567:
544:
847:
40:
1486:
955:
rejected P&G's attempted injunction. P&G was criticized for attempting to "preserve its market share by denigrating Boniva". Judge Crotty wrote that "Roche was clearly entitled to respond with its own data, provided that the data was truthfully and accurately presented".
951:. The manufacturers of Boniva, a rival bisphosphonate, were accused in the suit of causing a "serious public health risk" through misrepresentation of scientific findings. In a ruling on September 7, 2006, U.S. District Judge
1227:
195:
1536:
150:
697:
1511:
1220:
1082:
1213:
919:
under the trade names
Actonel, Atelvia, and Benet. It is also available in a preparation that includes a calcium carbonate supplement, as Actonel with Calcium.
62:
653:
1135:
1096:
639:
1433:
1526:
1058:
1033:
894:
673:
868:
996:
959:
In 2006, P&G faced controversy over its handling of clinical research involving risedronate (News
Reports and discussion).
306:
180:
80:
872:
916:
1476:
1117:
1521:
1516:
1338:
428:
523:
99:
857:
1330:
730:
876:
861:
1506:
967:
377:
1371:
136:
562:
1143:
252:
681:
InChI=1S/C7H11NO7P2/c9-7(16(10,11)12,17(13,14)15)4-6-2-1-3-8-5-6/h1-3,5,9H,4H2,(H2,10,11,12)(H2,13,14,15)
368:
484:
272:
1194:
928:
734:
539:
323:
143:
1381:
1182:
1076:
110:
505:
1436:
1064:
1054:
1029:
1023:
940:
495:
221:
208:
52:
1205:
579:
332:
234:
437:
1531:
1490:
1343:
1318:
1298:
1288:
1283:
1278:
1273:
1268:
1263:
948:
944:
908:
820:
802:
792:
782:
772:
417:
262:
242:
72:
566:
543:
1313:
1308:
1250:
963:
952:
932:
912:
762:
722:
1500:
1441:
555:
1446:
1241:
1237:
726:
163:
158:
1157:
357:
1419:
1411:
1364:
846:
31:
725:. It slows down the cells which break down bone. It's used to treat or prevent
1453:
1426:
1404:
936:
615:
408:
1068:
1463:
1396:
285:
66:
1258:
388:
94:
397:
39:
1458:
343:
17:
966:
drugs, risedronate appears to be associated with the rare side effect
475:
290:
970:, often preceded by dental procedures inducing trauma to the bone.
464:
457:
1389:
638:
629:
511:
448:
202:
1209:
215:
89:
840:
740:
It was patented in 1984 and approved for medical use in 1998.
528:
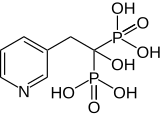
314:(1-hydroxy-1-phosphono-2-pyridin-3-yl-ethyl)phosphonic acid
189:
121:
704:
1474:
1352:
1329:
1249:
1118:"Boniva advertising 'not misleading' says US judge"
991:
989:
987:
985:
983:
627:
614:
578:
573:
554:
522:
494:
474:
447:
427:
407:
387:
376:
367:
342:
322:
297:
284:
271:
261:
251:
241:
233:
179:
174:
149:
135:
109:
79:
61:
51:
46:
997:"Actonel- risedronate sodium tablet, film coated"
1537:Drugs developed by Takeda Pharmaceutical Company
356:
331:
939:false claims lawsuit against rival drugmakers
1221:
8:
98:
30:
875:. Unsourced material may be challenged and
226:In general: ℞ (Prescription only)
1512:Farnesyl pyrophosphate synthase inhibitors
1228:
1214:
1206:
1081:: CS1 maint: location missing publisher (
565:
542:
416:
895:Learn how and when to remove this message
436:
747:
1481:
979:
678:
658:
538:
396:
311:
1074:
1028:. John Wiley & Sons. p. 523.
556:
29:
504:
483:
71:
7:
1201:. U.S. National Library of Medicine.
1189:. U.S. National Library of Medicine.
1160:. Scientific-misconduct.blogspot.com
873:adding citations to reliable sources
510:
162:
1434:Parathyroid hormone-related protein
463:
456:
347:
1051:Essentials of medical pharmacology
605:
25:
1049:Tripathi KD (30 September 2013).
947:claiming false advertising about
661:OC(Cc1cccnc1)(P(=O)(O)O)P(=O)(O)O
1484:
845:
717:, often used as its sodium salt
596:
590:
38:
1053:(Seventh ed.). New Delhi.
1022:Fischer J, Ganellin CR (2006).
907:It is produced and marketed by
686:Key:IIDJRNMFWXDHID-UHFFFAOYSA-N
57:Actonel, Atelvia, Benet, others
599:
584:
1:
1025:Analogue-based Drug Discovery
1158:"Scientific Misconduct Blog"
1136:"Actonel Case Media Reports"
1331:Bone morphogenetic proteins
1553:
1140:Scientific Misconduct Wiki
931:and its marketing partner
574:Chemical and physical data
1527:Drugs developed by AbbVie
1097:"P&G Press statement"
694:
669:
649:
302:
37:
968:osteonecrosis of the jaw
1372:Aluminium chlorohydrate
1236:Drugs for treatment of
1199:Drug Information Portal
1187:Drug Information Portal
731:Paget's disease of bone
1377:Dual action bone agent
962:In common with other
1517:Procter & Gamble
1360:Resorption inhibitor
1195:"Risedronate sodium"
869:improve this section
1522:3-Pyridyl compounds
1146:on 2 February 2009.
1124:. 8 September 2006.
832:Society and culture
750:
211:(Prescription only)
198:(Prescription only)
34:
1382:Strontium ranelate
915:, and in Japan by
748:
719:risedronate sodium
1472:
1471:
1183:"Risedronic acid"
1003:. 1 November 2019
905:
904:
897:
829:
828:
757:Relative potency
749:Relative potency
712:
711:
640:Interactive image
524:CompTox Dashboard
219:
206:
193:
125:
92:
27:Chemical compound
16:(Redirected from
1544:
1489:
1488:
1487:
1480:
1305:Non-nitrogenous
1230:
1223:
1216:
1207:
1202:
1190:
1169:
1168:
1166:
1165:
1154:
1148:
1147:
1142:. Archived from
1132:
1126:
1125:
1114:
1108:
1107:
1105:
1104:
1093:
1087:
1086:
1080:
1072:
1046:
1040:
1039:
1019:
1013:
1012:
1010:
1008:
993:
927:In January 2006
900:
893:
889:
886:
880:
849:
841:
751:
708:
707:
700:
642:
622:
607:
601:
598:
592:
586:
569:
558:
547:
546:
532:
530:
514:
508:
487:
467:
460:
440:
420:
400:
380:
360:
350:
349:
335:
276:
217:
214:
204:
201:
191:
188:
166:
123:
120:
102:
91:
88:
75:
42:
35:
33:
21:
1552:
1551:
1547:
1546:
1545:
1543:
1542:
1541:
1507:Bisphosphonates
1497:
1496:
1495:
1485:
1483:
1475:
1473:
1468:
1348:
1344:Eptotermin alfa
1339:Dibotermin alfa
1325:
1319:Tiludronic acid
1299:Zoledronic acid
1294:Risedronic acid
1289:Pamidronic acid
1284:Neridronic acid
1279:Minodronic acid
1274:Incadronic acid
1269:Ibandronic acid
1264:Alendronic acid
1251:Bisphosphonates
1245:
1234:
1193:
1181:
1178:
1173:
1172:
1163:
1161:
1156:
1155:
1151:
1134:
1133:
1129:
1116:
1115:
1111:
1102:
1100:
1095:
1094:
1090:
1073:
1061:
1048:
1047:
1043:
1036:
1021:
1020:
1016:
1006:
1004:
995:
994:
981:
976:
945:GlaxoSmithKline
925:
909:Warner Chilcott
901:
890:
884:
881:
866:
850:
839:
834:
754:Bisphosphonate
746:
715:Risedronic acid
703:
701:
698:(what is this?)
695:
690:
687:
682:
677:
676:
665:
662:
657:
656:
645:
620:
610:
604:
595:
589:
550:
526:
518:
490:
470:
443:
423:
403:
383:
363:
346:
338:
318:
315:
310:
309:
274:
253:Protein binding
243:Bioavailability
235:Pharmacokinetic
229:
170:
138:
131:
112:
105:
32:Risedronic acid
28:
23:
22:
15:
12:
11:
5:
1550:
1548:
1540:
1539:
1534:
1529:
1524:
1519:
1514:
1509:
1499:
1498:
1494:
1493:
1470:
1469:
1467:
1466:
1461:
1456:
1451:
1450:
1449:
1444:
1431:
1430:
1429:
1416:
1415:
1414:
1401:
1400:
1399:
1386:
1385:
1384:
1374:
1369:
1368:
1367:
1356:
1354:
1350:
1349:
1347:
1346:
1341:
1335:
1333:
1327:
1326:
1324:
1323:
1322:
1321:
1316:
1314:Clodronic acid
1311:
1309:Etidronic acid
1303:
1302:
1301:
1296:
1291:
1286:
1281:
1276:
1271:
1266:
1255:
1253:
1247:
1246:
1235:
1233:
1232:
1225:
1218:
1210:
1204:
1203:
1191:
1177:
1176:External links
1174:
1171:
1170:
1149:
1127:
1109:
1088:
1059:
1041:
1034:
1014:
978:
977:
975:
972:
964:bisphosphonate
953:Paul A. Crotty
933:Sanofi-Aventis
924:
921:
913:Sanofi-Aventis
903:
902:
885:September 2023
853:
851:
844:
838:
835:
833:
830:
827:
826:
823:
817:
816:
813:
809:
808:
805:
799:
798:
795:
789:
788:
785:
779:
778:
775:
769:
768:
765:
759:
758:
755:
745:
742:
733:. It is taken
723:bisphosphonate
710:
709:
692:
691:
689:
688:
685:
683:
680:
672:
671:
670:
667:
666:
664:
663:
660:
652:
651:
650:
647:
646:
644:
643:
635:
633:
625:
624:
618:
612:
611:
608:
602:
593:
587:
582:
576:
575:
571:
570:
560:
552:
551:
549:
548:
535:
533:
520:
519:
517:
516:
500:
498:
492:
491:
489:
488:
480:
478:
472:
471:
469:
468:
461:
453:
451:
445:
444:
442:
441:
433:
431:
425:
424:
422:
421:
413:
411:
405:
404:
402:
401:
393:
391:
385:
384:
382:
381:
373:
371:
365:
364:
362:
361:
353:
351:
340:
339:
337:
336:
328:
326:
320:
319:
317:
316:
313:
305:
304:
303:
300:
299:
295:
294:
288:
282:
281:
278:
269:
268:
265:
259:
258:
255:
249:
248:
245:
239:
238:
231:
230:
228:
227:
224:
212:
199:
185:
183:
177:
176:
172:
171:
169:
168:
155:
153:
147:
146:
141:
139:administration
133:
132:
130:
129:
127:
117:
115:
107:
106:
104:
103:
85:
83:
77:
76:
69:
59:
58:
55:
49:
48:
44:
43:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
1549:
1538:
1535:
1533:
1530:
1528:
1525:
1523:
1520:
1518:
1515:
1513:
1510:
1508:
1505:
1504:
1502:
1492:
1482:
1478:
1465:
1462:
1460:
1457:
1455:
1452:
1448:
1445:
1443:
1442:Abaloparatide
1440:
1439:
1438:
1435:
1432:
1428:
1425:
1424:
1423:
1421:
1417:
1413:
1410:
1409:
1408:
1406:
1402:
1398:
1395:
1394:
1393:
1391:
1387:
1383:
1380:
1379:
1378:
1375:
1373:
1370:
1366:
1363:
1362:
1361:
1358:
1357:
1355:
1351:
1345:
1342:
1340:
1337:
1336:
1334:
1332:
1328:
1320:
1317:
1315:
1312:
1310:
1307:
1306:
1304:
1300:
1297:
1295:
1292:
1290:
1287:
1285:
1282:
1280:
1277:
1275:
1272:
1270:
1267:
1265:
1262:
1261:
1260:
1257:
1256:
1254:
1252:
1248:
1243:
1239:
1238:bone diseases
1231:
1226:
1224:
1219:
1217:
1212:
1211:
1208:
1200:
1196:
1192:
1188:
1184:
1180:
1179:
1175:
1159:
1153:
1150:
1145:
1141:
1137:
1131:
1128:
1123:
1119:
1113:
1110:
1098:
1092:
1089:
1084:
1078:
1070:
1066:
1062:
1060:9789350259375
1056:
1052:
1045:
1042:
1037:
1035:9783527607495
1031:
1027:
1026:
1018:
1015:
1002:
998:
992:
990:
988:
986:
984:
980:
973:
971:
969:
965:
960:
957:
954:
950:
946:
942:
938:
934:
930:
923:Controversies
922:
920:
918:
914:
910:
899:
896:
888:
878:
874:
870:
864:
863:
859:
854:This section
852:
848:
843:
842:
836:
831:
824:
822:
819:
818:
814:
811:
810:
806:
804:
801:
800:
796:
794:
791:
790:
786:
784:
781:
780:
776:
774:
771:
770:
766:
764:
761:
760:
756:
753:
752:
743:
741:
738:
736:
732:
728:
724:
720:
716:
706:
699:
693:
684:
679:
675:
668:
659:
655:
648:
641:
637:
636:
634:
631:
626:
619:
617:
613:
583:
581:
577:
572:
568:
564:
561:
559:
557:ECHA InfoCard
553:
545:
541:
540:DTXSID2023563
537:
536:
534:
525:
521:
513:
512:RCSB PDB
507:
502:
501:
499:
497:
493:
486:
482:
481:
479:
477:
473:
466:
462:
459:
455:
454:
452:
450:
446:
439:
435:
434:
432:
430:
426:
419:
415:
414:
412:
410:
406:
399:
395:
394:
392:
390:
386:
379:
375:
374:
372:
370:
366:
359:
355:
354:
352:
345:
341:
334:
330:
329:
327:
325:
321:
312:
308:
301:
296:
292:
289:
287:
283:
279:
277:
270:
266:
264:
260:
256:
254:
250:
246:
244:
240:
236:
232:
225:
223:
213:
210:
200:
197:
187:
186:
184:
182:
178:
173:
165:
160:
157:
156:
154:
152:
148:
145:
142:
140:
134:
128:
119:
118:
116:
114:
108:
101:
96:
87:
86:
84:
82:
78:
74:
70:
68:
64:
60:
56:
54:
50:
47:Clinical data
45:
41:
36:
19:
1447:Teriparatide
1418:
1403:
1388:
1376:
1359:
1293:
1198:
1186:
1162:. Retrieved
1152:
1144:the original
1139:
1130:
1122:Pharma Times
1121:
1112:
1101:. Retrieved
1091:
1050:
1044:
1024:
1017:
1005:. Retrieved
1000:
961:
958:
926:
906:
891:
882:
867:Please help
855:
812:Risedronate
744:Pharmacology
739:
729:, and treat
727:osteoporosis
718:
714:
713:
702:
696:
273:Elimination
181:Legal status
175:Legal status
81:License data
1420:Cathepsin K
1412:Romosozumab
1365:Ipriflavone
1259:Nitrogenous
1099:. Uk.pg.com
837:Brand names
821:Zoledronate
803:Ibandronate
793:Alendronate
783:Pamidronate
773:Tiludronate
623: g·mol
563:100.116.436
333:105462-24-6
298:Identifiers
53:Trade names
1501:Categories
1454:Calcitonin
1427:Odanacatib
1405:Sclerostin
1164:2013-03-01
1103:2013-03-01
974:References
937:Lanham Act
763:Etidronate
628:3D model (
616:Molar mass
496:PDB ligand
438:KM2Z91756Z
409:ChemSpider
369:IUPHAR/BPS
324:CAS Number
307:IUPAC name
263:Metabolism
100:Risedronic
1464:Vitamin D
1437:analogues
1422:inhibitor
1407:inhibitor
1397:Denosumab
1392:inhibitor
1077:cite book
1069:868299888
856:does not
807:500-1000
485:ChEMBL923
293:and fecal
286:Excretion
275:half-life
137:Routes of
111:Pregnancy
73:Monograph
67:Drugs.com
1491:Medicine
1001:DailyMed
935:filed a
797:100-500
735:by mouth
705:(verify)
389:DrugBank
151:ATC code
144:By mouth
126: B3
113:category
95:DailyMed
1459:Calcium
1007:28 June
929:P&G
877:removed
862:sources
721:, is a
621:283.113
580:Formula
398:DB00884
344:PubChem
167:)
161: (
159:M05BA07
97::
18:Actonel
1532:Sanofi
1477:Portal
1067:
1057:
1032:
949:Boniva
917:Takeda
654:SMILES
476:ChEMBL
465:D03234
458:D00942
291:Kidney
222:℞-only
220:
207:
194:
93:
1390:RANKL
1353:Other
941:Roche
825:5000
815:1000
674:InChI
630:JSmol
503:RIS (
280:1.5 h
247:0.63%
1083:link
1065:OCLC
1055:ISBN
1030:ISBN
1009:2022
943:and
860:any
858:cite
787:100
506:PDBe
449:KEGG
429:UNII
418:5055
378:3176
358:5245
267:None
257:~24%
237:data
63:AHFS
1242:M05
871:by
777:10
529:EPA
348:CID
209:POM
164:WHO
1503::
1197:.
1185:.
1138:.
1120:.
1079:}}
1075:{{
1063:.
999:.
982:^
911:,
767:1
737:.
594:11
509:,
216:US
203:UK
196:S4
190:AU
122:AU
90:US
1479::
1244:)
1240:(
1229:e
1222:t
1215:v
1167:.
1106:.
1085:)
1071:.
1038:.
1011:.
898:)
892:(
887:)
883:(
879:.
865:.
632:)
609:2
606:P
603:7
600:O
597:N
591:H
588:7
585:C
531:)
527:(
515:)
218::
205::
192::
124::
65:/
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.