241:
threshold barrier for an elementary reaction. First, it is often unclear as to whether or not reaction does proceed in one step; threshold barriers that are averaged out over all elementary steps have little theoretical value. Second, even if the reaction being studied is elementary, a spectrum of individual collisions contributes to rate constants obtained from bulk ('bulb') experiments involving billions of molecules, with many different reactant collision geometries and angles, different translational and (possibly) vibrational energies—all of which may lead to different microscopic reaction rates.
439:. However, instead of modeling the temperature dependence of reaction rate phenomenologically, the Eyring equation models individual elementary steps of a reaction. Thus, for a multistep process, there is no straightforward relationship between the two models. Nevertheless, the functional forms of the Arrhenius and Eyring equations are similar, and for a one-step process, simple and chemically meaningful correspondences can be drawn between Arrhenius and Eyring parameters.
256:
268:
31:
1076:, where i, p and t refer respectively to initiation, propagation and termination steps. The propagation step normally has a very small activation energy, so that the overall value is negative if the activation energy for termination is larger than that for initiation. The normal range of overall activation energies for cationic polymerization varies from
324:. A catalyst increases the rate of reaction without being consumed in the reaction. In addition, the catalyst lowers the activation energy, but it does not change the energies of the original reactants or products, and so does not change equilibrium. Rather, the reactant energy and the product energy remain the same and only the
89:. For a chemical reaction to proceed at a reasonable rate, the temperature of the system should be high enough such that there exists an appreciable number of molecules with translational energy equal to or greater than the activation energy. The term "activation energy" was introduced in 1889 by the Swedish scientist
352:
binding energy released assists in achieving the unstable transition state. Reactions without catalysts need a higher input of energy to achieve the transition state. Non-catalyzed reactions do not have free energy available from active site stabilizing interactions, such as catalytic enzyme reactions.
351:
energy. A chemical reaction is able to manufacture a high-energy transition state molecule more readily when there is a stabilizing fit within the active site of a catalyst. The binding energy of a reaction is this energy released when favorable interactions between substrate and catalyst occur. The
655:
Elementary reactions exhibiting negative activation energies are typically barrierless reactions, in which the reaction proceeding relies on the capture of the molecules in a potential well. Increasing the temperature leads to a reduced probability of the colliding molecules capturing one another
240:
At a more advanced level, the net
Arrhenius activation energy term from the Arrhenius equation is best regarded as an experimentally determined parameter that indicates the sensitivity of the reaction rate to temperature. There are two objections to associating this activation energy with the
1301:
Terracciano, Anthony C; De
Oliveira, Samuel; Vazquez-Molina, Demetrius; Uribe-Romo, Fernando J; Vasu, Subith S; Orlovskaya, Nina (2017). "Effect of catalytically active Ce 0.8 Gd 0.2 O 1.9 coating on the heterogeneous combustion of methane within MgO stabilized ZrO 2 porous ceramics".
1367:
1608:
347:). Specific and favorable bonding occurs within the active site until the substrate forms to become the high-energy transition state. Forming the transition state is more favorable with the catalyst because the favorable stabilizing interactions within the active site
1359:
312:. The highest energy position (peak position) represents the transition state. With the catalyst, the energy required to enter transition state decreases, thereby decreasing the energy required to initiate the reaction.
123:
gives the quantitative basis of the relationship between the activation energy and the rate at which a reaction proceeds. From the equation, the activation energy can be found through the relation
190:
1074:
1002:
754:
645:
with increasing temperature. When following an approximately exponential relationship so the rate constant can still be fit to an
Arrhenius expression, this results in a negative value of
1239:
Wang, Jenqdaw; Raj, Rishi (1990). "Estimate of the
Activation Energies for Boundary Diffusion from Rate-Controlled Sintering of Pure Alumina, and Alumina Doped with Zirconia or Titania".
406:
holds. In transition state theory, a more sophisticated model of the relationship between reaction rates and the transition state, a superficially similar mathematical relationship, the
921:
829:
1266:
Kiraci, A; Yurtseven, H (2012). "Temperature
Dependence of the Raman Frequency, Damping Constant and the Activation Energy of a Soft-Optic Mode in Ferroelectric Barium Titanate".
1183:
298:
663:
Some multistep reactions can also have apparent negative activation energies. For example, the overall rate constant k for a two-step reaction A ⇌ B, B → C is given by k = k
331:
A catalyst is able to reduce the activation energy by forming a transition state in a more favorable manner. Catalysts, by nature, create a more "comfortable" fit for the
339:
of a catalyst. This energy is known as
Binding Energy. Upon binding to a catalyst, substrates partake in numerous stabilizing forces while within the active site (e.g.
1337:
656:(with more glancing collisions not leading to reaction as the higher momentum carries the colliding particles out of the potential well), expressed as a reaction
1740:
1530:
Mozurkewich, Michael; Benson, Sidney (1984). "Negative activation energies and curved
Arrhenius plots. 1. Theory of reactions over potential wells".
1217:
472:
are the
Boltzmann and Planck constants, respectively. Although the equations look similar, it is important to note that the Gibbs energy contains an
660:
that decreases with increasing temperature. Such a situation no longer leads itself to direct interpretations as the height of a potential barrier.
237:
can be evaluated from the variation in reaction rate coefficients as a function of temperature (within the validity of the
Arrhenius equation).
1398:
1952:
621:
The total free energy change of a reaction is independent of the activation energy however. Physical and chemical reactions can be either
335:
of a reaction to progress to a transition state. This is possible due to a release of energy that occurs when the substrate binds to the
1829:
1786:
1191:
1129:
126:
2093:
476:
term in addition to the enthalpic one. In the
Arrhenius equation, this entropic term is accounted for by the pre-exponential factor
2207:
1733:
1706:
1681:
1656:
1591:
1566:
1514:
1486:
1453:
42:. The blue flame sustains itself after the sparks stop because the continued combustion of the flame is now energetically favorable.
2001:
1996:
1806:
1167:
2161:
2166:
1089:
301:
1422:
1329:
2192:
1726:
1012:
1018:
2187:
2131:
1821:
926:
697:
1858:
1758:
687:
increases, so that k actually decreases with temperature corresponding to a negative observed activation energy.
834:
2088:
1104:
1099:
572:
for a reaction that proceeds over several hours at room temperature. Due to the relatively small magnitude of
1330:"General Chemistry Online: FAQ: Chemical change: What are some examples of reactions that involve catalysts?"
2136:
1937:
1008:
657:
361:
759:
1891:
1213:
223:
199:
2121:
2053:
1911:
1901:
481:
332:
2116:
1844:
274:
70:
2202:
2126:
2058:
2043:
1986:
1478:
630:
626:
378:
344:
309:
260:
86:
2197:
2151:
1921:
1750:
1283:
1109:
622:
367:
120:
114:
47:
480:. More specifically, we can write the Gibbs free energy of activation in terms of enthalpy and
320:; a catalyst composed only of protein and (if applicable) small molecule cofactors is termed an
2146:
2103:
2048:
1967:
1947:
1883:
1702:
1677:
1652:
1587:
1562:
1510:
1482:
1449:
1394:
1163:
1094:
561:. For a one-step unimolecular process whose half-life at room temperature is about 2 hours, Δ
74:
55:
1184:"Activation Energy and the Arrhenius Equation – Introductory Chemistry- 1st Canadian Edition"
1011:
reactions have negative activation energies so that the rate decreases with temperature. For
2078:
2027:
1981:
1539:
1470:
1311:
1275:
1248:
458:
381:
102:
90:
82:
27:
Minimum amount of energy that must be provided for a system to undergo a reaction or process
1137:
633:
of a reaction. The overall reaction energy change is not altered by the activation energy.
2156:
2068:
2017:
407:
316:
A substance that modifies the transition state to lower the activation energy is termed a
1471:
1863:
1852:
1315:
1252:
615:
66:
2181:
2111:
2083:
1991:
1942:
1916:
1287:
340:
39:
602:
The enthalpy, entropy and Gibbs energy of activation are more correctly written as Δ
17:
2063:
1869:
1776:
1766:
691:
450:
255:
207:
1493:... but we shall omit the standard state sign to avoid overburdening the notation.
1279:
2022:
1957:
831:
with a negative activation energy. This is explained by the two-step mechanism:
336:
267:
35:
1414:
2073:
1718:
250:
30:
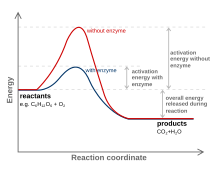
54:
is the minimum amount of energy that must be available to reactants for a
679:
is the equilibrium constant of the rapid first step. In some reactions, K
565:
is approximately 23 kcal/mol. This is also the roughly the magnitude of
317:
1543:
557:
is temperature independent, while here, there is a linear dependence on
73:(kcal/mol). Activation energy can be thought of as the magnitude of the
473:
1444:
Steinfeld, Jeffrey I.; Francisco, Joseph S.; Hase, William L. (1999).
618:. However, some authors omit the o in order to simplify the notation.
321:
215:
78:
599:
are often conflated and all referred to as the "activation energy".
1421:. IUPAC (International Union of Pure and Applied Chemistry). 2019.
584:
at ordinary temperatures for most reactions, in sloppy discourse,
1976:
254:
29:
614:
respectively, where the o indicates a quantity evaluated between
109:
Temperature dependence and the relation to the Arrhenius equation
1701:(2nd ed.). Blackie (USA: Chapman & Hall+). p. 88.
675:
is the rate constant of the rate-limiting slow second step and K
1722:
101:
Although less commonly used, activation energy also applies to
1796:
38:
provide the activation energy to initiate combustion in this
1393:. New York, NY: WH Freeman and Company. pp. 240–244.
990:
970:
954:
941:
909:
896:
742:
722:
457:
to denote the Gibbs energy of activation to achieve the
34:
The sparks created by striking steel against a piece of
553:
hold. Note, however, that in Arrhenius theory proper,
410:, is used to describe the rate constant of a reaction:
1022:
867:
1021:
929:
837:
762:
700:
277:
129:
503:. Then, for a unimolecular, one-step reaction, the
2102:
2036:
2010:
1966:
1930:
1882:
1843:
1820:
1757:
1699:
Polymers: Chemistry and Physics of Modern Materials
308:) with and without a catalyst, plotted against the
1651:(3rd ed.). John Wiley and Sons. p. 316.
1068:
996:
915:
823:
748:
629:, but the activation energy is not related to the
292:
184:
1214:"Energy in a Modern Society: XIV. Nuclear energy"
875:
874:
857:
856:
77:(sometimes called the energy barrier) separating
185:{\displaystyle k=Ae^{{-E_{\textrm {a}}}/{(RT)}}}
1624:The overall activation energy is negative if Ea
1419:IUPAC Gold Book (2nd edition, on-line version)
1069:{\displaystyle \textstyle E=E_{i}+E_{p}-E_{t}}
683:decreases with temperature more rapidly than k
1734:
997:{\displaystyle {\ce {N2O2 + O2 -> 2 NO2}}}
749:{\displaystyle {\ce {2 NO + O2 -> 2 NO2}}}
8:
1586:(8th ed.). W. H. Freeman. p. 822.
1448:(2nd ed.). Prentice Hall. p. 301.
356:Relationship with Gibbs energy of activation
271:The relationship between activation energy (
85:surface pertaining to the initial and final
1676:. Pearson. Benjamin-Cummings. p. *34.
1505:Laidler, Keith J.; Meiser, John H. (1982).
916:{\displaystyle {\ce {2 NO <=> N2O2}}}
1741:
1727:
1719:
1647:Moore, John W.; Pearson, Ralph G. (1981).
449:, the Eyring equation uses the concept of
377:) is used to describe the energy required
1559:Chemical kinetics and reaction mechanisms
1160:Chemical Kinetics and Reaction Mechanisms
1059:
1046:
1033:
1020:
989:
984:
979:
969:
964:
953:
948:
940:
935:
930:
928:
908:
903:
895:
890:
876:
869:
868:
866:
858:
851:
849:
848:
846:
842:
838:
836:
809:
804:
803:
798:
792:
779:
778:
772:
761:
741:
736:
731:
721:
716:
705:
701:
699:
283:
282:
276:
166:
161:
153:
152:
144:
143:
128:
1360:"The Arrhenius Law: Activation Energies"
266:
214:is the absolute temperature (usually in
1582:Atkins, Peter; de Paula, Julio (2006).
1469:Atkins, Peter; de Paula, Julio (2006).
1241:Journal of the American Ceramic Society
1121:
850:
1767:Unimolecular nucleophilic substitution
1609:"3.2.2: Pre-equilibrium Approximation"
1370:from the original on February 18, 2017
105:and various other physical phenomena.
1777:Bimolecular nucleophilic substitution
1477:(8th ed.). W.H.Freeman. p.
1425:from the original on 21 February 2020
824:{\displaystyle v=k\,\left^{2}\,\left}
7:
1672:Engel, Thomas; Reid, Philip (2006).
1830:Electrophilic aromatic substitution
1607:Kadir, Tamara (10 September 2020).
1015:, the overall activation energy is
384:, and the exponential relationship
1797:Nucleophilic internal substitution
1787:Nucleophilic aromatic substitution
1509:. Benjamin/Cummings. p. 381.
1316:10.1016/j.combustflame.2017.02.019
1253:10.1111/j.1151-2916.1990.tb05175.x
1220:from the original on 22 March 2019
806:
783:
780:
25:
1216:(Course). Ohio State University.
694:which is a termolecular reaction
641:In some cases, rates of reaction
58:to occur. The activation energy (
1953:Lindemann–Hinshelwood mechanism
1340:from the original on 2016-11-29
690:An example is the oxidation of
293:{\displaystyle E_{\textrm {a}}}
259:Example of an enzyme-catalysed
65:) of a reaction is measured in
2002:Outer sphere electron transfer
1997:Inner sphere electron transfer
1807:Nucleophilic acyl substitution
1446:Chemical Kinetics and Dynamics
973:
877:
852:
725:
370:, the term activation energy (
176:
167:
1:
2167:Diffusion-controlled reaction
1212:Kagan, Harris; Barrett, Tom.
1090:Activation energy asymptotics
1561:. McGraw-Hill. p. 121.
1391:Biochemistry - Ninth Edition
1280:10.1080/00150193.2012.707592
1822:Electrophilic substitutions
1557:Espenson, James H. (1981).
1013:chain-growth polymerization
2224:
2132:Energy profile (chemistry)
2094:More O'Ferrall–Jencks plot
1759:Nucleophilic substitutions
1584:Atkins' Physical Chemistry
1473:Atkins' Physical Chemistry
637:Negative activation energy
359:
248:
112:
2162:Michaelis–Menten kinetics
224:reaction rate coefficient
2208:Biochemistry terminology
2089:Potential energy surface
1968:Electron/Proton transfer
1853:Unimolecular elimination
1697:Cowie, J. M. G. (1991).
1415:"Enthalpy of activation"
1158:Espenson, James (1995).
1105:Autoignition temperature
1100:Mean kinetic temperature
2137:Transition state theory
1938:Intramolecular reaction
1864:Bimolecular elimination
1009:cationic polymerization
362:Transition state theory
226:. Even without knowing
1931:Unimolecular reactions
1892:Electrophilic addition
1649:Kinetics and mechanism
1070:
998:
917:
825:
750:
442:Instead of also using
328:is altered (lowered).
313:
294:
264:
200:pre-exponential factor
186:
43:
2122:Rate-determining step
2054:Reactive intermediate
1912:Free-radical addition
1902:Nucleophilic addition
1845:Elimination reactions
1389:Berg, Jeremy (2019).
1334:antoine.frostburg.edu
1071:
999:
918:
826:
751:
482:entropy of activation
295:
270:
258:
187:
71:kilocalories per mole
33:
2117:Equilibrium constant
1364:Chemistry LibreTexts
1304:Combustion and Flame
1078:40 to 60 kJ/mol
1019:
927:
835:
760:
698:
345:van der Waals forces
302:enthalpy of reaction
275:
127:
18:Energy of activation
2193:Reaction mechanisms
2127:Reaction coordinate
2059:Radical (chemistry)
2044:Elementary reaction
1987:Grotthuss mechanism
1751:reaction mechanisms
1544:10.1021/j150669a073
1130:"Activation Energy"
992:
972:
956:
943:
911:
898:
863:
744:
724:
461:. In the equation,
310:reaction coordinate
87:thermodynamic state
67:kilojoules per mole
50:of reaction rates,
2152:Arrhenius equation
1922:Oxidative addition
1884:Addition reactions
1674:Physical Chemistry
1507:Physical Chemistry
1110:Quantum tunnelling
1066:
1065:
994:
980:
960:
944:
931:
913:
899:
886:
882:
821:
756:. The rate law is
746:
732:
712:
368:Arrhenius equation
314:
290:
265:
202:for the reaction,
182:
121:Arrhenius equation
115:Arrhenius equation
44:
2188:Chemical kinetics
2175:
2174:
2147:Activated complex
2142:Activation energy
2104:Chemical kinetics
2049:Reaction dynamics
1948:Photodissociation
1538:(25): 6429–6435.
1400:978-1-319-11467-1
1095:Chemical kinetics
983:
963:
947:
934:
902:
889:
884:
845:
735:
715:
708:
326:activation energy
286:
206:is the universal
156:
103:nuclear reactions
75:potential barrier
56:chemical reaction
52:activation energy
16:(Redirected from
2215:
2079:Collision theory
2028:Matrix isolation
1982:Harpoon reaction
1859:E1cB-elimination
1743:
1736:
1729:
1720:
1713:
1712:
1694:
1688:
1687:
1669:
1663:
1662:
1644:
1638:
1637:
1621:
1619:
1613:Chem Libre Texts
1604:
1598:
1597:
1579:
1573:
1572:
1554:
1548:
1547:
1527:
1521:
1520:
1502:
1496:
1495:
1476:
1466:
1460:
1459:
1441:
1435:
1434:
1432:
1430:
1411:
1405:
1404:
1386:
1380:
1379:
1377:
1375:
1355:
1349:
1348:
1346:
1345:
1326:
1320:
1319:
1298:
1292:
1291:
1263:
1257:
1256:
1236:
1230:
1229:
1227:
1225:
1209:
1203:
1202:
1200:
1199:
1190:. Archived from
1180:
1174:
1173:
1155:
1149:
1148:
1146:
1145:
1136:. Archived from
1134:www.chem.fsu.edu
1126:
1079:
1075:
1073:
1072:
1067:
1064:
1063:
1051:
1050:
1038:
1037:
1003:
1001:
1000:
995:
993:
991:
988:
981:
971:
968:
961:
955:
952:
945:
942:
939:
932:
922:
920:
919:
914:
912:
910:
907:
900:
897:
894:
887:
885:
883:
881:
880:
873:
865:
864:
862:
855:
847:
843:
830:
828:
827:
822:
820:
816:
815:
814:
813:
797:
796:
791:
787:
786:
755:
753:
752:
747:
745:
743:
740:
733:
723:
720:
713:
706:
552:
523:
502:
459:transition state
453:and the symbol Δ
438:
405:
382:transition state
341:hydrogen bonding
299:
297:
296:
291:
289:
288:
287:
284:
191:
189:
188:
183:
181:
180:
179:
165:
160:
159:
158:
157:
154:
91:Svante Arrhenius
83:potential energy
21:
2223:
2222:
2218:
2217:
2216:
2214:
2213:
2212:
2178:
2177:
2176:
2171:
2157:Eyring equation
2098:
2069:Stereochemistry
2032:
2018:Solvent effects
2006:
1962:
1926:
1907:
1897:
1878:
1873:
1839:
1835:
1816:
1812:
1802:
1792:
1782:
1772:
1753:
1747:
1717:
1716:
1709:
1696:
1695:
1691:
1684:
1671:
1670:
1666:
1659:
1646:
1645:
1641:
1635:
1631:
1627:
1617:
1615:
1606:
1605:
1601:
1594:
1581:
1580:
1576:
1569:
1556:
1555:
1551:
1529:
1528:
1524:
1517:
1504:
1503:
1499:
1489:
1468:
1467:
1463:
1456:
1443:
1442:
1438:
1428:
1426:
1413:
1412:
1408:
1401:
1388:
1387:
1383:
1373:
1371:
1357:
1356:
1352:
1343:
1341:
1328:
1327:
1323:
1300:
1299:
1295:
1265:
1264:
1260:
1238:
1237:
1233:
1223:
1221:
1211:
1210:
1206:
1197:
1195:
1182:
1181:
1177:
1170:
1162:. McGraw-Hill.
1157:
1156:
1152:
1143:
1141:
1128:
1127:
1123:
1118:
1086:
1077:
1055:
1042:
1029:
1017:
1016:
925:
924:
833:
832:
805:
799:
774:
773:
758:
757:
696:
695:
686:
682:
678:
674:
670:
666:
651:
639:
616:standard states
590:
571:
535:
525:
514:
508:
485:
467:
448:
421:
411:
408:Eyring equation
399:
385:
376:
364:
358:
278:
273:
272:
253:
247:
236:
148:
139:
125:
124:
117:
111:
99:
64:
48:Arrhenius model
28:
23:
22:
15:
12:
11:
5:
2221:
2219:
2211:
2210:
2205:
2200:
2195:
2190:
2180:
2179:
2173:
2172:
2170:
2169:
2164:
2159:
2154:
2149:
2144:
2139:
2134:
2129:
2124:
2119:
2114:
2108:
2106:
2100:
2099:
2097:
2096:
2091:
2086:
2081:
2076:
2071:
2066:
2061:
2056:
2051:
2046:
2040:
2038:
2037:Related topics
2034:
2033:
2031:
2030:
2025:
2020:
2014:
2012:
2011:Medium effects
2008:
2007:
2005:
2004:
1999:
1994:
1989:
1984:
1979:
1973:
1971:
1964:
1963:
1961:
1960:
1955:
1950:
1945:
1940:
1934:
1932:
1928:
1927:
1925:
1924:
1919:
1914:
1909:
1905:
1899:
1895:
1888:
1886:
1880:
1879:
1877:
1876:
1871:
1867:
1861:
1856:
1849:
1847:
1841:
1840:
1838:
1837:
1833:
1826:
1824:
1818:
1817:
1815:
1814:
1810:
1804:
1800:
1794:
1790:
1784:
1780:
1774:
1770:
1763:
1761:
1755:
1754:
1748:
1746:
1745:
1738:
1731:
1723:
1715:
1714:
1707:
1689:
1682:
1664:
1657:
1639:
1633:
1629:
1625:
1599:
1592:
1574:
1567:
1549:
1522:
1515:
1497:
1487:
1461:
1454:
1436:
1406:
1399:
1381:
1358:Bui, Matthew.
1350:
1321:
1293:
1268:Ferroelectrics
1258:
1231:
1204:
1175:
1168:
1150:
1120:
1119:
1117:
1114:
1113:
1112:
1107:
1102:
1097:
1092:
1085:
1082:
1062:
1058:
1054:
1049:
1045:
1041:
1036:
1032:
1028:
1025:
987:
978:
975:
967:
959:
951:
938:
906:
893:
879:
872:
861:
854:
841:
819:
812:
808:
802:
795:
790:
785:
782:
777:
771:
768:
765:
739:
730:
727:
719:
711:
704:
684:
680:
676:
672:
668:
664:
649:
638:
635:
588:
569:
533:
512:
507:relationships
465:
446:
419:
397:
374:
360:Main article:
357:
354:
281:
249:Main article:
246:
243:
234:
178:
175:
172:
169:
164:
151:
147:
142:
138:
135:
132:
113:Main article:
110:
107:
98:
95:
62:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
2220:
2209:
2206:
2204:
2201:
2199:
2196:
2194:
2191:
2189:
2186:
2185:
2183:
2168:
2165:
2163:
2160:
2158:
2155:
2153:
2150:
2148:
2145:
2143:
2140:
2138:
2135:
2133:
2130:
2128:
2125:
2123:
2120:
2118:
2115:
2113:
2112:Rate equation
2110:
2109:
2107:
2105:
2101:
2095:
2092:
2090:
2087:
2085:
2084:Arrow pushing
2082:
2080:
2077:
2075:
2072:
2070:
2067:
2065:
2062:
2060:
2057:
2055:
2052:
2050:
2047:
2045:
2042:
2041:
2039:
2035:
2029:
2026:
2024:
2021:
2019:
2016:
2015:
2013:
2009:
2003:
2000:
1998:
1995:
1993:
1992:Marcus theory
1990:
1988:
1985:
1983:
1980:
1978:
1975:
1974:
1972:
1969:
1965:
1959:
1956:
1954:
1951:
1949:
1946:
1944:
1943:Isomerization
1941:
1939:
1936:
1935:
1933:
1929:
1923:
1920:
1918:
1917:Cycloaddition
1915:
1913:
1910:
1903:
1900:
1893:
1890:
1889:
1887:
1885:
1881:
1875:
1868:
1865:
1862:
1860:
1857:
1854:
1851:
1850:
1848:
1846:
1842:
1831:
1828:
1827:
1825:
1823:
1819:
1808:
1805:
1798:
1795:
1788:
1785:
1778:
1775:
1768:
1765:
1764:
1762:
1760:
1756:
1752:
1744:
1739:
1737:
1732:
1730:
1725:
1724:
1721:
1710:
1708:0-216-92980-6
1704:
1700:
1693:
1690:
1685:
1683:0-8053-3842-X
1679:
1675:
1668:
1665:
1660:
1658:0-471-03558-0
1654:
1650:
1643:
1640:
1636:
1614:
1610:
1603:
1600:
1595:
1593:0-7167-8759-8
1589:
1585:
1578:
1575:
1570:
1568:0-07-019667-2
1564:
1560:
1553:
1550:
1545:
1541:
1537:
1533:
1532:J. Phys. Chem
1526:
1523:
1518:
1516:0-8053-5682-7
1512:
1508:
1501:
1498:
1494:
1490:
1488:0-7167-8759-8
1484:
1480:
1475:
1474:
1465:
1462:
1457:
1455:0-13-737123-3
1451:
1447:
1440:
1437:
1424:
1420:
1416:
1410:
1407:
1402:
1396:
1392:
1385:
1382:
1369:
1365:
1361:
1354:
1351:
1339:
1335:
1331:
1325:
1322:
1317:
1313:
1309:
1305:
1297:
1294:
1289:
1285:
1281:
1277:
1273:
1269:
1262:
1259:
1254:
1250:
1246:
1242:
1235:
1232:
1219:
1215:
1208:
1205:
1194:on 2017-07-08
1193:
1189:
1188:opentextbc.ca
1185:
1179:
1176:
1171:
1165:
1161:
1154:
1151:
1140:on 2016-12-07
1139:
1135:
1131:
1125:
1122:
1115:
1111:
1108:
1106:
1103:
1101:
1098:
1096:
1093:
1091:
1088:
1087:
1083:
1081:
1060:
1056:
1052:
1047:
1043:
1039:
1034:
1030:
1026:
1023:
1014:
1010:
1005:
985:
976:
965:
957:
949:
936:
904:
891:
870:
859:
839:
817:
810:
800:
793:
788:
775:
769:
766:
763:
737:
728:
717:
709:
702:
693:
688:
661:
659:
658:cross section
653:
648:
644:
636:
634:
632:
628:
624:
619:
617:
613:
609:
605:
600:
598:
594:
587:
583:
579:
575:
568:
564:
560:
556:
550:
546:
542:
538:
532:
528:
522:
518:
511:
506:
501:
497:
493:
489:
483:
479:
475:
471:
464:
460:
456:
452:
445:
440:
436:
432:
428:
424:
418:
414:
409:
403:
396:
392:
388:
383:
380:
373:
369:
363:
355:
353:
350:
346:
342:
338:
334:
329:
327:
323:
319:
311:
307:
303:
279:
269:
262:
257:
252:
244:
242:
238:
233:
229:
225:
221:
217:
213:
209:
205:
201:
197:
192:
173:
170:
162:
149:
145:
140:
136:
133:
130:
122:
116:
108:
106:
104:
96:
94:
92:
88:
84:
80:
76:
72:
68:
61:
57:
53:
49:
41:
40:Bunsen burner
37:
32:
19:
2141:
2064:Molecularity
1698:
1692:
1673:
1667:
1648:
1642:
1623:
1616:. Retrieved
1612:
1602:
1583:
1577:
1558:
1552:
1535:
1531:
1525:
1506:
1500:
1492:
1472:
1464:
1445:
1439:
1427:. Retrieved
1418:
1409:
1390:
1384:
1374:February 17,
1372:. Retrieved
1366:. UC Davis.
1363:
1353:
1342:. Retrieved
1333:
1324:
1307:
1303:
1296:
1271:
1267:
1261:
1244:
1240:
1234:
1222:. Retrieved
1207:
1196:. Retrieved
1192:the original
1187:
1178:
1159:
1153:
1142:. Retrieved
1138:the original
1133:
1124:
1006:
692:nitric oxide
689:
662:
654:
646:
642:
640:
620:
611:
607:
603:
601:
596:
592:
585:
581:
577:
573:
566:
562:
558:
554:
548:
544:
540:
536:
530:
526:
520:
516:
509:
504:
499:
495:
491:
487:
477:
469:
462:
454:
451:Gibbs energy
443:
441:
434:
430:
426:
422:
416:
412:
401:
394:
390:
386:
379:to reach the
371:
365:
348:
330:
325:
315:
305:
239:
231:
227:
219:
211:
208:gas constant
203:
195:
193:
118:
100:
69:(kJ/mol) or
59:
51:
45:
2023:Cage effect
1958:RRKM theory
1874:elimination
1247:(5): 1172.
631:spontaneity
543:) exp(1 + Δ
505:approximate
337:active site
2203:Combustion
2182:Categories
1618:23 January
1344:2017-01-13
1224:15 October
1198:2018-04-05
1169:0070202605
1144:2017-01-13
1116:References
627:endergonic
261:exothermic
97:Other uses
2198:Catalysis
2074:Catalysis
1970:reactions
1310:: 32–39.
1288:121142463
1274:: 14–21.
1053:−
974:⟶
878:⇀
871:−
860:−
853:↽
726:⟶
671:, where k
623:exergonic
333:substrate
251:Catalysis
245:Catalysts
146:−
1423:Archived
1368:Archived
1338:Archived
1218:Archived
1084:See also
1007:Certain
643:decrease
474:entropic
429:) exp(−Δ
318:catalyst
263:reaction
1632:< Ea
595:, and Δ
366:In the
349:release
222:is the
218:), and
216:kelvins
198:is the
81:of the
46:In the
1749:Basic
1705:
1680:
1655:
1590:
1565:
1513:
1485:
1452:
1429:10 May
1397:
1286:
1166:
322:enzyme
300:) and
194:where
79:minima
1977:Redox
1813:Acyl)
1284:S2CID
610:and Δ
393:exp(−
36:flint
1866:(E2)
1855:(E1)
1703:ISBN
1678:ISBN
1653:ISBN
1628:+ Ea
1620:2022
1588:ISBN
1563:ISBN
1511:ISBN
1483:ISBN
1450:ISBN
1431:2020
1395:ISBN
1376:2017
1226:2021
1164:ISBN
923:and
580:and
524:and
468:and
119:The
1836:Ar)
1793:Ar)
1540:doi
1479:883
1312:doi
1308:180
1276:doi
1272:432
1249:doi
625:or
606:, Δ
591:, Δ
529:= (
515:= Δ
490:= Δ
415:= (
343:or
2184::
1904:(A
1894:(A
1832:(S
1809:(S
1803:i)
1799:(S
1789:(S
1783:2)
1779:(S
1773:1)
1769:(S
1634:−1
1622:.
1611:.
1536:88
1534:.
1491:.
1481:.
1417:.
1362:.
1336:.
1332:.
1306:.
1282:.
1270:.
1245:73
1243:.
1186:.
1132:.
1080:.
1004:.
982:NO
844:NO
734:NO
707:NO
652:.
582:RT
521:RT
519:+
494:−
484::
435:RT
433:/
425:/
402:RT
389:=
304:(Δ
230:,
210:,
93:.
1908:)
1906:N
1898:)
1896:E
1872:i
1870:E
1834:E
1811:N
1801:N
1791:N
1781:N
1771:N
1742:e
1735:t
1728:v
1711:.
1686:.
1661:.
1630:2
1626:1
1596:.
1571:.
1546:.
1542::
1519:.
1458:.
1433:.
1403:.
1378:.
1347:.
1318:.
1314::
1290:.
1278::
1255:.
1251::
1228:.
1201:.
1172:.
1147:.
1061:t
1057:E
1048:p
1044:E
1040:+
1035:i
1031:E
1027:=
1024:E
986:2
977:2
966:2
962:O
958:+
950:2
946:O
937:2
933:N
905:2
901:O
892:2
888:N
840:2
818:]
811:2
807:O
801:[
794:2
789:]
784:O
781:N
776:[
770:k
767:=
764:v
738:2
729:2
718:2
714:O
710:+
703:2
685:2
681:1
677:1
673:2
669:1
667:K
665:2
650:a
647:E
612:G
608:S
604:H
597:H
593:G
589:a
586:E
578:S
576:Δ
574:T
570:a
567:E
563:G
559:T
555:A
551:)
549:R
547:/
545:S
541:h
539:/
537:T
534:B
531:k
527:A
517:H
513:a
510:E
500:S
498:Δ
496:T
492:H
488:G
486:Δ
478:A
470:h
466:B
463:k
455:G
447:a
444:E
437:)
431:G
427:h
423:T
420:B
417:k
413:k
404:)
400:/
398:a
395:E
391:A
387:k
375:a
372:E
306:H
285:a
280:E
235:a
232:E
228:A
220:k
212:T
204:R
196:A
177:)
174:T
171:R
168:(
163:/
155:a
150:E
141:e
137:A
134:=
131:k
63:a
60:E
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.