77:
221:
632:
160:, whereas benzaldehyde is both a proton acceptor and donor. In this way it is possible to synthesise mixed benzoins, i.e. products with different groups on each half of the product. However, care should be taken to match a proton donating aldehyde with a proton accepting aldehyde to avoid undesired homo-dimerization.
3816:
236:
using cyanide or thiazolium catalysts. The reaction mechanism is the same as above, but it occurs in the reverse direction. This can allow the access of ketones otherwise difficult to produce.
149:
2932:
2877:
3645:
2987:
3137:
1771:
3866:
3640:
2742:
1466:
2512:
663:
3312:
1256:
3407:
1511:
3387:
2882:
2049:
1930:
1486:
3477:
185:
206:
is responsible for biosynthesis of acyloin-like compounds utilizing the benzoin addition. This coenzyme also contains a thiazolium moiety, which on
3232:
699:
593:
2064:
3710:
3167:
3660:
591:
D. Enders, O. Niemeier & T. Balensiefer (2006). "Asymmetric
Intramolecular Crossed-Benzoin Reactions by N-Heterocyclic Carbene Catalysis".
3272:
3252:
3212:
2019:
3806:
3731:
3615:
2227:
1631:
962:
636:
3801:
3630:
3287:
3142:
2772:
568:
Knight, Roland; Leeper, F. (1998). "Comparison of chiral thiazolium and triazolium salts as asymmetric catalysts for the benzoin addition".
2617:
1854:
2977:
2467:
2142:
3881:
3665:
2687:
232:
can be synthetically useful. If a benzoin or acyloin can be synthesized by another method, then they can be converted into the component
3242:
3876:
3590:
3452:
3207:
3766:
3237:
3152:
3122:
3102:
2967:
2962:
2337:
2262:
1905:
1859:
1726:
987:
3705:
3871:
3831:
3781:
3257:
3007:
2937:
1426:
156:
In this reaction, one aldehyde donates a proton and one aldehyde accepts a proton. Some aldehydes can only donate protons, such as
3467:
3072:
1965:
1686:
528:"CXXII.—Reactions involving the addition of hydrogen cyanide to carbon compounds. Part II. Cyanohydrins regarded as complex acids"
396:
F. Wöhler, J. Liebig (1832). "Untersuchungen über das
Radikal der Benzoesäure" [Studies on the radicals of benzoic acid].
3457:
997:
3625:
3382:
3332:
2122:
2054:
1945:
1521:
1276:
1201:
982:
3337:
3147:
2622:
2532:
656:
3911:
3695:
3635:
3037:
3012:
2922:
2502:
2382:
1416:
912:
3796:
3282:
3077:
1346:
157:
3901:
3487:
2997:
2507:
2452:
2297:
2257:
2089:
1844:
1561:
1411:
3861:
3422:
3377:
2867:
2722:
148:
3896:
3811:
3670:
3585:
3482:
2557:
2212:
1880:
1291:
852:
217:
salts. Triazolium salts were found to give greater enantiomeric excess than thiazolium salts. An example is shown below.
3786:
3761:
3746:
3442:
3307:
3262:
3027:
2572:
2422:
1636:
1316:
1261:
3791:
3736:
3267:
2682:
2397:
2392:
1885:
1701:
1691:
1406:
1266:
1216:
1211:
1186:
1092:
3960:
3846:
3447:
3367:
2982:
2947:
2792:
2217:
2177:
2074:
1849:
1601:
1546:
1146:
857:
847:
822:
3821:
3522:
3327:
2762:
2327:
2302:
2242:
1834:
1541:
1191:
1376:
3112:
2647:
2099:
1321:
1286:
882:
817:
649:
51:
3680:
3302:
2362:
2287:
1811:
1646:
1331:
1107:
1067:
812:
3921:
3826:
3560:
3532:
3502:
3417:
3347:
3277:
3197:
3097:
3057:
2752:
2372:
1671:
1666:
1128:
992:
3886:
3756:
3620:
3462:
3322:
2842:
1816:
1366:
1326:
1077:
3776:
3372:
3342:
3217:
3172:
3002:
2912:
2727:
2717:
2547:
2104:
2044:
2009:
1796:
1756:
1531:
1401:
917:
907:
837:
547:
502:
458:
20:
3352:
2332:
1356:
902:
782:
3565:
3856:
3715:
3507:
3432:
3412:
3132:
3082:
2942:
2907:
2847:
2777:
2079:
2059:
1791:
1711:
1606:
1566:
1536:
1471:
1341:
1251:
1241:
1117:
827:
334:
Enders, Dieter; Niemeier, Oliver; Henseler, Alexander (2007). "Organocatalysis by N-Heterocyclic
Carbenes".
142:
3595:
3317:
3067:
3047:
3022:
2972:
2887:
2862:
2817:
2787:
2767:
2737:
2702:
2657:
2632:
2607:
2492:
2417:
2197:
1890:
1826:
1626:
1351:
1271:
957:
932:
709:
704:
181:
3931:
2677:
1301:
3517:
3472:
3187:
3157:
3127:
3062:
3042:
2957:
2952:
2917:
2872:
2857:
2852:
2832:
2822:
2757:
2747:
2627:
2147:
1950:
1526:
1481:
1311:
1047:
767:
729:
485:
441:
398:
122:
977:
972:
3570:
3700:
3650:
3600:
3580:
3427:
3402:
3117:
3107:
2992:
2807:
2802:
2732:
2517:
2317:
2277:
2207:
2172:
2127:
2094:
1960:
1935:
1915:
1736:
1696:
1656:
1621:
1551:
1306:
1176:
1151:
689:
250:
180:; the reaction mechanism is essentially the same. These compounds are important in the synthesis of
3916:
3906:
3891:
3537:
3512:
3497:
3492:
3222:
3177:
3162:
3052:
3032:
2927:
2812:
2797:
2642:
2587:
2577:
2567:
2542:
2307:
2182:
2157:
2069:
1925:
1910:
1895:
1751:
1716:
1661:
1431:
1281:
1226:
1097:
1012:
872:
797:
255:
138:
62:
942:
3655:
3605:
3575:
3437:
3227:
3017:
2902:
2837:
2827:
2592:
2522:
2487:
2482:
2462:
2457:
2402:
2312:
2162:
2024:
2014:
1920:
1706:
1651:
1581:
1501:
1396:
1296:
1231:
1156:
1002:
867:
802:
169:
110:
102:
787:
76:
3392:
2712:
2597:
2562:
2527:
2472:
2427:
2387:
2342:
2322:
2272:
2267:
2237:
2222:
2132:
2039:
1975:
1940:
1766:
1641:
1516:
1441:
1421:
1336:
1171:
1166:
1112:
1022:
927:
887:
842:
724:
719:
684:
610:
351:
316:
213:
The asymmetric version of this reaction has been performed by utilizing chiral thiazolium and
58:
39:
31:
3926:
3771:
3741:
3685:
3610:
3542:
3297:
3247:
3092:
2897:
2672:
2667:
2612:
2602:
2377:
2187:
2167:
2137:
2034:
1970:
1955:
1786:
1741:
1731:
1721:
1616:
1596:
1591:
1576:
1571:
1451:
1446:
1386:
1371:
1361:
1206:
1196:
1062:
1052:
1042:
952:
947:
922:
862:
714:
673:
602:
573:
539:
494:
450:
415:
407:
378:
343:
306:
298:
260:
189:
173:
3836:
3527:
3362:
3357:
2652:
2637:
2582:
2537:
2497:
2447:
2412:
2407:
2352:
2347:
2282:
2232:
2152:
1980:
1864:
1839:
1801:
1776:
1761:
1746:
1681:
1556:
1506:
1496:
1476:
1436:
1246:
1236:
1221:
1017:
937:
762:
757:
523:
177:
114:
106:
807:
777:
3841:
3751:
3690:
2782:
2692:
2662:
2437:
2292:
2029:
1806:
1676:
1491:
1461:
1161:
1057:
832:
694:
311:
286:
245:
133:
group, which then adds to the second carbonyl group in a second nucleophilic addition.
118:
220:
137:
transfer and elimination of the cyanide ion affords benzoin as the product. This is a
3954:
3851:
3552:
3397:
3292:
3087:
2477:
2442:
2432:
2367:
2357:
2247:
2084:
1900:
1611:
1586:
1456:
1102:
1087:
1072:
967:
897:
877:
792:
207:
70:
439:[Contributions to the knowledge of some compounds from the benzoyl series].
2892:
2252:
2004:
1781:
1381:
1181:
1032:
1027:
892:
747:
196:
134:
1391:
1037:
1007:
772:
94:
3675:
3202:
2552:
641:
141:, which means that the distribution of products is determined by the relative
498:
454:
411:
382:
287:"Recent advances in N-heterocyclic carbene (NHC)-catalysed benzoin reactions"
90:
614:
606:
420:
355:
320:
69:. The catalytic version of the reaction involving cyanide was developed by
631:
1082:
752:
543:
265:
228:
Since the products of the reaction are thermodynamically controlled, the
214:
203:
200:
130:
126:
35:
742:
302:
98:
47:
43:
347:
577:
483:[Study of some decomposition products of bitter almond oil].
233:
66:
527:
480:
436:
437:"Beiträge zur Kenntniss einiger Verbindungen aus der Benzoylreihe"
219:
75:
19:
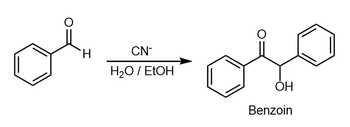
This article is about synthesis of
Benzoin. For other uses, see
1126:
645:
147:
117:. In the first step in this reaction, the cyanide anion (as
637:
animation of the
Benzoin condensation reaction mechanism
285:
Menon, Rajeev S.; Biju, Akkattu T.; Nair, Vijay (2016).
57:
The benzoin condensation was first reported in 1832 by
50:. In the classic example, benzaldehyde is converted to
481:"Ueber einige Zersetzungsprodukte des Bittermandelöls"
3817:
Erlenmeyer–Plöchl azlactone and amino-acid synthesis
3724:
3551:
3186:
2701:
2196:
2113:
1993:
1873:
1825:
1135:
184:. The analogous 1,4-addition of an aldehyde to an
2878:Divinylcyclopropane-cycloheptadiene rearrangement
125:. Rearrangement of the intermediate results in
3138:Thermal rearrangement of aromatic hydrocarbons
1772:Thermal rearrangement of aromatic hydrocarbons
3867:Lectka enantioselective beta-lactam synthesis
657:
532:Journal of the Chemical Society, Transactions
369:Roger Adams; C. S. Marvel (1921). "Benzoin".
8:
3646:Inverse electron-demand Diels–Alder reaction
1467:Heterogeneous metal catalyzed cross-coupling
224:Scheme 2. An intramolecular benzoin addition
2988:Lobry de Bruyn–Van Ekenstein transformation
3548:
1822:
1123:
664:
650:
642:
3478:Petrenko-Kritschenko piperidone synthesis
2933:Fritsch–Buttenberg–Wiechell rearrangement
419:
310:
3641:Intramolecular Diels–Alder cycloaddition
38:. The reaction generally occurs between
594:Angewandte Chemie International Edition
277:
145:of the products and starting material.
16:Reaction between two aromatic aldehydes
3661:Metal-centered cycloaddition reactions
3313:Debus–Radziszewski imidazole synthesis
1257:Bodroux–Chichibabin aldehyde synthesis
291:Beilstein Journal of Organic Chemistry
3807:Diazoalkane 1,3-dipolar cycloaddition
3711:Vinylcyclopropane (5+2) cycloaddition
3616:Diazoalkane 1,3-dipolar cycloaddition
3388:Hurd–Mori 1,2,3-thiadiazole synthesis
2883:Dowd–Beckwith ring-expansion reaction
2050:Hurd–Mori 1,2,3-thiadiazole synthesis
963:LFER solvent coefficients (data page)
7:
2618:Sharpless asymmetric dihydroxylation
1855:Methoxymethylenetriphenylphosphorane
2743:Allen–Millar–Trippett rearrangement
3882:Nitrone-olefin (3+2) cycloaddition
3877:Niementowski quinazoline synthesis
3666:Nitrone-olefin (3+2) cycloaddition
3591:Azide-alkyne Huisgen cycloaddition
3453:Niementowski quinazoline synthesis
3208:Azide-alkyne Huisgen cycloaddition
2513:Meerwein–Ponndorf–Verley reduction
2065:Leimgruber–Batcho indole synthesis
14:
3706:Trimethylenemethane cycloaddition
3408:Johnson–Corey–Chaykovsky reaction
3273:Cadogan–Sundberg indole synthesis
3253:Bohlmann–Rahtz pyridine synthesis
3213:Baeyer–Emmerling indole synthesis
2020:Cadogan–Sundberg indole synthesis
1512:Johnson–Corey–Chaykovsky reaction
152:Mechanism of the benzoin addition
46:, and results in formation of an
3802:Cook–Heilbron thiazole synthesis
3631:Hexadehydro Diels–Alder reaction
3458:Niementowski quinoline synthesis
3288:Cook–Heilbron thiazole synthesis
3233:Bischler–Möhlau indole synthesis
3143:Tiffeneau–Demjanov rearrangement
2773:Baker–Venkataraman rearrangement
1931:Horner–Wadsworth–Emmons reaction
1602:Mizoroki-Heck vs. Reductive Heck
1487:Horner–Wadsworth–Emmons reaction
998:Neighbouring group participation
630:
210:becomes a nucleophilic carbene.
168:The reaction can be extended to
121:) reacts with the aldehyde in a
3338:Fiesselmann thiophene synthesis
3168:Westphalen–Lettré rearrangement
3148:Vinylcyclopropane rearrangement
2978:Kornblum–DeLaMare rearrangement
2623:Epoxidation of allylic alcohols
2533:Noyori asymmetric hydrogenation
2468:Kornblum–DeLaMare rearrangement
2143:Gallagher–Hollander degradation
550:from the original on 2022-07-09
505:from the original on 2022-07-09
461:from the original on 2022-07-09
3797:Chichibabin pyridine synthesis
3283:Chichibabin pyridine synthesis
3243:Blum–Ittah aziridine synthesis
3078:Ring expansion and contraction
1347:Cross dehydrogenative coupling
570:J. Chem. Soc., Perkin Trans. 1
1:
3767:Bischler–Napieralski reaction
3725:Heterocycle forming reactions
3378:Hemetsberger indole synthesis
3238:Bischler–Napieralski reaction
3153:Wagner–Meerwein rearrangement
3123:Sommelet–Hauser rearrangement
3103:Seyferth–Gilbert homologation
2968:Ireland–Claisen rearrangement
2963:Hofmann–Martius rearrangement
2723:2,3-sigmatropic rearrangement
2338:Corey–Winter olefin synthesis
2263:Barton–McCombie deoxygenation
1906:Corey–Winter olefin synthesis
1860:Seyferth–Gilbert homologation
1727:Seyferth–Gilbert homologation
176:catalysis in the presence of
3872:Lehmstedt–Tanasescu reaction
3832:Gabriel–Colman rearrangement
3787:Bucherer carbazole synthesis
3782:Borsche–Drechsel cyclization
3762:Bernthsen acridine synthesis
3747:Bamberger triazine synthesis
3732:Algar–Flynn–Oyamada reaction
3443:Nazarov cyclization reaction
3308:De Kimpe aziridine synthesis
3263:Bucherer carbazole synthesis
3258:Borsche–Drechsel cyclization
3028:Nazarov cyclization reaction
3008:Meyer–Schuster rearrangement
2938:Gabriel–Colman rearrangement
2688:Wolffenstein–Böters reaction
2573:Reduction of nitro compounds
2423:Grundmann aldehyde synthesis
2228:Algar–Flynn–Oyamada reaction
1637:Olefin conversion technology
1632:Nozaki–Hiyama–Kishi reaction
1427:Gabriel–Colman rearrangement
1317:Claisen-Schmidt condensation
1262:Bouveault aldehyde synthesis
3847:Hantzsch pyridine synthesis
3626:Enone–alkene cycloadditions
3448:Nenitzescu indole synthesis
3368:Hantzsch pyridine synthesis
3333:Ferrario–Ackermann reaction
2983:Kowalski ester homologation
2948:Halogen dance rearrangement
2793:Benzilic acid rearrangement
2218:Akabori amino-acid reaction
2178:Von Braun amide degradation
2123:Barbier–Wieland degradation
2075:Nenitzescu indole synthesis
2055:Kharasch–Sosnovsky reaction
1946:Julia–Kocienski olefination
1850:Kowalski ester homologation
1547:Kowalski ester homologation
1522:Julia–Kocienski olefination
1277:Cadiot–Chodkiewicz coupling
1202:Aza-Baylis–Hillman reaction
1147:Acetoacetic ester synthesis
858:Dynamic binding (chemistry)
848:Conrotatory and disrotatory
823:Charge remote fragmentation
158:4-dimethylaminobenzaldehyde
3977:
3912:Robinson–Gabriel synthesis
3862:Kröhnke pyridine synthesis
3696:Retro-Diels–Alder reaction
3636:Imine Diels–Alder reaction
3423:Kröhnke pyridine synthesis
3038:Newman–Kwart rearrangement
3013:Mislow–Evans rearrangement
2923:Fischer–Hepp rearrangement
2868:Di-π-methane rearrangement
2648:Stephen aldehyde synthesis
2383:Eschweiler–Clarke reaction
2100:Williamson ether synthesis
1417:Fujiwara–Moritani reaction
1322:Combes quinoline synthesis
1287:Carbonyl olefin metathesis
988:More O'Ferrall–Jencks plot
913:Grunwald–Winstein equation
883:Electron-withdrawing group
818:Catalytic resonance theory
18:
3922:Urech hydantoin synthesis
3902:Pomeranz–Fritsch reaction
3827:Fischer oxazole synthesis
3561:1,3-Dipolar cycloaddition
3533:Urech hydantoin synthesis
3503:Reissert indole synthesis
3488:Pomeranz–Fritsch reaction
3418:Knorr quinoline synthesis
3348:Fischer oxazole synthesis
3278:Camps quinoline synthesis
3198:1,3-Dipolar cycloaddition
3098:Semipinacol rearrangement
3073:Ramberg–Bäcklund reaction
3058:Piancatelli rearrangement
2998:McFadyen–Stevens reaction
2753:Alpha-ketol rearrangement
2508:McFadyen–Stevens reaction
2453:Kiliani–Fischer synthesis
2373:Elbs persulfate oxidation
2298:Bouveault–Blanc reduction
2258:Baeyer–Villiger oxidation
2090:Schotten–Baumann reaction
1966:Ramberg–Bäcklund reaction
1845:Kiliani–Fischer synthesis
1687:Ramberg–Bäcklund reaction
1672:Pinacol coupling reaction
1667:Piancatelli rearrangement
1562:Liebeskind–Srogl coupling
1412:Fujimoto–Belleau reaction
1129:List of organic reactions
993:Negative hyperconjugation
738:
680:
65:during their research on
3897:Pictet–Spengler reaction
3812:Einhorn–Brunner reaction
3777:Boger pyridine synthesis
3671:Oxo-Diels–Alder reaction
3586:Aza-Diels–Alder reaction
3483:Pictet–Spengler reaction
3383:Hofmann–Löffler reaction
3373:Hegedus indole synthesis
3343:Fischer indole synthesis
3218:Bartoli indole synthesis
3173:Willgerodt rearrangement
3003:McLafferty rearrangement
2913:Ferrier carbocyclization
2728:2,3-Wittig rearrangement
2718:1,2-Wittig rearrangement
2558:Parikh–Doering oxidation
2548:Oxygen rebound mechanism
2213:Adkins–Peterson reaction
2105:Yamaguchi esterification
2045:Hegedus indole synthesis
2010:Bartoli indole synthesis
1881:Bamford–Stevens reaction
1797:Weinreb ketone synthesis
1757:Stork enamine alkylation
1532:Knoevenagel condensation
1402:Ferrier carbocyclization
1292:Castro–Stephens coupling
918:Hammett acidity function
908:Free-energy relationship
853:Curtin–Hammett principle
838:Conformational isomerism
499:10.1002/jlac.18400340205
455:10.1002/jlac.18390310312
412:10.1002/jlac.18320030302
383:10.15227/orgsyn.001.0033
113:was proposed in 1903 by
21:Benzoin (disambiguation)
3857:Knorr pyrrole synthesis
3792:Bucherer–Bergs reaction
3737:Allan–Robinson reaction
3716:Wagner-Jauregg reaction
3508:Ring-closing metathesis
3433:Larock indole synthesis
3413:Knorr pyrrole synthesis
3268:Bucherer–Bergs reaction
3133:Stieglitz rearrangement
3113:Skattebøl rearrangement
3083:Ring-closing metathesis
2943:Group transfer reaction
2908:Favorskii rearrangement
2848:Cornforth rearrangement
2778:Bamberger rearrangement
2683:Wolff–Kishner reduction
2503:Markó–Lam deoxygenation
2398:Fleming–Tamao oxidation
2393:Fischer–Tropsch process
2080:Oxymercuration reaction
2060:Knorr pyrrole synthesis
1886:Barton–Kellogg reaction
1792:Wagner-Jauregg reaction
1712:Ring-closing metathesis
1702:Reimer–Tiemann reaction
1692:Rauhut–Currier reaction
1607:Nef isocyanide reaction
1567:Malonic ester synthesis
1537:Knorr pyrrole synthesis
1472:High dilution principle
1407:Friedel–Crafts reaction
1342:Cross-coupling reaction
1267:Bucherer–Bergs reaction
1252:Blanc chloromethylation
1242:Blaise ketone synthesis
1217:Baylis–Hillman reaction
1212:Barton–Kellogg reaction
1187:Allan–Robinson reaction
1093:Woodward–Hoffmann rules
828:Charge-transfer complex
143:thermodynamic stability
3822:Feist–Benary synthesis
3596:Bradsher cycloaddition
3566:4+4 Photocycloaddition
3523:Simmons–Smith reaction
3468:Paternò–Büchi reaction
3328:Feist–Benary synthesis
3318:Dieckmann condensation
3068:Pummerer rearrangement
3048:Oxy-Cope rearrangement
3023:Myers allene synthesis
2973:Jacobsen rearrangement
2888:Electrocyclic reaction
2863:Demjanov rearrangement
2818:Buchner ring expansion
2788:Beckmann rearrangement
2768:Aza-Cope rearrangement
2763:Arndt–Eistert reaction
2738:Alkyne zipper reaction
2658:Transfer hydrogenation
2633:Sharpless oxyamination
2608:Selenoxide elimination
2493:Lombardo methylenation
2418:Griesbaum coozonolysis
2328:Corey–Itsuno reduction
2303:Boyland–Sims oxidation
2243:Angeli–Rimini reaction
1891:Boord olefin synthesis
1835:Arndt–Eistert reaction
1827:Homologation reactions
1627:Nitro-Mannich reaction
1542:Kolbe–Schmitt reaction
1352:Cross-coupling partner
1272:Buchner ring expansion
1192:Arndt–Eistert reaction
958:Kinetic isotope effect
705:Rearrangement reaction
607:10.1002/anie.200503885
230:retro benzoin addition
225:
182:heterocyclic compounds
153:
103:N-heterocyclic carbene
81:
3681:Pauson–Khand reaction
3518:Sharpless epoxidation
3473:Pechmann condensation
3353:Friedländer synthesis
3303:Davis–Beirut reaction
3158:Wallach rearrangement
3128:Stevens rearrangement
3063:Pinacol rearrangement
3043:Overman rearrangement
2958:Hofmann rearrangement
2953:Hayashi rearrangement
2918:Ferrier rearrangement
2873:Dimroth rearrangement
2858:Curtius rearrangement
2853:Criegee rearrangement
2833:Claisen rearrangement
2823:Carroll rearrangement
2758:Amadori rearrangement
2748:Allylic rearrangement
2628:Sharpless epoxidation
2363:Dess–Martin oxidation
2288:Bohn–Schmidt reaction
2148:Hofmann rearrangement
1951:Kauffmann olefination
1874:Olefination reactions
1812:Wurtz–Fittig reaction
1647:Palladium–NHC complex
1527:Kauffmann olefination
1482:Homologation reaction
1332:Corey–House synthesis
1312:Claisen rearrangement
1108:Yukawa–Tsuno equation
1068:Swain–Lupton equation
1048:Spherical aromaticity
983:Möbius–Hückel concept
768:Aromatic ring current
730:Substitution reaction
639:at Wikimedia Commons
486:Annalen der Pharmacie
442:Annalen der Pharmacie
399:Annalen der Pharmacie
223:
151:
123:nucleophilic addition
79:
3887:Paal–Knorr synthesis
3757:Barton–Zard reaction
3701:Staudinger synthesis
3651:Ketene cycloaddition
3621:Diels–Alder reaction
3601:Cheletropic reaction
3581:Alkyne trimerisation
3463:Paal–Knorr synthesis
3428:Kulinkovich reaction
3403:Jacobsen epoxidation
3323:Diels–Alder reaction
3118:Smiles rearrangement
3108:Sigmatropic reaction
2993:Lossen rearrangement
2843:Corey–Fuchs reaction
2808:Boekelheide reaction
2803:Bergmann degradation
2733:Achmatowicz reaction
2518:Methionine sulfoxide
2318:Clemmensen reduction
2278:Bergmann degradation
2208:Acyloin condensation
2173:Strecker degradation
2128:Bergmann degradation
2095:Ullmann condensation
1961:Peterson olefination
1936:Hydrazone iodination
1916:Elimination reaction
1817:Zincke–Suhl reaction
1737:Sonogashira coupling
1697:Reformatsky reaction
1657:Peterson olefination
1622:Nierenstein reaction
1552:Kulinkovich reaction
1367:Diels–Alder reaction
1327:Corey–Fuchs reaction
1307:Claisen condensation
1177:Alkyne trimerisation
1152:Acyloin condensation
1118:Σ-bishomoaromaticity
1078:Thorpe–Ingold effect
690:Elimination reaction
544:10.1039/CT9048501206
251:Acyloin condensation
3907:Prilezhaev reaction
3892:Pellizzari reaction
3571:(4+3) cycloaddition
3538:Van Leusen reaction
3513:Robinson annulation
3498:Pschorr cyclization
3493:Prilezhaev reaction
3223:Bergman cyclization
3178:Wolff rearrangement
3163:Weerman degradation
3053:Pericyclic reaction
3033:Neber rearrangement
2928:Fries rearrangement
2813:Brook rearrangement
2798:Bergman cyclization
2643:Staudinger reaction
2588:Rosenmund reduction
2578:Reductive amination
2543:Oppenauer oxidation
2333:Corey–Kim oxidation
2308:Cannizzaro reaction
2183:Weerman degradation
2158:Isosaccharinic acid
2070:Mukaiyama hydration
1926:Hofmann elimination
1911:Dehydrohalogenation
1896:Chugaev elimination
1717:Robinson annulation
1662:Pfitzinger reaction
1432:Gattermann reaction
1377:Wulff–Dötz reaction
1357:Dakin–West reaction
1282:Carbonyl allylation
1227:Bergman cyclization
1013:Kennedy J. P. Orton
933:Hammond's postulate
903:Flippin–Lodge angle
873:Electromeric effect
798:Beta-silicon effect
783:Baker–Nathan effect
256:Cannizzaro reaction
139:reversible reaction
73:in the late 1830s.
3961:Addition reactions
3656:McCormack reaction
3606:Conia-ene reaction
3438:Madelung synthesis
3228:Biginelli reaction
3018:Mumm rearrangement
2903:Favorskii reaction
2838:Cope rearrangement
2828:Chan rearrangement
2593:Rubottom oxidation
2523:Miyaura borylation
2488:Lipid peroxidation
2483:Lindgren oxidation
2463:Kornblum oxidation
2458:Kolbe electrolysis
2403:Fukuyama reduction
2313:Carbonyl reduction
2163:Marker degradation
2025:Diazonium compound
2015:Boudouard reaction
1994:Carbon-heteroatom
1921:Grieco elimination
1707:Rieche formylation
1652:Passerini reaction
1582:Meerwein arylation
1502:Hydroxymethylation
1397:Favorskii reaction
1297:Chan rearrangement
1232:Biginelli reaction
1157:Aldol condensation
1003:2-Norbornyl cation
978:Möbius aromaticity
973:Markovnikov's rule
868:Effective molarity
813:Bürgi–Dunitz angle
803:Bicycloaromaticity
303:10.3762/bjoc.12.47
226:
154:
111:reaction mechanism
85:Reaction mechanism
82:
3948:
3947:
3944:
3943:
3940:
3939:
3932:Wohl–Aue reaction
3576:6+4 Cycloaddition
3393:Iodolactonization
2713:1,2-rearrangement
2678:Wohl–Aue reaction
2598:Sabatier reaction
2563:Pinnick oxidation
2528:Mozingo reduction
2473:Leuckart reaction
2428:Haloform reaction
2343:Criegee oxidation
2323:Collins oxidation
2273:Benkeser reaction
2268:Bechamp reduction
2238:Andrussow process
2223:Alcohol oxidation
2133:Edman degradation
2040:Haloform reaction
1989:
1988:
1976:Takai olefination
1941:Julia olefination
1767:Takai olefination
1642:Olefin metathesis
1517:Julia olefination
1442:Grignard reaction
1422:Fukuyama coupling
1337:Coupling reaction
1302:Chan–Lam coupling
1172:Alkyne metathesis
1167:Alkane metathesis
1023:Phosphaethynolate
928:George S. Hammond
888:Electronic effect
843:Conjugated system
725:Stereospecificity
720:Stereoselectivity
685:Addition reaction
674:organic reactions
635:Media related to
572:(12): 1891–1894.
479:N. Zinin (1840).
435:N. Zinin (1839).
371:Organic Syntheses
348:10.1021/cr068372z
342:(12): 5606–5655.
127:polarity reversal
67:bitter almond oil
59:Justus von Liebig
32:addition reaction
3968:
3927:Wenker synthesis
3917:Stollé synthesis
3772:Bobbitt reaction
3742:Auwers synthesis
3686:Povarov reaction
3611:Cyclopropanation
3549:
3543:Wenker synthesis
3298:Darzens reaction
3248:Bobbitt reaction
3093:Schmidt reaction
2898:Enyne metathesis
2673:Whiting reaction
2668:Wharton reaction
2613:Shapiro reaction
2603:Sarett oxidation
2568:Prévost reaction
2378:Emde degradation
2188:Wohl degradation
2168:Ruff degradation
2138:Emde degradation
2035:Grignard reagent
1971:Shapiro reaction
1956:McMurry reaction
1823:
1787:Ullmann reaction
1752:Stollé synthesis
1742:Stetter reaction
1732:Shapiro reaction
1722:Sakurai reaction
1617:Negishi coupling
1597:Minisci reaction
1592:Michael reaction
1577:McMurry reaction
1572:Mannich reaction
1452:Hammick reaction
1447:Grignard reagent
1387:Enyne metathesis
1372:Doebner reaction
1362:Darzens reaction
1207:Barbier reaction
1197:Auwers synthesis
1124:
1098:Woodward's rules
1063:Superaromaticity
1053:Spiroaromaticity
953:Inductive effect
948:Hyperconjugation
923:Hammett equation
863:Edwards equation
715:Regioselectivity
666:
659:
652:
643:
634:
619:
618:
601:(9): 1463–1467.
588:
582:
581:
578:10.1039/A803635G
565:
559:
558:
556:
555:
520:
514:
513:
511:
510:
476:
470:
469:
467:
466:
432:
426:
425:
423:
393:
387:
386:
366:
360:
359:
336:Chemical Reviews
331:
325:
324:
314:
282:
261:Stetter reaction
190:Stetter reaction
178:thiazolium salts
107:thiazolium salts
89:The reaction is
80:Benzoin addition
63:Friedrich Wöhler
28:benzoin addition
3976:
3975:
3971:
3970:
3969:
3967:
3966:
3965:
3951:
3950:
3949:
3936:
3837:Gewald reaction
3720:
3547:
3528:Skraup reaction
3363:Graham reaction
3358:Gewald reaction
3189:
3182:
2704:
2697:
2653:Swern oxidation
2638:Stahl oxidation
2583:Riley oxidation
2538:Omega oxidation
2498:Luche reduction
2448:Jones oxidation
2413:Glycol cleavage
2408:Ganem oxidation
2353:Davis oxidation
2348:Dakin oxidation
2283:Birch reduction
2233:Amide reduction
2199:
2192:
2153:Hooker reaction
2115:
2109:
1997:
1995:
1985:
1981:Wittig reaction
1869:
1865:Wittig reaction
1840:Hooker reaction
1821:
1802:Wittig reaction
1777:Thorpe reaction
1762:Suzuki reaction
1747:Stille reaction
1682:Quelet reaction
1557:Kumada coupling
1507:Ivanov reaction
1497:Hydrovinylation
1477:Hiyama coupling
1437:Glaser coupling
1247:Blaise reaction
1237:Bingel reaction
1222:Benary reaction
1139:
1137:
1131:
1122:
1018:Passive binding
938:Homoaromaticity
788:Baldwin's rules
763:Antiaromaticity
758:Anomeric effect
734:
676:
670:
628:
623:
622:
590:
589:
585:
567:
566:
562:
553:
551:
522:
521:
517:
508:
506:
478:
477:
473:
464:
462:
434:
433:
429:
421:2027/hvd.hxdg3f
395:
394:
390:
368:
367:
363:
333:
332:
328:
284:
283:
279:
274:
242:
172:aldehydes with
166:
87:
24:
17:
12:
11:
5:
3974:
3972:
3964:
3963:
3953:
3952:
3946:
3945:
3942:
3941:
3938:
3937:
3935:
3934:
3929:
3924:
3919:
3914:
3909:
3904:
3899:
3894:
3889:
3884:
3879:
3874:
3869:
3864:
3859:
3854:
3849:
3844:
3842:Hantzsch ester
3839:
3834:
3829:
3824:
3819:
3814:
3809:
3804:
3799:
3794:
3789:
3784:
3779:
3774:
3769:
3764:
3759:
3754:
3752:Banert cascade
3749:
3744:
3739:
3734:
3728:
3726:
3722:
3721:
3719:
3718:
3713:
3708:
3703:
3698:
3693:
3691:Prato reaction
3688:
3683:
3678:
3673:
3668:
3663:
3658:
3653:
3648:
3643:
3638:
3633:
3628:
3623:
3618:
3613:
3608:
3603:
3598:
3593:
3588:
3583:
3578:
3573:
3568:
3563:
3557:
3555:
3546:
3545:
3540:
3535:
3530:
3525:
3520:
3515:
3510:
3505:
3500:
3495:
3490:
3485:
3480:
3475:
3470:
3465:
3460:
3455:
3450:
3445:
3440:
3435:
3430:
3425:
3420:
3415:
3410:
3405:
3400:
3395:
3390:
3385:
3380:
3375:
3370:
3365:
3360:
3355:
3350:
3345:
3340:
3335:
3330:
3325:
3320:
3315:
3310:
3305:
3300:
3295:
3290:
3285:
3280:
3275:
3270:
3265:
3260:
3255:
3250:
3245:
3240:
3235:
3230:
3225:
3220:
3215:
3210:
3205:
3200:
3194:
3192:
3184:
3183:
3181:
3180:
3175:
3170:
3165:
3160:
3155:
3150:
3145:
3140:
3135:
3130:
3125:
3120:
3115:
3110:
3105:
3100:
3095:
3090:
3085:
3080:
3075:
3070:
3065:
3060:
3055:
3050:
3045:
3040:
3035:
3030:
3025:
3020:
3015:
3010:
3005:
3000:
2995:
2990:
2985:
2980:
2975:
2970:
2965:
2960:
2955:
2950:
2945:
2940:
2935:
2930:
2925:
2920:
2915:
2910:
2905:
2900:
2895:
2890:
2885:
2880:
2875:
2870:
2865:
2860:
2855:
2850:
2845:
2840:
2835:
2830:
2825:
2820:
2815:
2810:
2805:
2800:
2795:
2790:
2785:
2783:Banert cascade
2780:
2775:
2770:
2765:
2760:
2755:
2750:
2745:
2740:
2735:
2730:
2725:
2720:
2715:
2709:
2707:
2703:Rearrangement
2699:
2698:
2696:
2695:
2693:Zinin reaction
2690:
2685:
2680:
2675:
2670:
2665:
2663:Wacker process
2660:
2655:
2650:
2645:
2640:
2635:
2630:
2625:
2620:
2615:
2610:
2605:
2600:
2595:
2590:
2585:
2580:
2575:
2570:
2565:
2560:
2555:
2550:
2545:
2540:
2535:
2530:
2525:
2520:
2515:
2510:
2505:
2500:
2495:
2490:
2485:
2480:
2475:
2470:
2465:
2460:
2455:
2450:
2445:
2440:
2438:Hydrogenolysis
2435:
2430:
2425:
2420:
2415:
2410:
2405:
2400:
2395:
2390:
2388:Étard reaction
2385:
2380:
2375:
2370:
2365:
2360:
2355:
2350:
2345:
2340:
2335:
2330:
2325:
2320:
2315:
2310:
2305:
2300:
2295:
2293:Bosch reaction
2290:
2285:
2280:
2275:
2270:
2265:
2260:
2255:
2250:
2245:
2240:
2235:
2230:
2225:
2220:
2215:
2210:
2204:
2202:
2198:Organic redox
2194:
2193:
2191:
2190:
2185:
2180:
2175:
2170:
2165:
2160:
2155:
2150:
2145:
2140:
2135:
2130:
2125:
2119:
2117:
2111:
2110:
2108:
2107:
2102:
2097:
2092:
2087:
2082:
2077:
2072:
2067:
2062:
2057:
2052:
2047:
2042:
2037:
2032:
2030:Esterification
2027:
2022:
2017:
2012:
2007:
2001:
1999:
1991:
1990:
1987:
1986:
1984:
1983:
1978:
1973:
1968:
1963:
1958:
1953:
1948:
1943:
1938:
1933:
1928:
1923:
1918:
1913:
1908:
1903:
1898:
1893:
1888:
1883:
1877:
1875:
1871:
1870:
1868:
1867:
1862:
1857:
1852:
1847:
1842:
1837:
1831:
1829:
1820:
1819:
1814:
1809:
1807:Wurtz reaction
1804:
1799:
1794:
1789:
1784:
1779:
1774:
1769:
1764:
1759:
1754:
1749:
1744:
1739:
1734:
1729:
1724:
1719:
1714:
1709:
1704:
1699:
1694:
1689:
1684:
1679:
1677:Prins reaction
1674:
1669:
1664:
1659:
1654:
1649:
1644:
1639:
1634:
1629:
1624:
1619:
1614:
1609:
1604:
1599:
1594:
1589:
1584:
1579:
1574:
1569:
1564:
1559:
1554:
1549:
1544:
1539:
1534:
1529:
1524:
1519:
1514:
1509:
1504:
1499:
1494:
1492:Hydrocyanation
1489:
1484:
1479:
1474:
1469:
1464:
1462:Henry reaction
1459:
1454:
1449:
1444:
1439:
1434:
1429:
1424:
1419:
1414:
1409:
1404:
1399:
1394:
1389:
1384:
1379:
1374:
1369:
1364:
1359:
1354:
1349:
1344:
1339:
1334:
1329:
1324:
1319:
1314:
1309:
1304:
1299:
1294:
1289:
1284:
1279:
1274:
1269:
1264:
1259:
1254:
1249:
1244:
1239:
1234:
1229:
1224:
1219:
1214:
1209:
1204:
1199:
1194:
1189:
1184:
1179:
1174:
1169:
1164:
1162:Aldol reaction
1159:
1154:
1149:
1143:
1141:
1136:Carbon-carbon
1133:
1132:
1127:
1121:
1120:
1115:
1113:Zaitsev's rule
1110:
1105:
1100:
1095:
1090:
1085:
1080:
1075:
1070:
1065:
1060:
1058:Steric effects
1055:
1050:
1045:
1040:
1035:
1030:
1025:
1020:
1015:
1010:
1005:
1000:
995:
990:
985:
980:
975:
970:
965:
960:
955:
950:
945:
940:
935:
930:
925:
920:
915:
910:
905:
900:
895:
890:
885:
880:
875:
870:
865:
860:
855:
850:
845:
840:
835:
830:
825:
820:
815:
810:
805:
800:
795:
790:
785:
780:
775:
770:
765:
760:
755:
750:
745:
739:
736:
735:
733:
732:
727:
722:
717:
712:
710:Redox reaction
707:
702:
697:
695:Polymerization
692:
687:
681:
678:
677:
671:
669:
668:
661:
654:
646:
627:
626:External links
624:
621:
620:
583:
560:
515:
493:(2): 186–192.
471:
449:(3): 329–332.
427:
406:(3): 249–282.
388:
361:
326:
276:
275:
273:
270:
269:
268:
263:
258:
253:
248:
246:Aldol addition
241:
238:
188:is called the
165:
162:
119:sodium cyanide
115:A. J. Lapworth
86:
83:
34:involving two
15:
13:
10:
9:
6:
4:
3:
2:
3973:
3962:
3959:
3958:
3956:
3933:
3930:
3928:
3925:
3923:
3920:
3918:
3915:
3913:
3910:
3908:
3905:
3903:
3900:
3898:
3895:
3893:
3890:
3888:
3885:
3883:
3880:
3878:
3875:
3873:
3870:
3868:
3865:
3863:
3860:
3858:
3855:
3853:
3852:Herz reaction
3850:
3848:
3845:
3843:
3840:
3838:
3835:
3833:
3830:
3828:
3825:
3823:
3820:
3818:
3815:
3813:
3810:
3808:
3805:
3803:
3800:
3798:
3795:
3793:
3790:
3788:
3785:
3783:
3780:
3778:
3775:
3773:
3770:
3768:
3765:
3763:
3760:
3758:
3755:
3753:
3750:
3748:
3745:
3743:
3740:
3738:
3735:
3733:
3730:
3729:
3727:
3723:
3717:
3714:
3712:
3709:
3707:
3704:
3702:
3699:
3697:
3694:
3692:
3689:
3687:
3684:
3682:
3679:
3677:
3674:
3672:
3669:
3667:
3664:
3662:
3659:
3657:
3654:
3652:
3649:
3647:
3644:
3642:
3639:
3637:
3634:
3632:
3629:
3627:
3624:
3622:
3619:
3617:
3614:
3612:
3609:
3607:
3604:
3602:
3599:
3597:
3594:
3592:
3589:
3587:
3584:
3582:
3579:
3577:
3574:
3572:
3569:
3567:
3564:
3562:
3559:
3558:
3556:
3554:
3553:Cycloaddition
3550:
3544:
3541:
3539:
3536:
3534:
3531:
3529:
3526:
3524:
3521:
3519:
3516:
3514:
3511:
3509:
3506:
3504:
3501:
3499:
3496:
3494:
3491:
3489:
3486:
3484:
3481:
3479:
3476:
3474:
3471:
3469:
3466:
3464:
3461:
3459:
3456:
3454:
3451:
3449:
3446:
3444:
3441:
3439:
3436:
3434:
3431:
3429:
3426:
3424:
3421:
3419:
3416:
3414:
3411:
3409:
3406:
3404:
3401:
3399:
3398:Isay reaction
3396:
3394:
3391:
3389:
3386:
3384:
3381:
3379:
3376:
3374:
3371:
3369:
3366:
3364:
3361:
3359:
3356:
3354:
3351:
3349:
3346:
3344:
3341:
3339:
3336:
3334:
3331:
3329:
3326:
3324:
3321:
3319:
3316:
3314:
3311:
3309:
3306:
3304:
3301:
3299:
3296:
3294:
3293:Cycloaddition
3291:
3289:
3286:
3284:
3281:
3279:
3276:
3274:
3271:
3269:
3266:
3264:
3261:
3259:
3256:
3254:
3251:
3249:
3246:
3244:
3241:
3239:
3236:
3234:
3231:
3229:
3226:
3224:
3221:
3219:
3216:
3214:
3211:
3209:
3206:
3204:
3201:
3199:
3196:
3195:
3193:
3191:
3188:Ring forming
3185:
3179:
3176:
3174:
3171:
3169:
3166:
3164:
3161:
3159:
3156:
3154:
3151:
3149:
3146:
3144:
3141:
3139:
3136:
3134:
3131:
3129:
3126:
3124:
3121:
3119:
3116:
3114:
3111:
3109:
3106:
3104:
3101:
3099:
3096:
3094:
3091:
3089:
3088:Rupe reaction
3086:
3084:
3081:
3079:
3076:
3074:
3071:
3069:
3066:
3064:
3061:
3059:
3056:
3054:
3051:
3049:
3046:
3044:
3041:
3039:
3036:
3034:
3031:
3029:
3026:
3024:
3021:
3019:
3016:
3014:
3011:
3009:
3006:
3004:
3001:
2999:
2996:
2994:
2991:
2989:
2986:
2984:
2981:
2979:
2976:
2974:
2971:
2969:
2966:
2964:
2961:
2959:
2956:
2954:
2951:
2949:
2946:
2944:
2941:
2939:
2936:
2934:
2931:
2929:
2926:
2924:
2921:
2919:
2916:
2914:
2911:
2909:
2906:
2904:
2901:
2899:
2896:
2894:
2891:
2889:
2886:
2884:
2881:
2879:
2876:
2874:
2871:
2869:
2866:
2864:
2861:
2859:
2856:
2854:
2851:
2849:
2846:
2844:
2841:
2839:
2836:
2834:
2831:
2829:
2826:
2824:
2821:
2819:
2816:
2814:
2811:
2809:
2806:
2804:
2801:
2799:
2796:
2794:
2791:
2789:
2786:
2784:
2781:
2779:
2776:
2774:
2771:
2769:
2766:
2764:
2761:
2759:
2756:
2754:
2751:
2749:
2746:
2744:
2741:
2739:
2736:
2734:
2731:
2729:
2726:
2724:
2721:
2719:
2716:
2714:
2711:
2710:
2708:
2706:
2700:
2694:
2691:
2689:
2686:
2684:
2681:
2679:
2676:
2674:
2671:
2669:
2666:
2664:
2661:
2659:
2656:
2654:
2651:
2649:
2646:
2644:
2641:
2639:
2636:
2634:
2631:
2629:
2626:
2624:
2621:
2619:
2616:
2614:
2611:
2609:
2606:
2604:
2601:
2599:
2596:
2594:
2591:
2589:
2586:
2584:
2581:
2579:
2576:
2574:
2571:
2569:
2566:
2564:
2561:
2559:
2556:
2554:
2551:
2549:
2546:
2544:
2541:
2539:
2536:
2534:
2531:
2529:
2526:
2524:
2521:
2519:
2516:
2514:
2511:
2509:
2506:
2504:
2501:
2499:
2496:
2494:
2491:
2489:
2486:
2484:
2481:
2479:
2478:Ley oxidation
2476:
2474:
2471:
2469:
2466:
2464:
2461:
2459:
2456:
2454:
2451:
2449:
2446:
2444:
2443:Hydroxylation
2441:
2439:
2436:
2434:
2433:Hydrogenation
2431:
2429:
2426:
2424:
2421:
2419:
2416:
2414:
2411:
2409:
2406:
2404:
2401:
2399:
2396:
2394:
2391:
2389:
2386:
2384:
2381:
2379:
2376:
2374:
2371:
2369:
2368:DNA oxidation
2366:
2364:
2361:
2359:
2358:Deoxygenation
2356:
2354:
2351:
2349:
2346:
2344:
2341:
2339:
2336:
2334:
2331:
2329:
2326:
2324:
2321:
2319:
2316:
2314:
2311:
2309:
2306:
2304:
2301:
2299:
2296:
2294:
2291:
2289:
2286:
2284:
2281:
2279:
2276:
2274:
2271:
2269:
2266:
2264:
2261:
2259:
2256:
2254:
2251:
2249:
2248:Aromatization
2246:
2244:
2241:
2239:
2236:
2234:
2231:
2229:
2226:
2224:
2221:
2219:
2216:
2214:
2211:
2209:
2206:
2205:
2203:
2201:
2195:
2189:
2186:
2184:
2181:
2179:
2176:
2174:
2171:
2169:
2166:
2164:
2161:
2159:
2156:
2154:
2151:
2149:
2146:
2144:
2141:
2139:
2136:
2134:
2131:
2129:
2126:
2124:
2121:
2120:
2118:
2112:
2106:
2103:
2101:
2098:
2096:
2093:
2091:
2088:
2086:
2085:Reed reaction
2083:
2081:
2078:
2076:
2073:
2071:
2068:
2066:
2063:
2061:
2058:
2056:
2053:
2051:
2048:
2046:
2043:
2041:
2038:
2036:
2033:
2031:
2028:
2026:
2023:
2021:
2018:
2016:
2013:
2011:
2008:
2006:
2003:
2002:
2000:
1996:bond forming
1992:
1982:
1979:
1977:
1974:
1972:
1969:
1967:
1964:
1962:
1959:
1957:
1954:
1952:
1949:
1947:
1944:
1942:
1939:
1937:
1934:
1932:
1929:
1927:
1924:
1922:
1919:
1917:
1914:
1912:
1909:
1907:
1904:
1902:
1901:Cope reaction
1899:
1897:
1894:
1892:
1889:
1887:
1884:
1882:
1879:
1878:
1876:
1872:
1866:
1863:
1861:
1858:
1856:
1853:
1851:
1848:
1846:
1843:
1841:
1838:
1836:
1833:
1832:
1830:
1828:
1824:
1818:
1815:
1813:
1810:
1808:
1805:
1803:
1800:
1798:
1795:
1793:
1790:
1788:
1785:
1783:
1780:
1778:
1775:
1773:
1770:
1768:
1765:
1763:
1760:
1758:
1755:
1753:
1750:
1748:
1745:
1743:
1740:
1738:
1735:
1733:
1730:
1728:
1725:
1723:
1720:
1718:
1715:
1713:
1710:
1708:
1705:
1703:
1700:
1698:
1695:
1693:
1690:
1688:
1685:
1683:
1680:
1678:
1675:
1673:
1670:
1668:
1665:
1663:
1660:
1658:
1655:
1653:
1650:
1648:
1645:
1643:
1640:
1638:
1635:
1633:
1630:
1628:
1625:
1623:
1620:
1618:
1615:
1613:
1612:Nef synthesis
1610:
1608:
1605:
1603:
1600:
1598:
1595:
1593:
1590:
1588:
1587:Methylenation
1585:
1583:
1580:
1578:
1575:
1573:
1570:
1568:
1565:
1563:
1560:
1558:
1555:
1553:
1550:
1548:
1545:
1543:
1540:
1538:
1535:
1533:
1530:
1528:
1525:
1523:
1520:
1518:
1515:
1513:
1510:
1508:
1505:
1503:
1500:
1498:
1495:
1493:
1490:
1488:
1485:
1483:
1480:
1478:
1475:
1473:
1470:
1468:
1465:
1463:
1460:
1458:
1457:Heck reaction
1455:
1453:
1450:
1448:
1445:
1443:
1440:
1438:
1435:
1433:
1430:
1428:
1425:
1423:
1420:
1418:
1415:
1413:
1410:
1408:
1405:
1403:
1400:
1398:
1395:
1393:
1390:
1388:
1385:
1383:
1380:
1378:
1375:
1373:
1370:
1368:
1365:
1363:
1360:
1358:
1355:
1353:
1350:
1348:
1345:
1343:
1340:
1338:
1335:
1333:
1330:
1328:
1325:
1323:
1320:
1318:
1315:
1313:
1310:
1308:
1305:
1303:
1300:
1298:
1295:
1293:
1290:
1288:
1285:
1283:
1280:
1278:
1275:
1273:
1270:
1268:
1265:
1263:
1260:
1258:
1255:
1253:
1250:
1248:
1245:
1243:
1240:
1238:
1235:
1233:
1230:
1228:
1225:
1223:
1220:
1218:
1215:
1213:
1210:
1208:
1205:
1203:
1200:
1198:
1195:
1193:
1190:
1188:
1185:
1183:
1180:
1178:
1175:
1173:
1170:
1168:
1165:
1163:
1160:
1158:
1155:
1153:
1150:
1148:
1145:
1144:
1142:
1138:bond forming
1134:
1130:
1125:
1119:
1116:
1114:
1111:
1109:
1106:
1104:
1103:Y-aromaticity
1101:
1099:
1096:
1094:
1091:
1089:
1088:Walsh diagram
1086:
1084:
1081:
1079:
1076:
1074:
1073:Taft equation
1071:
1069:
1066:
1064:
1061:
1059:
1056:
1054:
1051:
1049:
1046:
1044:
1043:Σ-aromaticity
1041:
1039:
1036:
1034:
1031:
1029:
1026:
1024:
1021:
1019:
1016:
1014:
1011:
1009:
1006:
1004:
1001:
999:
996:
994:
991:
989:
986:
984:
981:
979:
976:
974:
971:
969:
968:Marcus theory
966:
964:
961:
959:
956:
954:
951:
949:
946:
944:
943:Hückel's rule
941:
939:
936:
934:
931:
929:
926:
924:
921:
919:
916:
914:
911:
909:
906:
904:
901:
899:
898:Evelyn effect
896:
894:
891:
889:
886:
884:
881:
879:
878:Electron-rich
876:
874:
871:
869:
866:
864:
861:
859:
856:
854:
851:
849:
846:
844:
841:
839:
836:
834:
831:
829:
826:
824:
821:
819:
816:
814:
811:
809:
806:
804:
801:
799:
796:
794:
793:Bema Hapothle
791:
789:
786:
784:
781:
779:
776:
774:
771:
769:
766:
764:
761:
759:
756:
754:
751:
749:
746:
744:
741:
740:
737:
731:
728:
726:
723:
721:
718:
716:
713:
711:
708:
706:
703:
701:
698:
696:
693:
691:
688:
686:
683:
682:
679:
675:
667:
662:
660:
655:
653:
648:
647:
644:
640:
638:
633:
625:
616:
612:
608:
604:
600:
596:
595:
587:
584:
579:
575:
571:
564:
561:
549:
545:
541:
538:: 1206–1214.
537:
533:
529:
525:
519:
516:
504:
500:
496:
492:
489:(in German).
488:
487:
482:
475:
472:
460:
456:
452:
448:
445:(in German).
444:
443:
438:
431:
428:
422:
417:
413:
409:
405:
402:(in German).
401:
400:
392:
389:
384:
380:
376:
372:
365:
362:
357:
353:
349:
345:
341:
337:
330:
327:
322:
318:
313:
308:
304:
300:
296:
292:
288:
281:
278:
271:
267:
264:
262:
259:
257:
254:
252:
249:
247:
244:
243:
239:
237:
235:
231:
222:
218:
216:
211:
209:
208:deprotonation
205:
202:
198:
193:
191:
187:
183:
179:
175:
171:
163:
161:
159:
150:
146:
144:
140:
136:
132:
128:
124:
120:
116:
112:
108:
104:
100:
96:
92:
84:
78:
74:
72:
71:Nikolay Zinin
68:
64:
60:
55:
53:
49:
45:
42:aldehydes or
41:
37:
33:
29:
22:
2893:Ene reaction
2253:Autoxidation
2114:Degradation
2005:Azo coupling
1782:Ugi reaction
1382:Ene reaction
1182:Alkynylation
1033:Polyfluorene
1028:Polar effect
893:Electrophile
808:Bredt's rule
778:Baird's rule
748:Alpha effect
629:
598:
592:
586:
569:
563:
552:. Retrieved
535:
531:
524:Lapworth, A.
518:
507:. Retrieved
490:
484:
474:
463:. Retrieved
446:
440:
430:
403:
397:
391:
374:
370:
364:
339:
335:
329:
294:
290:
280:
229:
227:
212:
197:biochemistry
194:
167:
155:
95:nucleophiles
88:
56:
27:
25:
1392:Ethenolysis
1038:Ring strain
1008:Nucleophile
833:Clar's rule
773:Aromaticity
297:: 444–461.
3676:Ozonolysis
3203:Annulation
2553:Ozonolysis
672:Topics in
554:2019-06-28
509:2019-06-28
465:2020-09-11
272:References
215:triazolium
97:such as a
3190:reactions
2705:reactions
2200:reactions
2116:reactions
1998:reactions
1140:reactions
170:aliphatic
105:(usually
91:catalyzed
36:aldehydes
3955:Category
1083:Vinylogy
753:Annulene
700:Reagents
615:16389609
548:Archived
526:(1904).
503:Archived
459:Archived
356:17956132
321:27340440
266:Umpolung
240:See also
204:thiamine
201:coenzyme
131:carbonyl
44:glyoxals
40:aromatic
743:A value
312:4901930
234:ketones
129:of the
109:). The
99:cyanide
52:benzoin
48:acyloin
613:
377:: 33.
354:
319:
309:
199:, the
135:Proton
101:or an
30:is an
186:enone
164:Scope
611:PMID
352:PMID
317:PMID
174:base
61:and
26:The
603:doi
574:doi
540:doi
495:doi
451:doi
416:hdl
408:doi
379:doi
344:doi
340:107
307:PMC
299:doi
195:In
93:by
54:.
3957::
609:.
599:45
597:.
546:.
536:85
534:.
530:.
501:.
491:34
457:.
447:31
414:.
373:.
350:.
338:.
315:.
305:.
295:12
293:.
289:.
192:.
665:e
658:t
651:v
617:.
605::
580:.
576::
557:.
542::
512:.
497::
468:.
453::
424:.
418::
410::
404:3
385:.
381::
375:1
358:.
346::
323:.
301::
23:.
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.