675:
652:
31:
3458:
1126:
and decreases the amount of cholesterol normally available to liver cells. The lower levels of cholesterol in the liver cells leads them to absorb more cholesterol from circulation and thus lowering the levels of circulating cholesterol. It blocks the critical mediator of cholesterol absorption, the
2229:
Basha SJ, Naveed SA, Tiwari NK, Shashikumar D, Muzeeb S, Kumar TR, et al. (June 2007). "Concurrent determination of ezetimibe and its phase-I and II metabolites by HPLC with UV detection: quantitative application to various in vitro metabolic stability studies and for qualitative estimation in
1184:
Ezetimibe is primarily metabolized in the liver and the small intestine via glucuronide conjugation with subsequent renal and biliary excretion. Both the parent compound and its active metabolite are eliminated from plasma with a half-life around 22 hours, allowing for once-daily dosing. Ezetimibe
1800:
Catapano AL, Reiner Z, De Backer G, Graham I, Taskinen MR, Wiklund O, et al. (July 2011). "ESC/EAS Guidelines for the management of dyslipidaemias The Task Force for the management of dyslipidaemias of the
European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS)".
942:
Initiation of ezetimibe along with high-intensity statin therapy at the time of an acute coronary syndrome (ACS) event was associated with significantly better cholesterol reduction at day-7, 1-month, 3-months and 1-year post ACS event; which translated into significantly lower recurrent
2005:
Awad K, Mikhailidis DP, Katsiki N, Muntner P, Banach M (March 2018). "Effect of
Ezetimibe Monotherapy on Plasma Lipoprotein(a) Concentrations in Patients with Primary Hypercholesterolemia: A Systematic Review and Meta-Analysis of Randomized Controlled Trials".
1173:(45–71 ng/ml) of ezetimibe-glucuronide is attained within 1–2 hours. The concomitant administration of food (high-fat vs. nonfat meals) has no effect on the extent of absorption of ezetimibe. However, coadministration with a high-fat meal increases its C
1197:
5–6). Due to insufficient data, the manufacturer does not recommend ezetimibe for patients with moderate to severe hepatic impairment (Child-Pugh score 7–15). In patients with mild, moderate, or severe hepatic impairment, the mean
1096:
The incidence of overdose with ezetimibe is rare; subsequently, few data exist on the effects of overdose. However, an acute overdose of ezetimibe is expected to produce an exaggeration of its usual effects, leading to
1906:"Lipid modification: cardiovascular risk assessment and the modification of blood lipids for the primary and secondary prevention of cardiovascular disease | Guidance and guidelines | NICE"
747:
299:
2142:
Takada T, Yamanashi Y, Konishi K, Yamamoto T, Toyoda Y, Masuo Y, et al. (February 2015). "NPC1L1 is a key regulator of intestinal vitamin K absorption and a modulator of warfarin therapy".
1181:
cannot be determined, since ezetimibe is insoluble in aqueous media suitable for injection. Ezetimibe and its active metabolites are highly bound to human plasma proteins (90%).
224:
1852:"Executive summary of the Japan Atherosclerosis Society (JAS) guidelines for the diagnosis and prevention of atherosclerotic cardiovascular diseases in Japan -2012 version"
1464:
1404:
1300:
2297:
939:. Several treatment guidelines recommend adding ezetimibe in select high risk persons in whom LDL goals cannot be achieved by maximally tolerated statin alone.
799:
InChI=1S/C24H21F2NO3/c25-17-5-1-15(2-6-17)22(29)14-13-21-23(16-3-11-20(28)12-4-16)27(24(21)30)19-9-7-18(26)8-10-19/h1-12,21-23,28-29H,13-14H2/t21-,22+,23-/m1/s1
110:
1923:
Grundy SM, Arai H, Barter P, Bersot TP, Betteridge DJ, Carmena R, et al. (Expert
Dyslipidemia Panel of the International Atherosclerosis Society) (2014).
1169:) was 3.4–5.5 ng/ml. Following oral administration, ezetimibe is absorbed and extensively conjugated to a phenolic glucuronide (active metabolite). Mean C
943:
cardiovascular events (death from any cause, major ACS, non-fatal stroke, non-fatal myocardial infarction, and ischemic stroke) post the index event of ACS.
1434:
771:
3503:
3488:
2207:
2964:
2549:
1199:
935:. Combining ezetimibe with simvastatin had no effect on overall mortality but did lower the risk of heart attack or stroke in people with prior
2265:
2185:
2098:
1891:
1836:
1737:
3431:
3417:
3397:
3074:
3018:
2120:
2290:
1162:
Within 4–12 hours of the oral administration of a 10-mg dose to fasting adults, the attained mean ezetimibe peak plasma concentration (
3097:
877:
3493:
1380:
991:
791:
2119:. U.S. National Library of Medicine, National Institutes of Health, U.S. Department of Health and Human Services. 27 October 2014.
1202:
values for total ezetimibe are increased about 1.7-fold, 3-to-4-fold, and 5-to-6-fold, respectively, compared to healthy subjects.
2328:
1925:"An International Atherosclerosis Society Position Paper: global recommendations for the management of dyslipidemia--full report"
897:
217:
1486:
1456:
1396:
1292:
2283:
421:
284:
142:
1597:
Savarese G, De
Ferrari GM, Rosano GM, Perrone-Filardi P (December 2015). "Safety and efficacy of ezetimibe: A meta-analysis".
1550:
1520:
1343:
3478:
970:
1966:"Impact of early initiation of ezetimibe in patients with acute coronary syndrome: A systematic review and meta-analysis"
2553:
2319:
551:
3380:
3035:
631:
161:
3448:
1905:
1054:
1426:
1189:
isoenzymes, which explains its limited number of drug interactions. No dose adjustment is needed in patients with
1002:
The two contraindications to taking ezetimibe are a previous allergic reaction to it, including symptoms of rash,
3067:
2979:
2878:
2844:
2759:
2501:
1050:
870:
927:
has no effect on overall mortality or cardiovascular mortality, although it significantly reduces the risk of
500:
2199:
3424:
3107:
3023:
2903:
2898:
2873:
2840:
2802:
2586:
2505:
2441:
2368:
936:
198:
670:
3163:
2888:
2883:
1190:
1143:
1132:
924:
866:
862:
838:
367:
2670:
1249:"FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)"
946:
Ezetimibe is indicated in the United States as an add-on to dietary measures to reduce levels of certain
3483:
3320:
3214:
3168:
2893:
2360:
2259:
2179:
2092:
1885:
1830:
1731:
1034:
928:
858:
491:
994:
activity score but the available evidence indicates it does not improve outcomes of hepatic steatosis.
620:
3060:
2934:
2660:
2519:
1248:
911:. In 2022, it was the 79th most commonly prescribed medication in the United States, with more than 8
390:
3142:
2306:
1163:
1046:
850:
647:
446:
205:
3127:
3345:
3269:
2918:
2364:
2167:
2112:
2031:
908:
172:
2051:"Ezetimibe decreased nonalcoholic fatty liver disease activity score but not hepatic steatosis"
3404:
3112:
2913:
2582:
2539:
2437:
2247:
2159:
2080:
2023:
1987:
1946:
1873:
1818:
1782:
1719:
1668:
1614:
1376:
1106:
600:
323:
311:
100:
1686:
Cannon CP, Blazing MA, Giugliano RP, McCagg A, White JA, Theroux P, et al. (June 2015).
3498:
3158:
3117:
2239:
2151:
2070:
2062:
2015:
1977:
1936:
1863:
1810:
1772:
1764:
1709:
1699:
1658:
1648:
1606:
1194:
1058:
983:
A 2018 review found that ezetimibe used as sole treatment slightly lowered plasma levels of
687:
540:
455:
349:
48:
1222:
560:
3462:
3305:
3194:
3173:
2798:
2614:
1814:
1186:
1178:
1123:
907:
Ezetimibe was approved for medical use in the United States in 2002. It is available as a
377:
357:
2232:
Journal of
Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences
1427:"Liptruzet (ezetimibe and atorvastatin) tablets for oral useInitial U.S. Approval: 2013"
674:
651:
3360:
3102:
3083:
3029:
2755:
2595:
2075:
2050:
1777:
1752:
1663:
1636:
1457:"Roszet- rosuvastatin and ezetimibe tablet Roszet (- rosuvastatin and ezetimibe tablet"
1102:
1070:
984:
977:
955:
885:
1037:(≥1% of patients) associated with ezetimibe therapy include headache and/or diarrhea (
3472:
3310:
3224:
3137:
2402:
2392:
2035:
893:
758:
663:
336:
134:
3350:
3249:
3132:
3122:
3004:
2680:
2675:
2642:
2633:
2605:
2534:
2485:
2475:
2455:
2450:
2310:
2171:
842:
480:
267:
262:
257:
252:
247:
242:
237:
232:
2243:
1542:
2155:
1850:
Teramoto T, Sasaki J, Ishibashi S, Birou S, Daida H, Dohi S, et al. (2013).
1610:
1572:
1512:
1335:
120:
3410:
3325:
3279:
3274:
3259:
3254:
3244:
3219:
2984:
2959:
2939:
2908:
2816:
2788:
2783:
2778:
2773:
2768:
2720:
2715:
2705:
2700:
2665:
2572:
2562:
2490:
2480:
2460:
2410:
2377:
1038:
1022:
1014:
1007:
962:
881:
128:
1982:
1965:
1941:
1924:
3340:
3335:
3330:
3289:
3284:
3239:
3199:
3178:
2999:
2974:
2954:
2949:
2944:
2853:
2826:
2821:
2811:
2725:
2695:
2690:
2685:
2619:
2567:
2529:
2470:
2465:
2387:
2382:
2345:
2275:
2019:
1151:
1147:
1139:
1135:
1073:) have been reported and are included as warnings on the label for ezetimibe.
1062:
1003:
967:
854:
834:
723:
531:
212:
3264:
3229:
3204:
3052:
2710:
2397:
2350:
1909:
1081:
901:
889:
403:
114:
2251:
2163:
2084:
2027:
1991:
1950:
1877:
1822:
1786:
1768:
1723:
1672:
1618:
2066:
1704:
1687:
22:
3355:
3234:
2994:
2989:
2969:
2741:
2514:
2406:
2049:
Lee HY, Jun DW, Kim HJ, Oh H, Saeed WK, Ahn H, et al. (March 2019).
1714:
1098:
1085:
1066:
511:
156:
1653:
880:, joint pain, diarrhea, and tiredness. Serious side effects may include
520:
2836:
2652:
1042:
1018:
466:
1868:
1851:
30:
2524:
2433:
2332:
1128:
1077:
947:
932:
846:
611:
580:
2424:
1688:"Ezetimibe Added to Statin Therapy after Acute Coronary Syndromes"
746:
737:
591:
383:
1010:, and severe liver disease, especially when taken with a statin.
571:
317:
3056:
2279:
330:
151:
1256:
1573:"Ezetimibe Drug Usage Statistics, United States, 2013 - 2022"
1637:"Ezetimibe therapy: mechanism of action and clinical update"
1487:"Nexlizet- bempedoic acid and ezetimibe tablet, film coated"
1013:
Ezetimibe may have significant medication interactions with
636:
1041:). Infrequent adverse effects (0.1–1% of patients) include
845:. Generally it is used together with dietary changes and a
293:
183:
84:
60:
51:
1122:
Ezetimibe inhibits the absorption of cholesterol from the
1964:
Mahajan K, Nagendra L, Dhall A, Dutta D (February 2024).
1084:
uptake, the use of ezetimibe can lead to side effects in
849:. Alone, it is less preferred than a statin. It is taken
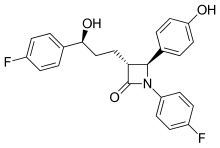
437:)-1-(4-fluorophenyl)-3--4-(4-hydroxyphenyl)azetidin-2-one
72:
66:
2200:"Ezetimibe - National Library of Medicine HSDB Database"
1375:(76 ed.). Pharmaceutical Press. 2018. p. 196.
306:
987:, but the effect was not large enough to be important.
816:
3446:
57:
87:
75:
63:
3389:
3373:
3298:
3187:
3151:
3090:
2927:
2866:
2835:
2797:
2754:
2734:
2651:
2641:
2632:
2604:
2581:
2548:
2499:
2432:
2423:
2359:
2327:
2318:
757:
735:
722:
686:
681:
662:
630:
610:
590:
570:
550:
530:
510:
499:
490:
465:
445:
412:
402:
389:
376:
366:
356:
348:
283:
278:
223:
211:
197:
171:
141:
127:
109:
99:
81:
69:
54:
42:
37:
1217:
1215:
1185:lacks significant inhibitor or inducer effects on
876:The most commonly reported adverse events include
1342:. American Society of Health-System Pharmacists.
1287:
1285:
1283:
1281:
1279:
1277:
1275:
1273:
1069:may occur. Cases of muscle problems (myalgia and
779:Fc1ccc(cc1)(O)CC4C(=O)N(c2ccc(F)cc2)4c3ccc(O)cc3
479:
454:
3068:
2291:
1154:complex involved in trafficking cholesterol.
8:
160:
21:
1397:"Vytorin- ezetimibe and simvastatin tablet"
888:, depression, and muscle breakdown. Use in
3295:
3075:
3061:
3053:
2648:
2638:
2429:
2324:
2298:
2284:
2276:
1330:
1328:
1326:
1324:
1322:
1320:
1318:
1057:) results. Rarely (<0.1% of patients),
961:Mixed hyperlipidemia, in combination with
673:
650:
539:
29:
2965:Magnesium pyridoxal 5-phosphate glutamate
2074:
1981:
1940:
1867:
1856:Journal of Atherosclerosis and Thrombosis
1776:
1713:
1703:
1662:
1652:
896:is of unclear safety. Ezetimibe works by
559:
16:Medication used to treat high cholesterol
1513:"Ezetimibe (Zetia) Use During Pregnancy"
1373:British national formulary : BNF 76
923:Adding ezetimibe to statin treatment of
3453:
2055:The Korean Journal of Internal Medicine
1751:Alenghat FJ, Davis AM (February 2019).
1635:Phan BA, Dayspring TD, Toth PP (2012).
1630:
1628:
1367:
1365:
1363:
1361:
1336:"Ezetimibe Monograph for Professionals"
1211:
796:
776:
763:164 to 166 °C (327 to 331 °F)
646:
519:
426:
133:
2257:
2177:
2090:
1883:
1828:
1729:
1227:Therapeutic Goods Administration (TGA)
973:, in combination with specific statins
664:
20:
3398:Merck Manual of Diagnosis and Therapy
1815:10.1016/j.atherosclerosis.2011.06.011
619:
599:
119:
7:
266:
256:
246:
236:
1692:The New England Journal of Medicine
1641:Vascular Health and Risk Management
1599:International Journal of Cardiology
1553:from the original on 30 August 2024
1467:from the original on 14 August 2022
1437:from the original on 14 August 2022
1407:from the original on 14 August 2022
579:
470:
3504:Drugs developed by Schering-Plough
3489:Drugs developed by Merck & Co.
2264:: CS1 maint: overridden setting (
2184:: CS1 maint: overridden setting (
2097:: CS1 maint: overridden setting (
1890:: CS1 maint: overridden setting (
1835:: CS1 maint: overridden setting (
1736:: CS1 maint: overridden setting (
1523:from the original on 13 April 2019
878:upper respiratory tract infections
14:
2329:Cholesterol absorption inhibitors
2210:from the original on 12 June 2018
1753:"Management of Blood Cholesterol"
1346:from the original on 17 June 2019
992:non-alcoholic fatty liver disease
898:decreasing cholesterol absorption
3456:
2206:. National Library of Medicine.
2123:from the original on 5 July 2016
1303:from the original on 10 May 2021
710:
704:
698:
218:Cholesterol absorption inhibitor
47:
804:Key:OLNTVTPDXPETLC-XPWALMASSA-N
2144:Science Translational Medicine
1929:Journal of Clinical Lipidology
853:. It is also available in the
713:
692:
1:
2244:10.1016/j.jchromb.2007.02.053
1193:or mild hepatic dysfunction (
971:familial hypercholesterolemia
2156:10.1126/scitranslmed.3010329
1611:10.1016/j.ijcard.2015.08.103
829:, sold under the brand name
3381:Merck Headquarters Building
3520:
1983:10.1016/j.ejim.2024.02.004
1942:10.1016/j.jacl.2013.12.005
682:Chemical and physical data
3013:
2980:Omega−3-acid ethyl esters
2020:10.1007/s40265-018-0870-1
1976:: S0953-6205(24)00049-9.
1293:"Zetia- ezetimibe tablet"
812:
787:
767:
417:
28:
3494:4-Fluorophenyl compounds
2879:Bempedoic acid/ezetimibe
2500:Niacin and derivatives (
1131:(NPC1L1) protein on the
958:, alone or with a statin
871:ezetimibe/bempedoic acid
2904:Fenofibrate/simvastatin
2899:Fenofibrate/pravastatin
2874:Amlodipine/atorvastatin
990:Ezetimibe improves the
915:million prescriptions.
3164:Cubist Pharmaceuticals
2889:Ezetimibe/rosuvastatin
2884:Ezetimibe/atorvastatin
2361:Bile acid sequestrants
1769:10.1001/jama.2019.0015
1191:chronic kidney disease
1133:gastrointestinal tract
1129:Niemann-Pick C1-like 1
1035:adverse drug reactions
925:high blood cholesterol
867:ezetimibe/rosuvastatin
863:ezetimibe/atorvastatin
839:high blood cholesterol
3321:Ezetimibe/simvastatin
3215:Ezetimibe/simvastatin
3169:H. K. Mulford Company
3084:Merck & Co., Inc.
2894:Ezetimibe/simvastatin
2307:Lipid-lowering agents
2067:10.3904/kjim.2017.194
1705:10.1056/NEJMoa1410489
1543:"The Top 300 of 2022"
1463:. 15 September 2021.
1433:. 30 September 2016.
1177:by 38%. The absolute
1138:cells, as well as in
929:myocardial infarction
859:ezetimibe/simvastatin
408:Kidney 11%, fecal 78%
3479:Hypolipidemic agents
2935:Alipogene tiparvovec
2661:Aluminium clofibrate
2520:Aluminium nicotinate
1912:on 19 November 2014.
3091:Corporate directors
2671:Choline fenofibrate
1654:10.2147/VHRM.S33664
1493:. 24 September 2021
1299:. 26 January 2011.
1223:"AusPAR: Ezetimibe"
1118:Mechanism of action
1047:liver function test
843:lipid abnormalities
833:among others, is a
326:(Prescription only)
302:(Prescription only)
25:
3346:Rocuronium bromide
3270:rVSV-ZEBOV vaccine
3040:Never to phase III
2919:Niacin/simvastatin
2909:Niacin/laropiprant
2204:toxnet.nlm.nih.gov
909:generic medication
841:and certain other
3444:
3443:
3405:The Merck Manuals
3369:
3368:
3050:
3049:
2914:Niacin/lovastatin
2862:
2861:
2750:
2749:
2628:
2627:
2583:ATP citrate lyase
2540:Nicotinyl alcohol
2438:HMG-CoA reductase
2419:
2418:
1869:10.5551/jat.15792
1698:(25): 2387–2397.
1146:and interrupts a
1061:reactions (rash,
998:Contraindications
824:
823:
748:Interactive image
632:CompTox Dashboard
382:Intestinal wall,
334:
321:
309:
297:
187:
154:
3511:
3461:
3460:
3459:
3452:
3296:
3159:Acceleron Pharma
3118:Rochelle Lazarus
3108:William Harrison
3077:
3070:
3063:
3054:
2799:PCSK9 inhibitors
2649:
2639:
2430:
2325:
2300:
2293:
2286:
2277:
2270:
2269:
2263:
2255:
2226:
2220:
2219:
2217:
2215:
2196:
2190:
2189:
2183:
2175:
2150:(275): 275ra23.
2139:
2133:
2132:
2130:
2128:
2109:
2103:
2102:
2096:
2088:
2078:
2046:
2040:
2039:
2002:
1996:
1995:
1985:
1970:Eur J Intern Med
1961:
1955:
1954:
1944:
1920:
1914:
1913:
1908:. Archived from
1902:
1896:
1895:
1889:
1881:
1871:
1847:
1841:
1840:
1834:
1826:
1797:
1791:
1790:
1780:
1748:
1742:
1741:
1735:
1727:
1717:
1707:
1683:
1677:
1676:
1666:
1656:
1632:
1623:
1622:
1594:
1588:
1587:
1585:
1583:
1569:
1563:
1562:
1560:
1558:
1539:
1533:
1532:
1530:
1528:
1509:
1503:
1502:
1500:
1498:
1483:
1477:
1476:
1474:
1472:
1453:
1447:
1446:
1444:
1442:
1423:
1417:
1416:
1414:
1412:
1393:
1387:
1386:
1369:
1356:
1355:
1353:
1351:
1332:
1313:
1312:
1310:
1308:
1289:
1268:
1267:
1265:
1263:
1253:nctr-crs.fda.gov
1245:
1239:
1238:
1236:
1234:
1219:
1195:Child-Pugh score
1158:Pharmacokinetics
1144:aminopeptidase N
1059:hypersensitivity
950:in people with:
914:
820:
819:
750:
730:
715:
712:
706:
700:
694:
677:
666:
655:
654:
640:
638:
623:
603:
583:
563:
543:
523:
503:
483:
473:
472:
458:
394:
339:
332:
329:
319:
316:
308:
305:
295:
292:
270:
260:
250:
240:
185:
182:
164:
153:
150:
137:
123:
94:
93:
90:
89:
86:
83:
78:
77:
74:
71:
68:
65:
62:
59:
56:
53:
33:
26:
24:
3519:
3518:
3514:
3513:
3512:
3510:
3509:
3508:
3469:
3468:
3467:
3457:
3455:
3447:
3445:
3440:
3385:
3365:
3306:Coppertone sign
3299:Schering-Plough
3294:
3183:
3174:Schering-Plough
3147:
3086:
3081:
3051:
3046:
3045:
3030:Clinical trials
3009:
2923:
2858:
2831:
2793:
2756:CETP inhibitors
2746:
2730:
2624:
2615:Dextrothyroxine
2600:
2577:
2544:
2495:
2415:
2355:
2314:
2304:
2274:
2273:
2256:
2228:
2227:
2223:
2213:
2211:
2198:
2197:
2193:
2176:
2141:
2140:
2136:
2126:
2124:
2111:
2110:
2106:
2089:
2048:
2047:
2043:
2004:
2003:
1999:
1963:
1962:
1958:
1922:
1921:
1917:
1904:
1903:
1899:
1882:
1849:
1848:
1844:
1827:
1803:Atherosclerosis
1799:
1798:
1794:
1750:
1749:
1745:
1728:
1685:
1684:
1680:
1634:
1633:
1626:
1596:
1595:
1591:
1581:
1579:
1571:
1570:
1566:
1556:
1554:
1541:
1540:
1536:
1526:
1524:
1511:
1510:
1506:
1496:
1494:
1485:
1484:
1480:
1470:
1468:
1455:
1454:
1450:
1440:
1438:
1425:
1424:
1420:
1410:
1408:
1403:. 1 June 2022.
1395:
1394:
1390:
1383:
1371:
1370:
1359:
1349:
1347:
1334:
1333:
1316:
1306:
1304:
1291:
1290:
1271:
1261:
1259:
1247:
1246:
1242:
1232:
1230:
1221:
1220:
1213:
1208:
1187:cytochrome P450
1179:bioavailability
1176:
1172:
1167:
1160:
1124:small intestine
1120:
1115:
1094:
1080:also regulates
1031:
1029:Adverse effects
1000:
921:
912:
815:
813:
808:
805:
800:
795:
794:
783:
780:
775:
774:
753:
728:
718:
709:
703:
697:
658:
634:
626:
606:
586:
566:
546:
526:
506:
486:
469:
461:
441:
438:
425:
424:
392:
368:Protein binding
358:Bioavailability
350:Pharmacokinetic
344:
337:
274:
200:
193:
174:
167:
80:
50:
46:
17:
12:
11:
5:
3517:
3515:
3507:
3506:
3501:
3496:
3491:
3486:
3481:
3471:
3470:
3466:
3465:
3442:
3441:
3439:
3438:
3437:
3436:
3429:
3422:
3415:
3401:
3393:
3391:
3387:
3386:
3384:
3383:
3377:
3375:
3371:
3370:
3367:
3366:
3364:
3363:
3361:Urofollitropin
3358:
3353:
3348:
3343:
3338:
3333:
3328:
3323:
3318:
3313:
3308:
3302:
3300:
3293:
3292:
3287:
3282:
3277:
3272:
3267:
3262:
3257:
3252:
3247:
3242:
3237:
3232:
3227:
3222:
3217:
3212:
3207:
3202:
3197:
3191:
3189:
3185:
3184:
3182:
3181:
3176:
3171:
3166:
3161:
3155:
3153:
3149:
3148:
3146:
3145:
3140:
3135:
3130:
3125:
3120:
3115:
3113:William Kelley
3110:
3105:
3103:Johnnetta Cole
3100:
3094:
3092:
3088:
3087:
3082:
3080:
3079:
3072:
3065:
3057:
3048:
3047:
3044:
3043:
3042:
3041:
3038:
3027:
3021:
3015:
3014:
3011:
3010:
3008:
3007:
3002:
2997:
2992:
2987:
2982:
2977:
2972:
2967:
2962:
2957:
2952:
2947:
2942:
2937:
2931:
2929:
2925:
2924:
2922:
2921:
2916:
2911:
2906:
2901:
2896:
2891:
2886:
2881:
2876:
2870:
2868:
2864:
2863:
2860:
2859:
2857:
2856:
2850:
2848:
2833:
2832:
2830:
2829:
2824:
2819:
2814:
2808:
2806:
2795:
2794:
2792:
2791:
2786:
2781:
2776:
2771:
2765:
2763:
2752:
2751:
2748:
2747:
2745:
2744:
2738:
2736:
2732:
2731:
2729:
2728:
2723:
2718:
2713:
2708:
2703:
2698:
2693:
2688:
2683:
2678:
2673:
2668:
2663:
2657:
2655:
2646:
2636:
2630:
2629:
2626:
2625:
2623:
2622:
2617:
2611:
2609:
2602:
2601:
2599:
2598:
2596:Bempedoic acid
2592:
2590:
2579:
2578:
2576:
2575:
2570:
2565:
2559:
2557:
2546:
2545:
2543:
2542:
2537:
2532:
2527:
2522:
2517:
2511:
2509:
2497:
2496:
2494:
2493:
2488:
2483:
2478:
2473:
2468:
2463:
2458:
2453:
2447:
2445:
2427:
2421:
2420:
2417:
2416:
2414:
2413:
2400:
2395:
2390:
2385:
2380:
2374:
2372:
2357:
2356:
2354:
2353:
2348:
2343:
2337:
2335:
2322:
2316:
2315:
2305:
2303:
2302:
2295:
2288:
2280:
2272:
2271:
2238:(1–2): 88–96.
2221:
2191:
2134:
2104:
2061:(2): 296–304.
2041:
2014:(4): 453–462.
1997:
1956:
1915:
1897:
1862:(6): 517–523.
1842:
1792:
1763:(8): 800–801.
1743:
1678:
1624:
1589:
1564:
1534:
1504:
1478:
1448:
1418:
1388:
1381:
1357:
1314:
1269:
1240:
1229:. 21 June 2022
1210:
1209:
1207:
1204:
1174:
1170:
1165:
1159:
1156:
1119:
1116:
1114:
1111:
1103:abdominal pain
1093:
1090:
1071:rhabdomyolysis
1045:and/or raised
1030:
1027:
999:
996:
985:lipoprotein(a)
981:
980:
978:sitosterolemia
974:
965:
959:
956:hyperlipidemia
920:
917:
886:liver problems
837:used to treat
822:
821:
810:
809:
807:
806:
803:
801:
798:
790:
789:
788:
785:
784:
782:
781:
778:
770:
769:
768:
765:
764:
761:
755:
754:
752:
751:
743:
741:
733:
732:
726:
720:
719:
716:
707:
701:
695:
690:
684:
683:
679:
678:
668:
660:
659:
657:
656:
643:
641:
628:
627:
625:
624:
616:
614:
608:
607:
605:
604:
596:
594:
588:
587:
585:
584:
576:
574:
568:
567:
565:
564:
556:
554:
548:
547:
545:
544:
536:
534:
528:
527:
525:
524:
516:
514:
508:
507:
505:
504:
496:
494:
488:
487:
485:
484:
476:
474:
463:
462:
460:
459:
451:
449:
443:
442:
440:
439:
428:
420:
419:
418:
415:
414:
410:
409:
406:
400:
399:
396:
387:
386:
380:
374:
373:
370:
364:
363:
360:
354:
353:
346:
345:
343:
342:
327:
314:
303:
289:
287:
281:
280:
276:
275:
273:
272:
229:
227:
221:
220:
215:
209:
208:
203:
201:administration
195:
194:
192:
191:
189:
179:
177:
169:
168:
166:
165:
147:
145:
139:
138:
131:
125:
124:
117:
107:
106:
103:
97:
96:
44:
40:
39:
35:
34:
15:
13:
10:
9:
6:
4:
3:
2:
3516:
3505:
3502:
3500:
3497:
3495:
3492:
3490:
3487:
3485:
3482:
3480:
3477:
3476:
3474:
3464:
3454:
3450:
3435:
3434:
3430:
3428:
3427:
3423:
3421:
3420:
3416:
3414:
3413:
3409:
3408:
3407:
3406:
3402:
3400:
3399:
3395:
3394:
3392:
3388:
3382:
3379:
3378:
3376:
3372:
3362:
3359:
3357:
3354:
3352:
3349:
3347:
3344:
3342:
3339:
3337:
3334:
3332:
3329:
3327:
3324:
3322:
3319:
3317:
3314:
3312:
3311:Desloratadine
3309:
3307:
3304:
3303:
3301:
3297:
3291:
3288:
3286:
3283:
3281:
3278:
3276:
3273:
3271:
3268:
3266:
3263:
3261:
3258:
3256:
3253:
3251:
3248:
3246:
3243:
3241:
3238:
3236:
3233:
3231:
3228:
3226:
3225:Fosaprepitant
3223:
3221:
3218:
3216:
3213:
3211:
3208:
3206:
3203:
3201:
3198:
3196:
3193:
3192:
3190:
3186:
3180:
3177:
3175:
3172:
3170:
3167:
3165:
3162:
3160:
3157:
3156:
3154:
3150:
3144:
3143:Peter Wendell
3141:
3139:
3138:Wendell Weeks
3136:
3134:
3131:
3129:
3126:
3124:
3121:
3119:
3116:
3114:
3111:
3109:
3106:
3104:
3101:
3099:
3098:Richard Clark
3096:
3095:
3093:
3089:
3085:
3078:
3073:
3071:
3066:
3064:
3059:
3058:
3055:
3039:
3037:
3034:
3033:
3031:
3028:
3025:
3022:
3020:
3017:
3016:
3012:
3006:
3003:
3001:
2998:
2996:
2993:
2991:
2988:
2986:
2983:
2981:
2978:
2976:
2973:
2971:
2968:
2966:
2963:
2961:
2958:
2956:
2953:
2951:
2948:
2946:
2943:
2941:
2938:
2936:
2933:
2932:
2930:
2926:
2920:
2917:
2915:
2912:
2910:
2907:
2905:
2902:
2900:
2897:
2895:
2892:
2890:
2887:
2885:
2882:
2880:
2877:
2875:
2872:
2871:
2869:
2865:
2855:
2852:
2851:
2849:
2846:
2842:
2838:
2834:
2828:
2825:
2823:
2820:
2818:
2815:
2813:
2810:
2809:
2807:
2804:
2800:
2796:
2790:
2787:
2785:
2782:
2780:
2777:
2775:
2772:
2770:
2767:
2766:
2764:
2761:
2757:
2753:
2743:
2740:
2739:
2737:
2733:
2727:
2724:
2722:
2719:
2717:
2714:
2712:
2709:
2707:
2704:
2702:
2699:
2697:
2694:
2692:
2689:
2687:
2684:
2682:
2679:
2677:
2674:
2672:
2669:
2667:
2664:
2662:
2659:
2658:
2656:
2654:
2650:
2647:
2644:
2643:PPAR agonists
2640:
2637:
2635:
2634:Blood vessels
2631:
2621:
2618:
2616:
2613:
2612:
2610:
2607:
2606:Thyromimetics
2603:
2597:
2594:
2593:
2591:
2588:
2584:
2580:
2574:
2571:
2569:
2566:
2564:
2561:
2560:
2558:
2555:
2551:
2547:
2541:
2538:
2536:
2533:
2531:
2528:
2526:
2523:
2521:
2518:
2516:
2513:
2512:
2510:
2507:
2503:
2498:
2492:
2489:
2487:
2484:
2482:
2479:
2477:
2474:
2472:
2469:
2467:
2464:
2462:
2459:
2457:
2454:
2452:
2449:
2448:
2446:
2443:
2439:
2435:
2431:
2428:
2426:
2422:
2412:
2408:
2404:
2403:Soluble fiber
2401:
2399:
2396:
2394:
2393:Colestyramine
2391:
2389:
2386:
2384:
2381:
2379:
2376:
2375:
2373:
2370:
2366:
2362:
2358:
2352:
2349:
2347:
2344:
2342:
2339:
2338:
2336:
2334:
2330:
2326:
2323:
2321:
2317:
2312:
2308:
2301:
2296:
2294:
2289:
2287:
2282:
2281:
2278:
2267:
2261:
2253:
2249:
2245:
2241:
2237:
2233:
2225:
2222:
2209:
2205:
2201:
2195:
2192:
2187:
2181:
2173:
2169:
2165:
2161:
2157:
2153:
2149:
2145:
2138:
2135:
2122:
2118:
2114:
2108:
2105:
2100:
2094:
2086:
2082:
2077:
2072:
2068:
2064:
2060:
2056:
2052:
2045:
2042:
2037:
2033:
2029:
2025:
2021:
2017:
2013:
2009:
2001:
1998:
1993:
1989:
1984:
1979:
1975:
1971:
1967:
1960:
1957:
1952:
1948:
1943:
1938:
1934:
1930:
1926:
1919:
1916:
1911:
1907:
1901:
1898:
1893:
1887:
1879:
1875:
1870:
1865:
1861:
1857:
1853:
1846:
1843:
1838:
1832:
1824:
1820:
1816:
1812:
1808:
1804:
1796:
1793:
1788:
1784:
1779:
1774:
1770:
1766:
1762:
1758:
1754:
1747:
1744:
1739:
1733:
1725:
1721:
1716:
1715:11573/1150638
1711:
1706:
1701:
1697:
1693:
1689:
1682:
1679:
1674:
1670:
1665:
1660:
1655:
1650:
1646:
1642:
1638:
1631:
1629:
1625:
1620:
1616:
1612:
1608:
1604:
1600:
1593:
1590:
1578:
1574:
1568:
1565:
1552:
1548:
1544:
1538:
1535:
1522:
1518:
1514:
1508:
1505:
1492:
1488:
1482:
1479:
1466:
1462:
1458:
1452:
1449:
1436:
1432:
1428:
1422:
1419:
1406:
1402:
1398:
1392:
1389:
1384:
1382:9780857113382
1378:
1374:
1368:
1366:
1364:
1362:
1358:
1345:
1341:
1337:
1331:
1329:
1327:
1325:
1323:
1321:
1319:
1315:
1302:
1298:
1294:
1288:
1286:
1284:
1282:
1280:
1278:
1276:
1274:
1270:
1258:
1254:
1250:
1244:
1241:
1228:
1224:
1218:
1216:
1212:
1205:
1203:
1201:
1196:
1192:
1188:
1182:
1180:
1168:
1157:
1155:
1153:
1149:
1145:
1141:
1137:
1134:
1130:
1125:
1117:
1112:
1110:
1108:
1104:
1100:
1091:
1089:
1087:
1083:
1079:
1074:
1072:
1068:
1064:
1060:
1056:
1052:
1048:
1044:
1040:
1036:
1028:
1026:
1024:
1020:
1016:
1011:
1009:
1005:
997:
995:
993:
988:
986:
979:
975:
972:
969:
966:
964:
960:
957:
953:
952:
951:
949:
944:
940:
938:
934:
930:
926:
918:
916:
910:
905:
903:
899:
895:
894:breastfeeding
891:
887:
883:
879:
874:
872:
868:
864:
860:
857:combinations
856:
852:
848:
844:
840:
836:
832:
828:
818:
811:
802:
797:
793:
786:
777:
773:
766:
762:
760:
759:Melting point
756:
749:
745:
744:
742:
739:
734:
727:
725:
721:
691:
689:
685:
680:
676:
672:
669:
667:
665:ECHA InfoCard
661:
653:
649:
648:DTXSID1044223
645:
644:
642:
633:
629:
622:
618:
617:
615:
613:
609:
602:
598:
597:
595:
593:
589:
582:
578:
577:
575:
573:
569:
562:
558:
557:
555:
553:
549:
542:
538:
537:
535:
533:
529:
522:
518:
517:
515:
513:
509:
502:
498:
497:
495:
493:
489:
482:
478:
477:
475:
468:
464:
457:
453:
452:
450:
448:
444:
436:
432:
427:
423:
416:
411:
407:
405:
401:
397:
395:
388:
385:
381:
379:
375:
371:
369:
365:
361:
359:
355:
351:
347:
340:
328:
325:
315:
313:
304:
301:
291:
290:
288:
286:
282:
277:
269:
264:
259:
254:
249:
244:
239:
234:
231:
230:
228:
226:
222:
219:
216:
214:
210:
207:
204:
202:
196:
190:
181:
180:
178:
176:
170:
163:
158:
149:
148:
146:
144:
140:
136:
132:
130:
126:
122:
118:
116:
112:
108:
105:Zetia, others
104:
102:
98:
92:
45:
43:Pronunciation
41:
38:Clinical data
36:
32:
27:
19:
3484:Beta-lactams
3432:
3425:
3418:
3411:
3403:
3396:
3390:Publications
3351:Temozolomide
3315:
3250:Omarigliptin
3209:
3152:Subsidiaries
3133:Samuel Thier
3128:Anne Tatlock
3123:Thomas Shenk
3005:Volanesorsen
2867:Combinations
2839:inhibitors (
2681:Clinofibrate
2676:Ciprofibrate
2585:inhibitors (
2552:inhibitors (
2535:Nicofuranose
2486:Rosuvastatin
2476:Pitavastatin
2456:Cerivastatin
2451:Atorvastatin
2340:
2260:cite journal
2235:
2231:
2224:
2212:. Retrieved
2203:
2194:
2180:cite journal
2147:
2143:
2137:
2125:. Retrieved
2117:Medline Plus
2116:
2107:
2093:cite journal
2058:
2054:
2044:
2011:
2007:
2000:
1973:
1969:
1959:
1935:(1): 29–60.
1932:
1928:
1918:
1910:the original
1900:
1886:cite journal
1859:
1855:
1845:
1831:cite journal
1806:
1802:
1795:
1760:
1756:
1746:
1732:cite journal
1695:
1691:
1681:
1644:
1640:
1602:
1598:
1592:
1580:. Retrieved
1576:
1567:
1555:. Retrieved
1546:
1537:
1525:. Retrieved
1516:
1507:
1495:. Retrieved
1490:
1481:
1469:. Retrieved
1460:
1451:
1439:. Retrieved
1430:
1421:
1409:. Retrieved
1400:
1391:
1372:
1348:. Retrieved
1339:
1305:. Retrieved
1296:
1260:. Retrieved
1252:
1243:
1231:. Retrieved
1226:
1183:
1161:
1142:; it blocks
1121:
1113:Pharmacology
1099:loose stools
1095:
1075:
1032:
1012:
1001:
989:
982:
945:
941:
937:heart attack
922:
919:Medical uses
906:
875:
830:
826:
825:
814:
434:
430:
398:19 h to 30 h
391:Elimination
285:Legal status
279:Legal status
143:License data
18:
3326:Famciclovir
3280:Sitagliptin
3275:Simvastatin
3260:Rizatriptan
3255:Raltegravir
3245:Montelukast
3220:Finasteride
3195:Alendronate
3026:from market
2985:Policosanol
2960:Lapaquistat
2940:Azacosterol
2817:Bococizumab
2789:Torcetrapib
2784:Obicetrapib
2779:Evacetrapib
2774:Dalcetrapib
2769:Anacetrapib
2721:Ronifibrate
2716:Pemafibrate
2706:Gemfibrozil
2701:Fenofibrate
2666:Bezafibrate
2573:Mitratapide
2563:Dirlotapide
2491:Simvastatin
2481:Pravastatin
2461:Fluvastatin
2411:glucomannan
2378:Colesevelam
2113:"Ezetimibe"
1809:(1): 3–46.
1647:: 415–427.
1605:: 247–252.
1140:hepatocytes
1039:steatorrhea
1023:fenofibrate
1021:other than
1015:ciclosporin
1008:anaphylaxis
976:Homozygous
963:fenofibrate
882:anaphylaxis
731: g·mol
671:100.207.996
601:CHEBI:49040
456:163222-33-1
413:Identifiers
129:MedlinePlus
101:Trade names
3473:Categories
3433:Geriatrics
3426:Veterinary
3374:Facilities
3341:Mometasone
3336:Loratadine
3331:Infliximab
3290:Vorinostat
3285:Vericiguat
3240:Lovastatin
3200:Aprepitant
3179:Viralytics
3000:Triparanol
2975:Mipomersen
2955:Darapladib
2950:Benfluorex
2945:Azalanstat
2854:Evinacumab
2827:Inclisiran
2822:Evolocumab
2812:Alirocumab
2726:Simfibrate
2696:Etofibrate
2691:Clofibride
2686:Clofibrate
2620:Resmetirom
2568:Lomitapide
2530:Niceritrol
2471:Mevastatin
2466:Lovastatin
2388:Colestipol
2383:Colestilan
2346:Hyzetimibe
1262:22 October
1206:References
1152:annexin A2
1148:caveolin 1
1136:epithelial
1063:angioedema
1004:angioedema
968:Homozygous
902:intestines
855:fixed-dose
835:medication
736:3D model (
724:Molar mass
621:ChEMBL1138
561:EOR26LQQ24
532:ChemSpider
492:IUPHAR/BPS
447:CAS Number
422:IUPAC name
378:Metabolism
362:35% to 65%
213:Drug class
3316:Ezetimibe
3265:Rofecoxib
3230:Indinavir
3210:Ezetimibe
3205:Ertapenem
3036:Phase III
3024:Withdrawn
2711:Nafenopin
2398:Colextran
2351:SCH-48461
2341:Ezetimibe
2036:207489460
1582:30 August
1557:30 August
1517:Drugs.com
1497:13 August
1471:13 August
1441:13 August
1411:13 August
1340:Drugs.com
1307:13 August
1088:therapy.
1082:vitamin K
1017:and with
890:pregnancy
827:Ezetimibe
404:Excretion
393:half-life
199:Routes of
173:Pregnancy
162:Ezetimibe
121:Monograph
115:Drugs.com
23:Ezetimibe
3463:Medicine
3356:Tibolone
3235:Losartan
3188:Products
2995:Tiadenol
2990:Probucol
2970:Meglutol
2742:GW501516
2653:Fibrates
2515:Acipimox
2407:psyllium
2405:such as
2320:GI tract
2252:17442643
2208:Archived
2164:25696002
2127:21 March
2121:Archived
2085:29551054
2028:29396832
1992:38336550
1951:24528685
1878:23665881
1823:21882396
1787:30715135
1724:26039521
1673:22910633
1619:26301648
1577:ClinCalc
1551:Archived
1547:ClinCalc
1527:13 April
1521:Archived
1491:DailyMed
1465:Archived
1461:DailyMed
1435:Archived
1431:DailyMed
1405:Archived
1401:DailyMed
1350:13 April
1344:Archived
1301:Archived
1297:DailyMed
1233:20 April
1092:Overdose
1086:warfarin
1067:myopathy
1019:fibrates
954:Primary
851:by mouth
817:(verify)
512:DrugBank
225:ATC code
206:By mouth
188: B3
175:category
157:DailyMed
3499:Phenols
2837:ANGPTL3
2434:Statins
2230:bile".
2172:5951911
2076:6406097
1778:6679800
1664:3402055
1107:fatigue
1043:myalgia
1033:Common
900:in the
729:409.433
688:Formula
521:DB00973
467:PubChem
372:>90%
341:Rx-only
338:WARNING
310::
265: (
263:C10BA02
255: (
253:C10BA06
245: (
243:C10BA05
241:)
235: (
233:C10AX09
159::
135:a603015
3449:Portal
3419:Manual
3019:WHO-EM
2735:Others
2608:(VLDL)
2525:Niacin
2365:resins
2333:NPC1L1
2250:
2214:29 May
2170:
2162:
2083:
2073:
2034:
2026:
1990:
1949:
1876:
1821:
1785:
1775:
1722:
1671:
1661:
1617:
1379:
1105:, and
1078:NPC1L1
1076:Since
1006:, and
948:lipids
933:stroke
913:
869:, and
847:statin
772:SMILES
612:ChEMBL
581:D01966
541:132493
481:150311
335:
322:
312:℞-only
298:
155:
95:
3412:Index
2928:Other
2645:(LDL)
2425:Liver
2168:S2CID
2032:S2CID
2008:Drugs
1065:) or
831:Zetia
792:InChI
738:JSmol
592:ChEBI
384:liver
2554:VLDL
2550:MTTP
2504:and
2409:and
2266:link
2248:PMID
2216:2018
2186:link
2160:PMID
2129:2018
2099:link
2081:PMID
2024:PMID
1988:PMID
1947:PMID
1892:link
1874:PMID
1837:link
1819:PMID
1783:PMID
1757:JAMA
1738:link
1720:PMID
1669:PMID
1615:PMID
1584:2024
1559:2024
1529:2019
1499:2022
1473:2022
1443:2022
1413:2022
1377:ISBN
1352:2019
1309:2022
1264:2023
1235:2024
931:and
892:and
572:KEGG
552:UNII
501:6816
352:data
111:AHFS
2845:HDL
2841:LDL
2803:LDL
2760:HDL
2587:LDL
2506:LDL
2502:HDL
2442:LDL
2369:LDL
2311:C10
2240:doi
2236:853
2152:doi
2071:PMC
2063:doi
2016:doi
1978:doi
1974:124
1937:doi
1864:doi
1811:doi
1807:217
1773:PMC
1765:doi
1761:321
1710:hdl
1700:doi
1696:372
1659:PMC
1649:doi
1607:doi
1603:201
1257:FDA
1200:AUC
1175:max
1171:max
1166:max
1055:AST
1051:ALT
637:EPA
471:CID
324:POM
268:WHO
258:WHO
248:WHO
238:WHO
3475::
3032::
2440:,
2331:,
2262:}}
2258:{{
2246:.
2234:.
2202:.
2182:}}
2178:{{
2166:.
2158:.
2146:.
2115:.
2095:}}
2091:{{
2079:.
2069:.
2059:34
2057:.
2053:.
2030:.
2022:.
2012:78
2010:.
1986:.
1972:.
1968:.
1945:.
1931:.
1927:.
1888:}}
1884:{{
1872:.
1860:20
1858:.
1854:.
1833:}}
1829:{{
1817:.
1805:.
1781:.
1771:.
1759:.
1755:.
1734:}}
1730:{{
1718:.
1708:.
1694:.
1690:.
1667:.
1657:.
1643:.
1639:.
1627:^
1613:.
1601:.
1575:.
1549:.
1545:.
1519:.
1515:.
1489:.
1459:.
1429:.
1399:.
1360:^
1338:.
1317:^
1295:.
1272:^
1255:.
1251:.
1225:.
1214:^
1109:.
1101:,
1025:.
904:.
884:,
873:.
865:,
861:,
702:21
696:24
433:,4
429:(3
331:US
318:UK
307:CA
300:S4
294:AU
261:)
251:)
184:AU
152:US
85:aɪ
79:,-
3451::
3076:e
3069:t
3062:v
2847:)
2843:/
2805:)
2801:(
2762:)
2758:(
2589:)
2556:)
2508:)
2444:)
2436:(
2371:)
2367:(
2363:/
2313:)
2309:(
2299:e
2292:t
2285:v
2268:)
2254:.
2242::
2218:.
2188:)
2174:.
2154::
2148:7
2131:.
2101:)
2087:.
2065::
2038:.
2018::
1994:.
1980::
1953:.
1939::
1933:8
1894:)
1880:.
1866::
1839:)
1825:.
1813::
1789:.
1767::
1740:)
1726:.
1712::
1702::
1675:.
1651::
1645:8
1621:.
1609::
1586:.
1561:.
1531:.
1501:.
1475:.
1445:.
1415:.
1385:.
1354:.
1311:.
1266:.
1237:.
1164:C
1150:–
1053:/
1049:(
740:)
717:3
714:O
711:N
708:2
705:F
699:H
693:C
639:)
635:(
435:S
431:R
333::
320::
296::
271:)
186::
113:/
91:/
88:b
82:m
76:b
73:ɪ
70:m
67:ɪ
64:t
61:ɛ
58:z
55:ˈ
52:ɛ
49:/
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.