Primary structure is characterized by the following: size of the primary particles, surface size, size and structure of the aggregates or chemical “composition” of the carbon black surface. These characteristics determine other carbon black features, such as adsorption properties, density, electrical conductivity, and absorption of UV radiation or visible light.
324:. It has been ascertained that the smaller the particles, the greater the size of their surface. Carbon black particle size is between 10 and 100 nm, while the surface particle size is between 20 and 1,500 m/g. Generally speaking, small carbon black particles with a high surface area are darker, have higher viscosity and lower wettability, are harder to disperse, retain greater conductivity and absorb UV radiation well.
263:
271:
22:
340:
Another significant characteristic of carbon black is its structure and the size of its aggregates. The size and complexity of the aggregate structure is determined by the volume of the carbon black spherical primary particles, which cluster together during the production process. The highly complex
189:
During the respective process, carbon black is captured into water through the method of scrubbing, thus creating carbon black water. The generated carbon black water with 7–15 g/L of carbon black is further processed at the production facility into several types of carbonaceous substrates. The main
150:
of what is referred to as partial oxidation, a process during which crude oil residues, such as vacuum residues from crude oil distillation or residues from the thermic cracking process, split due to the effects of the mixture of oxygen and water steam under high temperatures around 1,300 °C.
126:
or thermo-conductive characteristics of plastic materials and rubbers. By virtue of its pigmentation capabilities, it is also used for the production of special printing inks, paints and varnishes. Thanks to its advanced porous structure, it is also used as a catalyst carrier, and its notable
286:
Carbon black is essentially formed out of primary carbon, but its structure is much less arranged than that of, for example, graphite. Carbon black exists in the form of discrete particles, however, during the production process its spherical particles, also called primary particles, cluster
319:
The most important characteristic of carbon black is the size of its primary particles and the related surface area. The size of the primary particles describes the size of individual spherical particles that form a primary structure. The size of individual particles is determined using an
417:, such as Bare Conductive's Electric Paint, which can be used as a painted resistor element, a capacitive electrode, or as a conductor in designs that tolerate high resistivity when making circuits, as well as being painted onto gloves to allow people to use touchscreens in cold weather.
185:
formed from sulfurous compounds. Carbon black is formed as an undesired byproduct. The amount of carbon black grows as the injection's molecular weight increases. Methane gasification produces approx. 0.02% mass, crude oil residue gasification approx. 1-3% mass.
805:
Gubbels, F.; Jerome, R.; Teyssie, Ph; Vanlathem, E.; Deltour, R.; Calderone, A.; Parente, V.; Bredas, J. L. (2002-05-01). "Selective
Localization of Carbon Black in Immiscible Polymer Blends: A Useful Tool To Design Electrical Conductive Composites".
361:
groups appear on carbon black surfaces. These groups, which contain oxygen, can significantly affect chemical reactivity, wettability, carbon black catalytic characteristics, electric conductivity, etc.
86:) in its complex configuration, colloid dimensions and quasi-graphitic structure. Carbon black's purity and composition are practically free of inorganic pollutants and extractable organic substances.
495:
Pantea, Dana; Darmstadt, Hans; Kaliaguine, Serge; Roy, Christian (2003-07-15). "Electrical conductivity of conductive carbon blacks: influence of surface chemistry and topology".
114:
Carbon black can be characterized as a substance with over 97% amorphous carbon content. It is used extensively in many areas of industrial chemistry. It is often used in the
138:
on the visible spectrum, i.e. as a UV stabilizer for plastic materials, pigment in printer inks, paints and varnishes, or for coloring plastics, rubbers and sealants.
833:
Gubbels, R.; and collective (1994). "Selective
Localization of Carbon Black in Immiscible Polymer Blends: A Useful Tool To Design Electrical Conductive Composites".
130:
Carbon black predominantly includes a conductive type of carbon, which combines an extremely high specific surface and extensively developed structure –
703:
Herink, Tomáš; Raška, Stanislav; Nečesaný, František; Kubal, Petr (2008). "Application possibilities of the
Chezacarb soot produced at Unipetrol RPA".
98:– a specially produced type of carbon using the process of incomplete combustion with restricted oxygen access. The article addresses this type of
110:– auxiliary fuel (coal, hydrocarbons, crude oil) combustion product, which is considered to be a hazardous substance with carcinogenic properties.
770:
Tchoudakov, R.; Breuer, O.; Narkis, M.; Siegmann, A. (1996). "Conductive polymer blends with low carbon black loading: Polypropylene/polyamide".
674:
290:
721:
349:
Yet another noteworthy characteristic is carbon black's chemical surface composition. Chemisorbed complexes containing oxygen, such as
894:
367:
919:
899:
74:, carbon black is spherical in shape and arranged into aggregates and agglomerates. It differs from other carbon forms (
327:
306:
755:
Wiley, John (1997). "Agglomeration and electrical percolation behavior of carbon black dispersed in epoxy resin".
298:
924:
119:
611:
Bourrat, Xavier (1993-01-01). "Electrically conductive grades of carbon black: Structure and properties".
551:
Probst, Nicolaus; Grivei, Eusebiu (2002-02-01). "Structure and electrical properties of carbon black".
127:
sorption attributes are used for, in example, catching gaseous pollutants at waste incinerator plants.
842:
620:
560:
504:
51:
895:
http://www.unipetrolrpa.cz/CS/NabidkaProduktu/petrochemicke-produkty/chezacarb/Stranky/default.aspx
321:
32:
939:
920:
https://polymerchemistry.nouryon.com/products-applications/polymer-additives/conductive-blacks
900:
https://web.archive.org/web/20190209124223/https://www.orioncarbons.com/specialty-carbon-black
787:
670:
636:
576:
520:
457:
431:
182:
729:
438:(Ketjenblack), TIMCAL (Ensaco), BIRLA CARBON (Conductex) and ORION ENGINEERED CARBONS (XPB).
154:
Partial oxidation of various raw materials always creates a gaseous mixture containing CO, CO
850:
815:
779:
628:
568:
512:
174:
123:
944:
350:
846:
624:
564:
508:
405:
572:
516:
933:
632:
131:
914:
447:
135:
94:
83:
791:
640:
580:
524:
426:
Some of the world's main producers of electroconductive carbon black include
462:
452:
435:
427:
270:
147:
41:
925:
http://www.cabotcorp.com/solutions/applications/plastics/conductive-and-esd
21:
262:
79:
854:
819:
412:
390:
358:
354:
294:
Incorporated electroconductive carbon black in the polypropylene matrix
277:
Physical and chemical characteristics of electroconductive carbon black
167:
115:
75:
783:
261:
99:
71:
889:
398:
326:
305:
297:
289:
106:
374:
Picture: Diagram of carbon black structure and texture creation
904:
15:
909:
118:
and rubber manufacturing industries, where it improves
46:
36:
757:
Journal of
Applied Science: Applied Polymer Science
669:. Zlín: Czech Association of Industrial Chemistry.
555:. Third International Conference on Carbon Black.
915:http://www.imerys-graphite-and-carbon.com/brands/
722:"petroleum.cz, Výroba vodíku parciální oxidací"
89:A distinction is made between these two terms:
871:"World Market for Conductive Carbon Blacks".
8:
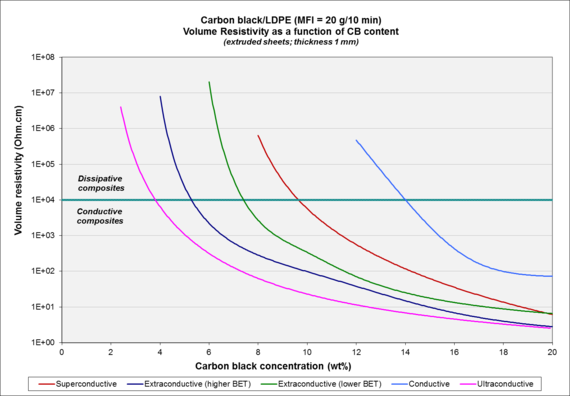
331:Electroconductive carbon black – Structure 3
310:Electroconductive carbon black – Structure 2
302:Electroconductive carbon black – Structure 1
269:
422:Electroconductive carbon black producers
197:
474:
194:Types of electroconductive carbon black
866:
864:
7:
750:
748:
746:
716:
714:
698:
696:
694:
692:
690:
688:
686:
660:
658:
656:
654:
652:
650:
606:
604:
602:
600:
598:
596:
594:
592:
590:
546:
544:
542:
540:
538:
536:
534:
490:
488:
486:
484:
482:
480:
478:
705:Chemical Sheets: Chemical Industry
14:
890:http://chezacarbcarbonblack.com/
366:
20:
1:
573:10.1016/S0008-6223(01)00174-9
517:10.1016/S0169-4332(03)00550-6
434:(Vulcan), DEGUSSA (Printex),
633:10.1016/0008-6223(93)90034-8
905:http://specialtyblacks.com/
772:Polymer Engineering Journal
961:
393:(compounds, concentrates)
146:Carbon black begins as a
910:https://birlacarbon.com/
665:Kliment, Josef (2008).
497:Applied Surface Science
345:Surface characteristics
120:electrical conductivity
35:, as no other articles
873:Notch Consulting Group
384:Composite applications
332:
311:
303:
295:
266:
236:extraconductive (cca)
225:superconductive (cca)
330:
309:
301:
293:
265:
855:10.1021/ma00085a049
847:1994MaMol..27.1972G
820:10.1021/ma00085a049
625:1993Carbo..31..287B
565:2002Carbo..40..201P
509:2003ApSS..217..181P
322:electron microscope
199:
70:Made up of primary
333:
312:
304:
296:
267:
198:
54:for suggestions.
44:to this page from
784:10.1002/pen.10528
778:(10): 1336–1346.
676:978-80-02-02004-2
458:Cabot Corporation
432:CABOT Corporation
257:
256:
214:conductive (cca)
68:
67:
952:
877:
876:
868:
859:
858:
841:(7): 1972–1974.
830:
824:
823:
814:(7): 1972–1974.
802:
796:
795:
767:
761:
760:
752:
741:
740:
738:
737:
728:. Archived from
726:www.petroleum.cz
718:
709:
708:
700:
681:
680:
662:
645:
644:
608:
585:
584:
548:
529:
528:
492:
370:
273:
247:ultraconductive
200:
63:
60:
49:
47:related articles
24:
16:
960:
959:
955:
954:
953:
951:
950:
949:
930:
929:
886:
881:
880:
870:
869:
862:
832:
831:
827:
804:
803:
799:
769:
768:
764:
754:
753:
744:
735:
733:
720:
719:
712:
702:
701:
684:
677:
664:
663:
648:
610:
609:
588:
550:
549:
532:
494:
493:
476:
471:
444:
424:
415:, paints, glues
381:
365:
347:
338:
317:
284:
279:
259:
196:
178:
171:
165:
161:
157:
144:
124:electromagnetic
64:
58:
55:
45:
42:introduce links
25:
12:
11:
5:
958:
956:
948:
947:
942:
932:
931:
928:
927:
922:
917:
912:
907:
902:
897:
892:
885:
884:External links
882:
879:
878:
860:
835:Macromolecules
825:
808:Macromolecules
797:
762:
742:
710:
682:
675:
646:
619:(2): 287–302.
586:
559:(2): 201–205.
530:
503:(1): 181–193.
473:
472:
470:
467:
466:
465:
460:
455:
450:
443:
440:
423:
420:
419:
418:
409:
402:
395:
380:
377:
346:
343:
337:
334:
316:
313:
283:
280:
278:
275:
255:
254:
251:
248:
244:
243:
240:
237:
233:
232:
229:
226:
222:
221:
218:
215:
211:
210:
207:
204:
195:
192:
176:
169:
163:
159:
155:
143:
140:
112:
111:
103:
66:
65:
52:Find link tool
28:
26:
19:
13:
10:
9:
6:
4:
3:
2:
957:
946:
943:
941:
938:
937:
935:
926:
923:
921:
918:
916:
913:
911:
908:
906:
903:
901:
898:
896:
893:
891:
888:
887:
883:
874:
867:
865:
861:
856:
852:
848:
844:
840:
836:
829:
826:
821:
817:
813:
809:
801:
798:
793:
789:
785:
781:
777:
773:
766:
763:
758:
751:
749:
747:
743:
732:on 2019-03-11
731:
727:
723:
717:
715:
711:
706:
699:
697:
695:
693:
691:
689:
687:
683:
678:
672:
668:
661:
659:
657:
655:
653:
651:
647:
642:
638:
634:
630:
626:
622:
618:
614:
607:
605:
603:
601:
599:
597:
595:
593:
591:
587:
582:
578:
574:
570:
566:
562:
558:
554:
547:
545:
543:
541:
539:
537:
535:
531:
526:
522:
518:
514:
510:
506:
502:
498:
491:
489:
487:
485:
483:
481:
479:
475:
468:
464:
461:
459:
456:
454:
451:
449:
446:
445:
441:
439:
437:
433:
430:(Chezacarb),
429:
421:
416:
414:
410:
408:
407:
403:
401:
400:
396:
394:
392:
388:
387:
386:
385:
378:
376:
375:
371:
369:
363:
360:
356:
352:
344:
342:
335:
329:
325:
323:
314:
308:
300:
292:
288:
281:
276:
274:
272:
264:
260:
252:
249:
246:
245:
241:
238:
235:
234:
230:
227:
224:
223:
219:
216:
213:
212:
208:
205:
203:Carbon black
202:
201:
193:
191:
187:
184:
180:
172:
152:
149:
141:
139:
137:
133:
132:microporosity
128:
125:
121:
117:
109:
108:
104:
101:
97:
96:
92:
91:
90:
87:
85:
81:
77:
73:
62:
59:November 2023
53:
48:
43:
39:
38:
34:
29:This article
27:
23:
18:
17:
872:
838:
834:
828:
811:
807:
800:
775:
771:
765:
756:
734:. Retrieved
730:the original
725:
704:
667:Carbon Black
666:
616:
612:
556:
552:
500:
496:
448:Carbon black
425:
411:
404:
397:
389:
383:
382:
379:Applications
373:
372:
364:
348:
339:
318:
285:
268:
258:
188:
153:
145:
136:UV radiation
129:
113:
105:
95:Carbon black
93:
88:
69:
56:
30:
934:Categories
736:2019-02-11
469:References
436:AKZO-Nobel
351:carboxylic
250:1300–1400
142:Production
50:; try the
37:link to it
940:Chemistry
792:0032-3888
641:0008-6223
581:0008-6223
525:0169-4332
463:AkzoNobel
453:Unipetrol
428:UNIPETROL
413:varnishes
282:Structure
253:> 600
239:800–1200
148:byproduct
40:. Please
442:See also
391:polymers
359:phenolic
355:quinonic
242:300–400
80:graphite
875:. 2012.
843:Bibcode
621:Bibcode
561:Bibcode
505:Bibcode
406:rubbers
336:Texture
315:Surface
116:plastic
76:diamond
945:Carbon
790:
707:: 102.
673:
639:
613:Carbon
579:
553:Carbon
523:
399:resins
100:carbon
72:carbon
33:orphan
31:is an
788:ISSN
671:ISBN
637:ISSN
577:ISSN
521:ISSN
231:180
228:250
220:100
217:120
209:OAN
206:BET
181:and
173:and
162:O, H
122:and
107:Soot
84:coke
851:doi
816:doi
780:doi
629:doi
569:doi
513:doi
501:217
357:or
183:COS
158:, H
936::
863:^
849:.
839:27
837:.
812:27
810:.
786:.
776:36
774:.
745:^
724:.
713:^
685:^
649:^
635:.
627:.
617:31
615:.
589:^
575:.
567:.
557:40
533:^
519:.
511:.
499:.
477:^
353:,
168:CH
166:,
82:,
78:,
857:.
853::
845::
822:.
818::
794:.
782::
759:.
739:.
679:.
643:.
631::
623::
583:.
571::
563::
527:.
515::
507::
179:S
177:2
175:H
170:4
164:2
160:2
156:2
102:.
61:)
57:(
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.