110:
133:
328:
359:
284:
214:
20:
1693:
1544:
1612:
1449:
54:
105:
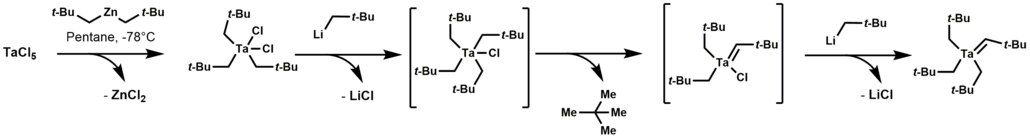
Tantalum alkylidene complexes arise by treating trialkyltantalum dichloride with alkyl lithium reagents. This reaction initially forms a thermally unstable tetraalkyl-monochloro-tantalum complex, which undergoes α-hydrogen elimination, followed by alkylation of the remaining chloride.
589:
Brennessel, William W.; Romanenkov, Alexander; Young, Victor G.; Ellis, John E. (2019). "Tantalum isocyanide complexes: TaI(CNDipp)6 (Dipp is 2,6-diisopropylphenyl) and ionic [Ta(CNDipp)7][Ta(CNDipp)6], a formal disproportionation product of the 17-electron Ta0 metalloradical
375:
of organotin compounds with tantalum(V) chloride. These organotantalum reagents promote the conjugate allylation of enones. Although the direct allylation of carbonyl groups is prevalent throughout the literature, little has been reported on the conjugate allylation of enones.
909:
Eisenberger, Patrick; Ayinla, Rashidat O.; Lauzon, Jean Michel P.; Schafer, Laurel L. (2009-10-19). "Tantalum–Amidate
Complexes for the Hydroaminoalkylation of Secondary Amines: Enhanced Substrate Scope and Enantioselective Chiral Amine Synthesis".
988:
Payne, Philippa R.; Garcia, Pierre; Eisenberger, Patrick; Yim, Jacky C.-H.; Schafer, Laurel L. (2013-05-03). "Tantalum
Catalyzed Hydroaminoalkylation for the Synthesis of α- and β-Substituted N-Heterocycles".
467:
McLain, S. J.; Wood, C. D.; Schrock, R. R. (1977-05-01). "Multiple metal-carbon bonds. 6. The reaction of niobium and tantalum neopentylidene complexes with simple olefins: a route to metallocyclopentanes".
765:
Takai, Kazuhiko; Kataoka, Y.; Utimoto, K. (1990-03-01). "Tantalum-alkyne complexes as synthetic intermediates. Stereoselective preparation of trisubstituted allylic alcohols from acetylenes and aldehydes".
730:
Bruck, M. A.; Copenhaver, A. S.; Wigley, D. E. (1987-10-01). "Alkyne cyclizations at reduced tantalum centers: synthesis and molecular structure of (.eta.6-C6Me6)Ta(O-2,6-i-Pr2C6H3)2Cl".
1032:
Shibata, Ikuya; Kano, Takeyoshi; Kanazawa, Nobuaki; Fukuoka, Shoji; Baba, Akio (2002-04-15). "Generation of
Organotantalum Reagents and Conjugate Addition to Enones".
953:
Dörfler, Jaika; Doye, Sven (2014-05-01). "A Commercially
Available Tantalum Catalyst for the Highly Regioselective Intermolecular Hydroaminoalkylation of Styrenes".
1127:
556:
J. E. Ellis; A. Davison (1976). "Tris[Bis(2-Methoxyethyl)Ether]Potassium and
Tetraphenylarsonium Hexacarbonylmetallates(1-) of Niobium and Tantalum".
633:
Pampaloni, G. (2010). "Aromatic hydrocarbons as ligands. Recent advances in the synthesis, the reactivity and the applications of bis(η6-arene) complexes".
320:. The chemistry developed by Maspero was later brought to fruition when Hartwig and Herzon reported the hydroaminoalkylation of olefins to form
45:. A wide variety of compound have been reported, initially with cyclopentadienyl and CO ligands. Oxidation states vary from Ta(V) to Ta(-I).
573:
520:
453:
1120:
660:
Labinger, Jay A.; Schwartz, Jeffrey; Townsend, John M. (1974-06-01). "Iodo- and hydridotantalum(III) complexes of dialkylacetylenes".
1830:
1045:
1113:
280:. Some tantalum-alkyne complexes are precursors to allylic alcohols. Tantalacyclopropenes are invoked as intermediates.
1825:
1682:
1677:
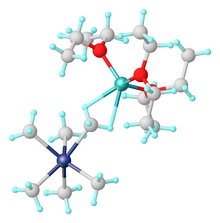
1672:
1667:
1662:
1657:
1652:
1647:
1642:
1637:
1632:
1627:
1617:
1560:
1454:
1375:
1370:
1227:
187:
1718:
1420:
1390:
1380:
1360:
1348:
1316:
1281:
1249:
1217:
1212:
1172:
503:
Endy Y.-J. Min; John E. Bercaw (2014). "Bis(η -Pentamethylcyclopentadienyl) Complexes of
Niobium and Tantalum".
340:
1187:
1151:
1105:
1763:
1758:
1753:
1748:
1743:
1738:
1733:
1728:
1723:
1708:
1698:
1549:
1524:
1519:
1504:
1489:
1469:
1415:
1343:
1326:
1276:
1271:
1266:
1261:
1237:
1197:
230:
under an atmosphere of CO gives the salts of . These same anions can be obtained by carbonylation of tantalum
168:
164:
132:
384:
Organotantalum compounds are of academic interest, but few or no commercial applications have been described.
327:
163:
Some of the first reported organotantalum complexes were cyclopentadienyl derivatives. These arise from the
1713:
1703:
1514:
1499:
1484:
1474:
1459:
1400:
1385:
1365:
1355:
1336:
1331:
1321:
1311:
1254:
1222:
1192:
1182:
358:
1622:
1537:
1479:
1442:
1437:
1425:
1405:
1395:
1301:
1296:
1291:
1232:
1207:
695:
Cotton, F. Albert; Hall, William T. (1979-08-01). "Reactions of tantalum(III) with alkynes and nitriles".
176:
172:
1430:
1242:
1177:
1167:
266:
109:
283:
1509:
1494:
1306:
1286:
1410:
66:
830:
615:
530:
348:
277:
31:
1092:
1057:
1049:
1014:
1006:
970:
935:
927:
891:
873:
822:
783:
747:
712:
677:
607:
569:
516:
485:
422:
250:
152:
1084:
1041:
998:
962:
919:
881:
865:
814:
775:
739:
704:
669:
642:
599:
561:
508:
477:
449:
440:
Kleinhenz, S.; Pfennig, V.; Seppelt, K. (1998). "Preparation and
Structures of , , , and ".
414:
344:
1075:
Yamamoto, Yoshinori; Asao, Naoki (1993-09-01). "Selective reactions using allylic metals".
213:
542:
372:
336:
321:
305:
70:
558:
355:
of the tantalum-carbon bond, and β-hydrogen abstraction affords the alkylamine product.
886:
849:
81:
1819:
1782:
42:
834:
619:
352:
405:
Schrock, Richard R. (1979-03-01). "Alkylidene complexes of niobium and tantalum".
19:
118:
565:
512:
646:
603:
454:
10.1002/(SICI)1521-3765(19980904)4:9<1687::AID-CHEM1687>3.0.CO;2-R
301:
238:
126:
1096:
1053:
1010:
974:
931:
877:
826:
787:
751:
716:
681:
489:
426:
262:
144:
1061:
1018:
966:
939:
923:
895:
611:
1046:
10.1002/1521-3773(20020415)41:8<1389::AID-ANIE1389>3.0.CO;2-D
818:
309:
140:
39:
1088:
779:
743:
708:
673:
481:
418:
148:
1002:
869:
1137:
122:
35:
850:"Direct, Catalytic Hydroaminoalkylation of Unactivated Olefins with
357:
326:
282:
231:
212:
131:
108:
53:
52:
175:. An example of this is the first transition metal trihydride,
1109:
801:
Clerici, Mario G.; Maspero, Federico (1980-01-01). "Catalytic
121:. They effect a number of reactions including: olefinations,
300:
Organotantalum compounds are invoked as intermediates in C-
125:
metathesis, hydroaminoalkylation of olefins, and conjugate
16:
Chemistry of compounds containing a carbon-to-tantalum bond
186:. More soluble and better developed are derivatives of
592:
560:. Inorganic Syntheses. Vol. 16. pp. 68–73.
1774:
151:react with tantalum alkylidene complexes to yield
848:Herzon, Seth B.; Hartwig, John F. (2007-05-01).
805:-Alkylation of Secondary Amines with Alkenes".
113:Synthesis of Tantalum Monoalkylidene Complexes
1121:
8:
331:Hartwig hydroaminoalkylation reaction scheme
76:Salts of are prepared by alkylation of TaF
1128:
1114:
1106:
362:Purposed mechanism of hydroaminoalkylation
1140:with other elements in the periodic table
885:
400:
398:
396:
249:Treatment of tantalum pentachloride with
858:Journal of the American Chemical Society
732:Journal of the American Chemical Society
697:Journal of the American Chemical Society
662:Journal of the American Chemical Society
470:Journal of the American Chemical Society
136:Tantalum Alkylidene Promoted Olefination
18:
912:Angewandte Chemie International Edition
392:
1157:
538:
528:
955:European Journal of Organic Chemistry
217:Tantalum Alkylidene Olefin Metathesis
7:
1799:Academic research, no widespread use
245:Tantalum arenes and alkyne complexes
276:Tantalum-alkyne complexes catalyze
49:Classes of organotantalum compounds
371:Organotantalum reagents arise via
222:Tantalum carbonyls and isocyanides
117:Tantalum alkylidene complexes are
14:
343:of the bisamide, which forms the
1691:
1610:
1542:
1447:
1147:
768:The Journal of Organic Chemistry
507:. Vol. 36. pp. 52–57.
635:Coordination Chemistry Reviews
505:Inorganic Syntheses: Volume 36
1:
407:Accounts of Chemical Research
1847:
566:10.1002/9780470132470.ch21
513:10.1002/9781118744994.ch11
241:complexes are also known.
188:pentamethylcyclopentadiene
159:Cyclopentadienyl complexes
1688:
1607:
1159:
1155:
1145:
647:10.1016/j.ccr.2009.05.014
604:10.1107/S2053229619000834
165:salt metathesis reactions
1831:Organometallic chemistry
296:Tantalum-amido complexes
287:Utimoto's Synthesis of (
169:sodium cyclopentadienide
62:Alkyl and aryl complexes
28:Organotantalum chemistry
1794:Many uses in chemistry
1789:Core organic chemistry
967:10.1002/ejoc.201400082
924:10.1002/anie.200903656
363:
341:β-hydrogen abstraction
332:
292:
218:
173:tantalum pentachloride
137:
114:
58:
24:
361:
330:
286:
267:aluminium trichloride
237:A number of tantalum
216:
135:
112:
56:
22:
819:10.1055/s-1980-29002
347:. Subsequent olefin
101:Alkylidene complexes
30:is the chemistry of
23:Tantalum-Carbon Bond
1089:10.1021/cr00022a010
780:10.1021/jo00293a008
744:10.1021/ja00255a056
709:10.1021/ja00511a064
674:10.1021/ja00819a047
482:10.1021/ja00452a064
419:10.1021/ar50135a004
278:cyclotrimerizations
67:Pentamethyltantalum
1826:Tantalum compounds
854:-Alkyl Arylamines"
364:
333:
293:
291:)-Allylic Alcohols
219:
138:
115:
59:
32:chemical compounds
25:
1813:
1812:
1769:
1768:
1034:Angewandte Chemie
1003:10.1021/ol400729v
961:(13): 2790–2797.
918:(44): 8361–8365.
870:10.1021/ja0718366
864:(21): 6690–6691.
738:(21): 6525–6527.
703:(17): 5094–5095.
668:(12): 4009–4011.
575:978-0-470-13247-0
522:978-1-118-74499-4
476:(10): 3519–3520.
251:hexamethylbenzene
226:Reduction of TaCl
153:olefin metathesis
1838:
1805:
1800:
1795:
1790:
1695:
1694:
1614:
1613:
1546:
1545:
1451:
1450:
1148:
1130:
1123:
1116:
1107:
1101:
1100:
1083:(6): 2207–2293.
1077:Chemical Reviews
1072:
1066:
1065:
1040:(8): 1447–1450.
1029:
1023:
1022:
997:(9): 2182–2185.
985:
979:
978:
950:
944:
943:
906:
900:
899:
889:
845:
839:
838:
798:
792:
791:
774:(6): 1707–1708.
762:
756:
755:
727:
721:
720:
692:
686:
685:
657:
651:
650:
641:(5–6): 402–419.
630:
624:
623:
586:
580:
579:
553:
547:
546:
540:
536:
534:
526:
500:
494:
493:
464:
458:
457:
437:
431:
430:
402:
345:metallaaziridine
306:secondary amines
69:was reported by
1846:
1845:
1841:
1840:
1839:
1837:
1836:
1835:
1816:
1815:
1814:
1809:
1808:
1803:
1798:
1793:
1788:
1770:
1692:
1611:
1543:
1448:
1141:
1134:
1104:
1074:
1073:
1069:
1031:
1030:
1026:
991:Organic Letters
987:
986:
982:
952:
951:
947:
908:
907:
903:
847:
846:
842:
800:
799:
795:
764:
763:
759:
729:
728:
724:
694:
693:
689:
659:
658:
654:
632:
631:
627:
588:
587:
583:
576:
555:
554:
550:
537:
527:
523:
502:
501:
497:
466:
465:
461:
439:
438:
434:
404:
403:
394:
390:
382:
373:transmetalation
369:
367:Transmetalation
339:may proceed by
337:catalytic cycle
319:
315:
298:
272:
260:
256:
247:
229:
224:
209:
205:
201:
197:
193:
190:such as Cp*TaCl
184:
180:
161:
103:
95:
91:
79:
71:Richard Schrock
64:
51:
17:
12:
11:
5:
1844:
1842:
1834:
1833:
1828:
1818:
1817:
1811:
1810:
1807:
1806:
1801:
1796:
1791:
1786:
1783:Chemical bonds
1779:
1778:
1776:
1772:
1771:
1767:
1766:
1761:
1756:
1751:
1746:
1741:
1736:
1731:
1726:
1721:
1716:
1711:
1706:
1701:
1696:
1689:
1686:
1685:
1680:
1675:
1670:
1665:
1660:
1655:
1650:
1645:
1640:
1635:
1630:
1625:
1620:
1615:
1608:
1605:
1604:
1600:
1599:
1596:
1593:
1590:
1587:
1584:
1581:
1578:
1575:
1572:
1569:
1566:
1563:
1558:
1555:
1552:
1547:
1540:
1535:
1531:
1530:
1527:
1522:
1517:
1512:
1507:
1502:
1497:
1492:
1487:
1482:
1477:
1472:
1467:
1462:
1457:
1452:
1445:
1440:
1434:
1433:
1428:
1423:
1418:
1413:
1408:
1403:
1398:
1393:
1388:
1383:
1378:
1373:
1368:
1363:
1358:
1353:
1351:
1346:
1340:
1339:
1334:
1329:
1324:
1319:
1314:
1309:
1304:
1299:
1294:
1289:
1284:
1279:
1274:
1269:
1264:
1259:
1257:
1252:
1246:
1245:
1240:
1235:
1230:
1225:
1220:
1215:
1210:
1204:
1203:
1200:
1195:
1190:
1185:
1180:
1175:
1170:
1164:
1163:
1160:
1158:
1156:
1154:
1146:
1143:
1142:
1135:
1133:
1132:
1125:
1118:
1110:
1103:
1102:
1067:
1024:
980:
945:
901:
840:
813:(4): 305–306.
793:
757:
722:
687:
652:
625:
598:(2): 135–140.
590:Ta(CNDipp)6".
581:
574:
548:
539:|journal=
521:
495:
459:
432:
391:
389:
386:
381:
378:
368:
365:
317:
313:
297:
294:
270:
258:
254:
246:
243:
227:
223:
220:
207:
203:
199:
195:
191:
182:
178:
160:
157:
102:
99:
98:
97:
96:→ Li + 5 LiF
93:
89:
82:methyl lithium
77:
63:
60:
57:Structure of .
50:
47:
15:
13:
10:
9:
6:
4:
3:
2:
1843:
1832:
1829:
1827:
1824:
1823:
1821:
1802:
1797:
1792:
1787:
1784:
1781:
1780:
1777:
1773:
1765:
1762:
1760:
1757:
1755:
1752:
1750:
1747:
1745:
1742:
1740:
1737:
1735:
1732:
1730:
1727:
1725:
1722:
1720:
1717:
1715:
1712:
1710:
1707:
1705:
1702:
1700:
1697:
1690:
1687:
1684:
1681:
1679:
1676:
1674:
1671:
1669:
1666:
1664:
1661:
1659:
1656:
1654:
1651:
1649:
1646:
1644:
1641:
1639:
1636:
1634:
1631:
1629:
1626:
1624:
1621:
1619:
1616:
1609:
1606:
1602:
1601:
1597:
1594:
1591:
1588:
1585:
1582:
1579:
1576:
1573:
1570:
1567:
1564:
1562:
1559:
1556:
1553:
1551:
1548:
1541:
1539:
1536:
1533:
1532:
1528:
1526:
1523:
1521:
1518:
1516:
1513:
1511:
1508:
1506:
1503:
1501:
1498:
1496:
1493:
1491:
1488:
1486:
1483:
1481:
1478:
1476:
1473:
1471:
1468:
1466:
1463:
1461:
1458:
1456:
1453:
1446:
1444:
1441:
1439:
1436:
1435:
1432:
1429:
1427:
1424:
1422:
1419:
1417:
1414:
1412:
1409:
1407:
1404:
1402:
1399:
1397:
1394:
1392:
1389:
1387:
1384:
1382:
1379:
1377:
1374:
1372:
1369:
1367:
1364:
1362:
1359:
1357:
1354:
1352:
1350:
1347:
1345:
1342:
1341:
1338:
1335:
1333:
1330:
1328:
1325:
1323:
1320:
1318:
1315:
1313:
1310:
1308:
1305:
1303:
1300:
1298:
1295:
1293:
1290:
1288:
1285:
1283:
1280:
1278:
1275:
1273:
1270:
1268:
1265:
1263:
1260:
1258:
1256:
1253:
1251:
1248:
1247:
1244:
1241:
1239:
1236:
1234:
1231:
1229:
1226:
1224:
1221:
1219:
1216:
1214:
1211:
1209:
1206:
1205:
1201:
1199:
1196:
1194:
1191:
1189:
1186:
1184:
1181:
1179:
1176:
1174:
1171:
1169:
1166:
1165:
1161:
1153:
1150:
1149:
1144:
1139:
1136:Compounds of
1131:
1126:
1124:
1119:
1117:
1112:
1111:
1108:
1098:
1094:
1090:
1086:
1082:
1078:
1071:
1068:
1063:
1059:
1055:
1051:
1047:
1043:
1039:
1035:
1028:
1025:
1020:
1016:
1012:
1008:
1004:
1000:
996:
992:
984:
981:
976:
972:
968:
964:
960:
956:
949:
946:
941:
937:
933:
929:
925:
921:
917:
913:
905:
902:
897:
893:
888:
883:
879:
875:
871:
867:
863:
859:
855:
853:
844:
841:
836:
832:
828:
824:
820:
816:
812:
808:
804:
797:
794:
789:
785:
781:
777:
773:
769:
761:
758:
753:
749:
745:
741:
737:
733:
726:
723:
718:
714:
710:
706:
702:
698:
691:
688:
683:
679:
675:
671:
667:
663:
656:
653:
648:
644:
640:
636:
629:
626:
621:
617:
613:
609:
605:
601:
597:
593:
585:
582:
577:
571:
567:
563:
559:
552:
549:
544:
532:
524:
518:
514:
510:
506:
499:
496:
491:
487:
483:
479:
475:
471:
463:
460:
455:
451:
447:
443:
436:
433:
428:
424:
420:
416:
413:(3): 98–104.
412:
408:
401:
399:
397:
393:
387:
385:
379:
377:
374:
366:
360:
356:
354:
350:
346:
342:
338:
329:
325:
323:
311:
307:
303:
295:
290:
285:
281:
279:
274:
268:
264:
252:
244:
242:
240:
235:
233:
221:
215:
211:
189:
185:
174:
170:
166:
158:
156:
154:
150:
146:
142:
134:
130:
129:of enones.
128:
124:
120:
111:
107:
100:
87:
86:
85:
83:
74:
72:
68:
61:
55:
48:
46:
44:
43:chemical bond
41:
37:
34:containing a
33:
29:
21:
1804:Bond unknown
1464:
1080:
1076:
1070:
1037:
1033:
1027:
994:
990:
983:
958:
954:
948:
915:
911:
904:
861:
857:
851:
843:
810:
806:
802:
796:
771:
767:
760:
735:
731:
725:
700:
696:
690:
665:
661:
655:
638:
634:
628:
595:
591:
584:
557:
551:
504:
498:
473:
469:
462:
445:
442:Chem. Eur. J
441:
435:
410:
406:
383:
380:Applications
370:
353:protonolysis
334:
312:using Ta(NMe
299:
288:
275:
248:
236:
225:
162:
139:
119:nucleophilic
116:
104:
75:
65:
27:
26:
448:(9): 1687.
322:alkylamines
234:complexes.
1820:Categories
388:References
302:alkylation
239:isocyanide
155:products.
127:allylation
1785:to carbon
1097:0009-2665
1054:1521-3757
1011:1523-7060
975:1099-0690
932:1521-3773
878:0002-7863
827:0039-7881
807:Synthesis
788:0022-3263
752:0002-7863
717:0002-7863
682:0002-7863
541:ignored (
531:cite book
490:0002-7863
427:0001-4842
349:insertion
310:1-alkenes
263:aluminium
202:, and Cp*
145:propylene
73:in 1974.
1062:19750774
1019:23600625
940:19787670
896:17474747
835:94579838
620:73450348
612:30720451
141:Ethylene
92:+ 6 LiCH
40:tantalum
1603:
887:2590937
149:styrene
1775:Legend
1138:carbon
1095:
1060:
1052:
1017:
1009:
973:
938:
930:
894:
884:
876:
833:
825:
786:
750:
715:
680:
618:
610:
572:
519:
488:
425:
269:gives
265:, and
147:, and
123:olefin
80:using
36:carbon
831:S2CID
616:S2CID
308:with
232:arene
194:, Cp*
1093:ISSN
1058:PMID
1050:ISSN
1015:PMID
1007:ISSN
971:ISSN
959:2014
936:PMID
928:ISSN
892:PMID
874:ISSN
823:ISSN
811:1980
784:ISSN
748:ISSN
713:ISSN
678:ISSN
608:PMID
570:ISBN
543:help
517:ISBN
486:ISSN
423:ISSN
335:The
198:TaCl
171:and
38:-to-
1749:CEs
1744:CCf
1739:CBk
1734:CCm
1729:CAm
1724:CPu
1719:CNp
1709:CPa
1704:CTh
1683:CYb
1678:CTm
1673:CEr
1668:CHo
1663:CDy
1658:CTb
1653:CGd
1648:CEu
1643:CSm
1638:CPm
1633:CNd
1628:CPr
1623:CCe
1618:CLa
1598:Og
1595:Ts
1592:Lv
1589:Mc
1586:Fl
1583:Nh
1580:Cn
1577:Rg
1574:Ds
1571:Mt
1568:Hs
1565:Bh
1561:CSg
1557:Db
1554:Rf
1538:CRa
1534:Fr
1529:Rn
1525:CAt
1520:CPo
1515:CBi
1510:CPb
1505:CTl
1500:CHg
1495:CAu
1490:CPt
1485:CIr
1480:COs
1475:CRe
1465:CTa
1460:CHf
1455:CLu
1443:CBa
1438:CCs
1431:CXe
1421:CTe
1416:CSb
1411:CSn
1406:CIn
1401:CCd
1396:CAg
1391:CPd
1386:CRh
1381:CRu
1376:CTc
1371:CMo
1366:CNb
1361:CZr
1349:CSr
1344:CRb
1337:CKr
1332:CBr
1327:CSe
1322:CAs
1317:CGe
1312:CGa
1307:CZn
1302:CCu
1297:CNi
1292:CCo
1287:CFe
1282:CMn
1277:CCr
1267:CTi
1262:CSc
1255:CCa
1243:CAr
1238:CCl
1223:CSi
1218:CAl
1213:CMg
1208:CNa
1202:Ne
1173:CBe
1168:CLi
1162:He
1085:doi
1042:doi
1038:114
999:doi
963:doi
920:doi
882:PMC
866:doi
862:129
815:doi
776:doi
740:doi
736:109
705:doi
701:101
670:doi
643:doi
639:254
600:doi
562:doi
509:doi
478:doi
450:doi
415:doi
304:of
261:),
206:TaH
181:TaH
167:of
88:TaF
1822::
1764:No
1759:Md
1754:Fm
1714:CU
1699:Ac
1550:Lr
1470:CW
1426:CI
1356:CY
1272:CV
1250:CK
1233:CS
1228:CP
1198:CF
1193:CO
1188:CN
1183:CC
1178:CB
1152:CH
1091:.
1081:93
1079:.
1056:.
1048:.
1036:.
1013:.
1005:.
995:15
993:.
969:.
957:.
934:.
926:.
916:48
914:.
890:.
880:.
872:.
860:.
856:.
829:.
821:.
809:.
782:.
772:55
770:.
746:.
734:.
711:.
699:.
676:.
666:96
664:.
637:.
614:.
606:.
596:75
594:.
568:.
535::
533:}}
529:{{
515:.
484:.
474:99
472:.
444:.
421:.
411:12
409:.
395:^
351:,
324::
273:.
257:Me
253:(C
210:.
177:Cp
143:,
84::
1129:e
1122:t
1115:v
1099:.
1087::
1064:.
1044::
1021:.
1001::
977:.
965::
942:.
922::
898:.
868::
852:N
837:.
817::
803:C
790:.
778::
754:.
742::
719:.
707::
684:.
672::
649:.
645::
622:.
602::
578:.
564::
545:)
525:.
511::
492:.
480::
456:.
452::
446:4
429:.
417::
318:5
316:)
314:2
289:E
271:2
259:6
255:6
228:5
208:3
204:2
200:2
196:2
192:4
183:3
179:2
94:3
90:5
78:5
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.