838:
2402:
2253:
2321:
2158:
863:
28:
140:
788:
1615:
Arrowsmith, Merle; Crimmin, Mark R.; Barrett, Anthony G. M.; Hill, Michael S.; Kociok-KöHn, Gabriele; Procopiou, Panayiotis A. (2011). "Cation Charge
Density and Precatalyst Selection in Group 2-Catalyzed Aminoalkene Hydroamination".
906:
with lithium biphenylide) with allyl halides. These allylbarium compounds react with carbonyl compounds. Such reagents are more alpha-selective and more stereoselective than the related
Grignards or organocalcium compounds. The
587:
1369:
Ruhlandt-Senge, Karin; Bartlett, Ruth A.; Olmstead, Marilyn M.; Power, Philip P. (1993). "Synthesis and structural characterization of the beryllium compounds , , and .cntdot.PhMe and determination of the structure of ".
1101:
Reuben D. Rieke; Tse-Chong Wu; Loretta I. Rieke (1995). "Highly
Reactive Calcium for the Preparation of Organocalcium Reagents: 1-Adamantyl Calcium Halides and Their Addition to Ketones: 1-(1-Adamantyl)cyclohexanol".
159:
Mixed alkyl/aryl-halide compounds, which contain a single C-M bond and a C-X bond, are typically prepared by oxidative addition. Magnesium-containing compounds of this configuration are known as the
1796:
Williams, R. A.; Hanusa, T. P.; Huffman, J. C. (1988). "Solid state structure of bis(pentamethylcyclopentadienyl)barium, (Me5C5)2Ba; the first X-ray crystal structure of an organobarium complex".
1151:
Coates, G. E.; Francis, B. R. (1971). "Preparation of base-free beryllium alkyls from trialkylboranes. Dineopentylberyllium, bis(trimethylsilylmethyl)beryllium, and an ethylberyllium hydride".
1643:
Miyoshi, N.; Kamiura, K.; Oka, H.; Kita, A.; Kuwata, R.; Ikehara, D.; Wada, M. (2004). "The
Barbier-Type Alkylation of Aldehydes with Alkyl Halides in the Presence of Metallic Strontium".
614:
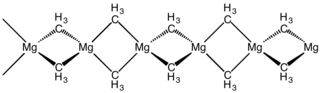
110:
and many compounds (particularly dialklys) are polymeric in solid or liquid states with highly complex structures in solution, though in the gaseous state they are often monomeric.
837:
1519:"Stable 'Inverse' Sandwich Complex with Unprecedented Organocalcium(I): Crystal Structures of and " Sven Krieck, Helmar Görls, Lian Yu, Markus Reiher and Matthias Westerhausen
82:
with higher and lower states being rare, and are less electronegative than carbon. However, as the group two elements (with the exception of beryllium) have considerably low
1670:
Miyoshi, N.; Ikehara, D.; Kohno, T.; Matsui, A.; Wada, M. (2005). "The
Chemistry of Alkylstrontium Halide Analogues: Barbier-type Alkylation of Imines with Alkyl Halides".
1742:
Yanagisawa, A.; Habaue, S.; Yamamoto, H. (1991). "Allylbarium in organic synthesis: unprecedented .alpha.-selective and stereospecific allylation of carbonyl compounds".
1769:
Yanagisawa, A.; Habaue, S.; Yasue, K.; Yamamoto, H. (1994). "Allylbarium
Reagents: Unprecedented Regio- and Stereoselective Allylation Reactions of Carbonyl Compounds".
297:
The distinctive feature of the
Grignard reagents is their formation from the organic halide and magnesium metal. Most other group II organic compounds are generated by
1836:
1499:
Lichtenberg, C., Jochmann, P., Spaniol, T. P. and Okuda, J. (2011), "The
Allylcalcium Monocation: A Bridging Allyl Ligand with a Non-Bent Coordination Geometry".
1228:
Almenningen, A; Haaland, Arne; Lusztyk, Janusz (1979). "The molecular structure of beryllocene, (C5H5)2Be. A reinvestigation by gas phase electron diffraction".
241:, the other organo-group 2 compound are almost exclusively of academic interest. Organoberyllium chemistry is limited due to the cost and toxicity of beryllium.
1697:
Miyoshi, N.; Matsuo, T.; Wada, M. (2005). "The
Chemistry of Alkylstrontium Halide Analogues, Part 2: Barbier-Type Dialkylation of Esters with Alkyl Halides".
283:(by exchange reaction of diethyl beryllium with triallyl boron), bis(1,3-trimethylsilylallyl)beryllium and Be(mes)2. Ligands can also be aryls and alkynyls.
163:, though some calcium Grignard's are known and more reactive and sensitive to decomposition. Calcium grignard's must be pre-activated prior to synthesis.
374:
1000:
1829:
1201:
Nugent, KW; Beattie, JK; Hambley, TW; Snow, MR (1984). "A precise low-temperature crystal structure of Bis(cyclopentadienyl)beryllium".
47:
2534:
1024:
317:. These simplified formulas are deceptive: Grignard reagents generally exist as dietherates, RMgX(ether)2. As such they obey the
1350:
Synthesis and structural characterization of the beryllium compounds , , and .cntdot.PhMe and determination of the structure of
357:
1822:
1352:
Karin
Ruhlandt-Senge, Ruth A. Bartlett, Marilyn M. Olmstead, and Philip P. Power Inorganic Chemistry 1993 32 (9), 1724-1728
301:, which limits their accessibility. The formation of the Grignard reagents has received intense scrutiny. It proceeds by a
1174:
Fischer, Ernst Otto; Hofmann, Hermann P. (1959). "Über Aromatenkomplexe von Metallen, XXV. Di-cyclopentadienyl-beryllium".
1576:"Butenyl-Substituted Alkaline-Earth Metallocenes: A First Step towards Olefin Complexes of the Alkaline-Earth Metals"
2391:
2386:
2381:
2376:
2371:
2366:
2361:
2356:
2351:
2346:
2341:
2336:
2326:
2269:
2163:
2084:
2079:
1936:
1288:
Wiegand, G.; Thiele, K.-H. (1974). "Ein Beitrag zur Existenz von Allylberyllium- und Allylaluminiumverbindungen".
783:{\displaystyle {\ce {{\overset {allylpotassium}{2KC3H5}}+{\overset {calcium\ iodide}{CaI2}}->{(C3H5)2Ca}+2KI}}}
2427:
2129:
2099:
2089:
2069:
2057:
2025:
1990:
1958:
1926:
1921:
1881:
1479:"Bis(allyl)calcium" Phillip Jochmann, Thomas S. Dols, Thomas P. Spaniol, Lionel Perrin, Laurent Maron, Jun Okuda
1397:
Morosin, B; Howatson, J. (1971). "The crystal structure of dimeric methyl-1-propynyl- beryllium-trimethylamine".
266:
254:
1896:
1860:
1814:
1315:
Chmely, Stephen C.; Hanusa, Timothy P.; Brennessel, William W. (2010). "Bis(1,3-trimethylsilylallyl)beryllium".
2472:
2467:
2462:
2457:
2452:
2447:
2442:
2437:
2432:
2417:
2407:
2258:
2233:
2228:
2213:
2198:
2178:
2173:
2124:
2052:
2035:
1985:
1980:
1975:
1970:
1946:
1906:
944:
353:
302:
170:
116:
compounds with group 2 elements are rare, but some do exist. Bis(cyclopentadienyl)beryllium or beryllocene (Cp
963:
Borislav Bogdanovic (1988). "Magnesium Anthracene Systems and their Application in Synthesis and Catalysis".
2422:
2412:
2223:
2208:
2193:
2183:
2168:
2109:
2094:
2074:
2064:
2045:
2040:
2030:
2020:
1963:
1931:
829:
310:
305:
process. For less reactive organic halides, activated forms of magnesium have been produced in the form of
121:
103:
1901:
1891:
2331:
2246:
2188:
2151:
2146:
2134:
2114:
2104:
2010:
2005:
2000:
1941:
1916:
314:
324:
Grignard reagents participate in the Schlenk equilibrium. Exploiting this reaction is a way to generate
2139:
1951:
1886:
1876:
1547:
2218:
2203:
2015:
1995:
329:
1426:"Preparation of t-Butyl-3-Bromo-5-Formylbenzoate Through Selective Metal-Halogen Exchange Reactions"
597:
2119:
1038:
213:
107:
1456:"Dimethylcalcium" Benjamin M. Wolf, Christoph Stuhl, Cäcilia Maichle-Mössmer, and Reiner Anwander
886:), with H atoms omitted. Even with bulky alkyl substituents, Ba coordinates to three THF ligands.
272:
1134:
D. Naglav, M. R. Buchner, G. Bendt, F. Kraus, S. Schulz, Angew. Chem. Int. Ed. 2016, 55, 10562.
898:
compounds of the type (allyl)BaCl can be prepared by reaction of activated barium (Rieke method
1726:
1597:
1521:
1332:
1020:
996:
819:
325:
147:
143:
83:
55:
1801:
1778:
1751:
1706:
1679:
1652:
1625:
1587:
1555:
1530:
1504:
1484:
1464:
1437:
1406:
1379:
1353:
1324:
1297:
1268:
1237:
1210:
1183:
1156:
1135:
1111:
1081:
1073:
1046:
972:
856:
593:
292:
238:
191:
160:
75:
71:
67:
51:
39:
596:)calcium(I). Bis(allyl)calcium was described in 2009. It forms in a metathesis reaction of
102:, but with some ionic bond characteristics owing to the attached carbon bearing a negative
306:
298:
95:
91:
79:
43:
17:
601:
275:. Examples of known organoberyllium compounds are dineopentylberyllium, beryllocene (Cp
50:. By far the most common group 2 organometallic compounds are the magnesium-containing
1410:
1241:
1050:
166:
There are three key reaction pathways for dialkyl and diaryl group 2 metal compounds.
58:. Other organometallic group 2 compounds are typically limited to academic interests.
2528:
2491:
1545:"Organometallic Compounds of the Heavier s-Block Elements—What Next?" J. David Smith
903:
365:
361:
99:
1730:
1086:
582:{\displaystyle \mathrm {Ca_{2}+2\ LiCH_{3}\longrightarrow Ca(CH_{3})_{2}+2\ Li} }
908:
337:
146:
is a polymer built up from 3-center, 2-electron bonded bridging methyl groups.
113:
1273:
1256:
1077:
605:
318:
87:
1442:
1425:
1301:
1187:
1115:
832:
have been shown to coordinate to calcium(II), strontium(II), and barium(II):
932:
798:
794:
333:
246:
245:
is nontoxic and cheap but organocalcium compounds are difficult to prepare,
1710:
1601:
1592:
1575:
1559:
1508:
1488:
1336:
1328:
1139:
90:-like, if not entirely ionic for the heavier barium compounds. The lighter
132:
Mg) is a regular metallocene, bis(pentamethylcyclopentadienyl)calcium (Cp)
106:. This higher ionic character and bond polarization tends to produce high
1805:
1468:
1160:
1782:
1755:
1683:
1656:
1383:
1357:
976:
271:
Beryllium derivatives and reagents are often prepared by alkylation of
242:
1629:
1534:
1214:
1846:
1257:"Crystal structure of bis(cyclopentadienyl)beryllium at −120 °C"
825:
694:
332:. This orange solid is used as a source of highly active magnesium.
250:
711:
1153:
Journal of the Chemical Society A: Inorganic, Physical, Theoretical
1064:
Snow, A.I.; Rundle, R.E. (1951). "Structure of Dimethylberyllium".
899:
862:
861:
253:
compounds even more so. One use for these type of compounds is in
138:
125:
27:
26:
1036:
Weiss, E. (1964). "Die Kristallstruktur des Dimethylmagnesiums".
328:. Beyond Grignard reagents, another organomagnesium compound is
237:
Although organomagnesium compounds are widespread in the form of
1132:
Off the Beaten Track—A Hitchhiker's Guide to Beryllium Chemistry
139:
1818:
912:
844:
Organocalcium compounds have been investigated as catalysts.
70:(also referred to as the alkaline earth metals) contain two
336:-magnesium serves as a source for the butadiene dianion.
128:, is so-called slipped η/η sandwich. While magnesocene (Cp
1424:
Arredondo, Juan D.; Li, Hongmei; Balsells, Jaume (2012).
762:
749:
736:
673:
649:
636:
1798:
Journal of the Chemical Society, Chemical Communications
1574:
H. Schumann; S. Schutte; H.-J. Kroth; D. Lentz (2004).
1724:
Comprehensive organic functional group transformations
1255:
Wong, C. H.; Lee, T. Y.; Chao, K. J.; Lee, S. (1972).
797:. This compound is also reported to give access to an
86:
the resulting C-M bonds are more highly polarized and
617:
377:
2483:
822:with two calcium atoms at either side of an arene.
818:The compound also described in 2009 is an inverse
78:organometallic compounds. Both readily assume a +2
1290:Zeitschrift für anorganische und allgemeine Chemie
782:
581:
855:compounds have been reported as intermediates in
31:Magnesium anthracenide with three thf ligands.
1830:
1013:Organometallics : A Concise Introduction
8:
1569:
1567:
566:
530:
427:
391:
340:of magnesium are also well known, e.g LiMgBu
1837:
1823:
1815:
1849:with other elements in the periodic table
1645:Bulletin of the Chemical Society of Japan
1591:
1483:Volume 48 Issue 31, Pages 5715–5719 2009
1441:
1272:
1085:
775:
761:
756:
748:
743:
735:
730:
722:
720:
712:
705:
699:
689:
672:
667:
661:
648:
643:
635:
630:
625:
619:
618:
616:
569:
559:
549:
503:
493:
468:
440:
430:
420:
410:
378:
376:
1771:Journal of the American Chemical Society
1744:Journal of the American Chemical Society
1127:
1125:
592:A well known organocalcium compound is (
1501:Angewandte Chemie International Edition
1481:Angewandte Chemie International Edition
1463:, Volume 140, Issue 6, Pages 2373–2383
1317:Angewandte Chemie International Edition
955:
1866:
990:Comprehensive Organometallic Chemistry
74:, their chemistries have similarities
1699:European Journal of Organic Chemistry
7:
2508:Academic research, no widespread use
309:. Examples of Grignard reagents are
1399:Journal of Organometallic Chemistry
1230:Journal of Organometallic Chemistry
48:main group organometallic chemistry
546:
542:
536:
533:
527:
521:
518:
490:
486:
480:
477:
465:
461:
458:
455:
407:
403:
397:
394:
388:
382:
379:
136:Ca is bent with an angle of 147°.
25:
2400:
2319:
2251:
2156:
1856:
1261:Acta Crystallographica Section B
992:by Mike Mingos, Robert Crabtree
836:
358:calcium bis(trimethylsilyl)amide
36:Group 2 organometallic chemistry
1203:Australian Journal of Chemistry
352:Dimethylcalcium is obtained by
98:compounds are often considered
752:
723:
575:
556:
539:
524:
500:
483:
474:
437:
417:
400:
385:
1:
1411:10.1016/S0022-328X(00)87485-9
1242:10.1016/S0022-328X(00)92065-5
1051:10.1016/S0022-328X(00)82217-2
965:Accounts of Chemical Research
1019:) from Wiley-VCH: Weinheim.
1011:C. Elschenbroich, A. Salzer
919:Ba has also been reported.
2551:
931:compound is the gas-phase
290:
264:
150:adopts the same structure.
2397:
2316:
1868:
1864:
1854:
1274:10.1107/S0567740872004820
1078:10.1107/S0365110X51001100
267:Organoberyllium chemistry
255:chemical vapor deposition
54:which are widely used in
18:Organostrontium chemistry
2535:Organometallic chemistry
1529:, 131 (8), pp 2977–2985
1443:10.15227/orgsyn.089.0460
1302:10.1002/zaac.19744050111
1188:10.1002/cber.19590920233
1116:10.15227/orgsyn.072.0147
945:Organometallic chemistry
830:cyclopentadienyl ligands
1087:2027/mdp.39015095081207
866:Structure of Ba(CH(tms)
311:phenylmagnesium bromide
122:molecular dipole moment
46:. It is a subtheme to
2503:Many uses in chemistry
2498:Core organic chemistry
1711:10.1002/ejoc.200500484
1593:10.1002/anie.200460927
1560:10.1002/anie.200901506
1509:10.1002/anie.201100073
1489:10.1002/anie.200901743
1329:10.1002/anie.201001866
1140:10.1002/anie.201601809
1066:Acta Crystallographica
887:
784:
718:
583:
315:ethylmagnesium bromide
151:
32:
1548:Angew. Chem. Int. Ed.
865:
785:
690:
584:
142:
30:
1806:10.1039/C39880001045
1580:Angew. Chem. Int. Ed
1469:10.1021/jacs.7b12984
1161:10.1039/J19710001308
793:The bonding mode is
615:
375:
330:magnesium anthracene
108:coordination numbers
40:organic derivativess
1783:10.1021/ja00093a010
1756:10.1021/ja00023a058
1684:10.1246/cl.2005.760
1657:10.1246/bcsj.77.341
1384:10.1021/ic00061a031
1372:Inorganic Chemistry
1358:10.1021/ic00061a031
1039:J. Organomet. Chem.
977:10.1021/ar00151a002
764:
751:
738:
717:
710:
675:
651:
638:
214:Schlenk equilibrium
1731:Charles Wayne Rees
1729:, Otto Meth-Cohn,
1176:Chemische Berichte
888:
780:
739:
726:
721:
663:
639:
626:
608:off-white powder:
579:
273:beryllium chloride
152:
33:
2522:
2521:
2478:
2477:
1727:Alan R. Katritzky
1672:Chemistry Letters
1630:10.1021/om101063m
1586:(45): 6208–6211.
1535:10.1021/ja808524y
1522:J. Am. Chem. Soc.
1503:, 50: 5753–5756.
1458:J. Am. Chem. Soc.
1430:Organic Syntheses
1215:10.1071/CH9841601
1001:978-0-08-044590-8
820:sandwich compound
778:
767:
742:
729:
715:
708:
687:
685:
682:
679:
666:
656:
655:
642:
629:
517:
454:
326:dimethylmagnesium
239:Grignard reagents
161:Grignard reagents
148:Dimethylberyllium
144:Dimethylmagnesium
84:electronegativity
72:valence electrons
56:organic chemistry
52:Grignard reagents
16:(Redirected from
2542:
2514:
2509:
2504:
2499:
2404:
2403:
2323:
2322:
2255:
2254:
2160:
2159:
1857:
1839:
1832:
1825:
1816:
1810:
1809:
1793:
1787:
1786:
1766:
1760:
1759:
1739:
1733:
1721:
1715:
1714:
1694:
1688:
1687:
1667:
1661:
1660:
1640:
1634:
1633:
1624:(6): 1493–1506.
1612:
1606:
1605:
1595:
1571:
1562:
1554:, 48, 6597–6599
1543:
1537:
1517:
1511:
1497:
1491:
1477:
1471:
1454:
1448:
1447:
1445:
1421:
1415:
1414:
1394:
1388:
1387:
1366:
1360:
1347:
1341:
1340:
1312:
1306:
1305:
1285:
1279:
1278:
1276:
1252:
1246:
1245:
1225:
1219:
1218:
1198:
1192:
1191:
1171:
1165:
1164:
1148:
1142:
1129:
1120:
1119:
1098:
1092:
1091:
1089:
1061:
1055:
1054:
1033:
1027:
1009:
1003:
987:
981:
980:
960:
840:
789:
787:
786:
781:
779:
776:
768:
765:
763:
760:
755:
750:
747:
740:
737:
734:
727:
719:
716:
713:
709:
706:
704:
703:
688:
686:
683:
680:
677:
674:
671:
664:
662:
657:
653:
652:
650:
647:
640:
637:
634:
627:
620:
604:as a stable non-
588:
586:
585:
580:
578:
574:
573:
564:
563:
554:
553:
515:
508:
507:
498:
497:
473:
472:
452:
445:
444:
435:
434:
425:
424:
415:
414:
293:Grignard reagent
281:diallylberyllium
221:2 RMX → MR
192:transmetallation
182:+ R-Y → MR
80:oxidation states
68:group 2 elements
21:
2550:
2549:
2545:
2544:
2543:
2541:
2540:
2539:
2525:
2524:
2523:
2518:
2517:
2512:
2507:
2502:
2497:
2479:
2401:
2320:
2252:
2157:
1850:
1843:
1813:
1795:
1794:
1790:
1768:
1767:
1763:
1741:
1740:
1736:
1722:
1718:
1696:
1695:
1691:
1669:
1668:
1664:
1642:
1641:
1637:
1618:Organometallics
1614:
1613:
1609:
1573:
1572:
1565:
1544:
1540:
1518:
1514:
1498:
1494:
1478:
1474:
1455:
1451:
1423:
1422:
1418:
1396:
1395:
1391:
1368:
1367:
1363:
1348:
1344:
1314:
1313:
1309:
1287:
1286:
1282:
1254:
1253:
1249:
1227:
1226:
1222:
1200:
1199:
1195:
1173:
1172:
1168:
1150:
1149:
1145:
1130:
1123:
1100:
1099:
1095:
1063:
1062:
1058:
1035:
1034:
1030:
1010:
1006:
988:
984:
962:
961:
957:
953:
941:
927:The only known
925:
918:
893:
885:
881:
877:
873:
869:
853:Organostrontium
850:
848:Organostrontium
814:
808:
804:
801:polymeric (CaCH
695:
676:
621:
613:
612:
565:
555:
545:
499:
489:
464:
436:
426:
416:
406:
373:
372:
350:
343:
307:Rieke magnesium
299:salt metathesis
295:
289:
287:Organomagnesium
278:
269:
263:
261:Organoberyllium
235:
228:
224:
206:
202:
185:
181:
157:
135:
131:
119:
96:organomagnesium
92:organoberyllium
64:
62:Characteristics
44:group 2 element
23:
22:
15:
12:
11:
5:
2548:
2546:
2538:
2537:
2527:
2526:
2520:
2519:
2516:
2515:
2510:
2505:
2500:
2495:
2492:Chemical bonds
2488:
2487:
2485:
2481:
2480:
2476:
2475:
2470:
2465:
2460:
2455:
2450:
2445:
2440:
2435:
2430:
2425:
2420:
2415:
2410:
2405:
2398:
2395:
2394:
2389:
2384:
2379:
2374:
2369:
2364:
2359:
2354:
2349:
2344:
2339:
2334:
2329:
2324:
2317:
2314:
2313:
2309:
2308:
2305:
2302:
2299:
2296:
2293:
2290:
2287:
2284:
2281:
2278:
2275:
2272:
2267:
2264:
2261:
2256:
2249:
2244:
2240:
2239:
2236:
2231:
2226:
2221:
2216:
2211:
2206:
2201:
2196:
2191:
2186:
2181:
2176:
2171:
2166:
2161:
2154:
2149:
2143:
2142:
2137:
2132:
2127:
2122:
2117:
2112:
2107:
2102:
2097:
2092:
2087:
2082:
2077:
2072:
2067:
2062:
2060:
2055:
2049:
2048:
2043:
2038:
2033:
2028:
2023:
2018:
2013:
2008:
2003:
1998:
1993:
1988:
1983:
1978:
1973:
1968:
1966:
1961:
1955:
1954:
1949:
1944:
1939:
1934:
1929:
1924:
1919:
1913:
1912:
1909:
1904:
1899:
1894:
1889:
1884:
1879:
1873:
1872:
1869:
1867:
1865:
1863:
1855:
1852:
1851:
1844:
1842:
1841:
1834:
1827:
1819:
1812:
1811:
1788:
1761:
1734:
1716:
1689:
1662:
1635:
1607:
1563:
1538:
1512:
1492:
1472:
1449:
1416:
1389:
1361:
1342:
1323:(34): 5870–4.
1307:
1280:
1247:
1220:
1193:
1166:
1143:
1121:
1093:
1056:
1045:(4): 314–321.
1028:
1004:
982:
971:(7): 261–267.
954:
952:
949:
948:
947:
940:
937:
924:
921:
916:
892:
889:
883:
879:
875:
871:
867:
849:
846:
842:
841:
810:
806:
802:
791:
790:
774:
771:
759:
754:
746:
733:
725:
702:
698:
693:
670:
660:
654:allylpotassium
646:
633:
624:
602:calcium iodide
598:allylpotassium
590:
589:
577:
572:
568:
562:
558:
552:
548:
544:
541:
538:
535:
532:
529:
526:
523:
520:
514:
511:
506:
502:
496:
492:
488:
485:
482:
479:
476:
471:
467:
463:
460:
457:
451:
448:
443:
439:
433:
429:
423:
419:
413:
409:
405:
402:
399:
396:
393:
390:
387:
384:
381:
349:
346:
341:
288:
285:
276:
265:Main article:
262:
259:
234:
231:
230:
229:
226:
222:
218:
217:
209:
208:
204:
203:+ M → MR
200:
196:
195:
188:
187:
183:
179:
175:
174:
156:
153:
133:
129:
117:
63:
60:
38:refers to the
24:
14:
13:
10:
9:
6:
4:
3:
2:
2547:
2536:
2533:
2532:
2530:
2511:
2506:
2501:
2496:
2493:
2490:
2489:
2486:
2482:
2474:
2471:
2469:
2466:
2464:
2461:
2459:
2456:
2454:
2451:
2449:
2446:
2444:
2441:
2439:
2436:
2434:
2431:
2429:
2426:
2424:
2421:
2419:
2416:
2414:
2411:
2409:
2406:
2399:
2396:
2393:
2390:
2388:
2385:
2383:
2380:
2378:
2375:
2373:
2370:
2368:
2365:
2363:
2360:
2358:
2355:
2353:
2350:
2348:
2345:
2343:
2340:
2338:
2335:
2333:
2330:
2328:
2325:
2318:
2315:
2311:
2310:
2306:
2303:
2300:
2297:
2294:
2291:
2288:
2285:
2282:
2279:
2276:
2273:
2271:
2268:
2265:
2262:
2260:
2257:
2250:
2248:
2245:
2242:
2241:
2237:
2235:
2232:
2230:
2227:
2225:
2222:
2220:
2217:
2215:
2212:
2210:
2207:
2205:
2202:
2200:
2197:
2195:
2192:
2190:
2187:
2185:
2182:
2180:
2177:
2175:
2172:
2170:
2167:
2165:
2162:
2155:
2153:
2150:
2148:
2145:
2144:
2141:
2138:
2136:
2133:
2131:
2128:
2126:
2123:
2121:
2118:
2116:
2113:
2111:
2108:
2106:
2103:
2101:
2098:
2096:
2093:
2091:
2088:
2086:
2083:
2081:
2078:
2076:
2073:
2071:
2068:
2066:
2063:
2061:
2059:
2056:
2054:
2051:
2050:
2047:
2044:
2042:
2039:
2037:
2034:
2032:
2029:
2027:
2024:
2022:
2019:
2017:
2014:
2012:
2009:
2007:
2004:
2002:
1999:
1997:
1994:
1992:
1989:
1987:
1984:
1982:
1979:
1977:
1974:
1972:
1969:
1967:
1965:
1962:
1960:
1957:
1956:
1953:
1950:
1948:
1945:
1943:
1940:
1938:
1935:
1933:
1930:
1928:
1925:
1923:
1920:
1918:
1915:
1914:
1910:
1908:
1905:
1903:
1900:
1898:
1895:
1893:
1890:
1888:
1885:
1883:
1880:
1878:
1875:
1874:
1870:
1862:
1859:
1858:
1853:
1848:
1845:Compounds of
1840:
1835:
1833:
1828:
1826:
1821:
1820:
1817:
1807:
1803:
1799:
1792:
1789:
1784:
1780:
1776:
1772:
1765:
1762:
1757:
1753:
1749:
1745:
1738:
1735:
1732:
1728:
1725:
1720:
1717:
1712:
1708:
1704:
1700:
1693:
1690:
1685:
1681:
1677:
1673:
1666:
1663:
1658:
1654:
1650:
1646:
1639:
1636:
1631:
1627:
1623:
1619:
1611:
1608:
1603:
1599:
1594:
1589:
1585:
1581:
1577:
1570:
1568:
1564:
1561:
1557:
1553:
1550:
1549:
1542:
1539:
1536:
1532:
1528:
1524:
1523:
1516:
1513:
1510:
1506:
1502:
1496:
1493:
1490:
1486:
1482:
1476:
1473:
1470:
1466:
1462:
1459:
1453:
1450:
1444:
1439:
1435:
1431:
1427:
1420:
1417:
1412:
1408:
1404:
1400:
1393:
1390:
1385:
1381:
1377:
1373:
1365:
1362:
1359:
1355:
1351:
1346:
1343:
1338:
1334:
1330:
1326:
1322:
1318:
1311:
1308:
1303:
1299:
1295:
1291:
1284:
1281:
1275:
1270:
1266:
1262:
1258:
1251:
1248:
1243:
1239:
1235:
1231:
1224:
1221:
1216:
1212:
1208:
1204:
1197:
1194:
1189:
1185:
1181:
1177:
1170:
1167:
1162:
1158:
1154:
1147:
1144:
1141:
1137:
1133:
1128:
1126:
1122:
1117:
1113:
1109:
1105:
1097:
1094:
1088:
1083:
1079:
1075:
1072:(4): 348–52.
1071:
1067:
1060:
1057:
1052:
1048:
1044:
1041:
1040:
1032:
1029:
1026:
1025:3-527-28165-7
1022:
1018:
1014:
1008:
1005:
1002:
998:
995:
991:
986:
983:
978:
974:
970:
966:
959:
956:
950:
946:
943:
942:
938:
936:
934:
930:
922:
920:
914:
910:
905:
904:barium iodide
901:
897:
890:
864:
860:
858:
854:
847:
845:
839:
835:
834:
833:
831:
827:
823:
821:
816:
813:
800:
796:
772:
769:
757:
744:
731:
700:
696:
691:
668:
658:
644:
631:
622:
611:
610:
609:
607:
603:
599:
595:
570:
560:
550:
512:
509:
504:
494:
469:
449:
446:
441:
431:
421:
411:
371:
370:
369:
367:
366:diethyl ether
363:
362:methyllithium
359:
355:
348:Organocalcium
347:
345:
339:
338:Ate complexes
335:
331:
327:
322:
320:
316:
312:
308:
304:
300:
294:
286:
284:
282:
274:
268:
260:
258:
256:
252:
248:
244:
240:
232:
220:
219:
215:
211:
210:
198:
197:
193:
190:
189:
177:
176:
172:
169:
168:
167:
164:
162:
154:
149:
145:
141:
137:
127:
123:
115:
111:
109:
105:
104:dipole moment
101:
97:
93:
89:
85:
81:
77:
73:
69:
61:
59:
57:
53:
49:
45:
41:
37:
29:
19:
2513:Bond unknown
1800:(15): 1045.
1797:
1791:
1777:(14): 6130.
1774:
1770:
1764:
1750:(23): 8955.
1747:
1743:
1737:
1723:
1719:
1705:(20): 4253.
1702:
1698:
1692:
1675:
1671:
1665:
1648:
1644:
1638:
1621:
1617:
1610:
1583:
1579:
1551:
1546:
1541:
1526:
1520:
1515:
1500:
1495:
1480:
1475:
1460:
1457:
1452:
1433:
1429:
1419:
1402:
1398:
1392:
1375:
1371:
1364:
1349:
1345:
1320:
1316:
1310:
1293:
1289:
1283:
1264:
1260:
1250:
1233:
1229:
1223:
1206:
1202:
1196:
1179:
1175:
1169:
1152:
1146:
1131:
1107:
1103:
1096:
1069:
1065:
1059:
1042:
1037:
1031:
1016:
1012:
1007:
993:
989:
985:
968:
964:
958:
929:organoradium
928:
926:
923:Organoradium
896:Organobarium
895:
894:
891:Organobarium
878:(tms = Si(CH
857:Barbier-type
852:
851:
843:
828:tethered to
824:
817:
811:
792:
591:
356:reaction of
351:
323:
296:
280:
270:
236:
165:
158:
120:Be), with a
112:
65:
35:
34:
1296:: 101–108.
1267:(6): 1662.
1209:(8): 1601.
909:metallocene
859:reactions.
114:Metallocene
1678:(6): 760.
1651:(2): 341.
1236:(3): 271.
1182:(2): 482.
1104:Org. Synth
1015:(2nd Ed) (
951:References
815:compound.
606:pyrophoric
354:metathesis
319:octet rule
291:See also:
171:metathesis
2494:to carbon
933:acetylide
900:reduction
701:∘
475:⟶
334:Butadiene
247:strontium
233:Compounds
155:Synthesis
2529:Category
1602:15549740
1378:: 1724.
1337:20575128
1155:: 1308.
939:See also
692:→
212:via the
100:covalent
76:group 12
2312:
1436:: 460.
1110:: 147.
826:Olefins
678:calcium
243:Calcium
124:of 2.2
66:As the
42:of any
2484:Legend
1847:carbon
1600:
1335:
1023:
999:
684:iodide
681:
516:
453:
251:barium
186:+ Y-X'
1405:: 7.
874:(thf)
279:Be),
88:ionic
1703:2005
1598:PMID
1552:2009
1527:2009
1461:2018
1333:PMID
1021:ISBN
1017:1992
997:ISBN
994:2007
805:CHCH
600:and
360:and
313:and
249:and
225:+ MX
207:+ M'
94:and
2458:CEs
2453:CCf
2448:CBk
2443:CCm
2438:CAm
2433:CPu
2428:CNp
2418:CPa
2413:CTh
2392:CYb
2387:CTm
2382:CEr
2377:CHo
2372:CDy
2367:CTb
2362:CGd
2357:CEu
2352:CSm
2347:CPm
2342:CNd
2337:CPr
2332:CCe
2327:CLa
2307:Og
2304:Ts
2301:Lv
2298:Mc
2295:Fl
2292:Nh
2289:Cn
2286:Rg
2283:Ds
2280:Mt
2277:Hs
2274:Bh
2270:CSg
2266:Db
2263:Rf
2247:CRa
2243:Fr
2238:Rn
2234:CAt
2229:CPo
2224:CBi
2219:CPb
2214:CTl
2209:CHg
2204:CAu
2199:CPt
2194:CIr
2189:COs
2184:CRe
2174:CTa
2169:CHf
2164:CLu
2152:CBa
2147:CCs
2140:CXe
2130:CTe
2125:CSb
2120:CSn
2115:CIn
2110:CCd
2105:CAg
2100:CPd
2095:CRh
2090:CRu
2085:CTc
2080:CMo
2075:CNb
2070:CZr
2058:CSr
2053:CRb
2046:CKr
2041:CBr
2036:CSe
2031:CAs
2026:CGe
2021:CGa
2016:CZn
2011:CCu
2006:CNi
2001:CCo
1996:CFe
1991:CMn
1986:CCr
1976:CTi
1971:CSc
1964:CCa
1952:CAr
1947:CCl
1932:CSi
1927:CAl
1922:CMg
1917:CNa
1911:Ne
1882:CBe
1877:CLi
1871:He
1802:doi
1779:doi
1775:116
1752:doi
1748:113
1707:doi
1680:doi
1653:doi
1626:doi
1588:doi
1556:doi
1531:doi
1505:doi
1485:doi
1465:doi
1438:doi
1407:doi
1380:doi
1354:doi
1325:doi
1298:doi
1294:405
1269:doi
1238:doi
1234:170
1211:doi
1184:doi
1157:doi
1136:doi
1112:doi
1082:hdl
1074:doi
1047:doi
973:doi
913:Cp*
902:of
714:THF
665:CaI
364:in
303:SET
199:M'R
2531::
2473:No
2468:Md
2463:Fm
2423:CU
2408:Ac
2259:Lr
2179:CW
2135:CI
2065:CY
1981:CV
1959:CK
1942:CS
1937:CP
1907:CF
1902:CO
1897:CN
1892:CC
1887:CB
1861:CH
1773:.
1746:.
1701:.
1676:34
1674:.
1649:77
1647:.
1622:30
1620:.
1596:.
1584:43
1582:.
1578:.
1566:^
1525:,
1434:89
1432:.
1428:.
1403:29
1401:.
1376:32
1374:.
1331:.
1321:49
1319:.
1292:.
1265:28
1263:.
1259:.
1232:.
1207:37
1205:.
1180:92
1178:.
1124:^
1108:72
1106:.
1080:.
1068:.
969:21
967:.
935:.
777:KI
766:Ca
697:25
628:KC
594:Cp
368::
344:.
321:.
257:.
178:MX
1838:e
1831:t
1824:v
1808:.
1804::
1785:.
1781::
1758:.
1754::
1713:.
1709::
1686:.
1682::
1659:.
1655::
1632:.
1628::
1604:.
1590::
1558::
1533::
1507::
1487::
1467::
1446:.
1440::
1413:.
1409::
1386:.
1382::
1356::
1339:.
1327::
1304:.
1300::
1277:.
1271::
1244:.
1240::
1217:.
1213::
1190:.
1186::
1163:.
1159::
1138::
1118:.
1114::
1090:.
1084::
1076::
1070:4
1053:.
1049::
1043:2
979:.
975::
917:2
915:)
911:(
884:3
882:)
880:3
876:3
872:2
870:)
868:2
812:n
809:)
807:2
803:2
799:η
795:η
773:2
770:+
758:2
753:)
745:5
741:H
732:3
728:C
724:(
707:C
669:2
659:+
645:5
641:H
632:3
623:2
576:]
571:2
567:}
561:3
557:)
551:3
547:H
543:C
540:(
537:i
534:S
531:{
528:N
525:[
522:i
519:L
513:2
510:+
505:2
501:)
495:3
491:H
487:C
484:(
481:a
478:C
470:3
466:H
462:C
459:i
456:L
450:2
447:+
442:2
438:]
432:2
428:}
422:3
418:)
412:3
408:H
404:C
401:(
398:i
395:S
392:{
389:N
386:[
383:a
380:C
342:3
277:2
227:2
223:2
216::
205:2
201:2
194::
184:2
180:2
173::
134:2
130:2
126:D
118:2
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.