814:
301:
226:
28:
802:
37:
970:
65:
871:
56:
623:
466:
1017:
733:, which provides the N-C-C-S backbone of the ring. Thiamine does not fit this pattern however. Several biosynthesis routes lead to the thiazole ring as required for the formation of thiamine. Sulfur of the thiazole is derived from cysteine. In anaerobic bacteria, the CN group is derived from dehydroglycine.
1387:
Alajarín, M.; Cabrera, J.; Pastor, A.; Sánchez-Andrada, P.; Bautista, D. (2006). "On the
Cycloaddition of 2-Aminothiazoles and Dimethyl Acetylenedicarboxylate. Experimental and Computational Evidence of a Thermal Disrotatory Ring Opening of Fused Cyclobutenes".
676:
dyes contain benzothiazole subunits: Algol Yellow 8 (CAS# ), Algol Yellow GC (CAS# ), Indanthren Rubine B (CAS# ), Indanthren Blue CLG (CAS# , and
Indanthren Blue CLB (CAS#). These thiazole dye are used for dyeing
657:, oxazoles are not. It is found in naturally occurring peptides, and utilised in the development of peptidomimetics (i.e. molecules that mimic the function and structure of peptides).
479:
776:
813:
350:
865:-oxide is able to shift the reactivity to reliably favor the 2-position, and allows for these reactions to be carried out under much more mild conditions.
544:
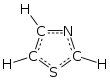
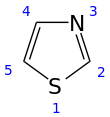
that contains both sulfur and nitrogen. The term 'thiazole' also refers to a large family of derivatives. Thiazole itself is a pale yellow liquid with a
637:
Thiazoles are found in a variety of specialized products, often fused with benzene derivatives, the so-called benzothiazoles. In addition to vitamin B
1126:
1088:
614:. The calculated pi-electron density marks C5 as the primary site for electrophilic substitution, and C2-H as susceptible to deprotonation.
668:
are marketed for control of various agricultural pests. Another widely used thiazole derivative is the non-steroidal anti-inflammatory drug
1340:
Campeau, Louis-Charles; Bertrand-Laperle, Mégan; Leclerc, Jean-Philippe; Villemure, Elisia; Gorelsky, Serge; Fagnou, Keith (2008-03-01).
781:
1251:
Dondoni, A.; Merino, P. (1995). "Diastereoselective
Homologation of D-(R)-Glyceraldehyde Acetonide using 2-(Trimethylsilyl)thiazole".
1232:
1063:
315:
1008:
771:
react at this site, replacing the proton. 2-Lithiothiazoles are also generated by metal-halogen exchange from 2-bromothiazole.
1469:
486:
258:
279:
969:
801:
424:
775:
611:
1464:
1305:
710:
443:
1046:
Zoltewicz, J. A.; Deady, L. W. (1978). "Quaternization of
Heteroaromatic Compounds: Quantitative Aspects".
870:
823:
221:
956:
768:
1268:
541:
183:
1424:
Arduengo, A. J.; Goerlich, J. R.; Marshall, W. J. (1997). "A Stable
Thiazol-2-ylidene and Its Dimer".
996:
928:
917:
83:
296:
109:
1459:
1105:
909:
1113:. Topics in Heterocyclic Chemistry. Vol. 48. Springer Berlin Heidelberg. pp. 235–266.
610:
chemical shift of the ring protons, which absorb between 7.27 and 8.77 ppm, indicating a strong
927:, but in general at high temperatures due to favorable aromatic stabilization of the reactant;
1406:
1369:
1361:
1322:
1228:
1205:
1144:
1122:
1084:
1059:
905:
838:
690:
1435:
1398:
1353:
1314:
1256:
1195:
1153:
1114:
1051:
992:
785:
714:
630:
588:
504:
373:
759:
with strong bases occurs at C2-H. The negative charge on this position is stabilized as an
267:
27:
1426:
1281:
1171:
Kriek, M.; Martins, F.; Leonardi, R.; Fairhurst, S. A.; Lowe, D. J.; Roach, P. L. (2007).
203:
143:
119:
693:
of thiazoles. Prominent is the
Hantzsch thiazole synthesis, which is a reaction between
300:
225:
163:
595:
457:
1055:
64:
1453:
1390:
1342:"C2, C5, and C4 Azole N -Oxide Direct Arylation Including Room-Temperature Reactions"
924:
897:
756:
718:
717:. Thiazoles can be accessed by acylation of 2-aminothiolates, often available by the
706:
702:
673:
665:
646:
414:
214:
36:
1172:
1004:
846:
1177:: An Investigation of the Substrates and Purified Proteins Required for Activity
247:
1081:
The
Chemistry of Heterocycles: Structure, Reactions, Syntheses, and Applications
793:
764:
654:
599:
55:
1439:
1026:
984:
948:
913:
694:
642:
607:
401:
194:
1365:
1341:
1260:
1157:
963:
in a 6-electron electrocyclic ring, closing before extruding the sulfur atom.
850:
746:
698:
669:
661:
650:
626:
580:
1410:
1373:
1326:
1209:
1200:
1118:
622:
936:
842:
730:
560:
545:
885:
854:
849:; some of the oxidation takes place at sulfur, leading to non-aromatic
827:
603:
584:
557:
234:
1402:
1357:
1016:
1318:
932:
882:
789:
678:
174:
861:-oxides are useful in Palladium-catalysed C-H arylations, where the
594:
Being planar thiazoles are characterized by significant pi-electron
456:
Except where otherwise noted, data are given for materials in their
1295:
Amir, E.; Rozen, S. (2006). "Easy Access to the Family of
Thiazole
951:
intermediate in a formal cycloaddition to a cyclobutene, then to a
1015:
834:
760:
621:
576:
154:
142:
132:
745:
of 2.5 for the conjugate acid, thiazoles are far less basic than
1104:
Mak, Jeffrey Y. W.; Xu, Weijun; Fairlie, David P. (2015-01-01).
935:
are followed by extrusion of sulfur, and the endproduct is a
713:, thiazoles arise by the condensation of α-aminonitrile with
968:
869:
812:
800:
510:
284:
63:
54:
35:
26:
1227:(3 ed.). Essex, England: Addison Wesley. p. 414.
525:
519:
513:
419:
116 to 118 °C (241 to 244 °F; 389 to 391 K)
1020:
Structure of thiazoles (left) and thiazolium salts (right)
660:
Commercial significant thiazoles include mainly dyes and
701:. For example, 2,4-dimethylthiazole is synthesized from
556:
NS. The thiazole ring is notable as a component of the
474:
522:
947:(DMAD) to a pyridine was found to proceed through a
528:
507:
729:Thiazoles are generally formed via reactions of
246:
1142:George Schwarz (1945). "2,4-Dimethylthiazole".
991:cation. Thiazolium salts are catalysts in the
118:
8:
1048:Advances in Heterocyclic Chemistry Volume 22
653:. Whereas thiazoles are well represented in
618:Occurrence of thiazoles and thiazolium salts
1246:
1244:
817:Thiazole Nucleophilic Aromatic Substitution
645:. Other important thiazole derivatives are
516:
939:; in one study, a very mild reaction of a
299:
224:
202:
15:
1199:
896:aldehyde takes place with, respectively,
689:Various laboratory methods exist for the
266:
1346:Journal of the American Chemical Society
606:. This aromaticity is evidenced by the
1038:
833:; many oxidizing agents exist, such as
355:
320:
295:
1277:
1266:
548:-like odor and the molecular formula C
215:
327:Key: FZWLAAWBMGSTSO-UHFFFAOYSA-N
182:
162:
7:
649:, for example, the firefly chemical
587:. Thiazole can also be considered a
782:Electrophilic aromatic substitution
664:. Thifluzamide, Tricyclazole, and
337:Key: FZWLAAWBMGSTSO-UHFFFAOYAI
237:
1079:Eicher, T.; Hauptmann, S. (2003).
1009:transition metal carbene complexes
571:Molecular and electronic structure
324:InChI=1S/C3H3NS/c1-2-5-3-4-1/h1-3H
14:
1050:. Vol. 22. pp. 71–121.
1003:-alkyl thiazolium salts give the
987:of thiazoles at nitrogen forms a
602:, moreso than the corresponding
334:InChI=1/C3H3NS/c1-2-5-3-4-1/h1-3H
774:
641:, the thiazole ring is found in
591:when part of a larger molecule.
503:
464:
391:
385:
945:dimethyl acetylenedicarboxylate
460:(at 25 °C , 100 kPa).
394:
379:
1:
1056:10.1016/S0065-2725(08)60103-8
575:Thiazoles are members of the
1223:Thomas L. Gilchrist (1997).
1029:is a thiazolium-based drug.
826:gives the aromatic thiazole
579:, heterocycles that include
50:
22:
1486:
957:electrocyclic ring opening
1440:10.1002/jlac.199719970213
941:2-(dimethylamino)thiazole
792:group, as illustrated in
629:is a thiazole-containing
454:
437:2.5 (of conjugate acid)
366:
346:
311:
102:
94:
82:
77:
49:
21:
1261:10.15227/orgsyn.072.0021
1173:"Thiazole Synthase from
1158:10.15227/orgsyn.025.0035
961:7-thia-2-azanorcaradiene
612:diamagnetic ring current
598:and have some degree of
1306:Chemical Communications
923:Thiazoles can react in
769:organolithium compounds
711:Cook-Heilbron synthesis
444:Magnetic susceptibility
1276:Cite journal requires
1225:Heterocyclic Chemistry
1201:10.1074/jbc.M700782200
1021:
974:
973:Thiazole cycloaddition
875:
818:
806:
634:
68:
59:
40:
31:
1470:Simple aromatic rings
1119:10.1007/7081_2015_176
1019:
972:
929:Diels-Alder reactions
873:
824:Oxidation at nitrogen
816:
804:
625:
542:heterocyclic compound
67:
58:
39:
30:
1299:-oxides using HOF·CH
999:. Deprotonation of
997:Benzoin condensation
918:Mercury(II) chloride
805:Thiazole bromination
84:Preferred IUPAC name
1194:(24): 17413–17423.
409: g·mol
18:
1022:
975:
910:sodium borohydride
876:
874:Thiazole oxidation
819:
807:
784:at C5 but require
635:
540:, is a 5-membered
487:Infobox references
69:
60:
41:
32:
16:
1403:10.1021/jo060664c
1397:(14): 5328–5339.
1358:10.1021/ja7107068
1352:(11): 3276–3277.
1313:(21): 2262–2264.
1145:Organic Syntheses
1128:978-3-319-49117-2
1107:Peptidomimetics I
1090:978-3-527-30720-3
906:organic reduction
839:hypofluorous acid
837:; a novel one is
786:activating groups
691:organic synthesis
495:Chemical compound
493:
492:
450:-50.55·10 cm/mol
280:CompTox Dashboard
144:Interactive image
73:
72:
45:
44:
1477:
1444:
1443:
1421:
1415:
1414:
1384:
1378:
1377:
1337:
1331:
1330:
1319:10.1039/b602594c
1292:
1286:
1285:
1279:
1274:
1272:
1264:
1248:
1239:
1238:
1220:
1214:
1213:
1203:
1185:
1175:Escherichia coli
1168:
1162:
1160:
1139:
1133:
1132:
1112:
1101:
1095:
1094:
1076:
1070:
1069:
1043:
993:Stetter reaction
980:Thiazolium salts
955:in a 4-electron
888:; conversion of
778:
715:carbon disulfide
672:. The following
631:anti-cancer drug
589:functional group
535:
534:
531:
530:
527:
524:
521:
518:
515:
512:
509:
477:
471:
468:
467:
408:
396:
393:
387:
381:
374:Chemical formula
304:
303:
288:
286:
270:
250:
239:
228:
217:
206:
186:
166:
146:
122:
51:
23:
19:
1485:
1484:
1480:
1479:
1478:
1476:
1475:
1474:
1450:
1449:
1448:
1447:
1427:Liebigs Annalen
1423:
1422:
1418:
1386:
1385:
1381:
1339:
1338:
1334:
1302:
1294:
1293:
1289:
1275:
1265:
1250:
1249:
1242:
1235:
1222:
1221:
1217:
1183:
1170:
1169:
1165:
1141:
1140:
1136:
1129:
1110:
1103:
1102:
1098:
1091:
1078:
1077:
1073:
1066:
1045:
1044:
1040:
1035:
982:
904:-methylation),
752:
744:
739:
727:
687:
640:
620:
573:
566:
555:
551:
506:
502:
496:
489:
484:
483:
482: ?)
473:
469:
465:
461:
447:
433:
406:
390:
384:
376:
362:
359:
354:
353:
342:
339:
338:
335:
329:
328:
325:
319:
318:
307:
289:
282:
273:
253:
240:
209:
189:
169:
149:
136:
125:
112:
98:
90:
89:
12:
11:
5:
1483:
1481:
1473:
1472:
1467:
1465:Aromatic bases
1462:
1452:
1451:
1446:
1445:
1434:(2): 365–374.
1416:
1379:
1332:
1300:
1287:
1278:|journal=
1240:
1233:
1215:
1163:
1134:
1127:
1096:
1089:
1071:
1064:
1037:
1036:
1034:
1031:
1024:
1023:
981:
978:
977:
976:
965:
964:
959:and then to a
953:1,3-thiazepine
925:cycloadditions
921:
881:Thiazoles are
878:
877:
841:prepared from
821:
820:
809:
808:
750:
742:
738:
735:
726:
723:
686:
683:
647:benzothiazoles
638:
619:
616:
596:delocalization
572:
569:
564:
553:
549:
494:
491:
490:
485:
463:
462:
458:standard state
455:
452:
451:
448:
442:
439:
438:
435:
431:
421:
420:
417:
411:
410:
404:
398:
397:
388:
382:
377:
372:
369:
368:
364:
363:
361:
360:
357:
349:
348:
347:
344:
343:
341:
340:
336:
333:
332:
330:
326:
323:
322:
314:
313:
312:
309:
308:
306:
305:
292:
290:
278:
275:
274:
272:
271:
263:
261:
255:
254:
252:
251:
243:
241:
233:
230:
229:
219:
211:
210:
208:
207:
199:
197:
191:
190:
188:
187:
179:
177:
171:
170:
168:
167:
159:
157:
151:
150:
148:
147:
139:
137:
130:
127:
126:
124:
123:
115:
113:
108:
105:
104:
100:
99:
96:
92:
91:
87:
86:
80:
79:
75:
74:
71:
70:
61:
47:
46:
43:
42:
33:
13:
10:
9:
6:
4:
3:
2:
1482:
1471:
1468:
1466:
1463:
1461:
1458:
1457:
1455:
1441:
1437:
1433:
1429:
1428:
1420:
1417:
1412:
1408:
1404:
1400:
1396:
1393:
1392:
1391:J. Org. Chem.
1383:
1380:
1375:
1371:
1367:
1363:
1359:
1355:
1351:
1347:
1343:
1336:
1333:
1328:
1324:
1320:
1316:
1312:
1308:
1307:
1298:
1291:
1288:
1283:
1270:
1262:
1258:
1254:
1247:
1245:
1241:
1236:
1234:0-582-27843-0
1230:
1226:
1219:
1216:
1211:
1207:
1202:
1197:
1193:
1189:
1188:J. Biol. Chem
1182:
1180:
1176:
1167:
1164:
1159:
1155:
1151:
1147:
1146:
1138:
1135:
1130:
1124:
1120:
1116:
1109:
1108:
1100:
1097:
1092:
1086:
1082:
1075:
1072:
1067:
1065:9780120206223
1061:
1057:
1053:
1049:
1042:
1039:
1032:
1030:
1028:
1018:
1014:
1013:
1012:
1010:
1006:
1005:free carbenes
1002:
998:
994:
990:
986:
979:
971:
967:
966:
962:
958:
954:
950:
946:
942:
938:
934:
930:
926:
922:
919:
915:
911:
907:
903:
899:
898:methyl iodide
895:
891:
887:
884:
880:
879:
872:
868:
867:
866:
864:
860:
856:
852:
848:
845:and water in
844:
840:
836:
832:
830:
825:
815:
811:
810:
803:
799:
798:
797:
795:
791:
787:
783:
779:
777:
772:
770:
766:
762:
758:
757:Deprotonation
754:
748:
736:
734:
732:
724:
722:
720:
719:Herz reaction
716:
712:
708:
707:chloroacetone
704:
703:thioacetamide
700:
696:
692:
684:
682:
680:
675:
674:anthroquinone
671:
667:
666:Thiabendazole
663:
658:
656:
652:
648:
644:
632:
628:
624:
617:
615:
613:
609:
605:
601:
597:
592:
590:
586:
582:
578:
570:
568:
562:
559:
547:
543:
539:
533:
500:
488:
481:
476:
459:
453:
449:
445:
441:
440:
436:
430:
426:
423:
422:
418:
416:
415:Boiling point
413:
412:
405:
403:
400:
399:
378:
375:
371:
370:
365:
356:
352:
345:
331:
321:
317:
310:
302:
298:
297:DTXSID2059776
294:
293:
291:
281:
277:
276:
269:
265:
264:
262:
260:
257:
256:
249:
245:
244:
242:
236:
232:
231:
227:
223:
220:
218:
216:ECHA InfoCard
213:
212:
205:
201:
200:
198:
196:
193:
192:
185:
181:
180:
178:
176:
173:
172:
165:
161:
160:
158:
156:
153:
152:
145:
141:
140:
138:
134:
129:
128:
121:
117:
116:
114:
111:
107:
106:
101:
93:
85:
81:
76:
66:
62:
57:
53:
52:
48:
38:
34:
29:
25:
24:
20:
1431:
1425:
1419:
1394:
1389:
1382:
1349:
1345:
1335:
1310:
1304:
1296:
1290:
1269:cite journal
1252:
1224:
1218:
1191:
1187:
1178:
1174:
1166:
1149:
1143:
1137:
1106:
1099:
1080:
1074:
1047:
1041:
1025:
1000:
988:
983:
960:
952:
949:zwitterionic
944:
940:
901:
893:
889:
862:
858:
847:acetonitrile
828:
822:
780:
773:
765:Hauser bases
755:
740:
728:
725:Biosynthesis
688:
659:
655:biomolecules
636:
593:
574:
538:1,3-thiazole
537:
498:
497:
428:
103:Identifiers
95:Other names
88:1,3-Thiazole
857:: Thiazole
794:bromination
695:haloketones
600:aromaticity
367:Properties
222:100.005.475
184:ChEMBL15605
164:CHEBI:43732
1454:Categories
1033:References
1027:Alagebrium
989:thiazolium
985:Alkylation
914:hydrolysis
788:such as a
699:thioamides
662:fungicides
643:epothilone
581:imidazoles
402:Molar mass
268:320RCW8PEF
195:ChemSpider
131:3D model (
110:CAS Number
1460:Thiazoles
1366:0002-7863
1083:. Wiley.
920:in water.
851:sulfoxide
747:imidazole
741:With a pK
737:Reactions
709:. In the
685:Synthesis
670:Meloxicam
651:luciferin
627:Bleomycin
17:Thiazole
1411:16808523
1374:18302383
1327:16718323
1210:17403671
1179:in vitro
995:and the
937:pyridine
886:synthons
843:fluorine
731:cysteine
604:oxazoles
585:oxazoles
561:thiamine
546:pyridine
499:Thiazole
446:(χ)
120:288-47-1
97:Thiazole
933:alkynes
892:to the
855:sulfone
558:vitamin
480:what is
478: (
425:Acidity
358:n1ccsc1
235:PubChem
1409:
1372:
1364:
1325:
1255:: 21.
1231:
1208:
1152:: 35.
1125:
1087:
1062:
912:, and
890:R-thia
883:formyl
831:-oxide
790:methyl
753:=7).
679:cotton
577:azoles
536:), or
475:verify
472:
351:SMILES
175:ChEMBL
78:Names
1303:CN".
1184:(PDF)
1111:(PDF)
943:with
931:with
916:with
908:with
894:R-CHO
835:mCPBA
761:ylide
608:H NMR
407:85.12
316:InChI
155:ChEBI
133:JSmol
1432:1997
1407:PMID
1370:PMID
1362:ISSN
1323:PMID
1311:2006
1282:help
1229:ISBN
1206:PMID
1123:ISBN
1085:ISBN
1060:ISBN
1007:and
767:and
705:and
697:and
583:and
259:UNII
248:9256
204:8899
1436:doi
1399:doi
1354:doi
1350:130
1315:doi
1257:doi
1196:doi
1192:282
1154:doi
1115:doi
1052:doi
749:(pK
567:).
285:EPA
238:CID
1456::
1430:.
1405:.
1395:71
1368:.
1360:.
1348:.
1344:.
1321:.
1309:.
1273::
1271:}}
1267:{{
1253:72
1243:^
1204:.
1190:.
1186:.
1150:25
1148:.
1121:.
1058:.
1011:.
796::
763:.
721:.
681:.
563:(B
526:oʊ
514:aɪ
434:)
427:(p
1442:.
1438::
1413:.
1401::
1376:.
1356::
1329:.
1317::
1301:3
1297:N
1284:)
1280:(
1263:.
1259::
1237:.
1212:.
1198::
1181:"
1161:.
1156::
1131:.
1117::
1093:.
1068:.
1054::
1001:N
902:N
900:(
863:N
859:N
853:/
829:N
751:a
743:a
639:1
633:.
565:1
554:3
552:H
550:3
532:/
529:l
523:z
520:ə
517:.
511:θ
508:ˈ
505:/
501:(
470:Y
432:a
429:K
395:S
392:N
389:3
386:H
383:3
380:C
287:)
283:(
135:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.