270:
form the amorphous phase. Mannitol is the first pharmaceutical substance featuring polyamorphism. In addition to the regular amorphous phase, a second amorphous phase can be prepared at room temperature and pressure. This new phase has substantially lower energy, lower density and higher glass transition temperature. Since mannitol is widely used in pharmaceutical tablet formulations, mannitol polyamorphism offers a powerful tool to engineer the property and behavior of tablets.
64:
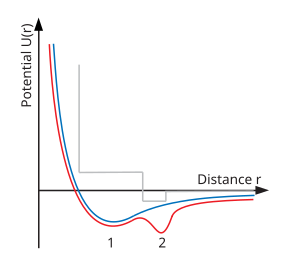
71:, which exhibits the ordinary liquid–gas critical point. The red line is a double well type potential, which is proposed for polyamorphous systems. The grey line, is a representative of the soft core square well potentials, which in atomisitc simulations exhibit liquid–liquid transitions and a second critical point. The numbers 1 and 2 correspond to the 1st and second minima in the potentials.
31:
210:
high-density amorphous ice is warmed up to 165 K not at low pressures but keeping the 1.6 GPa compression, and then cooled back to 77 K, then another amorphous ice is produced, which has even higher density of 1.25 g/cm at 1 bar. All those amorphous forms have very different vibrational lattice spectra and intermolecular distances. A similar abrupt liquid-amorphous
142:
The stable liquid state unlike most glasses and amorphous solids, is a thermodynamically stable equilibrium state. Thus new liquid–liquid or fluid-fluid transitions in the stable liquid (or fluid) states are more easily analysed than transitions in amorphous solids where arguments are complicated by
138:
Polyamorphism may apply to all amorphous states, i.e. glasses, other amorphous solids, supercooled liquids, ordinary liquids or fluids. A liquid–liquid transition however, is one that occurs only in the liquid state (red line in the phase diagram, top right). In this article liquid–liquid transitions
230:
melts are another system reported to exhibit polyamorphism. Observation of a liquid–liquid phase transition in the supercooled liquid has been reported. Though this is disputed in the literature. Polyamorphism has also been reported in Yttria-Alumina glasses. Yttria-Alumina melts quenched from about
209:
temperature (77 K) converts them to the high-density amorphous ice. Upon releasing the pressure, this phase is stable and has density of 1.17 g/cm at 77 K and 1 bar. Consequent warming to 127 K at ambient pressure transforms this phase to a low-density amorphous ice (0.94 g/cm at 1 bar). Yet, if the
160:
One physical explanation for polyamorphism is the existence of a double well inter-atomic pair potential (see lower right diagram). It is well known that the ordinary liquid–gas critical point appears when the inter-atomic pair potential contains a minimum. At lower energies (temperatures) particles
269:
Polyamorphism is also an important area in pharmaceutical science. The amorphous form of a drug typically has much better aqueous solubility (compared to the analogous crystalline form) but the actual local structure in an amorphous pharmaceutical can be different, depending on the method used to
214:
is predicted in liquid silicon when cooled under high pressures. This observation is based on first principles molecular dynamics computer simulations, and might be expected intuitively since tetrahedral amorphous carbon, silicon, and germanium are known to be structurally analogous to water.
50:
of crystalline materials, where different stable crystalline states (solid 1, 2 in diagram) of the same substance can exist (e.g. diamond and graphite are two polymorphs of carbon). Like the ordinary liquid–gas transition, the liquid–liquid transition is expected to end in a
151:
Liquid–liquid transitions were originally considered by
Rapoport in 1967 in order to explain high pressure melting curve maxima of some liquid metals. Rapoport's theory requires the existence of a melting curve maximum in polyamorphic systems.
55:. At temperatures beyond these critical points there is a continuous range of fluid states, i.e. the distinction between liquids and gasses is lost. If crystallisation is avoided the liquid–liquid transition can be extended into the metastable
239:). The two phases have the same average composition but different density, molecular structure and hardness. However whether the second phase is glassy or crystalline is also debated. Continuous changes in density were observed upon cooling
139:
are defined as transitions between two liquids of the same chemical substance. Elsewhere the term liquid–liquid transition may also refer to the more common transitions between liquid mixtures of different chemical composition.
161:
trapped in this minimum condense into the liquid state. At higher temperatures however, these particles can escape the well and the sharp definition between liquid and gas is lost.
620:; Salzmann, Christoph; Kohl, Ingrid; Mayer, Erwin; et al. (2001). "A second distinct structural "state" of high-density amorphous ice at 77 K and 1 bar".
231:
1900 °C at a rate ~400 °C/s, can form glasses containing a second co-existing phase. This happens for certain Y/Al ratios (about 20–40 mol% Y
737:
Benmore, C. J.; Hart, R.; Mei, Q.; Price, D.; et al. (2004). "Intermediate range chemical ordering in amorphous and liquid water, Si, and Ge".
165:
has shown that addition of a second well produces an additional transition between two different liquids (or fluids) with a second critical point.
371:
Franzese, G.; Malescio, G; Skibinsky, A; Buldyrev, SV; et al. (2001). "Generic mechanism for generating a liquid–liquid phase transition".
586:
Schober, H; Koza, M.; Tölle, A.; Fujara, F.; et al. (1997). "Amorphous polymorphism in ice investigated by inelastic neutron scattering".
481:
Mishima, O.; Calvert, L. D.; Whalley, E. (1985). "An apparently 1st-order transition between two amorphous phases of ice induced by pressure".
1114:
678:
247:. Although continuous density changes do not constitute a first order transition, they may be indicative of an underlying abrupt transition.
1130:
Kurita, Rei; Tanaka, Hajime (2005-07-13). "On the abundance and general nature of the liquid–liquid phase transition in molecular systems".
931:
Skinner, LB; Barnes, AC; Salmon, PS; Crichton, WA (2008). "Phase separation, crystallization and polyamorphism in the Y2O3-Al2O3 system".
90:
materials. Many amorphous substances can exist with different amorphous characteristics (e.g. polymers). However, polyamorphism requires
118:). Different local structures can produce amorphous phases of the same chemical composition with different physical properties such as
98:
between them. When such a transition occurs between two stable liquid states, a polyamorphic transition may also be referred to as a
126:
is one important example (see also examples below). Several of these transitions (including water) are expected to end in a second
38:, including an illustration of the liquid–liquid transition line proposed for several polyamorphous materials. This liquid–liquid
300:
295:
115:
83:
47:
127:
122:. In several cases sharp transitions have been observed between two different density amorphous states of the same material.
52:
1173:
Syme, Christopher D.; Mosses, Joanna; González-Jiménez, Mario; Shebanova, Olga; Walton, Finlay; Wynne, Klaas (2017).
1041:
Ha, Alice; Cohen, Itai; Zhao, Xiaolin; Lee, Michelle; et al. (1996). "Supercooled
Liquids and Polyamorphism†".
1351:
982:
Kurita, R. (2004-10-29). "Critical-Like
Phenomena Associated with Liquid-Liquid Transition in a Molecular Liquid".
305:
114:
periodic atomic ordering, there is still significant and varied local structure at inter-atomic length scales (see
1361:
1356:
888:
Aasland, S.; McMillan, P. F. (1994). "Density-driven liquid–liquid phase separation in the system AI2O3–Y2O3".
772:
Greaves, G; Wilding, MC; Fearn, S; Langstaff, D; Kargl, F; Cox, S; Van, QV; Majérus, O; et al. (2008).
68:
773:
190:
1304:
1251:
1186:
991:
940:
897:
846:
788:
746:
703:
629:
591:
537:
490:
390:
334:
325:
Mishima, O.; Mishima, Osamu (1998). "The relationship between liquid, supercooled and glassy water".
256:
182:
162:
1155:
1085:
1023:
964:
913:
870:
812:
694:
Morishita, T. (2004). "High
Density Amorphous Form and Polyamorphic Transformations of Silicon".
645:
506:
463:
414:
380:
350:
56:
831:
774:"Detection of First-Order Liquid/Liquid Phase Transitions in Yttrium Oxide-Aluminum Oxide Melts"
1328:
1320:
1277:
1269:
1240:"Possible existence of two amorphous phases of d-mannitol related by a first-order transition"
1220:
1202:
1147:
1110:
1015:
1007:
956:
862:
804:
719:
674:
664:
455:
406:
244:
1104:
1312:
1259:
1210:
1194:
1139:
1077:
1050:
999:
948:
905:
854:
796:
754:
711:
637:
599:
545:
498:
445:
398:
342:
211:
111:
95:
39:
617:
290:
240:
206:
952:
46:
transition between low and high density liquids (labelled 1 and 2). This is analogous to
1308:
1255:
1190:
995:
944:
901:
850:
792:
750:
707:
633:
595:
541:
494:
394:
338:
1215:
1174:
1143:
603:
1345:
202:
123:
43:
35:
1159:
1089:
1027:
968:
816:
649:
565:
467:
917:
874:
858:
510:
418:
354:
715:
63:
1081:
758:
450:
433:
178:
1324:
1273:
1206:
1151:
1068:
Poole, P. H. (1997). "Polymorphic Phase
Transitions in Liquids and Glasses".
1011:
173:
Polyamorphism has been experimentally observed or theoretically suggested in
1003:
800:
260:
79:
1332:
1281:
1224:
1019:
960:
866:
808:
723:
459:
410:
30:
255:
Polyamorphism has also been observed in organic compounds, such as liquid
17:
670:
385:
186:
27:
Ability of a substance to exist in more than one distinct amorphous state
227:
205:. Pressurizing conventional hexagonal ice crystals to about 1.6 GPa at
174:
119:
87:
1316:
1264:
1239:
1198:
1054:
830:
Barnes, AC; Skinner, LB; Salmon, PS; Bytchkov, A; et al. (2009).
550:
525:
909:
641:
502:
402:
285:
223:
67:
Schematic of interatomic pair potentials. The blue line is a typical
346:
280:
1238:
Zhu, Men; Wang, Jun-Qiang; Perepezko, John H.; Yu, Lian (2015).
143:
the non-equilibrium, non-ergodic nature of the amorphous state.
78:
is the ability of a substance to exist in several different
94:
amorphous states with a clear, discontinuous (first-order)
1295:
Zhu, Men; Yu, Lian (2017). "Polyamorphism of D-mannitol".
1175:"Frustration of crystallisation by a liquid–crystal phase"
434:"Polyamorphism: a pharmaceutical science perspective"
832:"Liquid/Liquid Phase Transitions in Yttria-Alumina"
526:"Model for melting curve maxima at high pressure"
432:Hancock, BC; Shalaev, EY; Shamblin, SL (2002).
259:at temperatures between 210 K and 226 K and
8:
110:Even though amorphous materials exhibit no
1263:
1214:
549:
449:
384:
266:at temperatures between 120 K and 140 K.
201:The most famous case of polyamorphism is
438:The Journal of Pharmacy and Pharmacology
62:
29:
317:
366:
364:
82:modifications. It is analogous to the
1106:Disordered materials: an introduction
7:
1132:Journal of Physics: Condensed Matter
622:Physical Chemistry Chemical Physics
25:
1043:The Journal of Physical Chemistry
301:polymorphism (materials science)
296:structure of liquids and glasses
116:structure of liquids and glasses
1297:The Journal of Chemical Physics
1244:The Journal of Chemical Physics
666:Structural chemistry of glasses
566:"Anomalous properties of water"
953:10.1088/0953-8984/20/20/205103
859:10.1103/PhysRevLett.103.225702
197:Water and structural analogues
100:liquid–liquid phase transition
1:
716:10.1103/PhysRevLett.93.055503
604:10.1016/S0921-4526(97)00749-7
1082:10.1126/science.275.5298.322
69:Lennard-Jones type potential
53:liquid-liquid critical point
1144:10.1088/0953-8984/17/27/L01
588:Physica B: Condensed Matter
1378:
759:10.1103/PhysRevB.72.132201
451:10.1211/002235702320266343
306:Pair distribution function
933:J. Phys.: Condens. Matter
219:Oxide liquids and glasses
169:Examples of polyamorphism
134:Liquid–liquid transitions
1109:. Springer. p. 65.
42:would be a first order,
1004:10.1126/science.1103073
839:Physical Review Letters
801:10.1126/science.1160766
1103:Paolo M. Ossi (2006).
156:Double well potentials
146:
72:
60:
524:Rapoport, E. (1967).
193:-forming substances.
66:
34:Pressure–temperature
33:
590:. 241–243: 897–902.
189:, and in some other
1309:2017JChPh.146x4503Z
1256:2015JChPh.142x4504Z
1191:2017NatSR...742439S
996:2004Sci...306..845K
945:2008JPCM...20t5103S
902:1994Natur.369..633A
851:2009PhRvL.103v5702B
793:2008Sci...322..566G
751:2005PhRvB..72m2201B
708:2004PhRvL..93e5503M
634:2001PCCP....3.5355L
596:1997PhyB..241..897S
542:1967JChPh..46.2891R
536:(2891): 2891–2895.
495:1985Natur.314...76M
395:2001Natur.409..692F
339:1998Natur.396..329M
257:triphenyl phosphite
183:triphenyl phosphate
163:Molecular modelling
1179:Scientific Reports
745:(132201): 132201.
663:K. J. Rao (2002).
73:
61:
57:supercooled liquid
1352:Phase transitions
1317:10.1063/1.4989961
1265:10.1063/1.4922543
1199:10.1038/srep42439
1138:(27): L293–L302.
1116:978-3-540-29609-6
1076:(5298): 322–323.
1055:10.1021/jp9530820
990:(5697): 845–848.
680:978-0-08-043958-7
551:10.1063/1.1841150
251:Organic materials
245:germanium dioxide
191:molecular network
147:Rapoport's theory
16:(Redirected from
1369:
1362:Amorphous solids
1357:Phases of matter
1337:
1336:
1292:
1286:
1285:
1267:
1235:
1229:
1228:
1218:
1170:
1164:
1163:
1127:
1121:
1120:
1100:
1094:
1093:
1065:
1059:
1058:
1038:
1032:
1031:
979:
973:
972:
928:
922:
921:
910:10.1038/369633a0
885:
879:
878:
836:
827:
821:
820:
787:(5901): 566–70.
778:
769:
763:
762:
734:
728:
727:
702:(55503): 55503.
691:
685:
684:
660:
654:
653:
642:10.1039/b108676f
618:Loerting, Thomas
614:
608:
607:
583:
577:
576:
574:
572:
562:
556:
555:
553:
521:
515:
514:
503:10.1038/314076a0
478:
472:
471:
453:
429:
423:
422:
403:10.1038/35055514
388:
386:cond-mat/0102029
368:
359:
358:
322:
212:phase transition
96:phase transition
40:phase transition
21:
1377:
1376:
1372:
1371:
1370:
1368:
1367:
1366:
1342:
1341:
1340:
1294:
1293:
1289:
1237:
1236:
1232:
1172:
1171:
1167:
1129:
1128:
1124:
1117:
1102:
1101:
1097:
1067:
1066:
1062:
1040:
1039:
1035:
981:
980:
976:
930:
929:
925:
887:
886:
882:
834:
829:
828:
824:
776:
771:
770:
766:
736:
735:
731:
696:Phys. Rev. Lett
693:
692:
688:
681:
673:. p. 120.
662:
661:
657:
616:
615:
611:
585:
584:
580:
570:
568:
564:
563:
559:
523:
522:
518:
480:
479:
475:
431:
430:
426:
379:(6821): 692–5.
370:
369:
362:
324:
323:
319:
315:
310:
291:Amorphous solid
276:
253:
241:silicon dioxide
238:
234:
221:
207:liquid nitrogen
199:
171:
158:
149:
136:
108:
28:
23:
22:
15:
12:
11:
5:
1375:
1373:
1365:
1364:
1359:
1354:
1344:
1343:
1339:
1338:
1303:(24): 244503.
1287:
1250:(24): 244504.
1230:
1165:
1122:
1115:
1095:
1060:
1033:
974:
939:(20): 205103.
923:
880:
845:(22): 225702.
822:
764:
729:
686:
679:
655:
609:
578:
557:
516:
473:
424:
360:
316:
314:
311:
309:
308:
303:
298:
293:
288:
283:
277:
275:
272:
252:
249:
236:
232:
220:
217:
198:
195:
170:
167:
157:
154:
148:
145:
135:
132:
128:critical point
107:
104:
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
1374:
1363:
1360:
1358:
1355:
1353:
1350:
1349:
1347:
1334:
1330:
1326:
1322:
1318:
1314:
1310:
1306:
1302:
1298:
1291:
1288:
1283:
1279:
1275:
1271:
1266:
1261:
1257:
1253:
1249:
1245:
1241:
1234:
1231:
1226:
1222:
1217:
1212:
1208:
1204:
1200:
1196:
1192:
1188:
1184:
1180:
1176:
1169:
1166:
1161:
1157:
1153:
1149:
1145:
1141:
1137:
1133:
1126:
1123:
1118:
1112:
1108:
1107:
1099:
1096:
1091:
1087:
1083:
1079:
1075:
1071:
1064:
1061:
1056:
1052:
1048:
1044:
1037:
1034:
1029:
1025:
1021:
1017:
1013:
1009:
1005:
1001:
997:
993:
989:
985:
978:
975:
970:
966:
962:
958:
954:
950:
946:
942:
938:
934:
927:
924:
919:
915:
911:
907:
903:
899:
896:(6482): 633.
895:
891:
884:
881:
876:
872:
868:
864:
860:
856:
852:
848:
844:
840:
833:
826:
823:
818:
814:
810:
806:
802:
798:
794:
790:
786:
782:
775:
768:
765:
760:
756:
752:
748:
744:
740:
733:
730:
725:
721:
717:
713:
709:
705:
701:
697:
690:
687:
682:
676:
672:
668:
667:
659:
656:
651:
647:
643:
639:
635:
631:
627:
623:
619:
613:
610:
605:
601:
597:
593:
589:
582:
579:
567:
561:
558:
552:
547:
543:
539:
535:
531:
530:J. Chem. Phys
527:
520:
517:
512:
508:
504:
500:
496:
492:
488:
484:
477:
474:
469:
465:
461:
457:
452:
447:
444:(8): 1151–2.
443:
439:
435:
428:
425:
420:
416:
412:
408:
404:
400:
396:
392:
387:
382:
378:
374:
367:
365:
361:
356:
352:
348:
347:10.1038/24540
344:
340:
336:
333:(6709): 329.
332:
328:
321:
318:
312:
307:
304:
302:
299:
297:
294:
292:
289:
287:
284:
282:
279:
278:
273:
271:
267:
265:
263:
258:
250:
248:
246:
242:
229:
225:
218:
216:
213:
208:
204:
203:amorphous ice
196:
194:
192:
188:
184:
180:
176:
168:
166:
164:
155:
153:
144:
140:
133:
131:
129:
125:
124:Amorphous ice
121:
117:
113:
105:
103:
101:
97:
93:
89:
85:
81:
77:
76:Polyamorphism
70:
65:
58:
54:
49:
45:
44:discontinuous
41:
37:
36:phase diagram
32:
19:
1300:
1296:
1290:
1247:
1243:
1233:
1185:(1): 42439.
1182:
1178:
1168:
1135:
1131:
1125:
1105:
1098:
1073:
1069:
1063:
1046:
1042:
1036:
987:
983:
977:
936:
932:
926:
893:
889:
883:
842:
838:
825:
784:
780:
767:
742:
739:Phys. Rev. B
738:
732:
699:
695:
689:
665:
658:
628:(24): 5355.
625:
621:
612:
587:
581:
569:. Retrieved
560:
533:
529:
519:
489:(6006): 76.
486:
482:
476:
441:
437:
427:
376:
372:
330:
326:
320:
268:
261:
254:
222:
200:
172:
159:
150:
141:
137:
109:
99:
92:two distinct
91:
84:polymorphism
75:
74:
48:polymorphism
88:crystalline
1346:Categories
313:References
179:phosphorus
112:long-range
18:Polyamorph
1325:0021-9606
1274:0021-9606
1207:2045-2322
1152:0953-8984
1012:0036-8075
571:30 August
177:, liquid
80:amorphous
1333:28668061
1282:26133438
1225:28209972
1160:94090829
1090:95734427
1028:29634533
1020:15514150
969:27352374
961:21694284
867:20366109
817:10368768
809:18948535
724:15323706
671:Elsevier
650:59485355
468:20047984
460:12195833
411:11217853
274:See also
264:-butanol
187:mannitol
106:Overview
1305:Bibcode
1252:Bibcode
1216:5314399
1187:Bibcode
1070:Science
1049:: 1–4.
992:Bibcode
984:Science
941:Bibcode
918:4325330
898:Bibcode
875:3493920
847:Bibcode
789:Bibcode
781:Science
747:Bibcode
704:Bibcode
630:Bibcode
592:Bibcode
538:Bibcode
511:4241205
491:Bibcode
419:4419993
391:Bibcode
355:4328846
335:Bibcode
228:alumina
175:silicon
120:density
59:regime.
1331:
1323:
1280:
1272:
1223:
1213:
1205:
1158:
1150:
1113:
1088:
1026:
1018:
1010:
967:
959:
916:
890:Nature
873:
865:
815:
807:
722:
677:
648:
509:
483:Nature
466:
458:
417:
409:
373:Nature
353:
327:Nature
286:Liquid
224:Yttria
1156:S2CID
1086:S2CID
1024:S2CID
965:S2CID
914:S2CID
871:S2CID
835:(PDF)
813:S2CID
777:(PDF)
646:S2CID
507:S2CID
464:S2CID
415:S2CID
381:arXiv
351:S2CID
281:Glass
1329:PMID
1321:ISSN
1278:PMID
1270:ISSN
1221:PMID
1203:ISSN
1148:ISSN
1111:ISBN
1016:PMID
1008:ISSN
957:PMID
863:PMID
805:PMID
720:PMID
675:ISBN
573:2015
456:PMID
407:PMID
1313:doi
1301:146
1260:doi
1248:142
1211:PMC
1195:doi
1140:doi
1078:doi
1074:275
1051:doi
1047:100
1000:doi
988:306
949:doi
906:doi
894:369
855:doi
843:103
797:doi
785:322
755:doi
712:doi
638:doi
600:doi
546:doi
499:doi
487:314
446:doi
399:doi
377:409
343:doi
331:396
243:or
86:of
1348::
1327:.
1319:.
1311:.
1299:.
1276:.
1268:.
1258:.
1246:.
1242:.
1219:.
1209:.
1201:.
1193:.
1181:.
1177:.
1154:.
1146:.
1136:17
1134:.
1084:.
1072:.
1045:.
1022:.
1014:.
1006:.
998:.
986:.
963:.
955:.
947:.
937:20
935:.
912:.
904:.
892:.
869:.
861:.
853:.
841:.
837:.
811:.
803:.
795:.
783:.
779:.
753:.
743:72
741:.
718:.
710:.
700:93
698:.
669:.
644:.
636:.
624:.
598:.
544:.
534:46
532:.
528:.
505:.
497:.
485:.
462:.
454:.
442:54
440:.
436:.
413:.
405:.
397:.
389:.
375:.
363:^
349:.
341:.
329:.
185:,
181:,
130:.
102:.
1335:.
1315::
1307::
1284:.
1262::
1254::
1227:.
1197::
1189::
1183:7
1162:.
1142::
1119:.
1092:.
1080::
1057:.
1053::
1030:.
1002::
994::
971:.
951::
943::
920:.
908::
900::
877:.
857::
849::
819:.
799::
791::
761:.
757::
749::
726:.
714::
706::
683:.
652:.
640::
632::
626:3
606:.
602::
594::
575:.
554:.
548::
540::
513:.
501::
493::
470:.
448::
421:.
401::
393::
383::
357:.
345::
337::
262:n
237:3
235:O
233:2
226:-
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.