835:
further increase of false positives in NYC DOHMH STD Clinics during the end of 2007 and beginning of 2008, their clinics opted to forgo further oral screenings, and instead reinstituted testing using finger-stick whole blood. Despite the increase in false positives in NYC DOHMH, the CDC still continues to support the use of noninvasive oral fluid specimens due to their popularity in health clinics and convenience of use. The director of the HIV control program for public health at
Seattle King county, reported OraQuick failed to spot at least 8 percent of 133 people found to be infected with a comparable diagnostic test. Strategies implemented to determine quality control and false positive rates were implemented. It is to be understood that any reactive OraQuick test result is a preliminary positive result and will always require a confirmatory test, regardless of the mean of testing (venipuncture whole blood, fingerstick whole blood or oral mucosal transudate fluid). Several other testing sites who did not experience a spike in false positive rates continue to use OraSure's OraQuick HIV Anti-body Testing.
314:(CDC) announced an initiative for voluntary, routine testing of all Americans aged 13–64 during health care encounters. An estimated 25% of infected individuals were unaware of their status; if successful, this effort was expected to reduce new infections by 30% per year. The CDC recommends elimination of requirements for written consent or extensive pre-test counseling as barriers to widespread routine testing. In 2006, the National Association of Community Health Centers implemented a model for offering free, rapid HIV testing to all patients between the ages of 13 and 64 during routine primary medical and dental care visits. The program increased testing rates, with 66% of the 17,237 patients involved in the study agreeing to testing (56% were tested for the first time). In September 2010, New York became the first state to require that hospitals and primary care providers offer an HIV test to all patients between the ages of 13 and 64 years. An evaluation of the law's impact found that it increased testing significantly throughout the state.
458:, and ENV gene-product groups are present, the result is positive. The three-gene-product approach to western blot interpretation has not been adopted for public health or clinical practice. Tests in which less than the required number of viral bands are detected are reported as indeterminate: a person who has an indeterminate result should be retested, as later tests may be more conclusive. Almost all HIV-infected persons with indeterminate western blot results will develop a positive result when tested in one month; persistently indeterminate results over a period of six months suggests the results are not due to HIV infection. In a generally healthy low-risk population, indeterminate results on western blot occur on the order of 1 in 5,000 patients. However, for those individuals who have had high-risk exposures to individuals where HIV-2 is most prevalent, Western Africa, an inconclusive western blot test may prove infection with HIV-2.
682:
if the pool tests positive, each sample is retested individually. Although this results in a dramatic decrease in cost, the dilution of the virus in the pooled samples decreases the effective sensitivity of the test, lengthening the window period by four days (assuming a 20-fold dilution, ~20hr virus doubling time, detection limit 50 copies/ml, making limit of detection 1,000 copies/ml). Since 2001, donated blood in the United States has been screened with nucleic-acid-based tests, shortening the window period between infection and detectability of disease to a median of 17 days (95% CI, 13–28 Days, assumes pooling of samples). A different version of this test is intended for use in conjunction with clinical presentation and other laboratory markers of disease progress for the management of
573:...the use of repeatedly reactive enzyme immunoassay followed by confirmatory western blot or immunofluorescent assay remains the standard method for diagnosing HIV-1 infection. A large study of HIV testing in 752 U.S. laboratories reported a sensitivity of 99.7% and specificity of 98.5% for enzyme immunoassay, and studies in U.S. blood donors reported specificities of 99.8% and greater than 99.99%. With confirmatory Western blot, the chance of a false-positive identification in a low-prevalence setting is about 1 in 250 000 (95% CI, 1 in 173 000 to 1 in 379 000).
450:. Some commercially prepared Western blot test kits contain the HIV proteins already on a cellulose acetate strip. Once the proteins are well-separated, they are transferred to a membrane and the procedure continues similar to an ELISA: the person's diluted serum is applied to the membrane and antibodies in the serum may attach to some of the HIV proteins. Antibodies that do not attach are washed away, and enzyme-linked antibodies with the capability to attach to the person's antibodies determine to which HIV proteins the person has antibodies.
615:). The chance that a positive test accurately indicates an HIV infection increases as the prevalence or rate of HIV infection increases in the population. Conversely, the negative predictive value will decrease as the HIV prevalence rises. Thus a positive test in a high-risk population, such as people who frequently engage in unprotected anal intercourse with unknown partners, is more likely to correctly represent HIV infection than a positive test in a very low-risk population, such as unpaid blood donors.
490:
426:
804:
555:
initially blamed on the influenza vaccines used during that flu season, but further investigation traced the cross-reactivity to several relatively non-specific test kits. A false positive result does not indicate a condition of significant risk to health. When the ELISA test is combined with
Western Blot, the rate of false positives is extremely low, and diagnostic accuracy is very high (see below).
42:
863:. While AIDS denialists focus on individual components of HIV testing, the combination of ELISA and western blot used for the diagnosis of HIV is remarkably accurate, with very low false-positive and -negative rates as described above. The views of AIDS denialists are based on highly selective analysis of mostly outdated scientific papers; there is broad
478:
597:
of these tests is extremely high, meaning that a negative test result will be correct more than 9,997 times in 10,000 (99.97% of the time). The very high negative predictive value of these tests is why the CDC recommends that a negative test result be considered conclusive evidence that an individual
834:
As a result of an increase in false positive rates with rapid oral HIV testing in 2005, New York City's
Department of Health and Mental Hygiene added the option of testing finger-stick whole blood after any reactive result, before using a western blot test to confirm the positive result. Following a
784:
In general, the lower the number of T cells the lower the immune system's function will be. Normal CD4 counts are between 500 and 1500 CD4+ T cells/microliter, and the counts may fluctuate in healthy people, depending on recent infection status, nutrition, exercise, and other factors. Women tend to
772:
occur. This use of a CD4 count as an AIDS criterion was introduced in 1992; the value of 200 was chosen because it corresponded with a greatly increased likelihood of opportunistic infection. Lower CD4 counts in people with AIDS are indicators that prophylaxis against certain types of opportunistic
747:
The South
African government announced a plan to start screening for HIV in secondary schools by March 2011. This plan was cancelled due to concerns it would invade pupils' privacy, schools typically don't have the facilities to securely store such information, and schools generally do not have the
681:
Nucleic-acid-based tests amplify and detect one or more of several target sequences located in specific HIV genes, such as HIV-I GAG, HIV-II GAG, HIV-env, or the HIV-pol. Since these tests are relatively expensive, the blood is screened by first pooling some 8–24 samples and testing these together;
559:
HIV antibody tests are highly sensitive, meaning they react preferentially with HIV antibodies, but not all positive or inconclusive HIV ELISA tests mean the person is infected by HIV. Risk history, and clinical judgement should be included in the assessment, and a confirmation test (western blot)
284:
Considerable controversy exists over the ethical obligations of health care providers to inform the sexual partners of individuals infected with HIV that they are at risk of contracting the virus. Some legal jurisdictions permit such disclosure, while others do not. More state funded testing sites
776:
Low CD4 T-cell counts are associated with a variety of conditions, including many viral infections, bacterial infections, parasitic infections, primary immunodeficiency, coccidioidomycosis, burns, trauma, intravenous injections of foreign proteins, malnutrition, over-exercising, pregnancy, normal
728:
to concentrate the virus, which is then opened to release its RNA. Special oligonucleotides that bind to viral RNA and to certain oligonucleotides bound to the wall of the vessel are added. In this way, viral RNA is fastened to the wall. Then new oligonucleotides that bind at several locations to
659:
In blood donation screening, this test is no longer used routinely in the US or the EU since the objective was to reduce the risk of false negatives in the window period. Nucleic acid testing (NAT) is more effective for this purpose, and p24 antigen testing is no longer indicated if a NAT test is
554:
Rare false positive results due to factors unrelated to HIV exposure are found more often with the ELISA test than with the western blot. False positives may be associated with medical conditions such as recent acute illnesses and allergies. A rash of false positive tests in the fall of 1991 was
288:
In developing countries, home-based HIV testing and counseling (HBHTC) is an emerging approach for addressing confidentiality issues. HBHTC allows individuals, couples, and families to learn their HIV status in the convenience and privacy of their home environment. Rapid HIV tests are most often
875:
There have been a number of cases of fraudulent tests being sold via mail order or the
Internet to the general public. In 1997, a California man was indicted on mail fraud and wire charges for selling supposed home test kits. In 2004, the US Federal Trade Commission asked Federal Express and US
519:
If no antibodies to HIV are detected, this does not mean the person has not been infected with HIV. It may take several months after HIV infection for the antibody response to reach detectable levels, during which time rapid testing for antibodies to HIV will not be indicative of true infection
767:
A CD4 count does not check for the presence of HIV. It is used to monitor immune system function in HIV-positive people. Declining CD4 T-cell counts are considered to be a marker of progression of HIV infection. A normal CD4 count can range from 500 cells/mm3 to 1000 cells/mm3. In HIV-positive
663:
In general diagnostics, p24 antigen tests are used for early detection of HIV, as p24 antigen rises soon after infection relative to antibodies, and the test is often used in combination with an antibody test to effectively cover a longer portion of the window period. It is less useful as a
729:
this RNA are added, and other oligonucleotides that bind at several locations to those oligonucleotides. This is done to amplify the signal. Finally, oligonucleotides that bind to the last set of oligonucleotides and that are bound to an enzyme are added; the enzyme action causes a
876:
Customs to confiscate shipments of the
Discreet home HIV test kits, produced by Gregory Stephen Wong of Vancouver, Canada. In February 2005, the US FDA issued a warning against using the rapid HIV test kits and other home use kits marketed by Globus Media of Montreal, Canada.
610:
and negative predictive value of all tests, including HIV tests, take into account the prior probability of having a disease along with the accuracy of the testing method to determine a new degree of belief that an individual has or does not have a disease (also known as the
733:, which allows quantification of the viral RNA in the original sample. Monitoring the effects of antiretroviral therapy by serial measurements of plasma HIV-1 RNA with this test has been validated for patients with viral loads greater than 25,000 copies per milliliter.
780:
This test is also used occasionally to estimate immune system function for people whose CD4 T cells are impaired for reasons other than HIV infection, which include several blood diseases, several genetic disorders, and the side effects of many chemotherapy drugs.
1429:
MacDonald K, Jackson J, Bowman R, Polesky H, Rhame F, Balfour H, Osterholm M (1989). "Performance characteristics of serologic tests for human immunodeficiency virus type 1 (HIV-1) antibody among
Minnesota blood donors. Public health and clinical implications".
297:
Testing that has only a number attached to the specimen that will be delivered for testing. Items that are confirmed positive will not have the HIV infected individual's name attached to the specimen. Sites that offer this service advertise this testing option.
655:
specific to the p24 protein are mixed with the person's blood. Any p24 protein in the person's blood will stick to the monoclonal antibody and an enzyme-linked antibody to the monoclonal antibodies to p24 causes a color change if p24 was present in the sample.
379:
is diluted 400-fold and applied to a plate to which HIV antigens have been attached. If antibodies to HIV are present in the serum, they may bind to these HIV antigens. The plate is then washed to remove all other components of the serum. A specially prepared
392:
for the enzyme is applied, and catalysis by the enzyme leads to a change in color or fluorescence. ELISA results are reported as a number; the most controversial aspect of this test is determining the "cut-off" point between a positive and negative result.
664:
standalone test, as it has low sensitivity and only works during the early time period after infection. The presence of p24 antigen diminishes as the body increases production of antibodies to the p24 protein, making p24 more difficult to detect later.
748:
capacity to provide counseling for HIV-positive pupils. In South Africa, anyone over the age of 12 may request an HIV test without parental knowledge or consent. Some 80,000 pupils in three provinces were tested under this programme before it ended.
2375:
1649:"Use of Nucleic Acid Tests on Pooled and Individual Samples from Donations of Whole Blood and Blood Components (including Source Plasma and Source Leukocytes) to Adequately and Appropriately Reduce the Risk of Transmission of HIV-1 and HCV"
535:
ELISA testing alone cannot be used to diagnose HIV, even if the test suggests a high probability that antibody to HIV-1 is present. In the United States, such ELISA results are not reported as "positive" unless confirmed by a western blot.
1612:
Farzadegan H, Vlahov D, Solomon L, Muñoz A, Astemborski J, Taylor E, Burnley A, Nelson K (1993). "Detection of human immunodeficiency virus type 1 infection by polymerase chain reaction in a cohort of seronegative intravenous drug users".
672:
A combination, or 4th generation assay, is designed to detect both the p24 antigen and HIV antibodies in a single test. Combination tests can detect HIV as early as 2–6 weeks after infection, and are recommended in laboratory testing.
214:) and more expeditious nucleic acid testing (NAT). These diagnostic tests are combined with careful donor selection. As of 2001, the risk of transfusion-acquired HIV in the US was approximately one in 2.5 million for each transfusion.
445:
within are placed into a slab of gel, to which an electric current is applied. Different proteins will move with different speeds in this field, depending on their size, while their electrical charge is leveled by a surfactant called
453:
There are no universal criteria for interpreting the western blot test: The number of viral bands that must be present may vary. If no viral bands are detected, the result is negative. If at least one viral band for each of the GAG,
344:
is implemented. Most people develop detectable antibodies approximately 18 to 30 days after exposure, although some do seroconvert later. The vast majority of people (99%) have detectable antibodies by two months after HIV exposure.
285:
are now using confidential forms of testing. This allows for monitoring of infected individuals easily, compared to anonymous testing that has a number attached to the positive test results. Controversy exists over privacy issues.
1182:
C B Hare, B L Pappalardo, M P Busch, B Phelps, S S Alexander, C Ramstead, J A Levy, F M Hecht (2004). "Negative HIV antibody test results among individuals treated with antiretroviral therapy (ART) during acute/early infection".
1238:
Mas A, Soriano V, Gutiérrez M, Fumanal F, Alonso A, González-Lahoz J (1997). "Reliability of a new recombinant immunoblot assay (RIBA HIV-1/HIV-2 SIA) as a supplemental (confirmatory) test for HIV-1 and HIV-2 infections".
601:
Of course, the actual numbers vary depending on the testing population. This is because interpreting of the results of any medical test (assuming no test is 100% accurate) depends upon the initial degree of belief, or the
289:
used, so results are available for the client between 15 and 30 minutes. Furthermore, when an HIV-positive result is communicated, the HTC provider can offer appropriate linkages for prevention, care, and treatment.
437:
is an antibody detection test. However, unlike the ELISA method, the viral proteins are separated first and immobilized. In subsequent steps, the binding of serum antibodies to specific HIV proteins is visualized.
2370:
1391:
Burke D, Brundage J, Redfield R, Damato J, Schable C, Putman P, Visintine R, Kim H (1988). "Measurement of the false positive rate in a screening program for human immunodeficiency virus infections".
569:
Modern HIV testing is highly accurate. The evidence regarding the risks and benefits of HIV screening was reviewed in July 2005 by the U.S. Preventive
Services Task Force. The authors concluded that:
1768:"Simultaneous detection of multiplex-amplified human immunodeficiency virus type 1 RNA, hepatitis C virus RNA, and hepatitis B virus DNA using a flow cytometer microsphere-based hybridization assay"
2682:
847:(a fringe group whose members believe that HIV either does not exist or is harmless). The accuracy of serologic testing has been verified by isolation and culture of HIV and by detection of HIV
2646:
1048:
721:
bound to the vessel wall, and are then made visible with a probe bound to an enzyme. The amount of virus in the sample can be quantified with sufficient accuracy to detect threefold changes.
634:
585:. Confirming the test result (i.e., by repeating the test, if this option is available) could reduce the ultimate likelihood of a false positive to about 1 result in 250,000 tests given. The
210:
In the US, the Food and Drug
Administration requires that all donated blood be screened for several infectious diseases, including HIV-1 and HIV-2, using a combination of antibody testing (
2220:
606:
that an individual has, or does not have a disease. Generally the prior probability is estimated using the prevalence of a disease within a population or at a given testing location. The
2365:
356:(PEP). Those patients must take ELISA tests at various intervals after the usual 28-day course of treatment, sometimes extending outside of the conservative window period of 6 months.
2737:
117:
The eclipse period is a variable period starting from HIV exposure in which no existing test can detect HIV. The median duration of the eclipse period in one study was 11.5 days. The
3333:
352:
during the window period can delay the formation of antibodies and extend the window period beyond 12 months. This was not the case with patients that underwent treatment with
3328:
2382:
166:, or the presence of antibodies directed to other infectious agents that may be antigenically similar to HIV can produce false positive results. Autoimmune diseases, such as
581:
rate given here for the inexpensive enzyme immunoassay screening tests indicates that, in 1,000 HIV test results of healthy individuals, about 15 of these results will be a
894:
This image of
Randall L. Tobias is used in a Korean news article suggesting that foreign English teachers residing in Korea are at risk for AIDS. Accessed 16 February 2010.
417:
or computer without additional support or battery power, and takes some fifteen minutes to analyse a drop of blood. The units cost approximately $ 34 each to manufacture.
508:
to aid in the diagnosis of HIV infection. These tests should be used in conjunction with the clinical status, history, and risk factors of the person being tested. The
3366:
2843:
384:" – an antibody that binds to human antibodies – is then applied to the plate, followed by another wash. This secondary antibody is chemically linked in advance to an
3415:
2316:
2216:
2203:
2717:
1160:
1138:
2589:
2408:
2358:
2306:
1919:
2702:
121:
is the time between HIV exposure and when an antibody or antigen test can detect HIV. The median window period for antibody/antigen testing is 18 days.
1120:
3400:
1734:
909:"Time Until Emergence of HIV Test Reactivity Following Infection With HIV-1: Implications for Interpreting Test Results and Retesting After Exposure"
2516:
2212:
1052:
3716:
348:
During the window period, an infected person can transmit HIV to others although their HIV infection may not be detectable with an antibody test.
242:
procedure determines the size of the antigens in the test kit binding to the antibodies. The combination of these two methods is highly accurate
2428:
2258:
183:
Tests selected to screen donor blood and tissue must provide a high degree of confidence that HIV will be detected if present (that is, a high
2677:
2175:
1956:
3359:
3108:
1470:"Evaluation of screened blood donations for human immunodeficiency virus type 1 infection by culture and DNA amplification of pooled cells"
322:
HIV antibody tests are specifically designed for routine diagnostic testing of adults; these tests are inexpensive and extremely accurate.
2796:
2029:
629:
2004:
1849:"Dynamics of HIV viremia and antibody seroconversion in plasma donors: Implications for diagnosis and staging of primary HIV infection"
3233:
2473:
523:
Although these tests have high specificity, false positives do occur. Any positive test result should be confirmed by a lab using the
1847:
Fiebig EW, Wright DJ, Rawal BD, Garrett PE, Schumacher RT, Peddada L, Heldebrant C, Smith R, Conrad A, Kleinman SH, Busch MP (2003).
1509:
Van de Perre P, Simonon A, Msellati P, Hitimana D, Vaira D, Bazubagira A, Van Goethem C, Stevens A, Karita E, Sondag-Thull D (1991).
1139:"Community Health Centers Integrate Rapid HIV Screening into Routine Primary Care, Leading to Significant Increases in Testing Rates"
340:, hence an interval of three weeks to six months between the time of HIV exposure and the production of measurable antibodies to HIV
2656:
2546:
1934:
1680:
1352:"False-positive HIV-1 test results in a low-risk screening setting of voluntary blood donation. Retrovirus Epidemiology Donor Study"
365:
2133:
1815:
1511:"Postnatal transmission of human immunodeficiency virus type 1 from mother to infant. A prospective cohort study in Kigali, Rwanda"
250:
The UNAIDS/WHO policy statement on HIV Testing states that conditions under which people undergo HIV testing must be anchored in a
2403:
1274:
Simonsen L, Buffington J (June 1995). "Multiple false reactions in viral antibody screening assays after influenza vaccination".
429:
Western blot test results. The first two strips are a negative and a positive control, respectively. The others are actual tests.
104:
3352:
3552:
3136:
2398:
1648:
512:
of Rapid Antibody Tests in low-risk populations has not been evaluated. These tests should be used in appropriate multi-test
551:
ELISA antibody test. This is why a confirmatory western blot is always used before reporting a "positive" HIV test result.
3198:
2712:
2692:
2423:
2311:
2081:
968:
946:
3311:
2584:
2572:
2453:
2413:
1711:
1653:
891:
167:
2779:
3083:
2707:
2636:
2594:
2338:
2296:
586:
578:
311:
3615:
3530:
2055:
2773:
2697:
2687:
2418:
2251:
714:
710:
992:
547:
ELISA antibody test is in fact infected with HIV. Sometimes, retesting the donor in several months will produce a
3504:
3247:
3215:
3203:
3156:
2732:
2231:
1909:
852:
706:
622:, reporting false-positive rates of 0.0004 to 0.0007 and false-negative rates of 0.003 in the general population.
607:
594:
509:
353:
204:
3088:
2938:
2811:
2499:
2458:
389:
207:
estimated that, as of 2000, inadequate blood screening had resulted in 1 million new HIV infections worldwide.
1981:
145:
All diagnostic tests have limitations, and sometimes their use may produce erroneous or questionable results.
1124:
543:
infected with HIV. It is therefore not possible to conclude that blood rejected for transfusion because of a
388:. Thus the plate will contain enzyme in proportion to the amount of secondary antibody bound to the plate. A
3306:
3178:
3053:
2971:
2873:
2438:
2433:
1161:"New York State Legislation Leads to More HIV Testing and Linking of HIV-Positive Patients to Followup Care"
769:
170:, have also rarely caused false positive results. Most false negative results are due to the window period.
3499:
1706:
593:
result. However, based upon the HIV prevalence rates at most testing centers within the United States, the
3646:
3018:
2991:
2981:
2903:
2888:
2786:
2606:
2536:
2343:
2333:
1741:
163:
3634:
3301:
3255:
3193:
3183:
3093:
3028:
2599:
2567:
2504:
2244:
725:
698:
612:
505:
497:
447:
1219:
Bartlett, JG. Serologic tests for the diagnosis of HIV infection, in UpToDate. Accessed 5 October 2006.
489:
425:
3629:
3270:
3188:
3103:
3078:
3063:
3001:
2953:
2933:
2858:
2853:
2838:
2579:
2511:
908:
864:
856:
589:
rating, likewise, indicates that, in 1,000 test results of HIV infected people, 3 will actually be a
539:
The ELISA antibody tests were developed to provide a high level of confidence that donated blood was
122:
3434:
1468:
Busch M, Eble B, Khayam-Bashi H, Heilbron D, Murphy E, Kwok S, Sninsky J, Perkins H, Vyas G (1991).
560:
should be administered. An individual with an inconclusive test should be re-tested at a later date.
3602:
3462:
3405:
3387:
3285:
3260:
3228:
3146:
3118:
3068:
2986:
2976:
2928:
2918:
2908:
2883:
2494:
2468:
2107:
1021:
JM Appel (June 2006). "Must My Doctor Tell My Partner? Rethinking Confidentiality in the HIV Era".
652:
482:
402:
349:
234:
combining two tests for HIV antibodies. If antibodies are detected by an initial test based on the
227:
223:
222:
Tests used for the diagnosis of HIV infection in a particular person require a high degree of both
184:
138:
132:
713:
unique to the virus's genome. After PCR amplification is complete, the resulting DNA products are
3690:
3487:
3275:
3265:
3173:
3168:
3141:
3128:
3113:
3098:
3048:
3011:
2996:
2948:
2943:
2923:
2893:
2868:
2828:
2820:
2752:
2747:
2616:
2541:
2443:
1878:
1594:
1099:
648:
381:
369:
211:
3721:
3448:
3223:
3073:
3043:
3023:
3006:
2913:
2878:
2863:
2848:
2833:
2611:
2521:
2478:
2348:
2033:
1870:
1797:
1630:
1586:
1532:
1491:
1447:
1408:
1373:
1332:
1291:
1256:
1091:
1030:
928:
603:
46:
1199:
768:
people, AIDS is officially diagnosed when the count drops below 200 cells/μL or when certain
3547:
3537:
3516:
3280:
3058:
3038:
3033:
2963:
2898:
2641:
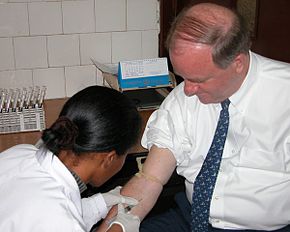
1860:
1787:
1779:
1622:
1576:
1522:
1481:
1439:
1400:
1363:
1327:
1322:
1310:
1283:
1248:
1083:
920:
501:
307:
272:
3585:
3420:
3410:
2791:
2531:
2301:
1816:"Instruction Manual – MPCR Kit for Human Immunodeficiency Virus (HIV) Type I/II, MaximBio"
1008:
996:
844:
718:
462:
372:(EIA), was the first screening test commonly employed for HIV. It has a high sensitivity.
2154:
1684:
1350:
Kleinman S, Busch M, Hall L, Thomson R, Glynn S, Gallahan D, Ownby H, Williams A (1998).
1311:"Screening for HIV: a review of the evidence for the U.S. Preventive Services Task Force"
907:
Delaney KP, Hanson DL, Masciotra S, Ethridge SF, Wesolowski L, Owen SM (1 January 2017).
520:
status. For most people, HIV antibodies reach a detectable level after two to six weeks.
1822:
1074:
Armstrong WS, Taege AJ (April 2007). "HIV screening for all: the new standard of care".
817:
Please help update this article to reflect recent events or newly available information.
2742:
2526:
2204:
Complete List of Donor Screening Assays for Infectious Agents and HIV Diagnostic Assays
1287:
730:
590:
582:
341:
331:
255:
155:
149:
1252:
3710:
3695:
3654:
3610:
3573:
3542:
3474:
3456:
3344:
2722:
2631:
1865:
1848:
1792:
1767:
761:
625:
619:
336:
118:
1882:
1783:
1735:"CDC Laboratory Testing for the Diagnosis of HIV Infection, Updated Recommendations"
1598:
947:"Keeping Blood Transfusions Safe: FDA's Multi-layered Protections for Donated Blood"
3579:
3566:
3511:
3442:
2757:
2448:
1914:
1103:
860:
694:
524:
434:
262:
258:. According to these principles, the conduct of HIV testing of individuals must be
251:
239:
196:
1658:
618:
Many studies have confirmed the accuracy of current methods of HIV testing in the
2005:"Seattle Department Finds OraSure HIV Test to Be Less Accurate Than Label Claims"
1895:
3669:
3592:
2661:
2562:
2353:
1527:
1510:
1486:
1469:
1443:
1404:
376:
72:
2085:
1626:
41:
3428:
3375:
2621:
2463:
2291:
737:
414:
200:
152:: The test incorrectly indicates that HIV is present in a non-infected person.
141:: The percentage of the results that will be negative when HIV is not present.
84:
2227:
1549:
Update: serologic testing for HIV-1 antibody – United States, 1988 and 1989.
1368:
1351:
932:
334:(no antibodies were detected despite the presence of HIV) results during the
17:
3379:
513:
455:
310:
is to screen all patients for HIV in all health care settings. In 2006, the
231:
1874:
1801:
1336:
1260:
1095:
1034:
2059:
1634:
1590:
1536:
1495:
1451:
1412:
1377:
1295:
1087:
158:: The test incorrectly indicates that HIV is absent in an infected person.
3674:
3664:
3395:
2727:
2626:
2323:
2271:
924:
410:
188:
135:: The percentage of the results that will be positive when HIV is present
68:
50:
1581:
1564:
49:, former U.S. Global AIDS Coordinator, being publicly tested for HIV in
3659:
3623:
3524:
2651:
989:
442:
192:
88:
1982:"False-Positive Oral Fluid Rapid HIV Tests – New York City, 2005–2008"
3560:
2767:
1049:"Getting in the Door: Home-Based HIV Testing and Counseling in Kenya"
406:
385:
76:
477:
1766:
Defoort JP, Martin M, Casano B, Prato S, Camilla C, Fert V (2000).
724:
In the Quantiplex bDNA or branched DNA test, plasma is placed in a
3492:
2236:
1228:
Scand J Infect Dis. 1992;24(4):419-21. Accessed 23 September 2008.
1200:"Test for HIV in just 15 minutes with this $ 34 smartphone dongle"
488:
476:
424:
235:
80:
892:"Blacklist of English teachers suspected of having AIDS pursued."
2801:
2762:
702:
441:
Specifically, cells that may be HIV-infected are opened and the
3348:
2240:
1910:"South Africa teaching unions criticise HIV testing in schools"
760:
is not an HIV test, but rather a procedure where the number of
516:
designed for statistical validation of rapid HIV test results.
2286:
2267:
848:
797:
690:
92:
64:
2683:
European and Developing Countries Clinical Trials Partnership
1309:
Chou R, Huffman LH, Fu R, Smits AK, Korthuis PT (July 2005).
999:. From the Puget Sound Blood Center. Accessed 5 October 2006.
777:
daily variation, psychological stress, and social isolation.
461:
The HIV proteins used in western blotting can be produced by
128:
Performance of medical tests is often described in terms of:
2209:
Fact sheets from the National Aids Trust ("NAT") in the UK:
2056:"Testing for HIV – Why and How | Free Testing Site HAF"
651:
of HIV (also known as CA), the capsid protein of the virus.
2111:
1163:. Agency for Healthcare Research and Quality. 10 April 2013
1141:. Agency for Healthcare Research and Quality. 5 April 2013
2366:
WHO disease staging system for HIV infection and disease
268:
Accompanied by counseling (for those who test positive);
2030:"Overview of False-Positive Oral Fluid Rapid HIV Tests"
3334:
List of HIV/AIDS cases and deaths registered by region
2003:
Henry J. Kaiser Family Foundation (5 September 2008).
2176:"Defective HIV Test Kit Marketer Settles FTC Charges"
2084:. Harlem United Community AIDS Center. Archived from
3683:
3645:
3601:
3473:
3386:
3329:
List of countries by HIV/AIDS adult prevalence rate
3294:
3246:
3214:
3155:
3127:
2962:
2819:
2810:
2718:
President's Emergency Plan for AIDS Relief (PEPFAR)
2703:
Joint United Nations Programme on HIV/AIDS (UNAIDS)
2670:
2555:
2487:
2391:
2279:
1683:. Eurosurveillance. 1 February 2005. Archived from
1565:"Increased sensitivity of HIV-1 antibody detection"
990:
Adverse reactions associated with blood transfusion
34:
230:. In the United States, this is achieved using an
1957:"Lymphocyte surface marker analysis - Immunology"
647:The p24 antigen test detects the presence of the
53:in an effort to reduce the stigma of being tested
1009:UNAIDS/WHO policy statement on HIV Testing (PDF)
125:(NAT) further reduces this period to 11.5 days.
3416:Fluorescent treponemal antibody absorption test
2317:National Addiction and HIV Data Archive Program
571:
3360:
2252:
8:
1463:
1461:
1424:
1422:
2409:Diffuse infiltrative lymphocytosis syndrome
2058:. Freehivtesting.hafnyc.org. Archived from
179:Screening donor blood and cellular products
3367:
3353:
3345:
3161:
2816:
2642:People With AIDS Self-Empowerment Movement
2259:
2245:
2237:
1563:Urnovitz H, Sturge J, Gottfried T (1997).
1115:
1113:
40:
3401:Venereal Disease Research Laboratory test
1864:
1791:
1580:
1526:
1485:
1367:
1326:
709:(PCR) process is then applied, using two
1922:from the original on 21 September 2013.
1707:"HIV Tests For Screening and Diagnosis"
1328:10.7326/0003-4819-143-1-200507050-00010
884:
109:AIDS is diagnosed separately from HIV.
63:are used to detect the presence of the
2108:"The scientific evidence for HIV/AIDS"
31:
2678:Discredited HIV/AIDS origins theories
785:have somewhat lower counts than men.
238:method, then a second test using the
7:
1185:The XV International AIDS Conference
902:
900:
493:Blood being taken for HIV rapid test
481:A woman demonstrates the use of the
2797:The Freddie Mercury Tribute Concert
2155:"The Controversy over HIV and AIDS"
2134:"The Evidence That HIV Causes AIDS"
701:(RT) to convert the viral RNA into
306:In the United States, one emerging
2213:General information on HIV testing
1288:10.1093/oxfordjournals.aje.a117374
1187:. pp. Abstract no. MoPeB3107.
843:HIV tests have been criticized by
668:Antigen/antibody combination tests
254:approach that pays due respect to
25:
2383:Countries by AIDS prevalence rate
2228:Bulk procurement of HIV test kits
1198:Mariella Moon (6 February 2015).
1023:Medicine and Health, Rhode Island
949:. US Food and Drug Administration
773:infections should be instituted.
752:Other tests used in HIV treatment
504:immunoassays intended for use in
366:enzyme-linked immunosorbent assay
2844:Democratic Republic of the Congo
2404:AIDS-defining clinical condition
1896:FDA summary of branched DNA test
1866:10.1097/00002030-200309050-00005
1772:Journal of Clinical Microbiology
802:
693:is extracted from the patient's
105:AIDS-defining clinical condition
2110:. AIDSTruth.org. Archived from
1784:10.1128/JCM.38.3.1066-1071.2000
867:that HIV is the cause of AIDS.
409:capable of testing for HIV and
187:is required). A combination of
3717:Infectious disease blood tests
3553:Orthopoxvirus inclusion bodies
1740:. 27 June 2014. Archived from
677:Nucleic acid-based tests (NAT)
433:Like the ELISA procedure, the
302:Routine testing recommendation
1:
2713:Misconceptions about HIV/AIDS
2693:International AIDS Conference
2647:HIV/AIDS in the porn industry
2590:Discrimination against people
1253:10.1016/s0955-3886(96)00078-1
1051:. Aidstar-One. Archived from
855:, which are widely accepted "
375:In an ELISA test, a person's
67:(HIV), the virus that causes
2573:Catholic Church and HIV/AIDS
2454:HIV Drug Resistance Database
1712:Food and Drug Administration
1654:Food and Drug Administration
913:Clinical Infectious Diseases
764:in the blood is determined.
473:Rapid or point-of-care tests
467:recombinant immunoblot assay
405:have produced an ELISA test
168:systemic lupus erythematosus
65:human immunodeficiency virus
2874:Côte d'Ivoire (Ivory Coast)
2708:Media portrayal of HIV/AIDS
2637:List of HIV-positive people
1908:Smith D (2 February 2011).
1528:10.1056/NEJM199108293250901
1487:10.1056/NEJM199107043250101
1444:10.7326/0003-4819-110-8-617
1405:10.1056/NEJM198810133191501
531:Interpreting antibody tests
312:Centers for Disease Control
275:of the person being tested.
3738:
2774:Reports from the Holocaust
2698:International AIDS Society
2688:Elton John AIDS Foundation
1898:, accessed 5 October 2006.
1011:, accessed 5 October 2006.
735:
689:In the RT-PCR test, viral
630:Excessive citations inline
413:. It is compatible to any
218:Diagnosis of HIV infection
203:in Western countries. The
102:
3505:Heterophile antibody test
3324:
3164:
2733:Treatment Action Campaign
2449:Tuberculosis co-infection
2312:disease progression rates
2232:World Health Organization
1551:MMWR Morb Mortal Wkly Rep
995:27 September 2006 at the
811:This section needs to be
707:polymerase chain reaction
608:positive predictive value
595:negative predictive value
510:positive predictive value
354:post-exposure prophylaxis
205:World Health Organization
39:
2459:Innate resistance to HIV
2429:Neurocognitive disorders
2180:Federal Trade Commission
1627:10.1093/infdis/168.2.327
1369:10.1001/jama.280.12.1080
770:opportunistic infections
330:Antibody tests may give
83:. Such tests may detect
2780:AIDS–Holocaust metaphor
2763:Gay Men's Health Crisis
2439:Opportunistic infection
635:considered for deletion
565:Accuracy of HIV testing
162:Nonspecific reactions,
3647:Bloodstream infections
3616:Sabin–Feldman dye test
3531:Warthin–Finkeldey cell
2607:HIV-affected community
2547:HIV and homosexual men
2230:instructions from the
575:
494:
486:
465:in a technique called
430:
350:Antiretroviral therapy
164:hypergammaglobulinemia
2787:Silence=Death Project
2585:Criminal transmission
2505:Multiple sex partners
2082:"Prevention Division"
2032:. CDC. Archived from
1127:on 16 September 2008.
1088:10.3949/ccjm.74.4.297
736:Further information:
699:reverse transcriptase
653:Monoclonal antibodies
613:posterior probability
506:point-of-care testing
492:
480:
448:sodium lauryl sulfate
428:
35:Diagnosis of HIV/AIDS
3630:Diagnosis of malaria
3109:United Arab Emirates
2580:Circumcision and HIV
2537:City of Hope Patient
2297:structure and genome
971:. American Red Cross
865:scientific consensus
697:and is treated with
686:-infected patients.
123:Nucleic acid testing
3603:Protozoan infection
3463:Anti-streptolysin O
3406:rapid plasma reagin
3388:Bacterial infection
2657:AIDS Memorial Quilt
2469:HIV-positive people
2036:on 14 December 2012
1747:on 28 February 2017
1582:10.1038/nm1197-1258
1241:Transfusion Science
598:does not have HIV.
500:antibody tests are
403:Columbia University
271:Conducted with the
3691:C-reactive protein
3500:Epstein–Barr virus
3488:Branched DNA assay
3380:infectious disease
3234:Dominican Republic
2617:HIV/AIDS denialism
2542:Women and HIV/AIDS
2532:The London Patient
2399:Signs and symptoms
2307:CDC classification
2088:on 19 October 2012
2062:on 4 November 2013
1715:. 30 November 2016
1055:on 3 November 2013
925:10.1093/cid/ciw666
871:Fraudulent testing
738:Viral load testing
495:
487:
431:
382:secondary antibody
370:enzyme immunoassay
256:ethical principles
199:tests are used by
27:Immunological test
3704:
3703:
3449:HelicoCARE direct
3342:
3341:
3320:
3319:
3242:
3241:
2723:The SING Campaign
2612:HIV/AIDS activism
2600:Cost of treatment
2522:Timothy Ray Brown
2517:World AIDS Museum
2217:Types of HIV test
1859:(13): 1871–1879.
1687:on 28 August 2008
832:
831:
604:prior probability
401:Researchers from
293:Anonymous testing
58:
57:
47:Randall L. Tobias
16:(Redirected from
3729:
3635:Schüffner's dots
3548:B type inclusion
3538:Inclusion bodies
3517:NS1 antigen test
3369:
3362:
3355:
3346:
3312:Papua New Guinea
3162:
2817:
2261:
2254:
2247:
2238:
2192:
2191:
2189:
2187:
2172:
2166:
2165:
2163:
2161:
2151:
2145:
2144:
2142:
2140:
2130:
2124:
2123:
2121:
2119:
2104:
2098:
2097:
2095:
2093:
2078:
2072:
2071:
2069:
2067:
2052:
2046:
2045:
2043:
2041:
2026:
2020:
2019:
2017:
2015:
2000:
1994:
1993:
1991:
1989:
1978:
1972:
1971:
1969:
1967:
1953:
1947:
1946:
1944:
1942:
1930:
1924:
1923:
1905:
1899:
1893:
1887:
1886:
1868:
1844:
1838:
1837:
1835:
1833:
1827:
1821:. Archived from
1820:
1812:
1806:
1805:
1795:
1778:(3): 1066–1071.
1763:
1757:
1756:
1754:
1752:
1746:
1739:
1731:
1725:
1724:
1722:
1720:
1703:
1697:
1696:
1694:
1692:
1677:
1671:
1670:
1668:
1666:
1657:. Archived from
1645:
1639:
1638:
1609:
1603:
1602:
1584:
1560:
1554:
1547:
1541:
1540:
1530:
1506:
1500:
1499:
1489:
1465:
1456:
1455:
1426:
1417:
1416:
1388:
1382:
1381:
1371:
1347:
1341:
1340:
1330:
1315:Ann. Intern. Med
1306:
1300:
1299:
1271:
1265:
1264:
1235:
1229:
1226:
1220:
1217:
1211:
1210:
1208:
1206:
1195:
1189:
1188:
1179:
1173:
1172:
1170:
1168:
1157:
1151:
1150:
1148:
1146:
1135:
1129:
1128:
1123:. Archived from
1121:"CDC fact sheet"
1117:
1108:
1107:
1076:Cleve Clin J Med
1071:
1065:
1064:
1062:
1060:
1045:
1039:
1038:
1018:
1012:
1006:
1000:
987:
981:
980:
978:
976:
965:
959:
958:
956:
954:
943:
937:
936:
904:
895:
889:
827:
824:
818:
806:
805:
798:
758:CD4 T-cell count
719:oligonucleotides
685:
638:
308:standard of care
273:informed consent
44:
32:
21:
3737:
3736:
3732:
3731:
3730:
3728:
3727:
3726:
3707:
3706:
3705:
3700:
3679:
3641:
3597:
3586:Councilman body
3475:Viral infection
3469:
3435:Weil–Felix test
3421:Abelin reaction
3411:Wassermann test
3382:
3373:
3343:
3338:
3316:
3290:
3238:
3210:
3151:
3123:
3069:Myanmar (Burma)
2958:
2806:
2792:Day Without Art
2738:The Global Fund
2666:
2595:Economic impact
2551:
2483:
2387:
2339:pathophysiology
2275:
2265:
2200:
2195:
2185:
2183:
2174:
2173:
2169:
2159:
2157:
2153:
2152:
2148:
2138:
2136:
2132:
2131:
2127:
2117:
2115:
2114:on 2 March 2012
2106:
2105:
2101:
2091:
2089:
2080:
2079:
2075:
2065:
2063:
2054:
2053:
2049:
2039:
2037:
2028:
2027:
2023:
2013:
2011:
2002:
2001:
1997:
1987:
1985:
1980:
1979:
1975:
1965:
1963:
1955:
1954:
1950:
1940:
1938:
1932:
1931:
1927:
1907:
1906:
1902:
1894:
1890:
1846:
1845:
1841:
1831:
1829:
1828:on 14 July 2010
1825:
1818:
1814:
1813:
1809:
1765:
1764:
1760:
1750:
1748:
1744:
1737:
1733:
1732:
1728:
1718:
1716:
1705:
1704:
1700:
1690:
1688:
1679:
1678:
1674:
1664:
1662:
1647:
1646:
1642:
1611:
1610:
1606:
1562:
1561:
1557:
1548:
1544:
1508:
1507:
1503:
1467:
1466:
1459:
1428:
1427:
1420:
1390:
1389:
1385:
1349:
1348:
1344:
1308:
1307:
1303:
1282:(11): 1089–96.
1273:
1272:
1268:
1237:
1236:
1232:
1227:
1223:
1218:
1214:
1204:
1202:
1197:
1196:
1192:
1181:
1180:
1176:
1166:
1164:
1159:
1158:
1154:
1144:
1142:
1137:
1136:
1132:
1119:
1118:
1111:
1073:
1072:
1068:
1058:
1056:
1047:
1046:
1042:
1020:
1019:
1015:
1007:
1003:
997:Wayback Machine
988:
984:
974:
972:
969:"Blood Testing"
967:
966:
962:
952:
950:
945:
944:
940:
906:
905:
898:
890:
886:
882:
873:
845:AIDS denialists
841:
828:
822:
819:
816:
807:
803:
796:
791:
754:
745:
740:
683:
679:
670:
645:
623:
567:
533:
485:rapid HIV test.
475:
463:recombinant DNA
423:
399:
362:
328:
320:
304:
295:
282:
280:Confidentiality
248:
220:
181:
176:
115:
107:
101:
54:
28:
23:
22:
15:
12:
11:
5:
3735:
3733:
3725:
3724:
3719:
3709:
3708:
3702:
3701:
3699:
3698:
3693:
3687:
3685:
3681:
3680:
3678:
3677:
3672:
3667:
3662:
3657:
3651:
3649:
3643:
3642:
3640:
3639:
3638:
3637:
3632:
3620:
3619:
3618:
3607:
3605:
3599:
3598:
3596:
3595:
3590:
3589:
3588:
3576:
3571:
3570:
3569:
3557:
3556:
3555:
3550:
3545:
3535:
3534:
3533:
3521:
3520:
3519:
3509:
3508:
3507:
3497:
3496:
3495:
3490:
3479:
3477:
3471:
3470:
3468:
3467:
3466:
3465:
3453:
3452:
3451:
3439:
3438:
3437:
3425:
3424:
3423:
3418:
3413:
3408:
3403:
3392:
3390:
3384:
3383:
3374:
3372:
3371:
3364:
3357:
3349:
3340:
3339:
3337:
3336:
3331:
3325:
3322:
3321:
3318:
3317:
3315:
3314:
3309:
3304:
3298:
3296:
3292:
3291:
3289:
3288:
3283:
3278:
3273:
3268:
3263:
3258:
3252:
3250:
3244:
3243:
3240:
3239:
3237:
3236:
3231:
3226:
3220:
3218:
3212:
3211:
3209:
3208:
3207:
3206:
3196:
3191:
3186:
3181:
3176:
3171:
3165:
3159:
3153:
3152:
3150:
3149:
3144:
3139:
3137:United Kingdom
3133:
3131:
3125:
3124:
3122:
3121:
3116:
3111:
3106:
3101:
3096:
3091:
3086:
3081:
3076:
3071:
3066:
3061:
3056:
3051:
3046:
3041:
3036:
3031:
3026:
3021:
3016:
3015:
3014:
3004:
2999:
2994:
2989:
2984:
2979:
2974:
2968:
2966:
2960:
2959:
2957:
2956:
2951:
2946:
2941:
2936:
2931:
2926:
2921:
2916:
2911:
2906:
2901:
2896:
2891:
2886:
2881:
2876:
2871:
2866:
2861:
2856:
2851:
2846:
2841:
2836:
2831:
2825:
2823:
2814:
2808:
2807:
2805:
2804:
2799:
2794:
2789:
2784:
2783:
2782:
2777:
2770:
2765:
2755:
2750:
2748:YAA/Youthforce
2745:
2743:World AIDS Day
2740:
2735:
2730:
2725:
2720:
2715:
2710:
2705:
2700:
2695:
2690:
2685:
2680:
2674:
2672:
2668:
2667:
2665:
2664:
2659:
2654:
2649:
2644:
2639:
2634:
2629:
2624:
2619:
2614:
2609:
2604:
2603:
2602:
2592:
2587:
2582:
2577:
2576:
2575:
2565:
2559:
2557:
2553:
2552:
2550:
2549:
2544:
2539:
2534:
2529:
2527:Berlin Patient
2524:
2519:
2514:
2509:
2508:
2507:
2497:
2491:
2489:
2485:
2484:
2482:
2481:
2476:
2471:
2466:
2461:
2456:
2451:
2446:
2444:Superinfection
2441:
2436:
2431:
2426:
2424:Cardiomyopathy
2421:
2416:
2411:
2406:
2401:
2395:
2393:
2389:
2388:
2386:
2385:
2380:
2379:
2378:
2376:Teens / Adults
2373:
2363:
2362:
2361:
2356:
2351:
2346:
2341:
2336:
2331:
2321:
2320:
2319:
2314:
2309:
2304:
2299:
2294:
2283:
2281:
2277:
2276:
2266:
2264:
2263:
2256:
2249:
2241:
2235:
2234:
2225:
2224:
2223:
2207:
2199:
2198:External links
2196:
2194:
2193:
2167:
2146:
2125:
2099:
2073:
2047:
2021:
1995:
1973:
1961:www.ouh.nhs.uk
1948:
1937:. M.news24.com
1925:
1900:
1888:
1839:
1807:
1758:
1726:
1698:
1681:"View Article"
1672:
1661:on 11 May 2009
1640:
1604:
1555:
1542:
1501:
1457:
1432:Ann Intern Med
1418:
1383:
1362:(12): 1080–5.
1342:
1301:
1276:Am J Epidemiol
1266:
1230:
1221:
1212:
1190:
1174:
1152:
1130:
1109:
1082:(4): 297–301.
1066:
1040:
1013:
1001:
982:
960:
938:
896:
883:
881:
878:
872:
869:
857:gold standards
840:
839:AIDS denialism
837:
830:
829:
810:
808:
801:
795:
792:
790:
787:
753:
750:
744:
741:
731:color reaction
678:
675:
669:
666:
644:
641:
591:false negative
583:false positive
566:
563:
562:
561:
532:
529:
474:
471:
422:
419:
398:
395:
361:
358:
342:seroconversion
332:false negative
327:
324:
319:
318:Antibody tests
316:
303:
300:
294:
291:
281:
278:
277:
276:
269:
266:
247:
244:
219:
216:
180:
177:
175:
172:
160:
159:
156:False negative
153:
150:False positive
143:
142:
136:
114:
111:
103:Main article:
100:
99:AIDS diagnosis
97:
56:
55:
45:
37:
36:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
3734:
3723:
3720:
3718:
3715:
3714:
3712:
3697:
3696:Procalcitonin
3694:
3692:
3689:
3688:
3686:
3682:
3676:
3673:
3671:
3668:
3666:
3663:
3661:
3658:
3656:
3655:Blood culture
3653:
3652:
3650:
3648:
3644:
3636:
3633:
3631:
3628:
3627:
3626:
3625:
3621:
3617:
3614:
3613:
3612:
3611:toxoplasmosis
3609:
3608:
3606:
3604:
3600:
3594:
3591:
3587:
3584:
3583:
3582:
3581:
3577:
3575:
3574:Cowdry bodies
3572:
3568:
3565:
3564:
3563:
3562:
3558:
3554:
3551:
3549:
3546:
3544:
3543:Downie bodies
3541:
3540:
3539:
3536:
3532:
3529:
3528:
3527:
3526:
3522:
3518:
3515:
3514:
3513:
3510:
3506:
3503:
3502:
3501:
3498:
3494:
3491:
3489:
3486:
3485:
3484:
3481:
3480:
3478:
3476:
3472:
3464:
3461:
3460:
3459:
3458:
3457:Streptococcus
3454:
3450:
3447:
3446:
3445:
3444:
3440:
3436:
3433:
3432:
3431:
3430:
3426:
3422:
3419:
3417:
3414:
3412:
3409:
3407:
3404:
3402:
3399:
3398:
3397:
3394:
3393:
3391:
3389:
3385:
3381:
3377:
3370:
3365:
3363:
3358:
3356:
3351:
3350:
3347:
3335:
3332:
3330:
3327:
3326:
3323:
3313:
3310:
3308:
3305:
3303:
3300:
3299:
3297:
3293:
3287:
3284:
3282:
3279:
3277:
3274:
3272:
3269:
3267:
3264:
3262:
3259:
3257:
3254:
3253:
3251:
3249:
3248:South America
3245:
3235:
3232:
3230:
3227:
3225:
3222:
3221:
3219:
3217:
3213:
3205:
3204:New York City
3202:
3201:
3200:
3199:United States
3197:
3195:
3192:
3190:
3187:
3185:
3182:
3180:
3177:
3175:
3172:
3170:
3167:
3166:
3163:
3160:
3158:
3157:North America
3154:
3148:
3145:
3143:
3140:
3138:
3135:
3134:
3132:
3130:
3126:
3120:
3117:
3115:
3112:
3110:
3107:
3105:
3102:
3100:
3097:
3095:
3092:
3090:
3087:
3085:
3082:
3080:
3077:
3075:
3072:
3070:
3067:
3065:
3062:
3060:
3057:
3055:
3052:
3050:
3047:
3045:
3042:
3040:
3037:
3035:
3032:
3030:
3027:
3025:
3022:
3020:
3017:
3013:
3010:
3009:
3008:
3005:
3003:
3000:
2998:
2995:
2993:
2990:
2988:
2985:
2983:
2980:
2978:
2975:
2973:
2970:
2969:
2967:
2965:
2961:
2955:
2952:
2950:
2947:
2945:
2942:
2940:
2937:
2935:
2932:
2930:
2927:
2925:
2922:
2920:
2917:
2915:
2912:
2910:
2907:
2905:
2902:
2900:
2897:
2895:
2892:
2890:
2887:
2885:
2882:
2880:
2877:
2875:
2872:
2870:
2867:
2865:
2862:
2860:
2857:
2855:
2852:
2850:
2847:
2845:
2842:
2840:
2837:
2835:
2832:
2830:
2827:
2826:
2824:
2822:
2818:
2815:
2813:
2809:
2803:
2800:
2798:
2795:
2793:
2790:
2788:
2785:
2781:
2778:
2776:
2775:
2771:
2769:
2766:
2764:
2761:
2760:
2759:
2756:
2754:
2751:
2749:
2746:
2744:
2741:
2739:
2736:
2734:
2731:
2729:
2726:
2724:
2721:
2719:
2716:
2714:
2711:
2709:
2706:
2704:
2701:
2699:
2696:
2694:
2691:
2689:
2686:
2684:
2681:
2679:
2676:
2675:
2673:
2669:
2663:
2660:
2658:
2655:
2653:
2650:
2648:
2645:
2643:
2640:
2638:
2635:
2633:
2632:Sex education
2630:
2628:
2625:
2623:
2620:
2618:
2615:
2613:
2610:
2608:
2605:
2601:
2598:
2597:
2596:
2593:
2591:
2588:
2586:
2583:
2581:
2578:
2574:
2571:
2570:
2569:
2566:
2564:
2561:
2560:
2558:
2554:
2548:
2545:
2543:
2540:
2538:
2535:
2533:
2530:
2528:
2525:
2523:
2520:
2518:
2515:
2513:
2510:
2506:
2503:
2502:
2501:
2498:
2496:
2493:
2492:
2490:
2486:
2480:
2477:
2475:
2472:
2470:
2467:
2465:
2462:
2460:
2457:
2455:
2452:
2450:
2447:
2445:
2442:
2440:
2437:
2435:
2432:
2430:
2427:
2425:
2422:
2420:
2417:
2415:
2414:Lipodystrophy
2412:
2410:
2407:
2405:
2402:
2400:
2397:
2396:
2394:
2390:
2384:
2381:
2377:
2374:
2372:
2369:
2368:
2367:
2364:
2360:
2357:
2355:
2352:
2350:
2347:
2345:
2342:
2340:
2337:
2335:
2332:
2330:
2327:
2326:
2325:
2322:
2318:
2315:
2313:
2310:
2308:
2305:
2303:
2300:
2298:
2295:
2293:
2290:
2289:
2288:
2285:
2284:
2282:
2278:
2273:
2269:
2262:
2257:
2255:
2250:
2248:
2243:
2242:
2239:
2233:
2229:
2226:
2222:
2218:
2214:
2211:
2210:
2208:
2205:
2202:
2201:
2197:
2181:
2177:
2171:
2168:
2156:
2150:
2147:
2135:
2129:
2126:
2113:
2109:
2103:
2100:
2087:
2083:
2077:
2074:
2061:
2057:
2051:
2048:
2035:
2031:
2025:
2022:
2010:
2006:
1999:
1996:
1983:
1977:
1974:
1962:
1958:
1952:
1949:
1936:
1929:
1926:
1921:
1917:
1916:
1911:
1904:
1901:
1897:
1892:
1889:
1884:
1880:
1876:
1872:
1867:
1862:
1858:
1854:
1850:
1843:
1840:
1824:
1817:
1811:
1808:
1803:
1799:
1794:
1789:
1785:
1781:
1777:
1773:
1769:
1762:
1759:
1743:
1736:
1730:
1727:
1714:
1713:
1708:
1702:
1699:
1686:
1682:
1676:
1673:
1660:
1656:
1655:
1650:
1644:
1641:
1636:
1632:
1628:
1624:
1621:(2): 327–31.
1620:
1616:
1608:
1605:
1600:
1596:
1592:
1588:
1583:
1578:
1574:
1570:
1566:
1559:
1556:
1553:1990; 39:380.
1552:
1546:
1543:
1538:
1534:
1529:
1524:
1520:
1516:
1512:
1505:
1502:
1497:
1493:
1488:
1483:
1479:
1475:
1471:
1464:
1462:
1458:
1453:
1449:
1445:
1441:
1438:(8): 617–21.
1437:
1433:
1425:
1423:
1419:
1414:
1410:
1406:
1402:
1399:(15): 961–4.
1398:
1394:
1387:
1384:
1379:
1375:
1370:
1365:
1361:
1357:
1353:
1346:
1343:
1338:
1334:
1329:
1324:
1320:
1316:
1312:
1305:
1302:
1297:
1293:
1289:
1285:
1281:
1277:
1270:
1267:
1262:
1258:
1254:
1250:
1246:
1242:
1234:
1231:
1225:
1222:
1216:
1213:
1201:
1194:
1191:
1186:
1178:
1175:
1162:
1156:
1153:
1140:
1134:
1131:
1126:
1122:
1116:
1114:
1110:
1105:
1101:
1097:
1093:
1089:
1085:
1081:
1077:
1070:
1067:
1054:
1050:
1044:
1041:
1036:
1032:
1028:
1024:
1017:
1014:
1010:
1005:
1002:
998:
994:
991:
986:
983:
970:
964:
961:
948:
942:
939:
934:
930:
926:
922:
918:
914:
910:
903:
901:
897:
893:
888:
885:
879:
877:
870:
868:
866:
862:
858:
854:
850:
846:
838:
836:
826:
814:
809:
800:
799:
793:
788:
786:
782:
778:
774:
771:
765:
763:
759:
751:
749:
742:
739:
734:
732:
727:
722:
720:
716:
712:
708:
704:
700:
696:
692:
687:
676:
674:
667:
665:
661:
657:
654:
650:
643:Antigen tests
642:
640:
636:
632:
631:
627:
621:
620:United States
616:
614:
609:
605:
599:
596:
592:
588:
584:
580:
574:
570:
564:
558:
557:
556:
552:
550:
546:
542:
537:
530:
528:
526:
521:
517:
515:
511:
507:
503:
499:
491:
484:
479:
472:
470:
468:
464:
459:
457:
451:
449:
444:
439:
436:
427:
420:
418:
416:
412:
408:
404:
396:
394:
391:
387:
383:
378:
373:
371:
367:
359:
357:
355:
351:
346:
343:
339:
338:
337:window period
333:
326:Window period
325:
323:
317:
315:
313:
309:
301:
299:
292:
290:
286:
279:
274:
270:
267:
264:
261:
260:
259:
257:
253:
245:
243:
241:
237:
233:
229:
225:
217:
215:
213:
208:
206:
202:
198:
194:
190:
186:
178:
173:
171:
169:
165:
157:
154:
151:
148:
147:
146:
140:
137:
134:
131:
130:
129:
126:
124:
120:
119:window period
112:
110:
106:
98:
96:
94:
90:
86:
82:
78:
74:
70:
66:
62:
52:
48:
43:
38:
33:
30:
19:
18:HIV screening
3622:
3580:yellow fever
3578:
3567:Negri bodies
3559:
3523:
3512:Dengue fever
3482:
3455:
3443:Helicobacter
3441:
3427:
3099:Taiwan (ROC)
3089:Saudi Arabia
2939:South Africa
2772:
2758:Larry Kramer
2500:Epidemiology
2328:
2221:Home testing
2184:. Retrieved
2179:
2170:
2158:. Retrieved
2149:
2137:. Retrieved
2128:
2116:. Retrieved
2112:the original
2102:
2090:. Retrieved
2086:the original
2076:
2064:. Retrieved
2060:the original
2050:
2038:. Retrieved
2034:the original
2024:
2014:10 September
2012:. Retrieved
2008:
1998:
1986:. Retrieved
1976:
1964:. Retrieved
1960:
1951:
1939:. Retrieved
1935:"Die Burger"
1933:Die Burger.
1928:
1915:The Guardian
1913:
1903:
1891:
1856:
1852:
1842:
1830:. Retrieved
1823:the original
1810:
1775:
1771:
1761:
1749:. Retrieved
1742:the original
1729:
1717:. Retrieved
1710:
1701:
1689:. Retrieved
1685:the original
1675:
1663:. Retrieved
1659:the original
1652:
1643:
1618:
1615:J Infect Dis
1614:
1607:
1575:(11): 1258.
1572:
1568:
1558:
1550:
1545:
1521:(9): 593–8.
1518:
1515:N Engl J Med
1514:
1504:
1477:
1474:N Engl J Med
1473:
1435:
1431:
1396:
1393:N Engl J Med
1392:
1386:
1359:
1355:
1345:
1321:(1): 55–73.
1318:
1314:
1304:
1279:
1275:
1269:
1247:(1): 63–69.
1244:
1240:
1233:
1224:
1215:
1203:. Retrieved
1193:
1184:
1177:
1165:. Retrieved
1155:
1143:. Retrieved
1133:
1125:the original
1079:
1075:
1069:
1057:. Retrieved
1053:the original
1043:
1029:(6): 223–4.
1026:
1022:
1016:
1004:
985:
973:. Retrieved
963:
951:. Retrieved
941:
919:(1): 53–59.
916:
912:
887:
874:
861:microbiology
842:
833:
820:
812:
783:
779:
775:
766:
757:
755:
746:
723:
717:to specific
688:
680:
671:
662:
658:
646:
628:
617:
600:
576:
572:
568:
553:
548:
544:
540:
538:
534:
525:western blot
522:
518:
496:
466:
460:
452:
440:
435:western blot
432:
421:Western blot
400:
397:ELISA dongle
374:
368:(ELISA), or
363:
347:
335:
329:
321:
305:
296:
287:
283:
263:Confidential
252:human rights
249:
246:Human rights
240:western blot
221:
209:
197:nucleic acid
182:
161:
144:
127:
116:
108:
60:
59:
29:
3670:Parasitemia
3593:Tzanck test
3483:HIV testing
3376:Blood tests
3307:New Zealand
3179:El Salvador
3084:Philippines
3054:North Korea
3007:China (PRC)
2972:Afghanistan
2662:Product Red
2563:AIDS orphan
2419:Nephropathy
2354:vaccination
2182:. June 2005
762:CD4 T-cells
660:performed.
649:p24 protein
587:sensitivity
579:specificity
502:qualitative
228:specificity
224:sensitivity
201:blood banks
185:sensitivity
139:Specificity
133:Sensitivity
113:Terminology
3711:Categories
3429:Rickettsia
3019:East Timor
2992:Bangladesh
2982:Azerbaijan
2904:Mozambique
2889:Madagascar
2622:Red ribbon
2464:Serostatus
2392:Conditions
2344:prevention
2334:management
2292:Lentivirus
2186:5 November
2092:2 November
2066:2 November
2040:2 November
2009:TheBodyPro
1988:2 November
1941:2 November
1832:4 December
1691:2 November
1665:2 November
1480:(1): 1–5.
1205:6 February
1059:2 November
975:12 October
953:12 October
880:References
823:March 2016
794:Oral tests
789:Criticisms
726:centrifuge
715:hybridized
514:algorithms
415:smartphone
174:Principles
85:antibodies
3302:Australia
3256:Argentina
3216:Caribbean
3194:Nicaragua
3184:Guatemala
3094:Sri Lanka
3029:Indonesia
2812:Locations
2753:"Free Me"
2479:Pregnancy
2474:Nutrition
2329:diagnosis
1984:. Cdc.gov
933:1058-4838
743:Screening
633:is being
390:substrate
232:algorithm
61:HIV tests
3722:HIV/AIDS
3675:Algaemia
3665:Fungemia
3396:syphilis
3271:Colombia
3189:Honduras
3104:Thailand
3079:Pakistan
3064:Malaysia
3002:Cambodia
2954:Zimbabwe
2934:Tanzania
2859:Ethiopia
2854:Eswatini
2839:Botswana
2728:Solidays
2627:Safe sex
2568:Religion
2512:Timeline
2434:Pruritus
2371:Children
2349:research
2324:HIV/AIDS
2302:subtypes
2160:13 March
2139:13 March
2118:13 March
1966:19 April
1920:Archived
1883:33073998
1875:12960819
1802:10698998
1599:22007455
1337:15998755
1261:10174294
1096:17438679
1035:16875013
993:Archived
626:template
549:negative
545:positive
483:OraQuick
469:(RIBA).
443:proteins
411:syphilis
189:antibody
89:antigens
69:HIV/AIDS
51:Ethiopia
3684:General
3660:Viremia
3624:malaria
3525:measles
3295:Oceania
3286:Uruguay
3261:Bolivia
3229:Jamaica
3147:Ukraine
3119:Vietnam
2987:Bahrain
2977:Armenia
2929:Senegal
2919:Nigeria
2909:Namibia
2884:Lesotho
2671:Culture
2652:HIV.gov
2495:History
2488:History
1751:8 March
1719:8 March
1635:8335969
1591:9359701
1569:Nat Med
1537:1812850
1496:2046708
1452:2648922
1413:3419477
1378:9757856
1296:7539579
1104:1898168
813:updated
711:primers
639:
193:antigen
3561:rabies
3276:Guyana
3266:Brazil
3174:Mexico
3169:Canada
3142:Russia
3129:Europe
3114:Turkey
3049:Jordan
3012:Yunnan
2997:Bhutan
2949:Zambia
2944:Uganda
2924:Rwanda
2894:Malawi
2869:Guinea
2829:Angola
2821:Africa
2768:ACT UP
2556:Social
2274:topics
1881:
1873:
1800:
1790:
1633:
1597:
1589:
1535:
1494:
1450:
1411:
1376:
1335:
1294:
1259:
1167:10 May
1145:10 May
1102:
1094:
1033:
931:
705:. The
695:plasma
407:dongle
386:enzyme
77:saliva
3493:mChip
3224:Haiti
3074:Nepal
3044:Japan
3024:India
2914:Niger
2879:Kenya
2864:Ghana
2849:Egypt
2834:Benin
2206:– FDA
1879:S2CID
1826:(PDF)
1819:(PDF)
1793:86341
1745:(PDF)
1738:(PDF)
1595:S2CID
1100:S2CID
859:" in
684:HIV-1
624:‹The
498:Rapid
377:serum
360:ELISA
236:ELISA
91:, or
81:urine
79:, or
73:serum
71:, in
3378:for
3281:Peru
3059:Laos
3039:Iraq
3034:Iran
2964:Asia
2899:Mali
2802:Ayds
2359:PrEP
2272:AIDS
2188:2014
2162:2012
2141:2012
2120:2012
2094:2013
2068:2013
2042:2013
2016:2020
1990:2013
1968:2021
1943:2013
1871:PMID
1853:AIDS
1834:2013
1798:PMID
1753:2017
1721:2017
1693:2013
1667:2013
1631:PMID
1587:PMID
1533:PMID
1492:PMID
1448:PMID
1409:PMID
1374:PMID
1356:JAMA
1333:PMID
1292:PMID
1257:PMID
1207:2015
1169:2013
1147:2013
1092:PMID
1061:2013
1031:PMID
977:2013
955:2013
929:ISSN
756:The
703:cDNA
577:The
364:The
226:and
195:and
2287:HIV
2280:HIV
2268:HIV
1861:doi
1788:PMC
1780:doi
1623:doi
1619:168
1577:doi
1523:doi
1519:325
1482:doi
1478:325
1440:doi
1436:110
1401:doi
1397:319
1364:doi
1360:280
1323:doi
1319:143
1284:doi
1280:141
1249:doi
1084:doi
921:doi
853:PCR
851:by
849:RNA
691:RNA
541:not
456:POL
212:EIA
93:RNA
3713::
2219:–
2215:–
2178:.
2007:.
1959:.
1918:.
1912:.
1877:.
1869:.
1857:17
1855:.
1851:.
1796:.
1786:.
1776:38
1774:.
1770:.
1709:.
1651:.
1629:.
1617:.
1593:.
1585:.
1571:.
1567:.
1531:.
1517:.
1513:.
1490:.
1476:.
1472:.
1460:^
1446:.
1434:.
1421:^
1407:.
1395:.
1372:.
1358:.
1354:.
1331:.
1317:.
1313:.
1290:.
1278:.
1255:.
1245:18
1243:.
1112:^
1098:.
1090:.
1080:74
1078:.
1027:89
1025:.
927:.
917:64
915:.
911:.
899:^
637:.›
527:.
191:,
95:.
87:,
75:,
3368:e
3361:t
3354:v
2270:/
2260:e
2253:t
2246:v
2190:.
2164:.
2143:.
2122:.
2096:.
2070:.
2044:.
2018:.
1992:.
1970:.
1945:.
1885:.
1863::
1836:.
1804:.
1782::
1755:.
1723:.
1695:.
1669:.
1637:.
1625::
1601:.
1579::
1573:3
1539:.
1525::
1498:.
1484::
1454:.
1442::
1415:.
1403::
1380:.
1366::
1339:.
1325::
1298:.
1286::
1263:.
1251::
1209:.
1171:.
1149:.
1106:.
1086::
1063:.
1037:.
979:.
957:.
935:.
923::
825:)
821:(
815:.
380:"
265:;
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.