215:, each carbon atom uses only 3 of its 4 outer energy level electrons in covalently bonding to three other carbon atoms in a plane. Each carbon atom contributes one electron to a delocalized system of electrons that is also a part of the chemical bonding. The delocalized electrons are free to move throughout the plane. For this reason, graphite conducts electricity along the planes of carbon atoms, but does not conduct in a direction at
31:
256:
In the methane molecule, ab initio calculations show bonding character in four molecular orbitals, sharing the electrons uniformly among all five atoms. There are two orbital levels, a bonding molecular orbital formed from the 2s orbital on carbon and triply degenerate bonding molecular orbitals from
165:
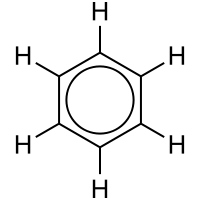
ring is often graphically indicated by a circle. The fact that the six C-C bonds are equidistant is one indication that the electrons are delocalized; if the structure were to have isolated double bonds alternating with discrete single bonds, the bond would likewise have alternating longer and
211:
atom are 'localized' between the atoms in covalent bonding. The movement of electrons is restricted and diamond does not conduct an electric current. In
196:) in a "sea" of delocalized electrons. This means that the electrons are free to move throughout the structure, and gives rise to properties such as
116:
234:
242:
188:
Delocalized electrons also exist in the structure of solid metals. Metallic structure consists of aligned positive
320:
325:
197:
250:
270:
171:
140:
93:
257:
each of the 2p orbitals on carbon. The localized sp orbitals corresponding to each individual bond in
146:
258:
167:
112:
108:
246:
101:
280:
238:
228:
127:
123:
97:
89:
183:
241:
that, in general, extend over an entire molecule and have the symmetry of the molecule.
30:
314:
74:
216:
275:
46:
17:
261:
can be obtained from a linear combination of the four molecular orbitals.
212:
84:
is general and can have slightly different meanings in different fields:
58:
54:
38:
300:
204:
158:
150:
34:
208:
193:
27:
Electrons that are not associated with a single atom or covalent bond
66:
29:
70:
189:
62:
130:
electrons that have extended over several adjacent atoms.
249:
of the delocalized orbitals, given by an appropriate
170:, delocalization in benzene is represented by
8:
292:
69:that are not associated with a single
7:
235:ab initio quantum chemistry methods
25:
207:all four outer electrons of each
37:, with the delocalization of the
1:
342:
226:
181:
138:
41:indicated by the circle
251:unitary transformation
42:
271:Aromatic ring current
245:may then be found as
178:Electrical conduction
141:Resonance (chemistry)
117:electrical conduction
51:delocalized electrons
33:
239:delocalized orbitals
172:resonance structures
166:shorter lengths. In
147:simple aromatic ring
259:valence bond theory
247:linear combinations
168:valence bond theory
109:solid-state physics
243:Localized orbitals
223:Molecular orbitals
219:to the plane.
102:aromatic compounds
98:conjugated systems
43:
281:Solvated electron
229:Molecular orbital
128:molecular orbital
124:quantum chemistry
90:organic chemistry
16:(Redirected from
333:
321:Chemical bonding
305:
299:IUPAC Gold Book
297:
184:Metallic bonding
115:that facilitate
21:
341:
340:
336:
335:
334:
332:
331:
330:
326:Electron states
311:
310:
309:
308:
298:
294:
289:
267:
231:
225:
186:
180:
164:
143:
137:
126:, it refers to
111:, it refers to
92:, it refers to
28:
23:
22:
15:
12:
11:
5:
339:
337:
329:
328:
323:
313:
312:
307:
306:
302:delocalization
291:
290:
288:
285:
284:
283:
278:
273:
266:
263:
227:Main article:
224:
221:
182:Main article:
179:
176:
162:
155:delocalization
139:Main article:
136:
133:
132:
131:
120:
113:free electrons
105:
82:delocalization
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
338:
327:
324:
322:
319:
318:
316:
304:
303:
296:
293:
286:
282:
279:
277:
274:
272:
269:
268:
264:
262:
260:
254:
252:
248:
244:
240:
236:
230:
222:
220:
218:
214:
210:
206:
201:
199:
195:
191:
185:
177:
175:
173:
169:
160:
156:
152:
148:
142:
134:
129:
125:
121:
118:
114:
110:
106:
103:
99:
95:
91:
87:
86:
85:
83:
78:
76:
75:covalent bond
72:
68:
64:
60:
56:
52:
48:
40:
36:
32:
19:
301:
295:
255:
232:
217:right angles
202:
198:conductivity
187:
154:
144:
81:
79:
50:
44:
159:π electrons
18:Delocalized
315:Categories
287:References
161:over the C
276:Electride
233:Standard
135:Resonance
94:resonance
80:The term
65:or solid
55:electrons
47:chemistry
39:electrons
265:See also
237:lead to
213:graphite
59:molecule
205:diamond
194:cations
157:of six
151:benzene
145:In the
35:Benzene
209:carbon
153:, the
73:or a
67:metal
57:in a
190:ions
100:and
71:atom
53:are
203:In
149:of
122:In
107:In
96:in
88:In
63:ion
45:In
317::
253:.
200:.
174:.
77:.
61:,
49:,
192:(
163:6
119:.
104:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.