540:
3591:
426:
1431:
1172:
383:
2762:
619:
880:
3214:
3718:
596:
2925:
2572:
2360:
3420:
813:
178:
3164:
1032:
687:
265:
1043:
1509:. Whilst they have found that these ligands do not activate the metal catalyst as much as phosphine ligands they often result in more robust catalysts. Several catalytic systems have been looked into by Hermann and Enders, using catalysts containing imidazole and triazole carbene ligands, with moderate success. Grubbs has reported replacing a phosphine ligand (PCy
532:
3551:
654:
1126:
3306:
3733:. By way of example, a stable carbene prepared from potassium hydride can be filtered through a dry celite pad to remove excess KH (and resulting salts) from the reaction. On a relatively small scale, a suspension containing a stable carbene in solution can be allowed to settle and the supernatant solution pushed through a dried membrane
3468:
357:
629:-based carbenes are thermodynamically stable and have diagnostic C NMR chemical shift values between 210 and 220 ppm for the carbenic carbon. The X-ray structure of the triphenyl substituted carbene above shows an N–C–N bond angle of around 101°. The 5-methoxytriazole precursor to this carbene was made by the treatment of a
31:
288:
310:
481:
3495:
salt. Removing lithium ions resulting from deprotonation with reagents such as lithium diisopropylamide (LDA) can be especially problematic. Potassium and sodium salt by-products tend to precipitate from solution and can be removed. Lithium ions may be chemically removed by binding to species such as
721:
protection of the carbenic centre is limited especially when the N–C–X unit is part of a ring. These acyclic carbenes have diagnostic C NMR chemical shift values between 250 and 300 ppm for the carbenic carbon, further downfield than any other types of stable carbene. X-ray structures have shown
669:
Diaminocarbenes have diagnostic C NMR chemical shift values between 230 and 270 ppm for the carbenic atom. The X-ray structure of dihydroimidazole-2-ylidene shows a N–C–N bond angle of about 106°, whilst the angle of the acyclic carbene is 121°, both greater than those seen for imidazol-2-ylidenes.
3479:
The preparation of stable carbenes free from metal cations has been keenly sought to allow further study of the carbene species in isolation from these metals. Separating a carbene from a carbene-metal complex can be problematic due to the stability of the complex. Accordingly, it is preferable to
328:
This carbene, the forerunner of a large family of carbenes with the imidazol-2-ylidene core, is indefinitely stable at room temperature in the absence of oxygen and moisture. It melts at 240–241 °C without decomposition. The C NMR spectrum shows a signal at 211 ppm for the carbenic atom. The
3745:
as shown right can be an effective method of purification, although temperatures below 60 °C under high vacuum are preferable as these carbenes are relatively volatile and also could begin to decompose at these higher temperatures. Indeed, sublimation in some cases can give single crystals
661:
Unlike the aromatic imidazol-2-ylidenes or triazol-5-ylidenes, these carbenes appear not to be thermodynamically stable, as shown by the dimerisation of some unhindered cyclic and acyclic examples. Studies suggest that these carbenes dimerise via acid catalysed dimerisation (as in the
650:'s research group. In some of these compounds, the N–C–N unit is a member of a five- or six-membered non-aromatic ring, including a bicyclic example. In other examples, the adjacent nitrogens are connected only through the carbenic atom, and may or may not be part of separate rings.
933:
3411:
as strong bases has not been extensively studied, and have been unreliable for deprotonation of precursor salts. With non-aromatic salts, n-BuLi and PhLi can act as nucleophiles whilst t-BuLi can on occasion act as a source of hydride, reducing the salt with the generation of
698:. A thiazole based carbene (analogous to the carbene postulated by Breslow) has been prepared and characterised by X-ray crystallography. Other non-aromatic aminocarbenes with O, S and P atoms adjacent (i.e. alpha) to the carbene centre have been prepared, for example,
583:
Imidazole-based carbenes are thermodynamically stable and generally have diagnostic C NMR chemical shift values between 210 and 230 ppm for the carbenic carbon. Typically, X-ray structures of these molecules show N–C–N bond angles of 101–102°.
347:
In 2000, Bertrand obtained additional carbenes of the phosphanyl type, including (phosphanyl)(trifluoromethyl)carbene, stable in solution at -30 °C and a moderately stable (amino)(aryl)carbene with only one heteroatom adjacent to the carbenic atom.
3394:
at −40 °C has been reported for imidazole-based carbenes. Arduengo and coworkers managed to prepare a dihydroimidazol-2-ylidene using NaH. However, this method has not been applied to the preparation of diaminocarbenes. In some cases,
228:
and coworkers in 1964, who presented evidence that the dimer did not dissociate; and by
Winberg in 1965. However, subsequent experiments by Denk, Herrmann and others have confirmed this equilibrium, albeit in specific circumstances.
6289:
Michael Otto; Salvador
Conejero; Yves Canac; Vadim D. Romanenko; Valentyn Rudzevitch; Guy Bertrand (2004). "Mono- and Diaminocarbenes from Chloroiminium and -amidinium Salts: Synthesis of Metal-Free Bis(dimethylamino)carbene".
445:
backbone. The following year, the first acyclic persistent carbene demonstrated that stability did not even require a cyclic backbone. Unhindered derivatives of the hydrogenated and acyclic carbenes dimerized, suggesting that
4180:
H. W. Wanzlick; H. J. Schonherr (1970). "Chemie nucleophiler
Carbene, XVIII, 1) 1.3.4.5-Tetraphenyl-imidazoliumperchlorat" [Chemistry of nucleophilic carbenes, XVIII. 1) 1,3,4,5-Tetraphenylimidazolium perchlorate].
6202:
Roger W. Alder; Michael E. Blake; Simone Bufali; Craig P. Butts; A. Guy Orpen; Jan Schütz; Stuart J. Williams (2001). "Preparation of tetraalkylformamidinium salts and related species as precursors to stable carbenes".
1183:
salts, a protonated precursor species. Accordingly, this reaction can be acid catalysed. This reaction occurs because unlike imidazolium based carbenes, there is no loss of aromaticity in protonation of the carbene.
678:
There exist several variants of the stable carbenes above where one of the nitrogen atoms adjacent to the carbene center (the α nitrogens) has been replaced by an alternative heteroatom, such as oxygen, sulfur, or
3371:. However, these catalysts have proved ineffective for the preparation of non-imidazolium adducts as they tend to act as nucleophiles towards the precursor salts and in so doing are destroyed. The presence of
3574:
Vacuum pyrolysis, with the removal of neutral volatile byproducts i.e. methanol or chloroform, has been used to prepare dihydroimidazole and triazole based carbenes. Historically the removal of chloroform by
1001:-heterocyclic carbenes (rNHC). A variety of free carbenes can be isolated and are stable at room temperature. Other free carbenes are not stable and are susceptible to intermolecular decomposition pathways.
645:
The two families above can be seen as special cases of a broader class of compounds which have a carbenic atom bridging two nitrogen atoms. A range of such diaminocarbenes have been prepared principally by
993:-heterocyclic carbenes (NHCs) except that canonical resonance structures with the carbene depicted cannot be drawn without adding additional charges. Mesoionic carbenes are also referred to as abnormal
1107:
nature of these carbenes, which is lost upon dimerisation. In fact imidazol-2-ylidenes are so thermodynamically stable that only in highly constrained conditions are these carbenes forced to dimerise.
6004:
M. Scholl; T. M. Trnka; J. P. Morgan; R. H. Grubbs (1999). "Increased ring closing metathesis activity of ruthenium-based olefin metathesis catalysts coordinated with imidazolin-2-ylidene ligands".
3480:
make the carbene free from these metals in the first place. Indeed, some metal ions, rather than stabilising the carbene, have been implicated in the catalytic dimerisation of unhindered examples.
1560:
1529:
CHPh, and noted increased ring closing metathesis as well as exhibiting "a remarkable air and water stability". Molecules containing two and three carbene moieties have been prepared as potential
1498:
atoms adjacent to the carbene centre, and so are able to coordinate to even relatively electron deficient metals. Enders and
Hermann have shown that these carbenes are suitable replacements for
5795:
Wolf Janine; Böhlmann
Winfried; Findeisen Matthias; Gelbrich Thomas; Hofmann Hans-Jorg; Schulze Borbel (2007). "Reply to "Recently Reported Crystalline Isothiazole Carbenes: Myth or Reality"".
5417:
Itoh, T; Nakata, Y; Hirai, K; Tomioka, H (2006). "Triplet
Diphenylcarbenes Protected by Trifluoromethyl and Bromine Groups. A Triplet Carbene Surviving a Day in Solution at Room Temperature".
637:. This may indicate that these carbenes are less aromatic than imidazol-2-ylidenes, as the imidazolium precursors do not react with nucleophiles due to the resultant loss of aromaticity.
5904:
D. Enders; H. Gielen; G. Raabe; J. Runsink; J. H. Teles (1996). "Synthesis and
Stereochemistry of the First Chiral (Imidazolinylidene)- and (Triazolinylidene)palladium(II) Complexes".
4478:
Solé, Stéphane; Gornitzka, Heinz; Schoeller, Wolfgang W.; Bourissou, Didier; Bertrand, Guy (2001). "(Amino)(Aryl)Carbenes: Stable
Singlet Carbenes Featuring a Spectator Substituent".
3459:
works very cleanly for the deprotonation of all types of salts, except for unhindered formamidinium salts, where this base can act as a nucleophile to give a triaminomethane adduct.
615:
through loss of methanol from 2-methoxytriazoles. Only a limited range of these molecules have been reported, with the triphenyl substituted molecule being commercially available.
5131:
R. W. Alder; M. E. Blake; C. Bortolotti; S. Buffali; C. P. Butts; E. Lineham; J. M. Oliva; A. G. Orpen; M. J. Quayle (1999). "Complexation of stable carbenes with alkali metals".
3583:
was used by
Wanzlick in his early attempts to prepare dihydroimidazol-2-ylidenes but this method is not widely used. The Enders laboratory has used vacuum pyrolysis of adduct
3535:, is insoluble in the solvent. The elevated temperatures suggest that this method is not suitable for the preparation of unstable dimerising carbenes. A single example of the
3185:
Upon coordination to metal centers, the C carbene resonance usually shifts highfield, depending on the Lewis acidity of the complex fragment. Based on this observation, Huynh
6380:
Carbene
Chemistry: From Fleeting Intermediates to Powerful Reagents, (Chapter 4, Hideo Tomioka (triplet state); Chapter 5 (singlet state), Roger W. Alder) - ed. Guy Bertrand
592:
Depending on the arrangement of the three nitrogen atoms in triazol-5-ylidene, there are two possible isomers, namely 1,2,3-triazol-5-ylidenes and 1,2,4-triazol-5-ylidenes.
952:
and the trifluoromethyl groups on the phenyl rings, that shield the carbene and prevent or slow down the process of dimerization to a 1,1,2,2-tetra(phenyl)alkene. Based on
4433:
Christophe Buron; Heinz Gornitzka; Vadim Romanenko; Guy Bertrand (2000). "Stable Versions of Transient Push-Pull Carbenes: Extending Lifetimes from Nanoseconds to Weeks".
3171:
Those carbenes that have been isolated to date tend to be colorless solids with low melting points. These carbenes tend to sublime at low temperatures under high vacuum.
367:
In the modern understanding, the superficially unoccupied p-orbital on a (meta)stable carbene is not, in fact, fully empty. Instead, the carbene Lewis structures are in
5092:
D. Enders; K. Breuer; G. Raabe; J. Runsink; J. H. Teles; J. P. Melder; K. Ebel; S. Brode (1995). "Preparation, Structure, and Reactivity of 1,3,4-Triphenyl-4,5-dihydro-1
3455:
used to make the lithium amide. Titration of lithium amide can be used to determine the amount of hydroxide in solution. The deprotonation of precursor salts with metal
1553:
295:
These compounds were called "push-pull carbenes" in reference to the contrasting electron affinities of the phosphorus and silicon atoms. They exhibit both carbenic and
3332:
being deprotonated. The major drawback with this method of preparation is the problem of isolation of the free carbene from the metals ions used in their preparation.
1392:) probably proceed via deprotonation, resulting in the generation of a nucleophile (XR) which can attack the generated salt giving the impression of a H–X insertion.
469:. Indeed, most persistent carbenes are stabilized by two flanking nitrogen centers. The outliers include an aminothiocarbene and an aminooxycarbene, which use other
3193:-palladium(II)-carbene complexes. The use of a C-labeled N-heterocyclic carbene ligand also allows for the study of mixed carbene-phosphine complexes, which undergo
4405:
A. J. Arduengo; F. Davidson; H. V. R. Dias; J. R. Goerlich; D. Khasnis; W. J. Marshall; T. K. Prakasha (1997). "An Air Stable Carbene and Mixed Carbene "Dimers"".
1215:
have performed a range of organic reactions involving a triazol-5-ylidene. These reactions are outlined below and may be considered as a model for other carbenes.
1076:
values for the conjugate acids of several NHC families have been examined in aqueous solution. pKa values of triazolium ions lie in the range 16.5–17.8, around 3 p
473:, and room-temperature-stable bis(diisopropylamino)cyclopropenylidene, in which the carbene atom is connected to two carbon atoms in a three-member, aromatic,
1546:
299:
reactivity. An X-ray structure of this molecule has not been obtained and at the time of publication some doubt remained as to their exact carbenic nature.
450:
ImC: might be exceptional, rather than paradigmatic. But the behavior of the acyclic carbenes offered a tantalizing clue to the stabilization mechanism.
1039:
However, further work showed that diaminocarbenes will deprotonate the DMSO solvent, with the resulting anion reacting with the resulting amidinium salt.
3328:
Several bases and reaction conditions have been employed with varying success. The degree of success has been principally dependent on the nature of the
65:), but does not exhibit the tremendous instability typically associated with such moieties. The best-known examples and by far largest subgroup are the
453:
Unlike the cyclic derivatives, acyclic carbenes are flexible and bonds to the carbenic atom admit rotation. But bond rotation in the compound appeared
5524:
Higgins, Eleanor M.; Sherwood, Jennifer A.; Lindsay, Anita G.; Armstrong, James; Massey, Richard S.; Alder, Roger W.; O'Donoghue, Annmarie C. (2011).
99:. Excitation to a carbene structure then accounts for the carbene-like dimerization that some persistent carbenes undergo over the course of days.
1219:
785:. However, these compounds seem to exhibit some alkynic properties, and when published the exact carbenic nature of these red oils was in debate.
509:
ring, with the hydrogen in carbon 2 of the ring (between the two nitrogen atoms) removed, and other hydrogens replaced by various groups. These
5759:
DeHope, A; Lavallo, V; Donnadieu, B; Schoeller, WW; Bertrand, G (2007). "Recently reported crystalline isothiazole carbenes: Myth or reality".
3241:
for cross-coupling reactions. NHC-metal complexes, specifically Ag(I)-NHC complexes have been widely tested for their biological applications.
5483:"Proton Transfer Reactions of Triazol-3-ylidenes: Kinetic Acidities and Carbon Acid pKaValues for Twenty Triazolium Salts in Aqueous Solution"
4338:
Tapu, Daniela; Dixon, David A.; Roe, Christopher (12 August 2009). "13C NMR Spectroscopy of "Arduengo-type" Carbenes and Their Derivatives".
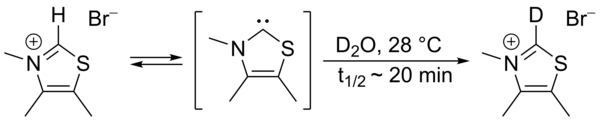
3729:. However, provided rigorously dry, relatively non-acidic and air-free materials are used, stable carbenes are reasonably robust to handling
4987:
W. A. Herrmann; C. Kocher; L. J. Goossen; G. R. J. Artus (1996). "Heterocyclic Carbenes: A High-Yielding Synthesis of Novel, Functionalized
516:
A considerable range of imidazol-2-ylidenes have been synthesised, including those in which the 1,3-positions have been functionalised with
557:
atoms for the two hydrogens at ring positions 4 and 5 yielded the first air-stable carbene. Its extra stability probably results from the
5026:-Heterocyclic Carbenes: Generation under Mild Conditions and Formation of Group 8–10 Transition Metal Complexes Relevant to Catalysis".
3325:
salts. The acyclic carbenes and the tetrahydropyrimidinyl based carbenes were prepared by deprotonation using strong homogeneous bases.
3488:
1471:
of elements gives some idea of the complexes which have been prepared, and in many cases these have been identified by single crystal
694:
In particular, the formal substitution of sulfur for one of the nitrogens in imidazole would yield the aromatic heterocyclic compound
6107:
Díez-González, Silvia; Marion, Nicolas; Nolan, Steven P. (2009-08-12). "N-Heterocyclic Carbenes in Late Transition Metal Catalysis".
1447:
Imidazol-2-ylidenes, triazol-5-ylidenes (and less so, diaminocarbenes) have been shown to coordinate to a plethora of elements, from
6469:
6095:
6075:
6385:
Reactive Intermediate Chemistry By Robert A. Moss, Matthew Platz, Maitland Jones (Chapter 8, Stable Singlet Carbenes, Guy Bertrand)
5976:; Lukas J. Goossen; Christian Köcher; Georg R. J. Artus (1996). "Chiral Heterocylic Carbenes in Asymmetric Homogeneous Catalysis".
4733:
Alder, RW; Chaker, L; Paolini, FP (2004). "Bis(diethylamino)carbene and the mechanism of dimerisation for simple diaminocarbenes".
4155:
3566:
to give imidazol-2-ylidenes or diaminocarbenes has not been widely used. The method was used to prepare dihydroimidazole carbenes.
3352:
by-products and excess hydride being removed by filtration. In practice this reaction is often too slow, requiring the addition of
872:
Although the figure below shows the two parts of the molecule in one flat plane, molecular geometry puts the two aromatic parts in
539:
3118:
250:
5653:
Enders, D.; Breuer, K.; Runsink, J.; Teles, J.H. (1996). "Chemical Reactions of the Stable Carbene 1,3,4-Triphenyl-4,5-dihydro-1
1099:. However, imidazol-2-ylidenes and triazol-5-ylidenes are thermodynamically stable and do not dimerise, and have been stored in
6384:
837:
core, a three-carbon ring with a double bond between the two atoms adjacent to the carbenic one. This family is exemplified by
6205:
5720:
Wolf, J; Böhlmann, W; Findeisen, M; Gelbrich, T; Hofmann, HJ; Schulze, B (2007). "Synthesis of stable isothiazole carbenes".
838:
419:
6159:
Garrison Jered C.; Youngs Wiley J. (2005). "Ag(I) N-Heterocyclic Carbene Complexes: Synthesis, Structure, and Application".
5390:
5331:
Tomioka, H; Iwamoto, E; Itakura, H; Hirai, K (2001). "Generation and characterization of a fairly stable triplet carbene".
3959:
Hans-Werner Wanzlick; E. Schikora (1960). "Ein neuer Zugang zur Carben-Chemie" [A new way into carbene chemistry].
5869:
Gernot Boche; Christof Hilf; Klaus Harms; Michael Marsch; John C. W. Lohrenz (1995). "Crystal Structure of the Dimeric (4-
3738:
3441:
817:
245:, proposed that these imidazole-based carbenes should be more stable than their 4,5-dihydro analogues, due to Hückel-type
3947:
3344:
would be the ideal base for deprotonating these precursor salts. The hydride should react irreversibly with the loss of
6034:"13C NMR Spectroscopic Determination of Ligand Donor Strengths Using N-Heterocyclic Carbene Complexes of Palladium(II)"
3142:. The reactions that such carbenes participate in are very different from those in which organic carbenes participate.
185:
This exchange was proposed to proceed via intermediacy of a thiazol-2-ylidene. In 2012 the isolation of the so-called
1114:
the respective diimidazolium salt. Only the deprotonation of the doubly tethered diimidazolium salt with the shorter
6379:
6474:
6459:
4591:
A. J. Arduengo; H. V. R. Dias; R. L. Harlow; M. Kline (1992). "Electronic stabilization of nucleophilic carbenes".
3603:
3151:
Persistent triplet state carbenes are likely to have very similar reactivity as other non-persistent triplet state
3129:
558:
344:
The first air-stable ylidic carbene, a chlorinated member of the imidazol-2-ylidene family, was obtained in 1997.
5246:
G. Bertrand; A. Igau; A. Baceiredo; G. Trinquier (1989). "trimethylsilylcarbene: A Stable Nucleophilic Carbene".
3174:
One of the more useful physical properties is the diagnostic chemical shift of the carbenic carbon atom in the C-
434:
197:
3741:
of stable carbenes can be difficult, due to the unavailability of suitable non-acidic polar solvents. Air-free
3484:
3437:
3114:
107:
5611:
Alder, Roger W.; Blake, Michael E.; Chaker, Leila; Harvey, Jeremy N.; Paolini, François; Schütz, Jan (2004).
5525:
4113:
Denk M. K.; Hatano K.; Ma M. (1999). "Nucleophilic Carbenes and the Wanzlick Equilibrium A Reinvestigation".
3178:
spectrum. Typically this peak is in the range between 200 and 300 ppm, where few other peaks appear in the C-
3742:
3396:
3238:
3182:
spectrum. An example is shown on the left for a cyclic diaminocarbene which has a carbenic peak at 238 ppm.
3139:
1164:) have been shown to dimerise, albeit quite slowly. This has been presumed to be due to the high barrier to
953:
6332:
Hopkinson, M. N.; Richter, C.; Schedler, M.; Glorius, F. (2014). "An Overview of N-Heterocyclic Carbenes".
4809:
R. W. Alder; M. E. Blake; J. M. Oliva (1999). "Diaminocarbenes; Calculation of Barriers to Rotation about C
3908:-Heterocyclic Carbenes: Generation and Reactivity of the Elusive 2,2-Diamino Enols (Breslow Intermediates)"
3846:
Ronald Breslow (1958). "On the Mechanism of Thiamine Action. IV.1 Evidence from Studies on Model Systems".
3367:, which increase the rate of reaction of this heterogeneous system, via the generation of tert-butoxide or
862:
1420:
895:
337:
in the ring of the carbene than in the parent imidazolium compound, indicating that there was very little
273:
4374:
Arduengo, Anthony J.; Harlow, Richard L.; Kline, Michael (January 1991). "A stable crystalline carbene".
6422:
6415:
For a review on the physico-chemical properties (electronics, sterics, ...) of N-heterocyclic carbenes:
6395:
5978:
5944:
5875:
5835:
5797:
5761:
5722:
5617:
5585:
5248:
5098:
4700:
4698:
M. K. Denk; A. Thadani; K. Hatano; A. J. Lough (1997). "Steric Stabilization of Nucleophilic Carbenes".
4663:
4143:
4031:
3912:
3456:
3322:
3302:
of carbene precursor salts with strong bases has proved a reliable route to almost all stable carbenes:
1679:
1472:
922:
525:
454:
368:
132:
6389:
R. W. Alder, in 'Diaminocarbenes: exploring structure and reactivity', ed. G. Bertrand, New York, 2002
4859:
A. J. Arduengo, J. R. Goerlich and W. J. Marshall (1997). "A Stable Thiazol-2-ylidene and Its Dimer".
4244:-bis-carbenoid, triply bonded species: synthesis of a stable λ-phosphino carbene-λ-phosphaacetylene".
3291:
Several approaches have been developed in order to prepare stable carbenes, these are outlined below.
1141:
interactions would have a significant destabilising effect. To avoid this electronic interaction, the
6343:
5973:
5935:
5826:
5342:
4938:
4824:
4489:
4444:
4057:
D. M. Lemal; R. A. Lovald; K. I. Kawano (1964). "Tetraaminoethylenes. The Question of Dissociation".
3969:
3781:
1589:
1578:
1175:"Least motion" (path A – not allowed) and "non-least motion" (path B) routes of carbene dimerisation.
1100:
1096:
858:
663:
438:
221:
193:
171:
143:
3590:
762:
725:
Carbenes that formally derive from imidazole-2-ylidenes by substitution of sulfur, oxygen, or other
6234:
4529:
3234:
1430:
945:
854:
54:
1171:
513:
are still the most stable and the most well studied and understood family of persistent carbenes.
382:
6367:
6261:
D. Kovacs; M. S. Lee; D. Olson; J. E. Jackson (1996). "Carbene-to-Carbene Oxygen Atom Transfer".
6184:
6141:
5938:; Martina Elison; Jakob Fischer; Christian Köcher; Georg R. J. Artus (1995). "Metal Complexes of
5851:
5659:
5565:
5366:
4861:
4183:
3805:
3726:
3353:
3266:
3135:
3125:
1452:
1349:
1025:
913:
Again the figure below is not an adequate representation of the actual molecular structure: both
834:
699:
510:
474:
360:
217:
3998:
H. W. Wanzlick; E. Schikora (1960). "Ein nucleophiles Carben" [A nucleophilic carbene].
1137:
on the carbenic carbon would be forced into close proximity. Presumably the resulting repulsive
703:
4923:
Lavallo, Vincent; Canac, Yves; Donnadieu, Bruno; Schoeller, Wolfgang W.; Bertrand, Guy (2006).
3444:) generally work well for the deprotonation of all types of salts, providing that not too much
769:. Thus it is possible that the reverse of this process might be occurring in similar carbenes.
425:
6464:
6438:
6359:
6308:
6292:
6263:
6176:
6133:
6125:
6091:
6071:
6006:
5777:
5738:
5635:
5557:
5506:
5490:
5435:
5419:
5358:
5313:
5287:
5220:
5189:
5158:
5059:
4964:
4929:
4894:
4746:
4626:
4593:
4505:
4480:
4460:
4435:
4407:
4376:
4356:
4312:
4279:
4246:
4211:
4159:
4115:
4087:
4059:
3930:
3876:
3848:
3797:
3631:
3532:
3445:
3341:
2467:
1514:
1357:
1092:
986:
981:
604:
580:
Molecules containing two and even three imidazol-2-ylidene groups have also been synthesised.
330:
254:
213:
5942:-Heterocyclic Carbenes – A New Structural Principle for Catalysts in Homogeneous Catalysis".
3737:. Stable carbenes are readily soluble in non-polar solvents such as hexane, and so typically
3547:
derived carbene to give the tetramethyldiaminocarbene and fluorenone has also been reported:
690:
Synthesised heteroamino carbenes (top and bottom right) and Bertrand's carbenes (bottom left)
158:
ring exchanges a hydrogen atom (attached to carbon 2 of the ring) for a furfural residue. In
6430:
6403:
6351:
6334:
6300:
6271:
6243:
6210:
6168:
6117:
6087:
6047:
6014:
5986:
5952:
5914:
5883:
5843:
5805:
5769:
5730:
5702:
5667:
5625:
5593:
5549:
5498:
5463:
5427:
5399:
5350:
5333:
5303:
5295:
5256:
5228:
5197:
5166:
5138:
5106:
5067:
5036:
5001:
4954:
4946:
4902:
4869:
4832:
4815:
4788:
4738:
4708:
4671:
4634:
4601:
4537:
4497:
4452:
4415:
4384:
4348:
4320:
4287:
4254:
4219:
4191:
4151:
4123:
4095:
4067:
4039:
4008:
3977:
3920:
3884:
3856:
3789:
3772:
3725:
Stable carbenes are very reactive, and so the minimum amount of handling is desirable using
3576:
3250:
2543:
2331:
2124:
1917:
1819:
1721:
1689:
1641:
1636:
1631:
1506:
1456:
1115:
932:
766:
750:
612:
574:
566:
403:
281:
277:
205:
201:
96:
50:
3902:
Berkessel A.; Elfert S.; Yatham V. R.; Neudörfl J.-M.; Schlörer N. E.; Teles J. H. (2012).
3483:
Shown right is an X-ray structure showing a complex between a diaminocarbene and potassium
3471:
Stable carbenes readily coordinate to metals; in this case a diaminocarbene coordinates to
1218:
237:
In 1970, Wanzlick's group generated imidazol-2-ylidene carbenes by the deprotonation of an
6038:
5694:
4925:"Cyclopropenylidenes: From Interstellar Space to an Isolated Derivative in the Laboratory"
3559:
3516:
3391:
1626:
1621:
1616:
1611:
1606:
1601:
1596:
1476:
806:
733:α-nitrogens are expected to be unstable, as they have the potential to dissociate into an
630:
618:
6228:
N. Kuhn; T. Kratz (1993). "Synthesis of Imidazol-2-ylidenes by Reduction of Imidazole-2(3
5692:-1,2,4-triazol-5-ylidene – applications of a stable carbene in synthesis and catalysis".
4661:
R. W. Alder; P. R. Allen; M. Murray; A. G. Orpen (1996). "Bis(diisopropylamino)carbene".
4523:
Lai Chun-Liang; Guo Wen-Hsin; Lee Ming-Tsung; Hu Ching-Han (2005). "Ligand properties of
3284:
In these cases, strong unhindered nucleophiles are avoided whether they are generated in
968:
of 158.8°. The planes of the phenyl groups are almost at right angles to each other (the
6347:
5346:
4942:
4892:
R. W. Alder; C. P. Butts; A. G. Orpen (1998). "Stable Aminooxy- and Aminothiocarbenes".
4828:
4493:
4448:
3973:
3785:
5385:
5308:
5278:
5218:
H. D. Hartzler (1972). "1,3-Dithiolium carbenes from acetylenes and carbon disulfide".
4959:
4924:
3823:
3734:
3449:
3258:
3226:
1569:
1468:
969:
926:
892:
608:
546:
485:
387:
258:
242:
225:
124:
6018:
5071:
4127:
4085:
H. E. Winberg; J. E. Carnahan; D. D. Coffman; M. Brown (1965). "Tetraaminoethylenes".
3213:
1203:("non-least motion"). Carbene dimerisation can be catalyzed by both acids and metals.
6453:
6145:
5569:
5541:
5458:
5133:
5028:
4993:
4783:
4541:
4291:
3702:
3536:
3492:
3424:
3408:
3368:
3299:
3278:
3189:
developed a new methodology to determine ligand donor strengths by C NMR analysis of
2591:
1539:
1491:
1448:
1385:
1365:
1192:
1188:
1180:
1165:
1138:
1111:
1055:
948:. As with the other carbenes, this species contains large bulky substituents, namely
850:
303:
79:
Modern theoretical analysis suggests that the term "persistent carbene" is in fact a
6188:
5855:
5688:
Enders, D.; Breuer, K.; Teles, J.H.; Ebel, K. (1997). "1,3,4-Triphenyl-4,5-dihydro-1
5456:
R. W. Alder; P. R. Allen; S. J. Williams (1995). "Stable carbenes as strong bases".
4559:
3717:
1031:
940:
Exposure to oxygen (a triplet diradical) converts this carbene to the corresponding
879:
686:
6420:
T. Dröge; F. Glorius (2010). "The Measure of All Rings - N-Heterocyclic Carbenes".
5370:
3961:
3357:
3274:
3202:
2955:
2792:
2657:
1651:
1584:
1337:
1063:
1042:
941:
899:
738:
128:
84:
83:. Persistent carbenes do not in fact have a carbene electronic structure in their
6371:
5873:-Butylthiazolato)(glyme)lithium: Carbene Character of a Formyl Anion Equivalent".
4501:
3826:(1957). "Mechanism of Thiamine Action: Participation of a Thiazolium Zwitterion".
3809:
595:
531:
6067:
4456:
3766:
Hopkinson, M. N.; Richter, C.; Schedler, M.; Glorius, F. (2014). "An Overview of
1179:
Diaminocarbenes do not truly dimerise, but rather form the dimer by reaction via
102:
Persistent carbenes in general, and Arduengo carbenes in particular, are popular
4555:
4310:
Arduengo, A.J.; Harlow, R.L.; Kline, M. (1991). "A stable crystalline carbene".
3501:
3375:
ions as an impurity in the metal hydride could also destroy non-aromatic salts.
3318:
3065:
3043:
3032:
2723:
2679:
2668:
1646:
1460:
1396:
957:
821:
647:
634:
470:
458:
372:
338:
334:
246:
238:
159:
92:
5403:
4624:
J. Arduengo; J. R. Goerlich; W. J. Marshall (1995). "A stable diaminocarbene".
3550:
3531:. A contributing factor to the success of this reaction is that the byproduct,
3419:
1195:
carbenes do not approach head to head ("least motion"), but rather the carbene
653:
497:
The following are examples of the classes of stable carbenes isolated to date:
386:
1,3,4,5-tetramethylimidazol-2-ylidene, a relatively unhindered carbene. (
17:
6109:
5906:
5671:
4873:
4340:
4000:
3828:
3329:
3163:
2869:
2847:
2814:
2734:
2646:
2613:
2580:
2200:
2189:
1874:
1534:
1211:
The chemistry of stable carbenes has not been fully explored. However, Enders
965:
918:
873:
812:
778:
680:
466:
209:
177:
155:
136:
58:
6129:
5918:
5706:
5040:
5005:
4195:
4012:
3981:
3746:
suitable for X-ray analysis. However, strong complexation to metal ions like
4950:
4141:
Böhm Volker P. W.; Herrmann Wolfgang A. (2000). "The Wanzlick Equilibrium".
3627:
3563:
3524:
3413:
3383:
3372:
3349:
3305:
3230:
3021:
2999:
2988:
2977:
2913:
2803:
2770:
2745:
2712:
2701:
2299:
2233:
2211:
2167:
2143:
2070:
1993:
1925:
1852:
1838:
1740:
1671:
1661:
1656:
1530:
1499:
1484:
1464:
1200:
1196:
1134:
1035:
Measurement of the pKa value for the conjugate acid of an imidazol-2-ylidene
961:
903:
888:
866:
765:
carbenes (i.e. where X = X = S), which then dimerise to give derivatives of
758:
726:
570:
506:
442:
399:
6442:
6434:
6407:
6363:
6312:
6180:
6137:
5990:
5956:
5887:
5847:
5809:
5781:
5773:
5742:
5734:
5639:
5630:
5612:
5597:
5561:
5510:
5439:
5362:
5317:
5260:
5110:
4968:
4750:
4712:
4675:
4509:
4464:
4360:
4163:
4043:
3934:
3925:
3903:
3801:
611:
ring are pictured below and were first prepared by Enders and coworkers by
543:
1,3-Dimesityl-4,5-dichloroimidazol-2-ylidene, the first air-stable carbene.
264:
5022:
W. A. Herrmann; M. Elison; J. Fischer; C. Kocher; G. R. J. Artus (1996). "
4813:–N Bonds, Barriers to Dimerization, Proton Affinities, and C NMR Shifts".
4156:
10.1002/1521-3773(20001117)39:22<4036::AID-ANIE4036>3.0.CO;2-L
3507:
Metal free carbenes have been prepared in several ways as outlined below:
249:. Wanzlick did not however isolate imidazol-2-ylidenes, but instead their
6247:
5482:
5467:
4240:
A. Igau; H. Grutzmacher; A. Baceiredo; G. Bertrand (1988). "Analogous α,α
3706:
3544:
3520:
3497:
3364:
3345:
3152:
3076:
2933:
2836:
2825:
2690:
2551:
2522:
2511:
2478:
2445:
2401:
2390:
2368:
2288:
2132:
2092:
1982:
1971:
1960:
1949:
1896:
1798:
1776:
1697:
1495:
1104:
1103:
in the absence of water and air for years. This is presumably due to the
1058:
gave 90% of the 2-substituted adduct, with only 10% of the corresponding
907:
816:
In the second step of this reaction sequence the proton is abstracted by
798:
777:
In Bertrand's persistent carbenes, the unsaturated carbon is bonded to a
714:
695:
626:
562:
554:
394:
Early workers attributed the stability of Arduengo carbenes to the bulky
167:
151:
80:
6355:
5232:
5201:
5170:
4638:
4605:
4388:
4324:
4277:
G. Bertrand; R. Reed (1994). "λ-Phosphinocarbenes λ-phosphaacetylenes".
4258:
4223:
4099:
4071:
3888:
3860:
3793:
3288:
or are present as an impurity in other reagents (such as LiOH in BuLi).
484:
Stable carbenes with oxygen or sulfur atoms bound to the carbenic atom (
5553:
5057:
H. V. R. Dias; W. C. Jin (1994). "A stable tridentate carbene ligand".
3747:
3623:
3387:
3270:
3054:
2966:
2944:
2902:
2880:
2858:
2635:
2624:
2602:
2500:
2434:
2412:
2379:
2339:
2255:
2222:
2178:
2156:
2114:
2103:
2081:
2059:
1936:
1863:
1729:
1666:
1502:
1142:
1125:
1047:
949:
782:
376:
280:. These species can be represented as either a λ-phosphinocarbene or λ-
62:
6304:
6275:
6172:
6121:
6052:
6033:
5502:
5431:
5299:
4906:
4836:
4419:
4352:
309:
287:
6214:
5354:
5142:
4792:
4742:
3379:
3262:
3222:
3010:
2891:
2781:
2562:
2423:
2350:
2310:
2266:
2244:
2037:
2026:
2015:
1885:
1827:
1787:
1765:
1711:
1488:
1480:
1407:) was questioned. The researchers were only able to isolate 2-imino-2
1059:
914:
883:
Delocalization in a stable triplet carbene reported by Tomioka (2001)
734:
718:
710:
411:
296:
212:. They conjectured that the carbene existed in equilibrium with its
163:
147:
103:
4029:
H. W. Wanzlick (1962). "Aspects of Nucleophilic Carbene Chemistry".
3467:
1156:
N–C–SR) and non-aromatic carbenes such as diaminocarbenes (such as R
5156:
R. Breslow (1957). "Rapid Deuterium Exchange in Thiazolium Salts".
4209:
R. Gleiter; R. Hoffmann (1968). "Stabilizing a singlet methylene".
3874:
R. Breslow (1957). "Rapid Deuterium Exchange in Thiazolium Salts".
356:
4527:-heterocyclic and Bertrand carbenes: A density functional study".
3716:
3589:
3549:
3472:
3466:
3418:
3304:
3212:
2533:
2321:
1907:
1754:
1429:
1217:
1170:
1124:
1041:
1030:
960:
of the divalent carbon atom to its neighbors is claimed to be 138
931:
878:
811:
802:
685:
652:
617:
594:
538:
530:
517:
480:
479:
462:
424:
381:
355:
308:
286:
263:
176:
88:
34:
1475:. Stable carbenes are believed to behave in a similar fashion to
706:
based carbenes have been characterised by X-ray crystallography.
302:
In 1991, Arduengo and coworkers crystallized a diaminocarbene by
3540:
3314:
3261:
of an imidazol-2-ylidene was measured at ca. 24) and react with
2489:
2456:
2048:
2004:
1809:
936:
A persistent triplet carbene (right), synthesized by Itoh (2006)
887:
In 2006 a triplet carbene was reported by the same group with a
521:
429:
Bis(diisopropylamino) carbene, the first acyclic stable carbene.
30:
5187:
H. D. Haztzler (1970). "Nucleophilic 1,3-dithiolium carbenes".
944:. The diphenylmethane compound is formed when it is trapped by
3528:
3254:
3179:
3175:
2277:
1110:
Chen and Taton made a doubly tethered diimidazol-2-ylidene by
1015:
291:
Alkyne and carbene resonances structures of Bertrand's carbene
3705:
decomposition of a diazomethane product via the expulsion of
853:, dimerizing when forced into triplet states. Nevertheless,
414:
groups also affords 1,3,4,5-tetramethylimidazol-2‑ylidene (Me
3317:, have been prepared by the deprotonation of the respective
5583:
T. A. Taton; P. Chen (1996). "A Stable Tetraazafulvalene".
3948:
Chemists Approach Elusive Breslow Intermediate Carmen Drahl
3138:
carbenes; in which carbenes are tethered to a metal and an
433:
In 1995, Arduengo's group obtained a carbene derivative of
6393:
M. Regitz (1996). "Stable Carbenes—Illusion or Reality?".
5283:-Heterocyclic Carbenes with Tunable Electronic Properties"
3128:
carbenes in which carbenes are tethered to a metal and an
505:
The first stable carbenes to be isolated were based on an
4777:-piperidyl)carbene and its slow dimerisation to tetrakis(
3515:
Another approach of preparing carbenes has relied on the
3399:
can be employed without the addition of a metal hydride.
3313:
Imidazol-2-ylidenes and dihydroimidazol-2-ylidenes, such
3309:
Deprotonation of precursor salts to give stable carbenes.
921:
with respect to each other. The carbene carbon has an sp-
3701:
Persistent triplet state carbenes have been prepared by
1091:
At one time, stable carbenes were thought to reversibly
722:
N–C–X bond angles of around 104° and 109° respectively.
181:
Deuterium exchange of the C2-proton of thiazolium salt.
5899:
5897:
1062:, indicating that these molecules are also reasonably
6088:
N-Heterocyclic carbenes in transition metal catalysis
196:
and coworkers conjectured that carbenes derived from
76:), in which nitrogen atoms flank the formal carbene.
3109:
Carbenes in organometallic chemistry & catalysis
406:
the carbene from dimerising. But replacement of the
1133:If this dimer existed as a dicarbene, the electron
861:
to produce a comparatively stable triplet carbene (
797:-heterocyclic carbene has a structure analogous to
1483:are said to be good σ-donors through the carbenic
1479:in their coordination properties to metals. These
1148:On the other hand, heteroamino carbenes (such as R
809:. This results in a planar six-electron compound.
268:Preparation and trapping of an imidazol-2-ylidene.
27:Type of carbene demonstrating particular stability
5096:-1,2,4-triazol-5-ylidene, a New Stable Carbene".
3630:salts to give a metal-free carbene and elemental
3277:addition, other non-aromatic salts are not (i.e.
1083:units more acidic than related imidazolium ions.
1014:The imidazol-2-ylidenes are strong bases, having
929:each conjugating with one of the aromatic rings.
4175:
4173:
3709:gas, at a wavelength of 300 nm in benzene.
524:, alkyloxy, alkylamino, alkylphosphino and even
465:on adjacent nitrogen atoms while preserving the
437:, proving that stability did not arise from the
5272:
5270:
5213:
5211:
5182:
5180:
4918:
4916:
5754:
5752:
5683:
5681:
5277:Präsang, C; Donnadieu, B; Bertrand, G (2005).
5087:
5085:
5083:
5081:
3265:. Clearly these reactions are performed using
1129:Dimerisation of tethered diimidazol-2-ylidenes
1050:as an NMR solvent can have unexpected results.
657:Synthesised cyclic and acyclic diaminocarbenes
4728:
4726:
4724:
4722:
4560:"What's in a name? Carbenes: a reality check"
3594:Preparation of carbenes via vacuum pyrolysis.
3491:was used as a strong base to deprotonate the
1554:
1403:) derived from an isothiazolium perchlorate (
1325:Dimethylacetylene dicarboxylate, THF, reflux
891:of 40 minutes. This carbene is prepared by a
142:(thiamine), was the catalyst involved in the
8:
5821:
5819:
4854:
4852:
4850:
4848:
4846:
4619:
4617:
4615:
4586:
4584:
4582:
4580:
4400:
4398:
4305:
4303:
4301:
4272:
4270:
4268:
4235:
4233:
3229:chemistry. One practical application is the
35:1,3-Dimesityl-imidazol-4,5-dihydro-2-ylidene
5968:
5966:
5930:
5928:
5613:"When and How Do Diaminocarbenes Dimerize?"
5279:"Stable Planar Six-π-Electron Six-Membered
4991:-Heterocyclic Carbenes in Liquid Ammonia".
4024:
4022:
3993:
3991:
3554:Preparation of carbenes by dechalcogenation
3273:. Although imidazolium salts are stable to
3103: No carbene complex with element known
547:View the 3D structure with external viewer.
5017:
5015:
4982:
4980:
4978:
1561:
1547:
1024: ≈ 24 for the conjugate acid in
761:derivatives is proposed to give transient
633:with sodium methoxide, which attacks as a
6051:
5629:
5451:
5449:
5307:
5126:
5124:
5122:
5120:
4958:
4887:
4885:
4883:
4804:
4802:
4768:
4766:
4764:
4762:
4760:
4656:
4654:
4652:
4650:
4648:
3924:
849:Persistent carbenes tend to exist in the
833:Another family of carbenes is based on a
5052:
5050:
4693:
4691:
4689:
4687:
4685:
3750:will in most cases prevent sublimation.
3162:
3095: Carbene complex with element known
1232:3,6-diphenyl-1,2,4,5-tetrazine, toluene
1222:
997:-heterocyclic carbenes (aNHC) or remote
29:
3758:
2587:
1487:, but poor π-acceptors due to internal
839:bis(diisopropylamino)cyclopropenylidene
352:Stabilization through adjacent orbitals
204:of the corresponding 2-trichloromethyl
4773:R. W. Alder; M. E. Blake (1997). "Bis(
3487:. This complex was formed when excess
3348:to give the desired carbene, with the
3269:, avoiding compounds of even moderate
2951:
2788:
2653:
876:positions with respect to each other.
3217:A second generation Grubbs' catalyst.
3061:
3039:
3028:
2719:
2675:
2664:
1054:Reaction of imidazol-2-ylidenes with
865:) in 2001. It has an unusually long
622:Examples of 1,2,4-triazol-5-ylidenes.
37:, a representative persistent carbene
7:
6068:N-Heterocyclic carbenes in synthesis
6032:Han Vinh Huynh; et al. (2009).
2865:
2843:
2810:
2730:
2642:
2609:
2576:
2196:
2185:
1513:) with an imidazol-2-ylidene in the
1122:–) resulted in the dicarbene dimer:
224:. This conjecture was challenged by
166:was found to rapidly exchange for a
3017:
2995:
2984:
2973:
2909:
2799:
2766:
2741:
2708:
2697:
2295:
2229:
2207:
2163:
2139:
2066:
1989:
1870:
1848:
1834:
1736:
1427:is no longer considered as stable.
1336:These carbenes tend to behave in a
906:light in benzene with expulsion of
553:In particular, substitution of two
3363:. These reagents generate soluble
3119:transition metal carbene complexes
3072:
2929:
2832:
2821:
2686:
2547:
2518:
2507:
2474:
2441:
2386:
2364:
2284:
2128:
2088:
1978:
1956:
1945:
1921:
1892:
127:proposed that a relatively stable
25:
5384:Michael Freemantle (2001-08-13).
3587:to generate a triazol-5-ylidene.
3340:One might believe that sodium or
3050:
2940:
2898:
2876:
2854:
2631:
2620:
2598:
2496:
2463:
2430:
2408:
2397:
2375:
2335:
2251:
2218:
2174:
2110:
2099:
2077:
2055:
1967:
1932:
1859:
1794:
1772:
1725:
1693:
1243:2 equiv., PhNCO, toluene, reflux
925:, the two remaining orthogonal p-
6327:Reviews on persistent carbenes:
5537:-heterocyclic carbenes in water"
4542:10.1016/j.jorganchem.2005.07.058
3006:
2962:
2923:
2887:
2777:
2760:
2570:
2558:
2419:
2358:
2346:
2262:
2240:
2152:
2033:
2022:
2011:
1823:
1707:
233:Isolation of persistent carbenes
5391:Chemical & Engineering News
5386:"Triplet Carbene has Long Life"
3386:hydride in a mixture of liquid
3147:Triplet state carbene chemistry
2529:
2317:
2306:
1903:
1881:
1783:
1761:
1224:Reactions of triazol-5-ylidene
569:on the carbon atom bearing the
565:substituents, which reduce the
461:character that would place the
324:-diadamantyl-imidazol-2-ylidene
154:. In this cycle, the vitamin's
6206:J. Chem. Soc., Perkin Trans. 1
3680:
3463:Metal-free carbene preparation
3113:Carbenes can be stabilised as
2485:
2452:
2044:
2000:
1805:
1750:
333:structure revealed longer N–C
1:
6019:10.1016/S0040-4039(99)00217-8
5072:10.1016/S0040-4039(00)76219-8
4502:10.1126/science.292.5523.1901
4128:10.1016/S0040-4039(99)00164-1
3442:tetramethylpiperidide (LiTMP)
2273:
1269:, toluene, or PhNCS, THF, RT
375:toward adjacent lone-pair or
5829:; Christian Köcher (1997). "
5533:s of the conjugate acids of
4457:10.1126/science.288.5467.834
4292:10.1016/0010-8545(94)03005-B
3427:salts with tert-butyllithium
1438:) was proved to be unstable.
1010:Basicity and nucleophilicity
577:through the sigma-backbone.
5657:-1,2,4-triazol-5-ylidene".
3697:Photochemical decomposition
1388:. The insertion reactions (
1187:Unlike the dimerisation of
789:Other nucleophilic carbenes
208:compounds with the loss of
6491:
5404:10.1021/cen-v079n033.p011a
3604:Bis(trimethylsilyl)mercury
3599:Bis(trimethylsilyl)mercury
3201:-isomerization due to the
3130:electron-withdrawing group
3121:fall into two categories:
2754:
1845:
1747:
1704:
1683:
1670:
1665:
1660:
1655:
1650:
1645:
1640:
1635:
1630:
1625:
1620:
1615:
1610:
1605:
1600:
1595:
1588:
1583:
1577:
1542:
979:
824:groups shield the carbene.
757:) with electron deficient
599:Triazol-5-ylidene isomers.
535:Stable imidazol-2-ylidenes
493:Classes of stable carbenes
341:character to these bonds.
306:of an imidazolium cation:
241:salt. Wanzlick as well as
220:derivative, the so-called
5833:-Heterocyclic Carbenes".
5672:10.1002/jlac.199619961212
5481:Massey Richard S (2012).
4874:10.1002/jlac.199719970213
3770:-Heterocyclic Carbenes".
3721:Sublimation of a carbene.
3562:of thioureas with molten
2922:
2759:
1288:, or Se, toluene, reflux
1199:attacks the empty carbon
435:dihydroimidazol-2-ylidene
198:dihydroimidazol-2-ylidene
6470:Organometallic chemistry
5919:10.1002/cber.19961291213
5707:10.1002/prac.19973390170
5041:10.1002/chem.19960020708
5006:10.1002/chem.19960021222
4196:10.1002/jlac.19707310121
4013:10.1002/cber.19610940905
3982:10.1002/ange.19600721409
3436:Lithium amides like the
3249:NHCs are often strongly
3221:NHCs are widely used as
772:
420:thermodynamically stable
108:organometallic chemistry
72:(NHC) (sometimes called
4951:10.1126/science.1126675
4735:Chemical Communications
3397:potassium tert-butoxide
3239:NHC-Palladium Complexes
3140:electron-donating group
1419:was proposed through a
859:electron delocalization
410:-adamantyl groups with
363:of the allylic system.
57:has a carbon atom with
6435:10.1002/anie.201001865
6408:10.1002/anie.199106741
5991:10.1002/anie.199628051
5957:10.1002/anie.199523711
5888:10.1002/anie.199504871
5848:10.1002/anie.199721621
5810:10.1002/anie.200702746
5774:10.1002/anie.200702272
5735:10.1002/anie.200604305
5631:10.1002/anie.200400654
5598:10.1002/anie.199610111
5261:10.1002/anie.198906211
5111:10.1002/anie.199510211
4713:10.1002/anie.199726071
4676:10.1002/anie.199611211
4564:Chemistry with a Twist
4044:10.1002/anie.196200751
3926:10.1002/anie.201205878
3722:
3595:
3555:
3476:
3438:diisopropylamide (LDA)
3428:
3310:
3218:
3168:
1439:
1421:rearrangement reaction
1333:
1176:
1130:
1095:through the so-called
1051:
1036:
989:(MICs) are similar to
937:
902:precursor by 300
884:
845:Triplet state carbenes
825:
691:
658:
623:
600:
550:
536:
489:
430:
391:
364:
325:
292:
276:and others isolated a
269:
251:coordination compounds
182:
70:-heterocyclic carbenes
38:
6423:Angew. Chem. Int. Ed.
6396:Angew. Chem. Int. Ed.
6066:S. P. Nolan (2006).
5979:Angew. Chem. Int. Ed.
5945:Angew. Chem. Int. Ed.
5876:Angew. Chem. Int. Ed.
5836:Angew. Chem. Int. Ed.
5798:Angew. Chem. Int. Ed.
5762:Angew. Chem. Int. Ed.
5723:Angew. Chem. Int. Ed.
5618:Angew. Chem. Int. Ed.
5586:Angew. Chem. Int. Ed.
5249:Angew. Chem. Int. Ed.
5099:Angew. Chem. Int. Ed.
4701:Angew. Chem. Int. Ed.
4664:Angew. Chem. Int. Ed.
4144:Angew. Chem. Int. Ed.
4032:Angew. Chem. Int. Ed.
3913:Angew. Chem. Int. Ed.
3720:
3593:
3553:
3470:
3457:hexamethyldisilazides
3422:
3308:
3216:
3167:Carbene peak in C NMR
3166:
3132:(usually a carbonyl),
1473:X-ray crystallography
1434:Isothiazole carbene (
1433:
1221:
1174:
1128:
1045:
1034:
935:
917:rings are positioned
882:
863:bis(9-anthryl)carbene
815:
737:(RC≡CR) and a carbon
689:
656:
641:Other diaminocarbenes
621:
598:
542:
534:
483:
428:
385:
359:
312:
290:
267:
180:
33:
6248:10.1055/s-1993-25902
5985:(23–24): 2805–2807.
5974:Wolfgang A. Herrmann
5936:Wolfgang A. Herrmann
5827:Wolfgang A. Herrmann
5468:10.1039/c39950001267
4781:-piperidyl)ethene".
4536:(24–25): 5867–5875.
1572:: Persistent carbene
1443:Carbene complexation
1415:). The intermediate
1395:The reported stable
1384:) as well as simple
1380:), cycloadditions (
1097:Wanzlick equilibrium
954:computer simulations
857:and associates used
674:Heteroamino carbenes
664:Wanzlick equilibrium
559:electron-withdrawing
402:substituents, which
222:Wanzlick equilibrium
194:Hans-Werner Wanzlick
187:Breslow intermediate
144:benzoin condensation
6356:10.1038/nature13384
6348:2014Natur.510..485H
6086:F. Glorius (2007)
5497:(50): 20421–20432.
5347:2001Natur.412..626T
5294:(29): 10182–10183.
5233:10.1021/ja00794a039
5202:10.1021/ja00708a058
5171:10.1021/ja01564a064
4943:2006Sci...312..722L
4901:(44): 11526–11527.
4829:1999JPCA..10311200A
4823:(50): 11200–11211.
4639:10.1021/ja00149a034
4633:(44): 11027–11028.
4606:10.1021/ja00040a007
4530:J. Organomet. Chem.
4494:2001Sci...292.1901S
4488:(5523): 1901–1903.
4449:2000Sci...288..834B
4414:(52): 12742–12749.
4389:10.1021/ja00001a054
4325:10.1021/ja00001a054
4259:10.1021/ja00227a028
4224:10.1021/ja01022a023
4100:10.1021/ja01087a040
4072:10.1021/ja01066a044
3974:1960AngCh..72..494W
3919:(49): 12370–12374.
3889:10.1021/ja01564a064
3861:10.1021/ja01547a064
3794:10.1038/nature13384
3786:2014Natur.510..485H
3727:air-free techniques
3622:reacts with chloro-
3378:Deprotonation with
3336:Metal hydride bases
3267:air-free techniques
3245:Preparation methods
3159:Physical properties
1453:main group elements
1350:insertion reactions
1299:Maleimide, THF, RT
1225:
1005:Chemical properties
946:cyclohexa-1,4-diene
829:Cyclopropenylidenes
805:atom replaced by a
773:Bertrand's carbenes
511:imidazol-2-ylidenes
501:Imidazol-2-ylidenes
55:resonance structure
5660:Liebigs Ann. Chem.
5554:10.1039/C0CC03367G
4862:Liebigs Ann. Chem.
4184:Liebigs Ann. Chem.
3723:
3596:
3556:
3477:
3475:to form a complex.
3448:is present in the
3429:
3323:dihydroimidazolium
3311:
3219:
3169:
1440:
1358:addition reactions
1334:
1223:
1177:
1131:
1052:
1037:
1026:dimethyl sulfoxide
987:Mesoionic carbenes
976:Mesoionic carbenes
938:
885:
835:cyclopropenylidene
826:
745:Non-amino carbenes
692:
659:
624:
605:triazol-5-ylidenes
601:
588:Triazol-5-ylidenes
551:
537:
490:
475:cyclopropenylidene
441:of the conjugated
431:
392:
365:
326:
293:
270:
218:tetraaminoethylene
183:
93:aromatic resonance
43:persistent carbene
39:
6475:Organic compounds
6460:Functional groups
6429:(39): 6940–6952.
6342:(7506): 485–496.
6305:10.1021/ja0393325
6293:J. Am. Chem. Soc.
6276:10.1021/ja961324j
6270:(34): 8144–8145.
6264:J. Am. Chem. Soc.
6209:(14): 1586–1593.
6173:10.1021/cr050004s
6167:(11): 3978–4008.
6122:10.1021/cr900074m
6053:10.1021/om900667d
6046:(18): 5395–5404.
6013:(12): 2247–2250.
6007:Tetrahedron Lett.
5951:(21): 2371–2374.
5913:(12): 1483–1488.
5842:(20): 2162–2187.
5768:(36): 6922–6925.
5729:(17): 3118–3121.
5666:(12): 2019–2028.
5624:(44): 5896–5911.
5503:10.1021/ja308420c
5491:J. Am. Chem. Soc.
5432:10.1021/ja056575j
5420:J. Am. Chem. Soc.
5341:(6847): 626–628.
5300:10.1021/ja052987g
5288:J. Am. Chem. Soc.
5227:(13): 4379–4387.
5221:J. Am. Chem. Soc.
5190:J. Am. Chem. Soc.
5159:J. Am. Chem. Soc.
5060:Tetrahedron Lett.
5000:(12): 1627–1636.
4937:(5774): 722–724.
4907:10.1021/ja9819312
4895:J. Am. Chem. Soc.
4837:10.1021/jp9934228
4787:(16): 1513–1514.
4737:(19): 2172–2173.
4707:(23): 2607–2609.
4670:(10): 1121–1123.
4627:J. Am. Chem. Soc.
4600:(14): 5530–5534.
4594:J. Am. Chem. Soc.
4443:(5467): 834–836.
4420:10.1021/ja973241o
4408:J. Am. Chem. Soc.
4377:J. Am. Chem. Soc.
4353:10.1021/cr800521g
4313:J. Am. Chem. Soc.
4280:Coord. Chem. Rev.
4253:(19): 6463–6466.
4247:J. Am. Chem. Soc.
4218:(20): 5457–5460.
4212:J. Am. Chem. Soc.
4150:(22): 4036–4038.
4122:(11): 2057–2060.
4116:Tetrahedron Lett.
4088:J. Am. Chem. Soc.
4066:(12): 2518–2519.
4060:J. Am. Chem. Soc.
3877:J. Am. Chem. Soc.
3855:(14): 3719–3726.
3849:J. Am. Chem. Soc.
3780:(7506): 485–496.
3739:recrystallisation
3533:potassium sulfide
3342:potassium hydride
3085:
3084:
1537:carbene ligands.
1515:olefin metathesis
1457:transition metals
1332:
1331:
982:Mesoionic carbene
200:were produced by
170:in a statistical
133:thiazol-2-ylidene
87:, but instead an
74:Arduengo carbenes
16:(Redirected from
6482:
6446:
6411:
6375:
6317:
6316:
6299:(4): 1016–1017.
6286:
6280:
6279:
6258:
6252:
6251:
6225:
6219:
6218:
6215:10.1039/b104110j
6199:
6193:
6192:
6156:
6150:
6149:
6116:(8): 3612–3676.
6104:
6098:
6084:
6078:
6064:
6058:
6057:
6055:
6029:
6023:
6022:
6001:
5995:
5994:
5970:
5961:
5960:
5932:
5923:
5922:
5901:
5892:
5891:
5866:
5860:
5859:
5823:
5814:
5813:
5792:
5786:
5785:
5756:
5747:
5746:
5717:
5711:
5710:
5685:
5676:
5675:
5650:
5644:
5643:
5633:
5608:
5602:
5601:
5592:(9): 1011–1013.
5580:
5574:
5573:
5548:(5): 1559–1561.
5521:
5515:
5514:
5487:
5478:
5472:
5471:
5453:
5444:
5443:
5414:
5408:
5407:
5381:
5375:
5374:
5355:10.1038/35088038
5328:
5322:
5321:
5311:
5274:
5265:
5264:
5243:
5237:
5236:
5215:
5206:
5205:
5196:(5): 1412–1413.
5184:
5175:
5174:
5165:(7): 1762–1763.
5153:
5147:
5146:
5143:10.1039/a808951e
5128:
5115:
5114:
5105:(9): 1021–1023.
5089:
5076:
5075:
5066:(9): 1365–1366.
5054:
5045:
5044:
5019:
5010:
5009:
4984:
4973:
4972:
4962:
4920:
4911:
4910:
4889:
4878:
4877:
4856:
4841:
4840:
4816:J. Phys. Chem. A
4806:
4797:
4796:
4793:10.1039/a703610h
4770:
4755:
4754:
4743:10.1039/b409112d
4730:
4717:
4716:
4695:
4680:
4679:
4658:
4643:
4642:
4621:
4610:
4609:
4588:
4575:
4574:
4572:
4570:
4552:
4546:
4545:
4520:
4514:
4513:
4475:
4469:
4468:
4430:
4424:
4423:
4402:
4393:
4392:
4371:
4365:
4364:
4347:(8): 3385–3407.
4335:
4329:
4328:
4307:
4296:
4295:
4274:
4263:
4262:
4243:
4237:
4228:
4227:
4206:
4200:
4199:
4177:
4168:
4167:
4138:
4132:
4131:
4110:
4104:
4103:
4094:(9): 2055–2056.
4082:
4076:
4075:
4054:
4048:
4047:
4026:
4017:
4016:
4007:(9): 2389–2393.
3995:
3986:
3985:
3956:
3950:
3945:
3939:
3938:
3928:
3899:
3893:
3892:
3883:(7): 1762–1763.
3871:
3865:
3864:
3843:
3837:
3836:
3820:
3814:
3813:
3763:
3682:
3669:
3668:
3667:
3577:vacuum pyrolysis
3570:Vacuum pyrolysis
3511:Dechalcogenation
3235:Grubbs' catalyst
3223:ancillary ligand
3104:
3102:
3096:
3094:
3081:
3079:
3070:
3068:
3059:
3057:
3048:
3046:
3037:
3035:
3026:
3024:
3015:
3013:
3004:
3002:
2993:
2991:
2982:
2980:
2971:
2969:
2960:
2958:
2949:
2947:
2938:
2936:
2927:
2926:
2918:
2916:
2907:
2905:
2896:
2894:
2885:
2883:
2874:
2872:
2863:
2861:
2852:
2850:
2841:
2839:
2830:
2828:
2819:
2817:
2808:
2806:
2797:
2795:
2786:
2784:
2775:
2773:
2764:
2763:
2750:
2748:
2739:
2737:
2728:
2726:
2717:
2715:
2706:
2704:
2695:
2693:
2684:
2682:
2673:
2671:
2662:
2660:
2651:
2649:
2640:
2638:
2629:
2627:
2618:
2616:
2607:
2605:
2596:
2594:
2585:
2583:
2574:
2573:
2567:
2565:
2556:
2554:
2538:
2536:
2527:
2525:
2516:
2514:
2505:
2503:
2494:
2492:
2483:
2481:
2472:
2470:
2461:
2459:
2450:
2448:
2439:
2437:
2428:
2426:
2417:
2415:
2406:
2404:
2395:
2393:
2384:
2382:
2373:
2371:
2362:
2361:
2355:
2353:
2344:
2342:
2326:
2324:
2315:
2313:
2304:
2302:
2293:
2291:
2282:
2280:
2271:
2269:
2260:
2258:
2249:
2247:
2238:
2236:
2227:
2225:
2216:
2214:
2205:
2203:
2194:
2192:
2183:
2181:
2172:
2170:
2161:
2159:
2148:
2146:
2137:
2135:
2119:
2117:
2108:
2106:
2097:
2095:
2086:
2084:
2075:
2073:
2064:
2062:
2053:
2051:
2042:
2040:
2031:
2029:
2020:
2018:
2009:
2007:
1998:
1996:
1987:
1985:
1976:
1974:
1965:
1963:
1954:
1952:
1941:
1939:
1930:
1928:
1912:
1910:
1901:
1899:
1890:
1888:
1879:
1877:
1868:
1866:
1857:
1855:
1843:
1841:
1832:
1830:
1814:
1812:
1803:
1801:
1792:
1790:
1781:
1779:
1770:
1768:
1759:
1757:
1745:
1743:
1734:
1732:
1716:
1714:
1702:
1700:
1563:
1556:
1549:
1540:
1507:catalytic cycles
1477:organophosphines
1314:CH=CHR, THF, RT
1226:
1191:carbenes, these
1145:units dimerise.
1116:methylene bridge
820:, while the two
767:tetrathiafulvene
751:carbon disulfide
749:The reaction of
613:vacuum pyrolysis
567:electron density
422:unhindered NHC.
322:
282:phosphaacetylene
278:phosphinocarbene
206:dihydroimidazole
202:vacuum pyrolysis
160:deuterated water
97:steric shielding
59:incomplete octet
51:organic molecule
21:
6490:
6489:
6485:
6484:
6483:
6481:
6480:
6479:
6450:
6449:
6419:
6392:
6331:
6325:
6323:Further reading
6320:
6288:
6287:
6283:
6260:
6259:
6255:
6227:
6226:
6222:
6201:
6200:
6196:
6158:
6157:
6153:
6106:
6105:
6101:
6085:
6081:
6065:
6061:
6039:Organometallics
6031:
6030:
6026:
6003:
6002:
5998:
5972:
5971:
5964:
5934:
5933:
5926:
5903:
5902:
5895:
5868:
5867:
5863:
5825:
5824:
5817:
5794:
5793:
5789:
5758:
5757:
5750:
5719:
5718:
5714:
5695:J. Prakt. Chem.
5687:
5686:
5679:
5652:
5651:
5647:
5610:
5609:
5605:
5582:
5581:
5577:
5532:
5523:
5522:
5518:
5485:
5480:
5479:
5475:
5455:
5454:
5447:
5416:
5415:
5411:
5383:
5382:
5378:
5330:
5329:
5325:
5276:
5275:
5268:
5245:
5244:
5240:
5217:
5216:
5209:
5186:
5185:
5178:
5155:
5154:
5150:
5130:
5129:
5118:
5091:
5090:
5079:
5056:
5055:
5048:
5021:
5020:
5013:
4986:
4985:
4976:
4922:
4921:
4914:
4891:
4890:
4881:
4858:
4857:
4844:
4812:
4808:
4807:
4800:
4772:
4771:
4758:
4732:
4731:
4720:
4697:
4696:
4683:
4660:
4659:
4646:
4623:
4622:
4613:
4590:
4589:
4578:
4568:
4566:
4558:(11 Sep 2016).
4554:
4553:
4549:
4522:
4521:
4517:
4477:
4476:
4472:
4432:
4431:
4427:
4404:
4403:
4396:
4373:
4372:
4368:
4337:
4336:
4332:
4309:
4308:
4299:
4276:
4275:
4266:
4241:
4239:
4238:
4231:
4208:
4207:
4203:
4179:
4178:
4171:
4140:
4139:
4135:
4112:
4111:
4107:
4084:
4083:
4079:
4056:
4055:
4051:
4028:
4027:
4020:
3997:
3996:
3989:
3958:
3957:
3953:
3946:
3942:
3901:
3900:
3896:
3873:
3872:
3868:
3845:
3844:
3840:
3822:
3821:
3817:
3765:
3764:
3760:
3756:
3715:
3699:
3691:
3687:
3683:
3677:
3673:
3666:
3663:
3662:
3661:
3659:
3657:
3653:
3649:
3645:
3641:
3634:. For example:
3621:
3617:
3613:
3609:
3601:
3572:
3560:desulfurisation
3517:desulfurisation
3513:
3465:
3434:
3405:
3338:
3297:
3247:
3211:
3161:
3149:
3117:species. These
3111:
3100:
3099:
3092:
3091:
3077:
3075:
3074:
3066:
3064:
3063:
3055:
3053:
3052:
3044:
3042:
3041:
3033:
3031:
3030:
3022:
3020:
3019:
3011:
3009:
3008:
3000:
2998:
2997:
2989:
2987:
2986:
2978:
2976:
2975:
2967:
2965:
2964:
2956:
2954:
2953:
2945:
2943:
2942:
2934:
2932:
2931:
2924:
2914:
2912:
2911:
2903:
2901:
2900:
2892:
2890:
2889:
2881:
2879:
2878:
2870:
2868:
2867:
2859:
2857:
2856:
2848:
2846:
2845:
2837:
2835:
2834:
2826:
2824:
2823:
2815:
2813:
2812:
2804:
2802:
2801:
2793:
2791:
2790:
2782:
2780:
2779:
2771:
2769:
2768:
2761:
2755:
2746:
2744:
2743:
2735:
2733:
2732:
2724:
2722:
2721:
2713:
2711:
2710:
2702:
2700:
2699:
2691:
2689:
2688:
2680:
2678:
2677:
2669:
2667:
2666:
2658:
2656:
2655:
2647:
2645:
2644:
2636:
2634:
2633:
2625:
2623:
2622:
2614:
2612:
2611:
2603:
2601:
2600:
2592:
2590:
2589:
2581:
2579:
2578:
2571:
2563:
2561:
2560:
2552:
2550:
2549:
2534:
2532:
2531:
2523:
2521:
2520:
2512:
2510:
2509:
2501:
2499:
2498:
2490:
2488:
2487:
2479:
2477:
2476:
2468:
2466:
2465:
2457:
2455:
2454:
2446:
2444:
2443:
2435:
2433:
2432:
2424:
2422:
2421:
2413:
2411:
2410:
2402:
2400:
2399:
2391:
2389:
2388:
2380:
2378:
2377:
2369:
2367:
2366:
2359:
2351:
2349:
2348:
2340:
2338:
2337:
2322:
2320:
2319:
2311:
2309:
2308:
2300:
2298:
2297:
2289:
2287:
2286:
2278:
2276:
2275:
2267:
2265:
2264:
2256:
2254:
2253:
2245:
2243:
2242:
2234:
2232:
2231:
2223:
2221:
2220:
2212:
2210:
2209:
2201:
2199:
2198:
2190:
2188:
2187:
2179:
2177:
2176:
2168:
2166:
2165:
2157:
2155:
2154:
2144:
2142:
2141:
2133:
2131:
2130:
2115:
2113:
2112:
2104:
2102:
2101:
2093:
2091:
2090:
2082:
2080:
2079:
2071:
2069:
2068:
2060:
2058:
2057:
2049:
2047:
2046:
2038:
2036:
2035:
2027:
2025:
2024:
2016:
2014:
2013:
2005:
2003:
2002:
1994:
1992:
1991:
1983:
1981:
1980:
1972:
1970:
1969:
1961:
1959:
1958:
1950:
1948:
1947:
1937:
1935:
1934:
1926:
1924:
1923:
1908:
1906:
1905:
1897:
1895:
1894:
1886:
1884:
1883:
1875:
1873:
1872:
1864:
1862:
1861:
1853:
1851:
1850:
1846:
1839:
1837:
1836:
1828:
1826:
1825:
1810:
1808:
1807:
1799:
1797:
1796:
1788:
1786:
1785:
1777:
1775:
1774:
1766:
1764:
1763:
1755:
1753:
1752:
1748:
1741:
1739:
1738:
1730:
1728:
1727:
1712:
1710:
1709:
1705:
1698:
1696:
1695:
1684:
1573:
1567:
1528:
1524:
1520:
1512:
1445:
1313:
1287:
1283:
1268:
1209:
1163:
1159:
1155:
1151:
1121:
1089:
1082:
1075:
1022:
1012:
1007:
984:
978:
869:of 19 minutes.
847:
831:
807:methylene group
791:
775:
756:
747:
713:and sulfur are
676:
643:
631:triazolium salt
590:
544:
503:
495:
463:positive charge
457:, suggesting a
449:
417:
354:
320:
313:Preparation of
235:
140:
121:
116:
45:(also known as
28:
23:
22:
18:Stable carbenes
15:
12:
11:
5:
6488:
6486:
6478:
6477:
6472:
6467:
6462:
6452:
6451:
6448:
6447:
6413:
6412:
6402:(6): 674–676.
6390:
6387:
6382:
6377:
6324:
6321:
6319:
6318:
6281:
6253:
6242:(6): 561–562.
6220:
6194:
6151:
6099:
6079:
6059:
6024:
5996:
5962:
5924:
5893:
5882:(4): 487–489.
5861:
5815:
5787:
5748:
5712:
5677:
5645:
5603:
5575:
5530:
5516:
5473:
5445:
5426:(3): 957–967.
5409:
5376:
5323:
5266:
5255:(5): 621–622.
5238:
5207:
5176:
5148:
5137:(3): 241–242.
5116:
5077:
5046:
5035:(7): 772–780.
5011:
4974:
4912:
4879:
4868:(2): 365–374.
4842:
4810:
4798:
4756:
4718:
4681:
4644:
4611:
4576:
4547:
4515:
4470:
4425:
4394:
4383:(1): 361–363.
4366:
4330:
4319:(1): 361–363.
4297:
4264:
4229:
4201:
4169:
4133:
4105:
4077:
4049:
4018:
3987:
3951:
3940:
3894:
3866:
3838:
3824:Ronald Breslow
3815:
3757:
3755:
3752:
3735:syringe filter
3714:
3711:
3698:
3695:
3694:
3693:
3689:
3685:
3679:
3675:
3671:
3664:
3655:
3651:
3647:
3643:
3639:
3619:
3615:
3611:
3607:
3600:
3597:
3571:
3568:
3512:
3509:
3464:
3461:
3433:
3430:
3404:
3401:
3337:
3334:
3296:
3293:
3259:conjugate acid
3246:
3243:
3227:organometallic
3210:
3207:
3160:
3157:
3148:
3145:
3144:
3143:
3133:
3115:organometallic
3110:
3107:
3106:
3105:
3097:
3089:
3083:
3082:
3071:
3060:
3049:
3038:
3027:
3016:
3005:
2994:
2983:
2972:
2961:
2950:
2939:
2928:
2920:
2919:
2908:
2897:
2886:
2875:
2864:
2853:
2842:
2831:
2820:
2809:
2798:
2787:
2776:
2765:
2757:
2756:
2752:
2751:
2740:
2729:
2718:
2707:
2696:
2685:
2674:
2663:
2652:
2641:
2630:
2619:
2608:
2597:
2586:
2575:
2568:
2557:
2546:
2540:
2539:
2528:
2517:
2506:
2495:
2484:
2473:
2462:
2451:
2440:
2429:
2418:
2407:
2396:
2385:
2374:
2363:
2356:
2345:
2334:
2328:
2327:
2316:
2305:
2294:
2283:
2272:
2261:
2250:
2239:
2228:
2217:
2206:
2195:
2184:
2173:
2162:
2151:
2149:
2138:
2127:
2121:
2120:
2109:
2098:
2087:
2076:
2065:
2054:
2043:
2032:
2021:
2010:
1999:
1988:
1977:
1966:
1955:
1944:
1942:
1931:
1920:
1914:
1913:
1902:
1891:
1880:
1869:
1858:
1847:
1844:
1833:
1822:
1816:
1815:
1804:
1793:
1782:
1771:
1760:
1749:
1746:
1735:
1724:
1718:
1717:
1706:
1703:
1692:
1686:
1685:
1682:
1675:
1674:
1669:
1664:
1659:
1654:
1649:
1644:
1639:
1634:
1629:
1624:
1619:
1614:
1609:
1604:
1599:
1594:
1592:
1587:
1582:
1575:
1574:
1570:Periodic table
1568:
1566:
1565:
1558:
1551:
1543:
1526:
1522:
1518:
1510:
1469:periodic table
1444:
1441:
1423:. The carbene
1386:deprotonations
1366:cycloadditions
1348:), performing
1330:
1329:
1326:
1323:
1320:
1318:
1315:
1311:
1308:
1304:
1303:
1300:
1297:
1294:
1292:
1289:
1285:
1281:
1278:
1274:
1273:
1270:
1266:
1263:
1260:
1258:
1255:
1252:
1248:
1247:
1244:
1241:
1238:
1236:
1233:
1230:
1208:
1205:
1168:dimerisation:
1161:
1157:
1153:
1149:
1119:
1088:
1085:
1080:
1073:
1020:
1011:
1008:
1006:
1003:
980:Main article:
977:
974:
972:being 85.7°).
970:dihedral angle
846:
843:
830:
827:
790:
787:
774:
771:
763:1,3-dithiolium
754:
746:
743:
739:dichalcogenide
675:
672:
642:
639:
609:1,2,4-triazole
589:
586:
561:effect of the
528:substituents:
502:
499:
494:
491:
447:
415:
353:
350:
259:isothiocyanate
243:Roald Hoffmann
234:
231:
189:was reported.
138:
135:derivative of
125:Ronald Breslow
120:
119:Early evidence
117:
115:
112:
91:stabilized by
53:whose natural
47:stable carbene
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
6487:
6476:
6473:
6471:
6468:
6466:
6463:
6461:
6458:
6457:
6455:
6444:
6440:
6436:
6432:
6428:
6425:
6424:
6418:
6417:
6416:
6409:
6405:
6401:
6398:
6397:
6391:
6388:
6386:
6383:
6381:
6378:
6373:
6369:
6365:
6361:
6357:
6353:
6349:
6345:
6341:
6337:
6336:
6330:
6329:
6328:
6322:
6314:
6310:
6306:
6302:
6298:
6295:
6294:
6285:
6282:
6277:
6273:
6269:
6266:
6265:
6257:
6254:
6249:
6245:
6241:
6237:
6236:
6231:
6224:
6221:
6216:
6212:
6208:
6207:
6198:
6195:
6190:
6186:
6182:
6178:
6174:
6170:
6166:
6162:
6155:
6152:
6147:
6143:
6139:
6135:
6131:
6127:
6123:
6119:
6115:
6112:
6111:
6103:
6100:
6097:
6096:3-540-36929-5
6093:
6089:
6083:
6080:
6077:
6076:3-527-31400-8
6073:
6069:
6063:
6060:
6054:
6049:
6045:
6041:
6040:
6035:
6028:
6025:
6020:
6016:
6012:
6009:
6008:
6000:
5997:
5992:
5988:
5984:
5981:
5980:
5975:
5969:
5967:
5963:
5958:
5954:
5950:
5947:
5946:
5941:
5937:
5931:
5929:
5925:
5920:
5916:
5912:
5909:
5908:
5900:
5898:
5894:
5889:
5885:
5881:
5878:
5877:
5872:
5865:
5862:
5857:
5853:
5849:
5845:
5841:
5838:
5837:
5832:
5828:
5822:
5820:
5816:
5811:
5807:
5803:
5800:
5799:
5791:
5788:
5783:
5779:
5775:
5771:
5767:
5764:
5763:
5755:
5753:
5749:
5744:
5740:
5736:
5732:
5728:
5725:
5724:
5716:
5713:
5708:
5704:
5700:
5697:
5696:
5691:
5684:
5682:
5678:
5673:
5669:
5665:
5662:
5661:
5656:
5649:
5646:
5641:
5637:
5632:
5627:
5623:
5620:
5619:
5614:
5607:
5604:
5599:
5595:
5591:
5588:
5587:
5579:
5576:
5571:
5567:
5563:
5559:
5555:
5551:
5547:
5544:
5543:
5542:Chem. Commun.
5538:
5536:
5529:
5520:
5517:
5512:
5508:
5504:
5500:
5496:
5493:
5492:
5484:
5477:
5474:
5469:
5465:
5461:
5460:
5459:Chem. Commun.
5452:
5450:
5446:
5441:
5437:
5433:
5429:
5425:
5422:
5421:
5413:
5410:
5405:
5401:
5397:
5393:
5392:
5387:
5380:
5377:
5372:
5368:
5364:
5360:
5356:
5352:
5348:
5344:
5340:
5336:
5335:
5327:
5324:
5319:
5315:
5310:
5305:
5301:
5297:
5293:
5290:
5289:
5284:
5282:
5273:
5271:
5267:
5262:
5258:
5254:
5251:
5250:
5242:
5239:
5234:
5230:
5226:
5223:
5222:
5214:
5212:
5208:
5203:
5199:
5195:
5192:
5191:
5183:
5181:
5177:
5172:
5168:
5164:
5161:
5160:
5152:
5149:
5144:
5140:
5136:
5135:
5134:Chem. Commun.
5127:
5125:
5123:
5121:
5117:
5112:
5108:
5104:
5101:
5100:
5095:
5088:
5086:
5084:
5082:
5078:
5073:
5069:
5065:
5062:
5061:
5053:
5051:
5047:
5042:
5038:
5034:
5031:
5030:
5029:Chem. Eur. J.
5025:
5018:
5016:
5012:
5007:
5003:
4999:
4996:
4995:
4994:Chem. Eur. J.
4990:
4983:
4981:
4979:
4975:
4970:
4966:
4961:
4956:
4952:
4948:
4944:
4940:
4936:
4932:
4931:
4926:
4919:
4917:
4913:
4908:
4904:
4900:
4897:
4896:
4888:
4886:
4884:
4880:
4875:
4871:
4867:
4864:
4863:
4855:
4853:
4851:
4849:
4847:
4843:
4838:
4834:
4830:
4826:
4822:
4818:
4817:
4805:
4803:
4799:
4794:
4790:
4786:
4785:
4784:Chem. Commun.
4780:
4776:
4769:
4767:
4765:
4763:
4761:
4757:
4752:
4748:
4744:
4740:
4736:
4729:
4727:
4725:
4723:
4719:
4714:
4710:
4706:
4703:
4702:
4694:
4692:
4690:
4688:
4686:
4682:
4677:
4673:
4669:
4666:
4665:
4657:
4655:
4653:
4651:
4649:
4645:
4640:
4636:
4632:
4629:
4628:
4620:
4618:
4616:
4612:
4607:
4603:
4599:
4596:
4595:
4587:
4585:
4583:
4581:
4577:
4565:
4561:
4557:
4551:
4548:
4543:
4539:
4535:
4532:
4531:
4526:
4519:
4516:
4511:
4507:
4503:
4499:
4495:
4491:
4487:
4483:
4482:
4474:
4471:
4466:
4462:
4458:
4454:
4450:
4446:
4442:
4438:
4437:
4429:
4426:
4421:
4417:
4413:
4410:
4409:
4401:
4399:
4395:
4390:
4386:
4382:
4379:
4378:
4370:
4367:
4362:
4358:
4354:
4350:
4346:
4343:
4342:
4334:
4331:
4326:
4322:
4318:
4315:
4314:
4306:
4304:
4302:
4298:
4293:
4289:
4285:
4282:
4281:
4273:
4271:
4269:
4265:
4260:
4256:
4252:
4249:
4248:
4236:
4234:
4230:
4225:
4221:
4217:
4214:
4213:
4205:
4202:
4197:
4193:
4189:
4186:
4185:
4176:
4174:
4170:
4165:
4161:
4157:
4153:
4149:
4146:
4145:
4137:
4134:
4129:
4125:
4121:
4118:
4117:
4109:
4106:
4101:
4097:
4093:
4090:
4089:
4081:
4078:
4073:
4069:
4065:
4062:
4061:
4053:
4050:
4045:
4041:
4037:
4034:
4033:
4025:
4023:
4019:
4014:
4010:
4006:
4003:
4002:
3994:
3992:
3988:
3983:
3979:
3975:
3971:
3967:
3964:
3963:
3955:
3952:
3949:
3944:
3941:
3936:
3932:
3927:
3922:
3918:
3915:
3914:
3909:
3907:
3904:"Umpolung by
3898:
3895:
3890:
3886:
3882:
3879:
3878:
3870:
3867:
3862:
3858:
3854:
3851:
3850:
3842:
3839:
3834:
3831:
3830:
3825:
3819:
3816:
3811:
3807:
3803:
3799:
3795:
3791:
3787:
3783:
3779:
3775:
3774:
3769:
3762:
3759:
3753:
3751:
3749:
3744:
3740:
3736:
3732:
3728:
3719:
3712:
3710:
3708:
3704:
3703:photochemical
3696:
3637:
3636:
3635:
3633:
3629:
3625:
3605:
3598:
3592:
3588:
3586:
3582:
3578:
3569:
3567:
3565:
3561:
3552:
3548:
3546:
3542:
3538:
3537:deoxygenation
3534:
3530:
3526:
3522:
3518:
3510:
3508:
3505:
3503:
3499:
3494:
3493:formamidinium
3490:
3486:
3481:
3474:
3469:
3462:
3460:
3458:
3454:
3453:-butyllithium
3452:
3447:
3443:
3439:
3431:
3426:
3425:formamidinium
3423:Reduction of
3421:
3417:
3415:
3410:
3409:alkyllithiums
3403:Alkyllithiums
3402:
3400:
3398:
3393:
3389:
3385:
3381:
3376:
3374:
3370:
3366:
3362:
3360:
3355:
3351:
3347:
3343:
3335:
3333:
3331:
3326:
3324:
3320:
3316:
3307:
3303:
3301:
3300:Deprotonation
3295:Deprotonation
3294:
3292:
3289:
3287:
3282:
3280:
3279:formamidinium
3276:
3272:
3268:
3264:
3260:
3257:value of the
3256:
3252:
3244:
3242:
3240:
3236:
3232:
3228:
3224:
3215:
3208:
3206:
3204:
3200:
3196:
3192:
3188:
3183:
3181:
3177:
3172:
3165:
3158:
3156:
3154:
3146:
3141:
3137:
3134:
3131:
3127:
3124:
3123:
3122:
3120:
3116:
3108:
3098:
3090:
3087:
3086:
3080:
3069:
3058:
3047:
3036:
3025:
3014:
3003:
2992:
2981:
2970:
2959:
2948:
2937:
2921:
2917:
2906:
2895:
2884:
2873:
2862:
2851:
2840:
2829:
2818:
2807:
2796:
2785:
2774:
2758:
2753:
2749:
2738:
2727:
2716:
2705:
2694:
2683:
2672:
2661:
2650:
2639:
2628:
2617:
2606:
2595:
2584:
2569:
2566:
2555:
2545:
2542:
2541:
2537:
2526:
2515:
2504:
2493:
2482:
2471:
2460:
2449:
2438:
2427:
2416:
2405:
2394:
2383:
2372:
2357:
2354:
2343:
2333:
2330:
2329:
2325:
2314:
2303:
2292:
2281:
2270:
2259:
2248:
2237:
2226:
2215:
2204:
2193:
2182:
2171:
2160:
2150:
2147:
2136:
2126:
2123:
2122:
2118:
2107:
2096:
2085:
2074:
2063:
2052:
2041:
2030:
2019:
2008:
1997:
1986:
1975:
1964:
1953:
1943:
1940:
1929:
1919:
1916:
1915:
1911:
1900:
1889:
1878:
1867:
1856:
1842:
1831:
1821:
1818:
1817:
1813:
1802:
1791:
1780:
1769:
1758:
1744:
1733:
1723:
1720:
1719:
1715:
1701:
1691:
1688:
1687:
1681:
1677:
1676:
1673:
1668:
1663:
1658:
1653:
1648:
1643:
1638:
1633:
1628:
1623:
1618:
1613:
1608:
1603:
1598:
1593:
1591:
1586:
1580:
1576:
1571:
1564:
1559:
1557:
1552:
1550:
1545:
1544:
1541:
1538:
1536:
1532:
1517:catalyst RuCl
1516:
1508:
1504:
1501:
1497:
1493:
1492:back-donation
1490:
1486:
1482:
1478:
1474:
1470:
1466:
1462:
1458:
1454:
1450:
1449:alkali metals
1442:
1437:
1432:
1428:
1426:
1422:
1418:
1414:
1410:
1406:
1402:
1398:
1393:
1391:
1387:
1383:
1379:
1375:
1371:
1367:
1363:
1359:
1355:
1351:
1347:
1343:
1339:
1327:
1324:
1321:
1319:
1316:
1309:
1306:
1305:
1301:
1298:
1295:
1293:
1290:
1279:
1276:
1275:
1271:
1264:
1261:
1259:
1256:
1253:
1250:
1249:
1245:
1242:
1239:
1237:
1234:
1231:
1228:
1227:
1220:
1216:
1214:
1206:
1204:
1202:
1198:
1194:
1193:singlet state
1190:
1189:triplet state
1185:
1182:
1181:formamidinium
1173:
1169:
1167:
1166:singlet state
1146:
1144:
1140:
1139:electrostatic
1136:
1127:
1123:
1117:
1113:
1112:deprotonating
1108:
1106:
1102:
1098:
1094:
1086:
1084:
1079:
1072:
1067:
1065:
1061:
1057:
1056:1-bromohexane
1049:
1044:
1040:
1033:
1029:
1027:
1023:
1019:
1009:
1004:
1002:
1000:
996:
992:
988:
983:
975:
973:
971:
967:
963:
959:
955:
951:
947:
943:
934:
930:
928:
924:
923:hybridisation
920:
916:
911:
909:
905:
901:
897:
896:decomposition
894:
893:photochemical
890:
881:
877:
875:
870:
868:
864:
860:
856:
855:Hideo Tomioka
852:
844:
842:
840:
836:
828:
823:
819:
814:
810:
808:
804:
800:
796:
788:
786:
784:
780:
770:
768:
764:
760:
752:
744:
742:
740:
736:
732:
728:
723:
720:
716:
712:
707:
705:
701:
697:
688:
684:
682:
673:
671:
667:
665:
655:
651:
649:
640:
638:
636:
632:
628:
620:
616:
614:
610:
607:based on the
606:
597:
593:
587:
585:
581:
578:
576:
572:
568:
564:
560:
556:
548:
541:
533:
529:
527:
523:
519:
514:
512:
508:
500:
498:
492:
487:
482:
478:
476:
472:
468:
464:
460:
456:
451:
444:
440:
436:
427:
423:
421:
413:
409:
405:
401:
397:
389:
384:
380:
378:
374:
370:
362:
358:
351:
349:
345:
342:
340:
336:
332:
323:
316:
311:
307:
305:
304:deprotonation
300:
298:
289:
285:
283:
279:
275:
266:
262:
260:
256:
252:
248:
244:
240:
232:
230:
227:
223:
219:
215:
211:
207:
203:
199:
195:
190:
188:
179:
175:
173:
169:
165:
161:
157:
153:
149:
145:
141:
134:
130:
126:
118:
113:
111:
109:
105:
100:
98:
94:
90:
86:
82:
77:
75:
71:
69:
64:
60:
56:
52:
48:
44:
36:
32:
19:
6426:
6421:
6414:
6399:
6394:
6339:
6333:
6326:
6296:
6291:
6284:
6267:
6262:
6256:
6239:
6233:
6232:)-thiones".
6229:
6223:
6204:
6197:
6164:
6160:
6154:
6113:
6108:
6102:
6082:
6070:, Wiley-VCH
6062:
6043:
6037:
6027:
6010:
6005:
5999:
5982:
5977:
5948:
5943:
5939:
5910:
5905:
5879:
5874:
5870:
5864:
5839:
5834:
5830:
5804:(36): 6926.
5801:
5796:
5790:
5765:
5760:
5726:
5721:
5715:
5698:
5693:
5689:
5663:
5658:
5654:
5648:
5621:
5616:
5606:
5589:
5584:
5578:
5545:
5540:
5534:
5527:
5519:
5494:
5489:
5476:
5462:(12): 1267.
5457:
5423:
5418:
5412:
5395:
5389:
5379:
5338:
5332:
5326:
5291:
5286:
5280:
5252:
5247:
5241:
5224:
5219:
5193:
5188:
5162:
5157:
5151:
5132:
5102:
5097:
5093:
5063:
5058:
5032:
5027:
5023:
4997:
4992:
4988:
4934:
4928:
4898:
4893:
4865:
4860:
4820:
4814:
4782:
4778:
4774:
4734:
4704:
4699:
4667:
4662:
4630:
4625:
4597:
4592:
4567:. Retrieved
4563:
4556:Rzepa, Henry
4550:
4533:
4528:
4524:
4518:
4485:
4479:
4473:
4440:
4434:
4428:
4411:
4406:
4380:
4375:
4369:
4344:
4339:
4333:
4316:
4311:
4283:
4278:
4250:
4245:
4215:
4210:
4204:
4187:
4182:
4147:
4142:
4136:
4119:
4114:
4108:
4091:
4086:
4080:
4063:
4058:
4052:
4038:(2): 75–80.
4035:
4030:
4004:
3999:
3965:
3962:Angew. Chem.
3960:
3954:
3943:
3916:
3911:
3905:
3897:
3880:
3875:
3869:
3852:
3847:
3841:
3832:
3827:
3818:
3777:
3771:
3767:
3761:
3730:
3724:
3713:Purification
3700:
3602:
3584:
3580:
3573:
3557:
3514:
3506:
3502:crown ethers
3482:
3478:
3450:
3435:
3432:Amides bases
3406:
3377:
3369:dimsyl anion
3358:
3339:
3327:
3312:
3298:
3290:
3285:
3283:
3275:nucleophilic
3248:
3220:
3209:Applications
3203:trans effect
3198:
3194:
3190:
3186:
3184:
3173:
3170:
3150:
3112:
1446:
1435:
1424:
1416:
1412:
1408:
1404:
1400:
1394:
1389:
1381:
1377:
1373:
1369:
1361:
1353:
1345:
1341:
1338:nucleophilic
1335:
1212:
1210:
1186:
1178:
1147:
1132:
1109:
1090:
1087:Dimerisation
1077:
1070:
1068:
1064:nucleophilic
1053:
1038:
1017:
1013:
998:
994:
990:
985:
942:benzophenone
939:
912:
900:diazomethane
886:
871:
848:
832:
794:
792:
776:
748:
730:
724:
708:
693:
677:
668:
660:
644:
625:
602:
591:
582:
579:
552:
515:
504:
496:
452:
432:
407:
395:
393:
373:dative bonds
366:
346:
343:
335:bond lengths
327:
318:
314:
301:
294:
274:Guy Bertrand
271:
236:
191:
186:
184:
146:that yields
129:nucleophilic
122:
101:
85:ground state
78:
73:
67:
66:
46:
42:
40:
6090:, Springer
5701:: 397–399.
4286:: 323–355.
4190:: 176–179.
3968:(14): 494.
3743:sublimation
3646:Si−Hg−Si(CH
3626:and chloro-
3614:Si-Hg-Si(CH
3579:of adducts
3407:The use of
3319:imidazolium
1505:in several
1461:lanthanides
1397:isothiazole
1152:N–C–OR or R
793:One stable
648:Roger Alder
635:nucleophile
471:heteroatoms
459:double bond
439:aromaticity
339:double bond
247:aromaticity
239:imidazolium
172:equilibrium
131:carbene, a
6454:Categories
6110:Chem. Rev.
5907:Chem. Ber.
5398:(33): 11.
4341:Chem. Rev.
4001:Chem. Ber.
3829:Chem. Ind.
3754:References
1535:tridentate
1207:Reactivity
1135:lone pairs
966:bond angle
962:picometers
919:orthogonal
874:orthogonal
822:cyclohexyl
779:phosphorus
727:chalcogens
704:oxyiminium
681:phosphorus
467:octet rule
379:orbitals.
210:chloroform
156:thiazolium
6235:Synthesis
6161:Chem. Rev
6146:206902952
6130:0009-2665
5570:205757477
3628:amidinium
3564:potassium
3525:potassium
3521:thioureas
3498:cryptands
3440:and the (
3414:isobutene
3384:potassium
3373:hydroxide
3365:catalysts
3350:inorganic
3330:precursor
3231:ruthenium
1531:bidentate
1500:phosphine
1494:from the
1485:lone pair
1465:actinides
1459:and even
1411:-thiete (
1399:carbene (
1340:fashion (
1201:p-orbital
1197:lone pair
889:half-life
867:half-life
801:with one
759:acetylene
741:(X=C=X).
575:induction
571:lone pair
507:imidazole
443:imidazole
418:ImC:), a
400:adamantyl
369:resonance
272:In 1988,
192:In 1960,
162:, the C2-
137:vitamin B
123:In 1957,
6465:Carbenes
6443:20715233
6364:24965649
6313:14746458
6189:43090499
6181:16277368
6138:19588961
5856:97336589
5782:17661300
5743:17372997
5640:15457494
5562:21116519
5511:23173841
5440:16417387
5363:11493917
5318:16028925
4969:16614171
4751:15467857
4510:11397943
4465:10796999
4361:19281270
4164:11093196
3935:23081675
3802:24965649
3707:nitrogen
3658:N=C(Cl)−
3545:fluorene
3346:hydrogen
3281:salts).
3153:carbenes
1581: →
1496:nitrogen
1254:RXH, RT
1105:aromatic
1101:solution
1093:dimerise
1028:(DMSO):
958:distance
927:orbitals
908:nitrogen
799:borazine
715:divalent
696:thiazole
627:Triazole
563:chlorine
555:chlorine
455:hindered
168:deuteron
152:furfural
81:misnomer
49:) is an
6344:Bibcode
5371:4373216
5343:Bibcode
5309:2440681
4960:2427275
4939:Bibcode
4930:Science
4825:Bibcode
4811:carbene
4490:Bibcode
4481:Science
4445:Bibcode
4436:Science
3970:Bibcode
3782:Bibcode
3748:lithium
3632:mercury
3624:iminium
3543:with a
3388:ammonia
3271:acidity
3233:-based
3136:Schrock
3126:Fischer
1678:↓
1503:ligands
1481:ligands
1317:25–68%
1302:47–84%
1291:54–68%
1272:71–90%
1257:95–97%
1143:carbene
1048:D6-DMSO
964:with a
950:bromine
851:singlet
783:silicon
404:prevent
377:pi-bond
297:alkynic
255:mercury
114:History
104:ligands
63:carbene
6441:
6372:672379
6370:
6362:
6335:Nature
6311:
6187:
6179:
6144:
6136:
6128:
6094:
6074:
5854:
5780:
5741:
5638:
5568:
5560:
5509:
5438:
5369:
5361:
5334:Nature
5316:
5306:
4967:
4957:
4749:
4569:15 Feb
4508:
4463:
4359:
4162:
3933:
3835:: 893.
3810:672379
3808:
3800:
3773:Nature
3731:per se
3684:+ 2(CH
3674:N−C−NR
3670:Cl → R
3380:sodium
3263:oxygen
3187:et al.
3101:
3093:
3088:Legend
1680:Period
1489:ligand
1213:et al.
1160:N–C–NR
1060:alkene
1046:Using
956:, the
915:phenyl
781:and a
735:alkyne
719:steric
711:oxygen
709:Since
573:, via
526:chiral
477:ring.
412:methyl
164:proton
148:furoin
6368:S2CID
6185:S2CID
6142:S2CID
5852:S2CID
5566:S2CID
5486:(PDF)
5367:S2CID
3806:S2CID
3539:of a
3523:with
3489:KHMDS
3473:KHMDS
3361:-BuOH
3253:(the
3251:basic
3195:trans
3191:trans
1579:Group
910:gas.
898:of a
818:LiTMP
803:boron
700:thio-
518:alkyl
371:with
331:X-ray
253:with
226:Lemal
214:dimer
150:from
89:ylide
6439:PMID
6360:PMID
6309:PMID
6240:1993
6177:PMID
6134:PMID
6126:ISSN
6092:ISBN
6072:ISBN
5871:tert
5778:PMID
5739:PMID
5664:1996
5636:PMID
5558:PMID
5507:PMID
5436:PMID
5359:PMID
5314:PMID
4965:PMID
4866:1997
4747:PMID
4571:2024
4506:PMID
4461:PMID
4357:PMID
4160:PMID
3931:PMID
3798:PMID
3692:SiCl
3678:+ Hg
3558:The
3541:urea
3485:HMDS
3446:LiOH
3354:DMSO
3321:and
3315:IMes
3286:situ
3237:and
1533:and
1521:(PCy
1467:. A
1463:and
1376:and
1364:),
1344:and
1328:21%
1246:92%
1235:92%
1118:(–CH
731:both
729:for
702:and
603:The
522:aryl
361:MO's
257:and
216:, a
6431:doi
6404:doi
6352:doi
6340:510
6301:doi
6297:126
6272:doi
6268:118
6244:doi
6211:doi
6169:doi
6165:105
6118:doi
6114:109
6048:doi
6015:doi
5987:doi
5953:doi
5915:doi
5911:129
5884:doi
5844:doi
5806:doi
5770:doi
5731:doi
5703:doi
5699:339
5668:doi
5626:doi
5594:doi
5550:doi
5499:doi
5495:134
5464:doi
5428:doi
5424:128
5400:doi
5351:doi
5339:412
5304:PMC
5296:doi
5292:127
5257:doi
5229:doi
5198:doi
5167:doi
5139:doi
5107:doi
5068:doi
5037:doi
5002:doi
4955:PMC
4947:doi
4935:312
4903:doi
4899:120
4870:doi
4833:doi
4821:103
4789:doi
4739:doi
4709:doi
4672:doi
4635:doi
4631:117
4602:doi
4598:114
4538:doi
4534:690
4498:doi
4486:292
4453:doi
4441:288
4416:doi
4412:119
4385:doi
4381:113
4349:doi
4345:109
4321:doi
4317:113
4288:doi
4284:137
4255:doi
4251:110
4220:doi
4192:doi
4188:731
4152:doi
4124:doi
4096:doi
4068:doi
4040:doi
4009:doi
3978:doi
3921:doi
3885:doi
3857:doi
3790:doi
3778:510
3681:(l)
3654:+ R
3638:(CH
3606:(CH
3529:THF
3527:in
3519:of
3500:or
3392:THF
3382:or
3356:or
3255:pKa
3225:in
3199:cis
3180:NMR
3176:NMR
3073:102
3062:101
3051:100
2742:118
2731:117
2720:116
2709:115
2698:114
2687:113
2676:112
2665:111
2654:110
2643:109
2632:108
2621:107
2610:106
2599:105
2588:104
2577:103
1356:),
1284:, S
753:(CS
666:).
106:in
95:or
61:(a
6456::
6437:.
6427:49
6400:30
6366:.
6358:.
6350:.
6338:.
6307:.
6238:.
6183:.
6175:.
6163:.
6140:.
6132:.
6124:.
6044:28
6042:.
6036:.
6011:40
5983:35
5965:^
5949:34
5927:^
5896:^
5880:34
5850:.
5840:36
5818:^
5802:46
5776:.
5766:46
5751:^
5737:.
5727:46
5680:^
5634:.
5622:43
5615:.
5590:35
5564:.
5556:.
5546:47
5539:.
5526:"p
5505:.
5488:.
5448:^
5434:.
5396:79
5394:.
5388:.
5365:.
5357:.
5349:.
5337:.
5312:.
5302:.
5285:.
5269:^
5253:28
5225:95
5210:^
5194:92
5179:^
5163:79
5119:^
5103:34
5080:^
5064:35
5049:^
5014:^
4977:^
4963:.
4953:.
4945:.
4933:.
4927:.
4915:^
4882:^
4845:^
4831:.
4819:.
4801:^
4759:^
4745:.
4721:^
4705:36
4684:^
4668:35
4647:^
4614:^
4579:^
4562:.
4504:.
4496:.
4484:.
4459:.
4451:.
4439:.
4397:^
4355:.
4300:^
4267:^
4232:^
4216:90
4172:^
4158:.
4148:39
4120:40
4092:87
4064:86
4021:^
4005:94
3990:^
3976:.
3966:72
3929:.
3917:51
3910:.
3881:79
3853:80
3833:26
3804:.
3796:.
3788:.
3776:.
3660:NR
3504:.
3416::
3205:.
3155:.
3078:No
3067:Md
3056:Fm
3045:Es
3040:99
3034:Cf
3029:98
3023:Bk
3018:97
3012:Cm
3007:96
3001:Am
2996:95
2990:Pu
2985:94
2979:Np
2974:93
2963:92
2957:Pa
2952:91
2946:Th
2941:90
2935:Ac
2930:89
2915:Yb
2910:70
2904:Tm
2899:69
2893:Er
2888:68
2882:Ho
2877:67
2871:Dy
2866:66
2860:Tb
2855:65
2849:Gd
2844:64
2838:Eu
2833:63
2827:Sm
2822:62
2816:Pm
2811:61
2805:Nd
2800:60
2794:Pr
2789:59
2783:Ce
2778:58
2772:La
2767:57
2747:Og
2736:Ts
2725:Lv
2714:Mc
2703:Fl
2692:Nh
2681:Cn
2670:Rg
2659:Ds
2648:Mt
2637:Hs
2626:Bh
2615:Sg
2604:Db
2593:Rf
2582:Lr
2564:Ra
2559:88
2553:Fr
2548:87
2535:Rn
2530:86
2524:At
2519:85
2513:Po
2508:84
2502:Bi
2497:83
2491:Pb
2486:82
2480:Tl
2475:81
2469:Hg
2464:80
2458:Au
2453:79
2447:Pt
2442:78
2436:Ir
2431:77
2425:Os
2420:76
2414:Re
2409:75
2398:74
2392:Ta
2387:73
2381:Hf
2376:72
2370:Lu
2365:71
2352:Ba
2347:56
2341:Cs
2336:55
2323:Xe
2318:54
2307:53
2301:Te
2296:52
2290:Sb
2285:51
2279:Sn
2274:50
2268:In
2263:49
2257:Cd
2252:48
2246:Ag
2241:47
2235:Pd
2230:46
2224:Rh
2219:45
2213:Ru
2208:44
2202:Tc
2197:43
2191:Mo
2186:42
2180:Nb
2175:41
2169:Zr
2164:40
2153:39
2145:Sr
2140:38
2134:Rb
2129:37
2116:Kr
2111:36
2105:Br
2100:35
2094:Se
2089:34
2083:As
2078:33
2072:Ge
2067:32
2061:Ga
2056:31
2050:Zn
2045:30
2039:Cu
2034:29
2028:Ni
2023:28
2017:Co
2012:27
2006:Fe
2001:26
1995:Mn
1990:25
1984:Cr
1979:24
1968:23
1962:Ti
1957:22
1951:Sc
1946:21
1938:Ca
1933:20
1922:19
1909:Ar
1904:18
1898:Cl
1893:17
1882:16
1871:15
1865:Si
1860:14
1854:Al
1849:13
1840:Mg
1835:12
1829:Na
1824:11
1811:Ne
1806:10
1742:Be
1731:Li
1713:He
1672:18
1667:17
1662:16
1657:15
1652:14
1647:13
1642:12
1637:11
1632:10
1455:,
1451:,
1436:2b
1425:2b
1401:2b
1372:,
1322:h
1307:d
1296:g
1277:c
1265:CS
1262:f
1251:b
1240:e
1229:a
1066:.
904:nm
841:.
717:,
683:.
520:,
486:3D
446:Me
388:3D
284::
261::
174:.
110:.
41:A
6445:.
6433::
6410:.
6406::
6376:.
6374:.
6354::
6346::
6315:.
6303::
6278:.
6274::
6250:.
6246::
6230:H
6217:.
6213::
6191:.
6171::
6148:.
6120::
6056:.
6050::
6021:.
6017::
5993:.
5989::
5959:.
5955::
5940:N
5921:.
5917::
5890:.
5886::
5858:.
5846::
5831:N
5812:.
5808::
5784:.
5772::
5745:.
5733::
5709:.
5705::
5690:H
5674:.
5670::
5655:H
5642:.
5628::
5600:.
5596::
5572:.
5552::
5535:N
5531:a
5528:K
5513:.
5501::
5470:.
5466::
5442:.
5430::
5406:.
5402::
5373:.
5353::
5345::
5320:.
5298::
5281:N
5263:.
5259::
5235:.
5231::
5204:.
5200::
5173:.
5169::
5145:.
5141::
5113:.
5109::
5094:H
5074:.
5070::
5043:.
5039::
5033:2
5024:N
5008:.
5004::
4998:2
4989:N
4971:.
4949::
4941::
4909:.
4905::
4876:.
4872::
4839:.
4835::
4827::
4795:.
4791::
4779:N
4775:N
4753:.
4741::
4715:.
4711::
4678:.
4674::
4641:.
4637::
4608:.
4604::
4573:.
4544:.
4540::
4525:N
4512:.
4500::
4492::
4467:.
4455::
4447::
4422:.
4418::
4391:.
4387::
4363:.
4351::
4327:.
4323::
4294:.
4290::
4261:.
4257::
4242:′
4226:.
4222::
4198:.
4194::
4166:.
4154::
4130:.
4126::
4102:.
4098::
4074:.
4070::
4046:.
4042::
4036:1
4015:.
4011::
3984:.
3980::
3972::
3937:.
3923::
3906:N
3891:.
3887::
3863:.
3859::
3812:.
3792::
3784::
3768:N
3690:3
3688:)
3686:3
3676:2
3672:2
3665:2
3656:2
3652:3
3650:)
3648:3
3644:3
3642:)
3640:3
3620:3
3618:)
3616:3
3612:3
3610:)
3608:3
3585:B
3581:A
3451:n
3390:/
3359:t
3197:-
2968:U
2544:7
2403:W
2332:6
2312:I
2158:Y
2125:5
1973:V
1927:K
1918:4
1887:S
1876:P
1820:3
1800:F
1795:9
1789:O
1784:8
1778:N
1773:7
1767:C
1762:6
1756:B
1751:5
1737:4
1726:3
1722:2
1708:2
1699:H
1694:1
1690:1
1627:9
1622:8
1617:7
1612:6
1607:5
1602:4
1597:3
1590:2
1585:1
1562:e
1555:t
1548:v
1527:2
1525:)
1523:3
1519:2
1511:3
1417:3
1413:4
1409:H
1405:1
1390:b
1382:a
1378:h
1374:g
1370:d
1368:(
1362:c
1360:(
1354:b
1352:(
1346:f
1342:e
1312:1
1310:R
1286:8
1282:2
1280:O
1267:2
1162:2
1158:2
1154:2
1150:2
1120:2
1081:a
1078:K
1074:a
1071:K
1069:p
1021:a
1018:K
1016:p
999:N
995:N
991:N
795:N
755:2
549:)
545:(
488:)
448:4
416:4
408:N
398:-
396:N
390:)
321:′
319:N
317:,
315:N
139:1
68:N
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.