321:
38:
212:
719:
359:
347:
136:
128:
302:, is angled upward, more liquid will have a chance to condense and flow back into the boiler leading to increased reflux. Typical results can increase production as high as 50% over the basic worm type condenser. The addition of a copper "boiling ball" in the path creates an area where expansion of gasses into the ball causes cooling and subsequent condensation and reflux. In a
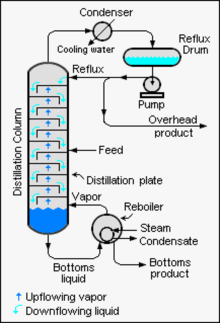
260:, where it cools until it condenses into a liquid. The separation can be enhanced with the addition of more trays (to a practical limitation of heat, flow, etc.). The process continues until all the most volatile components in the liquid feed boil out of the mixture. This point can be recognized by the rise in temperature shown on the thermometer. For
247:
on the glass platforms (known as plates or trays) inside the column and runs back down into the liquid below, thereby refluxing the upflowing distillate vapor. The hottest tray is at the bottom of the column and the coolest tray is at the top. At steady state conditions, the vapor and liquid on each
107:
In that context, reflux refers to the portion of the overhead liquid product from a distillation column or fractionator that is returned to the upper part of the column as shown in the schematic diagram of a typical industrial distillation column. Inside the column, the downflowing reflux liquid
163:
the reaction mixture; vapours produced from the mixture are condensed by the condenser, and return to the vessel through gravity. The purpose is to thermally accelerate the reaction by conducting it at an elevated, controlled temperature (i.e. the
320:
119:, the better is the column's separation of lower boiling materials from higher boiling materials. Conversely, for a given desired separation, the more reflux is provided, the fewer theoretical plates are required.
288:), or post distillation flavored spirits (gin, absinthe), a process of multiple distillations or charcoal filtering may be applied to obtain a product lacking in any suggestion of its original source material for
276:
may be used to ensure that higher boiling point components are returned to the flask while lighter elements are passed out to a secondary condenser. This is useful in producing high quality
536:
540:
478:
346:
650:
563:
452:
358:
306:, the addition of inert materials in the column (e.g., packing) creates surfaces for early condensation and leads to increased reflux.
718:
512:
487:
427:
881:
52:
of vapors and the return of this condensate to the system from which it originated. It is used in industrial and laboratory
876:
708:
249:
871:
779:
643:
257:
693:
37:
835:
810:
590:
825:
820:
800:
738:
383:
261:
232:
85:
28:
211:
805:
156:
866:
830:
636:
176:
743:
622:
393:
388:
240:
224:
81:
845:
418:
289:
235:. The liquid feed mixture to be distilled is placed into the round-bottomed flask along with a few
159:, which is typically open to the atmosphere at the top. The reaction vessel is heated in order to
840:
733:
556:
Chemical engineering design : principles, practice and economics of plant and process design
530:
445:
Handbook of laboratory distillation : with an introduction into the pilot plant distillation
378:
243:
is fitted into the top. As the mixture is heated and boils, vapor rises up the column. The vapor
152:
27:
This article is about using reflux in chemical engineering and chemistry. For other usage, see
774:
698:
569:
559:
518:
508:
483:
458:
448:
423:
144:
116:
65:
815:
703:
220:
216:
135:
688:
292:. The geometry of the still also plays a role in determining how much reflux occurs. In a
277:
678:
673:
200:
97:
112:
of the upflowing vapors thereby increasing the efficiency of the distillation column.
860:
748:
281:
256:
form all the way to the top. The vapor at the top of the column then passes into the
184:
169:
93:
618:
659:
334:
244:
236:
231:
The apparatus shown in the diagram represents a batch distillation as opposed to a
109:
89:
53:
49:
272:
By controlling the temperature of the condenser, often called a dephlegmator, a
101:
284:) are returned to the primary flask. For high quality neutral spirits (such as
215:
Laboratory apparatus using reflux to supply energy to chemical reactions. An
573:
462:
769:
337:
294:
196:
192:
180:
57:
522:
127:
447:( completely rev. 2nd ed.). Amsterdam: Elsevier Scientific Pub. Co.
188:
17:
764:
326:
223:
is used to carry out the condensation. Here the distillation head and
172:) and ambient pressure without losing large quantities of the mixture.
165:
160:
148:
330:
61:
340:
before it is distilled to give pure oxygen- and water-free toluene.
784:
285:
210:
187:
is not generally suitable, and alternatives such as a water bath,
134:
126:
36:
34:
Condensation of vapors and their return to where they originated
632:
628:
253:
41:
The reflux system in a typical industrial distillation column
558:. Sinnott, R. K. Amsterdam: Elsevier/Butterworth-Heinemann.
298:, if the tube leading from the boiler to the condenser, the
175:
The diagram shows a typical reflux apparatus. It includes a
131:
Laboratory reflux apparatus for heating a chemical reaction
80:
is very widely used in industries that utilize large-scale
179:
to indirectly heat the mixture. As many solvents used are
280:, while ensuring that less desirable components (such as
352:
Industrial fractionating columns all of which use reflux
264:, the feed mixture enters in the middle of the column.
595:
University of
Toronto Scarborough - Chemistry Online
793:
757:
726:
666:
252:. Only the most volatile of the vapors stays in
115:The more reflux provided for a given number of
644:
503:King, C. Judson (Cary Judson), 1934- (1980).
476:Perry, Robert H. & Green, Don W. (1984).
155:. This vessel is connected to a water-cooled
8:
535:: CS1 maint: multiple names: authors list (
585:
583:
651:
637:
629:
539:) CS1 maint: numeric names: authors list (
151:is placed in a suitable vessel, such as a
411:
409:
364:Organic synthesis apparatus using reflux
405:
316:
528:
507:(2d ed.). New York: McGraw-Hill.
219:is used as a receiving flask, while a
7:
479:Perry's Chemical Engineers' Handbook
25:
207:Reflux in laboratory distillation
72:Reflux in industrial distillation
717:
357:
345:
319:
268:Reflux in beverage distillation
619:Distillation column components
1:
482:(6th ed.). McGraw-Hill.
422:(1st ed.). McGraw-Hill.
48:is a technique involving the
139:Laboratory reflux apparatus.
123:Reflux in chemical reactions
68:over a long period of time.
898:
780:Spinning band distillation
227:are combined in one piece.
26:
715:
554:Towler, Gavin P. (2008).
416:Kister, Henry Z. (1992).
709:Vapor–liquid equilibrium
183:, direct heating with a
739:Continuous distillation
384:Fractional distillation
262:continuous distillation
233:continuous distillation
29:Reflux (disambiguation)
443:Krell, Erich. (1982).
228:
140:
132:
42:
882:Laboratory techniques
237:anti-bumping granules
214:
138:
130:
108:provides cooling and
56:. It is also used in
40:
877:Industrial processes
744:Fractionating column
727:Industrial processes
694:McCabe–Thiele method
623:Newcastle University
505:Separation processes
394:McCabe-Thiele method
389:Fractionating column
241:fractionating column
225:fractionating column
90:petroleum refineries
82:distillation columns
419:Distillation Design
278:alcoholic beverages
104:processing plants.
872:Chemical processes
758:Laboratory methods
734:Batch distillation
379:Batch distillation
229:
197:electric hot plate
153:round bottom flask
141:
133:
117:theoretical plates
43:
854:
853:
775:Rotary evaporator
699:Theoretical plate
625:, United Kingdom.
621:, Dr. Ming Tham,
591:"What is Reflux?"
565:978-0-08-055695-6
454:978-0-08-087549-1
329:is refluxed with
16:(Redirected from
889:
721:
704:Partial pressure
653:
646:
639:
630:
606:
605:
603:
601:
587:
578:
577:
551:
545:
544:
534:
526:
500:
494:
493:
473:
467:
466:
440:
434:
433:
413:
361:
349:
323:
221:Liebig condenser
217:Erlenmeyer flask
21:
897:
896:
892:
891:
890:
888:
887:
886:
857:
856:
855:
850:
789:
753:
722:
713:
689:Fenske equation
662:
657:
615:
613:Further reading
610:
609:
599:
597:
589:
588:
581:
566:
553:
552:
548:
527:
515:
502:
501:
497:
490:
475:
474:
470:
455:
442:
441:
437:
430:
415:
414:
407:
402:
375:
370:
369:
368:
365:
362:
353:
350:
341:
324:
312:
270:
209:
125:
98:chemical plants
74:
35:
32:
23:
22:
15:
12:
11:
5:
895:
893:
885:
884:
879:
874:
869:
859:
858:
852:
851:
849:
848:
843:
838:
833:
828:
823:
818:
813:
808:
803:
797:
795:
791:
790:
788:
787:
782:
777:
772:
767:
761:
759:
755:
754:
752:
751:
746:
741:
736:
730:
728:
724:
723:
716:
714:
712:
711:
706:
701:
696:
691:
686:
681:
676:
670:
668:
664:
663:
658:
656:
655:
648:
641:
633:
627:
626:
614:
611:
608:
607:
579:
564:
546:
513:
495:
488:
468:
453:
435:
428:
404:
403:
401:
398:
397:
396:
391:
386:
381:
374:
371:
367:
366:
363:
356:
354:
351:
344:
342:
325:
318:
315:
314:
313:
311:
308:
282:fusel alcohols
269:
266:
208:
205:
203:are employed.
201:heating mantle
124:
121:
73:
70:
33:
24:
14:
13:
10:
9:
6:
4:
3:
2:
894:
883:
880:
878:
875:
873:
870:
868:
865:
864:
862:
847:
844:
842:
839:
837:
834:
832:
829:
827:
824:
822:
819:
817:
814:
812:
809:
807:
804:
802:
799:
798:
796:
792:
786:
783:
781:
778:
776:
773:
771:
768:
766:
763:
762:
760:
756:
750:
749:Spinning cone
747:
745:
742:
740:
737:
735:
732:
731:
729:
725:
720:
710:
707:
705:
702:
700:
697:
695:
692:
690:
687:
685:
682:
680:
677:
675:
672:
671:
669:
665:
661:
654:
649:
647:
642:
640:
635:
634:
631:
624:
620:
617:
616:
612:
596:
592:
586:
584:
580:
575:
571:
567:
561:
557:
550:
547:
542:
538:
532:
524:
520:
516:
514:0-07-034612-7
510:
506:
499:
496:
491:
489:0-07-049479-7
485:
481:
480:
472:
469:
464:
460:
456:
450:
446:
439:
436:
431:
429:0-07-034909-6
425:
421:
420:
412:
410:
406:
399:
395:
392:
390:
387:
385:
382:
380:
377:
376:
372:
360:
355:
348:
343:
339:
336:
332:
328:
322:
317:
309:
307:
305:
301:
297:
296:
291:
287:
283:
279:
275:
267:
265:
263:
259:
255:
251:
246:
242:
238:
234:
226:
222:
218:
213:
206:
204:
202:
198:
194:
190:
186:
185:Bunsen burner
182:
178:
173:
171:
170:boiling point
167:
162:
158:
154:
150:
146:
143:A mixture of
137:
129:
122:
120:
118:
113:
111:
105:
103:
99:
95:
94:petrochemical
91:
87:
86:fractionators
83:
79:
71:
69:
67:
63:
59:
55:
54:distillations
51:
47:
39:
30:
19:
867:Distillation
846:Vacuum-based
683:
679:Dalton's law
674:Raoult's law
660:Distillation
598:. Retrieved
594:
555:
549:
504:
498:
477:
471:
444:
438:
417:
335:benzophenone
304:column still
303:
299:
293:
290:fermentation
274:reflux still
273:
271:
230:
174:
142:
114:
110:condensation
106:
77:
75:
50:condensation
45:
44:
841:Steam-based
836:Salt-effect
811:Destructive
600:October 21,
250:equilibrium
248:tray is at
102:natural gas
861:Categories
826:Fractional
821:Extractive
801:Azeotropic
794:Techniques
667:Principles
400:References
239:, and the
177:water bath
60:to supply
806:Catalytic
770:Kugelrohr
574:191735762
531:cite book
463:305628802
338:desiccant
295:pot still
258:condenser
245:condenses
193:sand bath
181:flammable
157:condenser
145:reactants
76:The term
66:reactions
58:chemistry
18:Refluxing
831:Reactive
373:See also
300:lyne arm
189:oil bath
88:such as
765:Alembic
523:4882985
327:Toluene
310:Gallery
254:gaseous
166:solvent
149:solvent
684:Reflux
572:
562:
521:
511:
486:
461:
451:
426:
331:sodium
100:, and
78:reflux
62:energy
46:Reflux
785:Still
286:vodka
602:2017
570:OCLC
560:ISBN
541:link
537:link
519:OCLC
509:ISBN
484:ISBN
459:OCLC
449:ISBN
424:ISBN
161:boil
147:and
96:and
84:and
816:Dry
199:or
168:'s
64:to
863::
593:.
582:^
568:.
533:}}
529:{{
517:.
457:.
408:^
195:,
191:,
92:,
652:e
645:t
638:v
604:.
576:.
543:)
525:.
492:.
465:.
432:.
333:-
31:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.