180:
240:
161:
330:
496:
410:
434:
465:
288:
315:
446:
390:
247:
This chelation is in contrast to the mechanism for formation of the over-addition product wherein collapse of the tetrahedral intermediate allows a second addition. The mechanistic conjecture on the part of
Weinreb was immediately accepted by the academic community, but it was not until 2006 that it
397:
Nonetheless, the
Weinreb–Nahm amide figures prominently into many syntheses, serving as an important coupling partner for various fragments. Shown below are key steps involving Weinreb amides in the synthesis of several natural products, including members of the
960:
Davies, S. G.; Goodwin, C. J.; Hepworth, D.; Roberts, P. M.; Thomson, J. E. (2010), "On the
Origins of Diastereoselectivity in the Alkylation of Enolates Derived from N-1-(1'-Naphthyl)ethyl-O-tert-butylhydroxamates: Chiral Weinreb Amide Equivalents",
195:
and can be reliably reacted to form new carbon–carbon bonds or converted into other functional groups. This method has been used in a number of syntheses, including macrosphelides A and B, amphidinolide J, and spirofungins A and B.
441:
Additionally, a one-pot magnesium–halogen exchange with subsequent arylation has been developed, showcasing the stability of the
Weinreb–Nahm amide and providing an operationally simple method for the synthesis of aryl ketones.
283:
Cl affords the corresponding
Weinreb amide in good yields. Alternatively, non-nucleophilic Grignard reagents such as isopropyl magnesium chloride can be used to activate the amine before addition of the ester.
784:
Martinelli, J. R.; Freckmann, D. M. M.; Buchwald, S. L. (2006), "Convenient Method for the
Preparation of Weinreb Amides via Pd-Catalyzed Aminocarbonylation of Aryl Bromides at Atmospheric Pressure",
342:
The standard conditions for the
Weinreb–Nahm ketone synthesis are known to tolerate a wide variety of functional groups elsewhere in the molecule, including alpha-halogen substitution, N-protected
168:
The major advantage of this method over addition of organometallic reagents to more typical acyl compounds is that it avoids the common problem of over-addition. For these latter reactions, two
904:
Whipple, W. L.; Reich, H. J. (1991), "Use of N,N'-dimethoxy-N,N'-dimethylurea as a carbonyl dication equivalent in organometallic addition reactions. Synthesis of unsymmetrical ketones",
386:
have been reported. However, with highly basic or sterically hindered nucleophiles, elimination of the methoxide moiety to release formaldehyde can occur as a significant side reaction.
758:
256:
In addition to the original procedure shown above (which may have compatibility issues for sensitive substrates), Weinreb amides can be synthesized from a variety of
426:
has been performed to avoid the sometimes harsh conditions required for addition of hydride reagents or organometallic compounds. This yields an N-methyl-N-methoxy-
1008:
932:
Sibi, M. P.; Sharma, R.; Paulson, K. L. (1992), "N,N′-Dimethoxy-N,N -Dimethylethanediamide: A Useful α-Oxo-N-Methoxy-N-Methylamide and 1,2-Diketone
Synthon",
568:
Paek, S.-M.; Seo, S.-Y.; Kim, S.-H.; Jung, J.-W.; Lee, Y.-S.; Jung, J.-K.; Suh, Y.-G. (2005), "Concise
Syntheses of (+)-Macrosphelides A and B",
179:
495:
187:
The
Weinreb–Nahm amide has since been adopted into regular use by organic chemists as a dependable method for the synthesis of ketones. These
738:
433:
329:
646:
Shimizu, T.; Satoh, T.; Murakoshi, K.; Sodeoka, M. (2005), "Asymmetric Total Synthesis of (−)-Spirofungin A and (+)-Spirofungin B",
160:
239:
963:
906:
725:
684:
287:
1013:
464:
314:
848:
Hisler, K.; Tripoli, R.; Murphy, J. A. (2006), "Reactions of Weinreb amides: formation of aldehydes by Wittig reactions",
820:
Graham, S. L.; Scholz, T. H. (1990), "A new mode of reactivity of N-methoxy-N-methylamides with strongly basic reagents",
409:
1003:
756:
Mentzel, M.; Hoffmann, H. M. R. (1997), "N-methoxy-N-methylamides (Weinreb amides) in modern organic synthesis",
509:
261:
150:
111:
69:
358:, sulfinates, and phosphonate esters. A wide variety of nucleophiles can be used in conjunction with the amide.
209:
445:
453:
More unusual reagents with multiple Weinreb–Nahm amide functional groups have been synthesized, serving as CO
176:
rather than a ketone or aldehyde. This occurs even if the equivalents of nucleophile are closely controlled.
1018:
876:
Conrad, K.; Hsiao, Y.; Miller, R. (2005), "A practical one-pot process for α-amino aryl ketone synthesis",
95:
169:
682:
Qu, B.; Collum, D. B. (2006), "Mechanism of Acylation of Lithium Phenylacetylide with a Weinreb Amide",
217:
134:
208:
to explain the selectivity shown in reactions of the Weinreb–Nahm amide. Their suggestion was that the
723:
Singh, J.; Satyamurthi, N.; Aidhen, I. S. (2000), "The Growing Synthetic Utility of Weinreb's Amide",
304:
934:
878:
850:
822:
539:
233:
173:
232:
group as shown. This intermediate is stable only at low temperatures, requiring a low-temperature
308:
205:
492:
alkylation followed by facile cleavage to the corresponding enantioenriched aldehyde or ketone.
980:
803:
701:
665:
626:
587:
473:
399:
389:
296:
130:
99:
91:
53:
43:
972:
943:
915:
887:
859:
831:
795:
767:
734:
693:
657:
618:
579:
548:
481:
363:
188:
537:
Nahm, S.; Weinreb, S. M. (1981), "N-methoxy-n-methylamides as effective acylating agents",
786:
648:
609:
570:
485:
423:
322:
Finally, an aminocarbonylation reaction reported by Stephen Buchwald allows conversion of
192:
154:
260:
compounds. The vast majority of these procedures utilize the commercially available salt
299:
reagents can also be used to prepare Weinreb–Nahm amides from carboxylic acids. Various
221:
126:
835:
552:
997:
947:
359:
107:
106:. The original reaction involved two subsequent substitutions: the conversion of an
607:
Barbazanges, M.; Meyer, C.; Cossy, J. (2008), "Total Synthesis of Amphidinolide J",
300:
891:
863:
383:
347:
430:
that converts to the corresponding ketone or aldehyde upon hydrolytic workup.
403:
355:
343:
771:
484:
that combines the functionality of the Weinreb amide with that of the Myers'
17:
739:
10.1002/(sici)1521-3897(200004)342:4<340::aid-prac340>3.0.co;2-1
367:
225:
984:
807:
705:
669:
630:
591:
138:
919:
489:
458:
427:
351:
272:
229:
103:
976:
799:
697:
661:
622:
583:
514:
477:
379:
371:
311:-based couplings have been reported specifically for this purpose.
268:
146:
142:
375:
323:
257:
494:
463:
444:
432:
408:
388:
328:
313:
286:
238:
178:
159:
264:, which is typically easier to handle than the free amine.
499:
Davies' Auxiliary with Weinreb–Nahm-like functionality
248:
was confirmed by spectroscopic and kinetic analyses.
437:
Reaction of Weinreb–Nahm amides with Wittig reagents
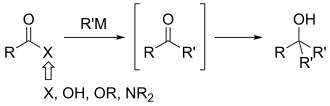
204:Weinreb and Nahm originally proposed the following
125:, and subsequent treatment of this species with an
137:. Nahm and Weinreb also reported the synthesis of
326:halides directly into aryl Weinreb–Nahm amides.
366:are most commonly employed; examples involving
759:Journal für Praktische Chemie/Chemiker-Zeitung
333:Aminocarbonylation to form Weinreb–Nahm amides
291:Example of syntheses from esters and lactones
8:
563:
561:
318:Example of Syntheses from Carboxyllic Acids
641:
639:
602:
600:
102:and Steven Nahm as a method to synthesize
31:
751:
749:
747:
718:
716:
714:
532:
530:
526:
488:auxiliary, allowing diastereoselective
262:N,O-dimethylhydroxylamine hydrochloride
468:Synthons based on Weinreb–Nahm-amides
422:Reaction of Weinreb–Nahm amides with
172:of the incoming group add to form an
7:
1009:Carbon-carbon bond forming reactions
413:Syntheses using Weinreb–Nahm amide
402:family of macrosphelides, and the
25:
191:are present in a large number of
164:The Weinreb–Nahm ketone synthesis
964:The Journal of Organic Chemistry
907:The Journal of Organic Chemistry
685:The Journal of Organic Chemistry
98:. It was discovered in 1981 by
90:is a chemical reaction used in
1:
836:10.1016/s0040-4039(00)97039-4
726:Journal für praktische Chemie
553:10.1016/s0040-4039(01)91316-4
216:below) formed as a result of
88:Weinreb–Nahm ketone synthesis
948:10.1016/0040-4039(92)88108-h
892:10.1016/j.tetlet.2005.09.183
864:10.1016/j.tetlet.2006.06.118
183:Overaddition of nucleophiles
1035:
449:One-pot arylation reaction
510:N,O-Dimethylhydroxylamine
224:reagent is stabilized by
76:
65:Organic Chemistry Portal
59:
35:Weinreb ketone synthesis
34:
772:10.1002/prac.19973390194
406:family of spirofungins.
210:tetrahedral intermediate
151:lithium aluminum hydride
84:Weinreb ketone synthesis
70:weinreb-ketone-synthesis
1014:Substitution reactions
500:
469:
450:
438:
414:
394:
334:
319:
292:
244:
184:
165:
119:-Dimethylhydroxylamine
498:
467:
448:
436:
412:
392:
332:
317:
290:
242:
218:nucleophilic addition
182:
163:
135:organolithium reagent
346:, α-β unsaturation,
305:hydroxybenzotriazole
935:Tetrahedron Letters
920:10.1021/jo00008a057
879:Tetrahedron Letters
851:Tetrahedron Letters
823:Tetrahedron Letters
540:Tetrahedron Letters
243:Chelation mechanism
96:carbon–carbon bonds
1004:Coupling reactions
501:
470:
451:
439:
415:
395:
335:
320:
309:triphenylphosphine
293:
245:
206:reaction mechanism
185:
166:
149:with an excess of
129:reagent such as a
123:Weinreb–Nahm amide
977:10.1021/jo902499s
942:(15): 1941–1944,
886:(49): 8587–8589,
858:(35): 6293–6295,
830:(44): 6269–6272,
800:10.1021/ol061902t
794:(21): 4843–4846,
698:10.1021/jo061223w
692:(18): 7117–7119,
662:10.1021/ol052039k
656:(25): 5573–5576,
623:10.1021/ol801708x
617:(20): 4489–4492,
584:10.1021/ol0508429
578:(15): 3159–3162,
547:(39): 3815–3818,
474:Stephen G. Davies
400:immunosuppressant
364:Grignard reagents
189:functional groups
100:Steven M. Weinreb
92:organic chemistry
80:
79:
54:Coupling reaction
44:Steven M. Weinreb
27:Chemical reaction
16:(Redirected from
1026:
988:
987:
971:(4): 1214–1227,
957:
951:
950:
929:
923:
922:
914:(8): 2911–2912,
901:
895:
894:
873:
867:
866:
845:
839:
838:
817:
811:
810:
781:
775:
774:
753:
742:
741:
720:
709:
708:
679:
673:
672:
643:
634:
633:
604:
595:
594:
565:
556:
555:
534:
515:Ketone#Synthesis
482:chiral auxiliary
297:peptide coupling
267:Treatment of an
193:natural products
131:Grignard reagent
72:
32:
21:
1034:
1033:
1029:
1028:
1027:
1025:
1024:
1023:
994:
993:
992:
991:
959:
958:
954:
931:
930:
926:
903:
902:
898:
875:
874:
870:
847:
846:
842:
819:
818:
814:
787:Organic Letters
783:
782:
778:
755:
754:
745:
722:
721:
712:
681:
680:
676:
649:Organic Letters
645:
644:
637:
610:Organic Letters
606:
605:
598:
571:Organic Letters
567:
566:
559:
536:
535:
528:
523:
506:
486:pseudoephedrine
480:has designed a
457:and α-diketone
456:
424:Wittig reagents
420:
340:
282:
278:
254:
202:
155:amide reduction
68:
28:
23:
22:
15:
12:
11:
5:
1032:
1030:
1022:
1021:
1019:Name reactions
1016:
1011:
1006:
996:
995:
990:
989:
952:
924:
896:
868:
840:
812:
776:
743:
710:
674:
635:
596:
557:
525:
524:
522:
519:
518:
517:
512:
505:
502:
454:
419:
416:
354:and lactones,
339:
336:
280:
276:
253:
250:
222:organometallic
201:
198:
127:organometallic
78:
77:
74:
73:
66:
62:
61:
57:
56:
51:
50:Reaction type
47:
46:
41:
37:
36:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
1031:
1020:
1017:
1015:
1012:
1010:
1007:
1005:
1002:
1001:
999:
986:
982:
978:
974:
970:
966:
965:
956:
953:
949:
945:
941:
937:
936:
928:
925:
921:
917:
913:
909:
908:
900:
897:
893:
889:
885:
881:
880:
872:
869:
865:
861:
857:
853:
852:
844:
841:
837:
833:
829:
825:
824:
816:
813:
809:
805:
801:
797:
793:
789:
788:
780:
777:
773:
769:
765:
761:
760:
752:
750:
748:
744:
740:
736:
732:
728:
727:
719:
717:
715:
711:
707:
703:
699:
695:
691:
687:
686:
678:
675:
671:
667:
663:
659:
655:
651:
650:
642:
640:
636:
632:
628:
624:
620:
616:
612:
611:
603:
601:
597:
593:
589:
585:
581:
577:
573:
572:
564:
562:
558:
554:
550:
546:
542:
541:
533:
531:
527:
520:
516:
513:
511:
508:
507:
503:
497:
493:
491:
487:
483:
479:
475:
466:
462:
460:
447:
443:
435:
431:
429:
425:
417:
411:
407:
405:
401:
393:Side reaction
391:
387:
385:
381:
377:
373:
369:
365:
361:
357:
353:
349:
345:
337:
331:
327:
325:
316:
312:
310:
306:
302:
298:
295:A variety of
289:
285:
274:
270:
265:
263:
259:
251:
249:
241:
237:
235:
231:
227:
223:
219:
215:
211:
207:
199:
197:
194:
190:
181:
177:
175:
171:
162:
158:
156:
152:
148:
144:
140:
136:
132:
128:
124:
120:
118:
114:
109:
108:acid chloride
105:
101:
97:
93:
89:
85:
75:
71:
67:
64:
63:
58:
55:
52:
49:
48:
45:
42:
39:
38:
33:
30:
19:
18:Weinreb amide
968:
962:
955:
939:
933:
927:
911:
905:
899:
883:
877:
871:
855:
849:
843:
827:
821:
815:
791:
785:
779:
763:
757:
730:
724:
689:
683:
677:
653:
647:
614:
608:
575:
569:
544:
538:
471:
452:
440:
421:
396:
384:nucleophiles
348:silyl ethers
341:
321:
301:carbodiimide
294:
266:
255:
246:
213:
203:
186:
167:
122:
121:, to form a
116:
112:
87:
83:
81:
60:Identifiers
40:Named after
29:
766:: 517–524,
344:amino acids
252:Preparation
170:equivalents
998:Categories
521:References
418:Variations
404:antibiotic
356:sulfonates
350:, various
472:Finally,
368:aliphatic
360:Lithiates
275:with AlMe
228:from the
226:chelation
200:Mechanism
143:reduction
139:aldehydes
985:20095549
808:17020317
706:16930080
670:16320994
631:18811171
592:16018610
504:See also
459:synthons
94:to make
733:: 340,
490:enolate
428:enamine
382:carbon
380:alkynyl
352:lactams
307:-, and
279:or AlMe
273:lactone
230:methoxy
220:by the
174:alcohol
145:of the
104:ketones
983:
806:
704:
668:
629:
590:
478:Oxford
378:, and
234:quench
372:vinyl
338:Scope
269:ester
153:(see
147:amide
110:with
981:PMID
804:PMID
702:PMID
666:PMID
627:PMID
588:PMID
376:aryl
362:and
324:aryl
258:acyl
82:The
973:doi
944:doi
916:doi
888:doi
860:doi
832:doi
796:doi
768:doi
764:339
735:doi
731:342
694:doi
658:doi
619:doi
580:doi
549:doi
476:of
303:-,
271:or
157:).
141:by
133:or
86:or
1000::
979:,
969:75
967:,
940:33
938:,
912:56
910:,
884:46
882:,
856:47
854:,
828:31
826:,
802:,
790:,
762:,
746:^
729:,
713:^
700:,
690:71
688:,
664:,
652:,
638:^
625:,
615:10
613:,
599:^
586:,
574:,
560:^
545:22
543:,
529:^
461:.
374:,
370:,
236:.
975::
946::
918::
890::
862::
834::
798::
792:8
770::
737::
696::
660::
654:7
621::
582::
576:7
551::
455:2
281:2
277:3
214:A
212:(
117:O
115:,
113:N
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.