1315:
715:
834:
621:
827:
805:
674:
587:
768:
861:
specifically, its 4fn electron configurations have almost no effect on its chemical reactivity and its electrostatic interactions require optimizing through ligand geometries. Moreover, the reactivity of the f-block element complexes relies heavily on their sterics. In other words, a sterically saturated structure offers the best stability, and so, both ligand size or metal size can be altered to modify the reactivity. These special properties allow the following reactions to occur.
875:
33:
910:
925:
860:
Ln, have extremely negative reduction potentials of -2.7 to -3.9 Volts versus the standard hydrogen electrode (NHE). Furthermore, in comparison with d-orbitals of transition metals, the radial extension of their 4f-orbitals are really small and limited, which greatly reduces the orbital effects. More
340:
U and its uranium-chloride bond (2.90 Å) is relatively longer than the uranium-chloride bonds of other analogues. Its existence also indicates that the larger f-block elements are capable of accommodating additional ligands in addition to the three cyclopentadienyl ligands resulting in the isolation
169:
Ln (Ln = La, Ce, Pr, Nd, Sm and Gd). However, their significance is limited more to their existences and structures than to their reactivity. The cyclopentadienyl ligands of f-block metallocenes were considered as inert ancillary ligands, only capable of enhancing their stability and solubility, but
1309:
Sm are able to provide strong reductivity and so this type of reaction was named as SIR. Due to the strong steric hindrance, one ligand cannot bind to the metal center at the ideal distance and so the complex is not stable. Thus, the anion is more inclined to become oxidized and leave the complex,
219:
Sm, making a breakthrough in f-block metallocenes, since both of these two organosamarium(II) complexes were unexpectedly found to participate in the coordination, activation and transformation of a variety of unsaturated compounds, including olefins, dinitrogen, internal alkynens, phosphaalkynes,
1287:
550:
187:, was introduced to prepare the lanthanide complexes with all metals in the series. Apart from improving the stability and solubility of the complexes, it was demonstrated to participate in organometallic reactions. Subsequently,
869:
Like alkyl group, the electron-rich ligand of f-block metallocenes can act as a nucleophile during organometallic reactions. For example, they can polymerize olefins, and participate in ring opening polymerizations, etc.
946:
395:
170:
not their reactivity. In addition, only late and small metals in the lanthanide series, i.e., elements from Sm to Lu, are trivalent metallocene complexes,
1314:
2186:
116:
54:
47:
97:
1282:{\displaystyle {\begin{array}{l}{\ce {(C5Me5)2Sm->{e^{-}}+{+}}}\\{\ce {(C5Me5)3Sm->{e^{-}}+{+}+1/2(C5Me5)2}}\\\end{array}}}
69:
221:
920:
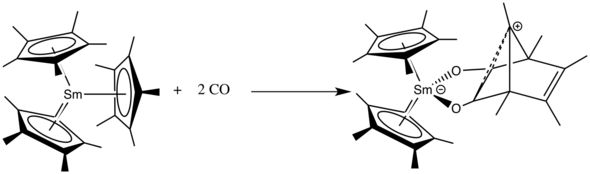
The f-block metallocenes are able to undergo insertion reactions of compounds like carbon monoxide, nitriles or isocyanates.
844:
Unlike d-block elements, f-block elements do not follow 18-electron rule due to their f-orbitals. The following complexes, (C
940:
Since f-block metallocenes are very electron-rich, they tend to lose one electron and a pentamethylcyclopentadienyl ligand.
76:
228:
and other trivalent f-block element complexes. Subsequently, tris(pentamethylcyclopentadienyl) lanthanide complexes, (C
157:
The first prepared and well-characterized f-block metallocenes were the tris(cyclopentadienyl) lanthanide complexes, (C
2191:
83:
188:
756:
M, such as THF, nitriles or isonitriles, should be avoided. Therefore, the following routes are possible options:
43:
714:
65:
833:
620:
826:
804:
673:
586:
1756:
Evans, W. J.; Drummond, D. K.; Chamberlain, L. R.; Doedens, R. J.; Bott, S. G.; Zhang, H.; Atwood, J. L.
767:
146:
142:
1927:
Blake, P. C.; Edelstein, N. M.; Hitchcock, P. B.; Kot, W. K.; Lappert, M. F.; Shalimoff, G. V.; Tian, S.
545:{\displaystyle {\begin{matrix}{}\\{\ce {{LnCl3}+ 3NaC5H5 -> {(C5H5)3Ln}+ 3NaCl}}\\{}\end{matrix}}}
240:
Ln, and their relevant complexes were synthesized from Sm complexes. These metallocenes included (C
2130:
Evans, W. J.; Davis, B. L.; Perotti, J. M.; Kozimor, S.; Ziller, J. W. Manuscript in preparation.
90:
138:
759:(i) For M=Ln including La, Ce, Pr, Nd and Gd, unsolvated cation route is preferred since
874:
389:
I. the synthesis of the first f-block metallocenes is described by following equation:
130:
2180:
1480:
Tilley, T. D.; Anderson, R. A.; Spencer, B.; Ruben, H.; Zalkin, A.; Templeton, D. H.
732:
M, the starting materials and the reaction conditions require optimizing to ensure (C
698:
An alternative unsolvated cation pathway prohibits THF during the reaction since (C
17:
224:(PAHs). Moreover, due to its strong reducing potential, it was used to synthesize
744:
M is the most favored product. In addition, compounds capable of reacting with (C
32:
2110:
Evans, W. J.; Ulibarri, T. A.; Chamberlain, L. R.; Ziller, J. W.; Alvarez, D.
909:
220:
carbon monoxide, carbon dioxide, isonitriles, diazine derivatives, imines and
797:
Ce requires the usage of silylated glassware since they are easily oxidized.
669:
Sm can also be prepared from trivalent precursors, without ring opening THF.
288:). Later, one tris(pentamethylcyclopentadienyl) f-element halide complex, (C
1616:
Evans, W. J.; Giarikos, D. G.; Robledo, C. B.; Leong, V. S.; Ziller, J. W.
924:
951:
300:
UCl, was successfully isolated as the intermediate of the formation of (C
905:, the Cp and other similar ligands can be removed in the following way.
679:
This solvated cation route generally allows the preparation of all (C
582:
Sm was prepared via exploratory Sm chemistry with cyclooctatetraene:
1656:
Evans, W. J.; Grate, J. W.; Hughes, L. A.; Zhang, H.; Atwood, J. L.
463:
1553:
Evans, W. J.; Chamberlain, L. R.; Ulibarri, T. A.; Ziller, J. W.
800:(ii) For M=actinide like U, solvated cation route can be used.
1410:; Hartley, F. R., Patai, S., Eds.; John Wiley: New York, 1982.
26:
1966:
Evans, W. J.; Forrestal, K. J.; Leman, J. T.; Ziller, J. W.
604:
Sm can be efficiently synthesized from a Sm precursor and (C
1716:
Evans, W. J.; Drummond, D. K.; Bott, S. G.; Atwood, J. L.
1884:
Evans, W. J.; Nyce, G. W.; Johnston, M. A.; Ziller, J. W.
1636:
Recknagel, A.; Stalke, D.; Roesky, H. W.; Edelmann, F. T.
1271:
1258:
1245:
1202:
1189:
1176:
1131:
1118:
1105:
1067:
1054:
1041:
996:
983:
970:
514:
501:
488:
455:
442:
421:
1596:
Evans, W. J.; Bloom, I.; Hunter, W. E.; Atwood, J. L.
400:
949:
398:
145:
metal and a set of electron-rich ligands such as the
642:) is used since it is more readily available than (C
1281:
885:Especially in the presence of Lewis acids like B(C
544:
174:In 1980, the pentamethylcyclopentadienyl ligand, C
2139:Evans, W. J.; Forrestal, K. J.; Ziller, J. W.
2011:Evans, W. J.; Forrestal, K. J.; Ziller, J. W.
1310:resulting in a highly reducing metal complex.
1819:Evans, W. J.; Gonzales, S. L.; Ziller, J. W.
1796:Evans, W. J.; Gonzales, S. L.; Ziller, J. W.
1573:Evans, W. J.; Ulibarri, T. A.; Ziller, J. W.
1533:Evans, W. J.; Ulibarri, T. A.; Ziller, J. W.
1520:Evans, W. J.; Ulibarri, T. A.; Ziller, J. W.
8:
2090:Evans, W. J.; DeCoster, D. M.; Greaves, J.
1986:Evans, W. J.; Seibel, C. A.; Ziller, J. W.
1676:Evans, W. J.; Seibel, C. A.; Ziller, J. W.
1500:Evans, W. J.; Hughes, L. A.; Hanusa, T. P.
1839:Evans, W. J.; Keyer, R. A.; Ziller, J. W.
191:and his coworkers successfully isolated (C
1946:Evans, W. J.; Nyce, G. W.; Ziller, J. W.
1270:
1265:
1257:
1252:
1244:
1239:
1231:
1212:
1201:
1196:
1188:
1183:
1175:
1170:
1162:
1157:
1155:
1145:
1140:
1130:
1125:
1117:
1112:
1104:
1099:
1091:
1089:
1077:
1066:
1061:
1053:
1048:
1040:
1035:
1027:
1022:
1020:
1010:
1005:
995:
990:
982:
977:
969:
964:
956:
954:
950:
948:
720:III. Generally, in order to synthesize (C
536:
527:
513:
508:
500:
495:
487:
482:
474:
472:
464:
458:
454:
449:
441:
436:
431:
420:
415:
410:
409:
403:
399:
397:
117:Learn how and when to remove this message
1326:
2066:
2064:
695:is known for all lanthanide elements.
328:UCl has a very similar structure as (C
53:Please improve this article by adding
2007:
2005:
2003:
1942:
1940:
1907:
1905:
1903:
1901:
1880:
1878:
1876:
1815:
1813:
1592:
1590:
7:
1476:
1474:
1472:
1378:
1376:
1355:
1353:
1332:
1330:
1297:Sterically crowded complexes like (C
1293:Sterically induced reduction (SIR)
25:
1439:Wolczanski, P. T.; Bercaw, J. E.
1359:Birmingham, J. M.; Wilkinson, G.
1336:Wilkinson, G.; Birmingham, J. M.
316:. It is worthy mentioning that (C
1313:
923:
908:
873:
832:
825:
803:
766:
713:
672:
619:
585:
222:polycyclic aromatic hydrocarbons
31:
1859:Evans, W. J.; Ulibarri, T. A.
1776:Evans, W. J.; Drummond, D. K.
1736:Evans, W. J.; Drummond, D. K.
1696:Evans, W. J.; Drummond, D. K.
1261:
1232:
1208:
1192:
1163:
1158:
1137:
1121:
1092:
1073:
1057:
1028:
1023:
1002:
986:
957:
592:(ii) Similar to method (i), (C
504:
475:
341:of the following complexes: (C
1:
55:secondary or tertiary sources
1461:J. Chem. Soc., Chem. Commun.
773:Notably, the synthesis of (C
763:complexes are too reactive.
1911:Evans, W. J.; Davis, B. J.
1638:Angew. Chem. Int. Ed. Engl.
1382:Evans, W. J.; Wayda, A. L.
654:Sm and does not react THF.
2208:
1404:The Chemistry of the Metal
2187:Organometallic compounds
1283:
710:Sm can ring open THF.
657:(iii) additionally, (C
546:
147:cyclopentadienyl anion
42:relies excessively on
2033:Adv. Organomet. Chem.
1284:
865:Alkyl-like reactivity
822:MZ with Z=X, H, etc.
555:II. Preparation of (C
547:
66:"F-block metallocene"
947:
396:
135:f-block metallocenes
1929:J. Organomet. Chem.
1841:J. Organomet. Chem.
1273:
1260:
1247:
1204:
1191:
1178:
1133:
1120:
1107:
1069:
1056:
1043:
998:
985:
972:
936:Ordinary reductions
810:IV. Synthesis of (C
691:Ln complexes since
626:In this pathway, (C
516:
503:
490:
469:
457:
444:
423:
18:F-Block Metallocene
2192:Sandwich compounds
1279:
1277:
1248:
1235:
1230:
1179:
1166:
1161:
1108:
1095:
1090:
1044:
1031:
1026:
973:
960:
955:
542:
540:
491:
478:
473:
445:
432:
411:
139:sandwich compounds
2161:Coord. Chem. Rev.
2141:J. Am. Chem. Soc.
2013:J. Am. Chem. Soc.
1988:J. Am. Chem. Soc.
1918:, 102, 2119−2136.
1886:J. Am. Chem. Soc.
1861:J. Am. Chem. Soc.
1821:J. Am. Chem. Soc.
1798:J. Am. Chem. Soc.
1778:J. Am. Chem. Soc.
1758:J. Am. Chem. Soc.
1738:J. Am. Chem. Soc.
1658:J. Am. Chem. Soc.
1598:J. Am. Chem. Soc.
1575:J. Am. Chem. Soc.
1555:J. Am. Chem. Soc.
1535:J. Am. Chem. Soc.
1522:J. Am. Chem. Soc.
1502:J. Am. Chem. Soc.
1421:Coord. Chem. Rev.
1361:J. Am. Chem. Soc.
1338:J. Am. Chem. Soc.
1251:
1238:
1229:
1207:
1182:
1169:
1144:
1136:
1111:
1098:
1072:
1047:
1034:
1009:
1001:
976:
963:
530:
519:
494:
481:
470:
467:
448:
435:
414:
141:consisting of an
127:
126:
119:
101:
16:(Redirected from
2199:
2171:
2157:
2151:
2137:
2131:
2128:
2122:
2108:
2102:
2088:
2082:
2068:
2059:
2058:, 46, 3435-3449.
2049:
2043:
2029:
2023:
2009:
1998:
1984:
1978:
1964:
1958:
1944:
1935:
1925:
1919:
1909:
1896:
1882:
1871:
1857:
1851:
1837:
1831:
1817:
1808:
1794:
1788:
1774:
1768:
1754:
1748:
1734:
1728:
1714:
1708:
1694:
1688:
1674:
1668:
1654:
1648:
1634:
1628:
1614:
1608:
1594:
1585:
1571:
1565:
1551:
1545:
1531:
1525:
1518:
1512:
1498:
1492:
1478:
1467:
1457:
1451:
1437:
1431:
1417:
1411:
1402:Evans, W. J. In
1400:
1394:
1380:
1371:
1357:
1348:
1334:
1317:
1288:
1286:
1285:
1280:
1278:
1274:
1272:
1269:
1264:
1259:
1256:
1249:
1246:
1243:
1236:
1222:
1218:
1217:
1216:
1211:
1205:
1203:
1200:
1195:
1190:
1187:
1180:
1177:
1174:
1167:
1151:
1150:
1149:
1142:
1134:
1132:
1129:
1124:
1119:
1116:
1109:
1106:
1103:
1096:
1084:
1083:
1082:
1081:
1076:
1070:
1068:
1065:
1060:
1055:
1052:
1045:
1042:
1039:
1032:
1016:
1015:
1014:
1007:
999:
997:
994:
989:
984:
981:
974:
971:
968:
961:
927:
912:
877:
836:
829:
807:
770:
717:
676:
623:
589:
570:(i) the first (C
551:
549:
548:
543:
541:
537:
531:
528:
520:
517:
515:
512:
507:
502:
499:
492:
489:
486:
479:
471:
468:
465:
459:
456:
453:
446:
443:
440:
433:
424:
422:
419:
412:
404:
189:William J. Evans
186:
185:
122:
115:
111:
108:
102:
100:
59:
35:
27:
21:
2207:
2206:
2202:
2201:
2200:
2198:
2197:
2196:
2177:
2176:
2175:
2174:
2158:
2154:
2138:
2134:
2129:
2125:
2112:Organometallics
2109:
2105:
2092:Macromolecules.
2089:
2085:
2069:
2062:
2050:
2046:
2030:
2026:
2010:
2001:
1985:
1981:
1968:Organometallics
1965:
1961:
1948:Organometallics
1945:
1938:
1926:
1922:
1910:
1899:
1883:
1874:
1858:
1854:
1838:
1834:
1818:
1811:
1795:
1791:
1775:
1771:
1755:
1751:
1735:
1731:
1718:Organometallics
1715:
1711:
1698:Organometallics
1695:
1691:
1675:
1671:
1655:
1651:
1635:
1631:
1618:Organometallics
1615:
1611:
1595:
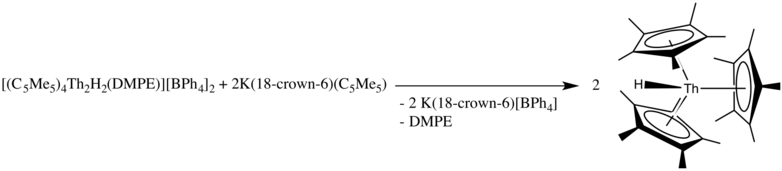
1588:
1572:
1568:
1552:
1548:
1532:
1528:
1519:
1515:
1499:
1495:
1479:
1470:
1458:
1454:
1441:Acc. Chem. Res.
1438:
1434:
1418:
1414:
1401:
1397:
1381:
1374:
1358:
1351:
1335:
1328:
1323:
1308:
1304:
1300:
1295:
1276:
1275:
1156:
1141:
1086:
1085:
1021:
1006:
945:
944:
938:
933:
931:Redox chemistry
918:
904:
900:
896:
892:
888:
883:
881:Ligand cleavage
867:
859:
855:
851:
847:
842:
821:
817:
813:
796:
792:
788:
784:
780:
776:
762:
755:
751:
747:
743:
739:
735:
731:
727:
723:
709:
705:
701:
694:
690:
686:
682:
668:
664:
660:
653:
649:
645:
641:
637:
633:
629:
615:
611:
607:
603:
599:
595:
581:
577:
573:
566:
562:
558:
539:
538:
533:
532:
406:
405:
394:
393:
387:
380:
376:
372:
368:
364:
360:
356:
352:
348:
344:
339:
335:
331:
327:
323:
319:
315:
311:
307:
303:
299:
295:
291:
287:
283:
279:
275:
271:
267:
263:
259:
255:
251:
247:
243:
239:
235:
231:
227:
218:
214:
210:
206:
202:
198:
194:
184:
181:
180:
179:
177:
173:
168:
164:
160:
155:
137:are a class of
123:
112:
106:
103:
60:
58:
52:
48:primary sources
36:
23:
22:
15:
12:
11:
5:
2205:
2203:
2195:
2194:
2189:
2179:
2178:
2173:
2172:
2152:
2132:
2123:
2103:
2083:
2060:
2044:
2024:
1999:
1979:
1959:
1936:
1920:
1897:
1872:
1852:
1832:
1809:
1789:
1769:
1749:
1729:
1709:
1689:
1669:
1649:
1629:
1609:
1586:
1566:
1546:
1526:
1513:
1493:
1468:
1459:Watson, P. L.
1452:
1432:
1412:
1395:
1372:
1349:
1325:
1324:
1322:
1319:
1306:
1302:
1298:
1294:
1291:
1290:
1289:
1268:
1263:
1255:
1242:
1234:
1228:
1225:
1221:
1215:
1210:
1199:
1194:
1186:
1173:
1165:
1160:
1154:
1148:
1139:
1128:
1123:
1115:
1102:
1094:
1088:
1087:
1080:
1075:
1064:
1059:
1051:
1038:
1030:
1025:
1019:
1013:
1004:
993:
988:
980:
967:
959:
953:
952:
937:
934:
932:
929:
917:
914:
902:
898:
894:
890:
886:
882:
879:
866:
863:
857:
853:
849:
845:
841:
838:
819:
815:
811:
794:
790:
786:
782:
778:
774:
760:
753:
749:
745:
741:
737:
733:
729:
725:
721:
707:
703:
699:
692:
688:
684:
680:
666:
662:
658:
651:
647:
643:
639:
635:
631:
627:
613:
609:
605:
601:
597:
593:
579:
575:
571:
564:
560:
556:
553:
552:
535:
534:
526:
523:
511:
506:
498:
485:
477:
462:
452:
439:
430:
427:
418:
408:
407:
402:
401:
386:
383:
378:
374:
370:
366:
362:
358:
354:
350:
346:
342:
337:
333:
329:
325:
321:
317:
313:
309:
305:
301:
297:
293:
289:
285:
281:
277:
273:
269:
265:
261:
257:
253:
249:
245:
241:
237:
233:
229:
225:
216:
212:
208:
204:
200:
196:
192:
182:
175:
171:
166:
162:
158:
154:
151:
131:organometallic
125:
124:
39:
37:
30:
24:
14:
13:
10:
9:
6:
4:
3:
2:
2204:
2193:
2190:
2188:
2185:
2184:
2182:
2169:
2165:
2162:
2159:Evans, W. J.
2156:
2153:
2149:
2145:
2142:
2136:
2133:
2127:
2124:
2120:
2116:
2113:
2107:
2104:
2100:
2096:
2093:
2087:
2084:
2080:
2076:
2073:
2070:Evans, W. J.
2067:
2065:
2061:
2057:
2054:
2051:Evans, W. J.
2048:
2045:
2041:
2037:
2034:
2031:Evans, W. J.
2028:
2025:
2021:
2017:
2014:
2008:
2006:
2004:
2000:
1996:
1992:
1989:
1983:
1980:
1976:
1972:
1969:
1963:
1960:
1956:
1952:
1949:
1943:
1941:
1937:
1933:
1930:
1924:
1921:
1917:
1914:
1908:
1906:
1904:
1902:
1898:
1894:
1890:
1887:
1881:
1879:
1877:
1873:
1869:
1865:
1862:
1856:
1853:
1849:
1845:
1842:
1836:
1833:
1829:
1825:
1822:
1816:
1814:
1810:
1806:
1802:
1799:
1793:
1790:
1786:
1782:
1779:
1773:
1770:
1766:
1762:
1759:
1753:
1750:
1746:
1742:
1739:
1733:
1730:
1726:
1722:
1719:
1713:
1710:
1706:
1702:
1699:
1693:
1690:
1686:
1682:
1679:
1673:
1670:
1666:
1662:
1659:
1653:
1650:
1646:
1642:
1639:
1633:
1630:
1626:
1622:
1619:
1613:
1610:
1606:
1602:
1599:
1593:
1591:
1587:
1583:
1579:
1576:
1570:
1567:
1563:
1559:
1556:
1550:
1547:
1543:
1539:
1536:
1530:
1527:
1523:
1517:
1514:
1510:
1506:
1503:
1497:
1494:
1490:
1486:
1483:
1477:
1475:
1473:
1469:
1465:
1462:
1456:
1453:
1449:
1445:
1442:
1436:
1433:
1429:
1425:
1422:
1416:
1413:
1409:
1405:
1399:
1396:
1392:
1388:
1385:
1379:
1377:
1373:
1369:
1365:
1362:
1356:
1354:
1350:
1346:
1342:
1339:
1333:
1331:
1327:
1320:
1318:
1316:
1311:
1292:
1266:
1253:
1240:
1226:
1223:
1219:
1213:
1197:
1184:
1171:
1152:
1146:
1126:
1113:
1100:
1078:
1062:
1049:
1036:
1017:
1011:
991:
978:
965:
943:
942:
941:
935:
930:
928:
926:
921:
915:
913:
911:
906:
880:
878:
876:
871:
864:
862:
839:
837:
835:
830:
828:
823:
808:
806:
801:
798:
771:
769:
764:
757:
718:
716:
711:
696:
677:
675:
670:
655:
624:
622:
617:
590:
588:
583:
568:
524:
521:
509:
496:
483:
460:
450:
437:
428:
425:
416:
392:
391:
390:
384:
382:
223:
190:
152:
150:
148:
144:
140:
136:
132:
121:
118:
110:
99:
96:
92:
89:
85:
82:
78:
75:
71:
68: –
67:
63:
62:Find sources:
56:
50:
49:
45:
40:This article
38:
34:
29:
28:
19:
2167:
2163:
2160:
2155:
2147:
2143:
2140:
2135:
2126:
2118:
2114:
2111:
2106:
2098:
2094:
2091:
2086:
2078:
2074:
2071:
2055:
2053:Inorg. Chem.
2052:
2047:
2039:
2035:
2032:
2027:
2019:
2015:
2012:
1994:
1990:
1987:
1982:
1974:
1970:
1967:
1962:
1954:
1950:
1947:
1931:
1928:
1923:
1915:
1912:
1892:
1888:
1885:
1867:
1863:
1860:
1855:
1847:
1843:
1840:
1835:
1827:
1823:
1820:
1804:
1800:
1797:
1792:
1784:
1780:
1777:
1772:
1764:
1760:
1757:
1752:
1744:
1740:
1737:
1732:
1724:
1720:
1717:
1712:
1704:
1700:
1697:
1692:
1684:
1680:
1678:Inorg. Chem.
1677:
1672:
1664:
1660:
1657:
1652:
1644:
1640:
1637:
1632:
1624:
1620:
1617:
1612:
1604:
1600:
1597:
1581:
1577:
1574:
1569:
1561:
1557:
1554:
1549:
1541:
1537:
1534:
1529:
1521:
1516:
1508:
1504:
1501:
1496:
1488:
1484:
1482:Inorg. Chem.
1481:
1463:
1460:
1455:
1447:
1443:
1440:
1435:
1427:
1423:
1420:
1419:King, R. B.
1415:
1407:
1403:
1398:
1390:
1386:
1384:Inorg. Chem.
1383:
1367:
1363:
1360:
1344:
1340:
1337:
1312:
1296:
939:
922:
919:
907:
884:
872:
868:
843:
831:
824:
809:
802:
799:
772:
765:
758:
719:
712:
697:
678:
671:
656:
625:
618:
591:
584:
569:
554:
388:
156:
134:
128:
113:
104:
94:
87:
80:
73:
61:
41:
1934:, 636, 124.
1408:Carbon Bond
133:chemistry,
2181:Categories
2072:Polyhedron
1913:Chem. Rev.
1321:References
916:Insertions
840:Reactivity
107:March 2016
77:newspapers
44:references
1147:−
1138:⟶
1012:−
1003:⟶
785:La and (C
385:Synthesis
369:Th and (C
2150:, 12635.
1895:, 12019.
1787:, 3329.
1747:, 7440.
461:→
2121:, 2124.
2101:, 7929.
2022:, 9273.
1997:, 6745.
1957:, 5489.
1870:, 4292.
1830:, 7423.
1807:, 2600.
1767:, 4983.
1727:, 2389.
1667:, 3728.
1627:, 5648.
1607:, 1401.
1584:, 6877.
1564:, 6423.
1544:, 2314.
1511:, 4270.
1491:, 2999.
1393:, 2190.
1347:, 6210.
203:Sm(THF)
153:History
143:f-block
91:scholar
2170:, 263.
2081:, 803.
2042:, 131.
1977:, 527.
1707:, 797.
1687:, 770.
1647:, 445.
1466:, 652.
1450:, 121.
1430:, 155.
638:Sm(OEt
353:UF, (C
256:Sm, (C
207:and (C
93:
86:
79:
72:
64:
1850:, 87.
1370:, 42.
897:or Al
381:ThH.
361:(TMS)
98:JSTOR
84:books
2164:2000
2144:1995
2115:1990
2095:1995
2075:1987
2056:2007
2036:1985
2016:1998
1991:1998
1971:1996
1951:2001
1932:2001
1916:2002
1889:2000
1864:1987
1844:1990
1824:1991
1801:1994
1781:1989
1761:1988
1741:1986
1721:1986
1701:1988
1681:1998
1661:1985
1641:1989
1621:2001
1601:1983
1578:1988
1558:1988
1538:1990
1524:219.
1505:1984
1485:1980
1464:1980
1444:1980
1424:1976
1387:1980
1364:1956
1341:1954
852:SiMe
616:Pb:
567:Sm:
529:NaCl
413:LnCl
280:(μ-C
268:Sm(C
252:Sm,
70:news
2168:206
2148:117
2020:120
1995:120
1893:122
1868:109
1848:394
1828:113
1805:116
1785:111
1765:110
1745:108
1665:107
1605:105
1582:110
1562:110
1542:112
1509:106
466:THF
434:NaC
312:UCl
276:),
129:In
46:to
2183::
2166:,
2146:,
2117:,
2099:28
2097:,
2077:,
2063:^
2040:24
2038:,
2018:,
2002:^
1993:,
1975:15
1973:,
1955:20
1953:,
1939:^
1900:^
1891:,
1875:^
1866:,
1846:,
1826:,
1812:^
1803:,
1783:,
1763:,
1743:,
1723:,
1703:,
1685:37
1683:,
1663:,
1645:28
1643:,
1625:20
1623:,
1603:,
1589:^
1580:,
1560:,
1540:,
1507:,
1489:19
1487:,
1471:^
1448:11
1446:,
1428:20
1426:,
1391:19
1389:,
1375:^
1368:78
1366:,
1352:^
1345:76
1343:,
1329:^
1301:Me
1250:Me
1206:Sm
1181:Me
1135:Sm
1110:Me
1071:Sm
1046:Me
1000:Sm
975:Me
901:Me
814:Me
789:Me
777:Me
748:Me
736:Me
724:Me
702:Me
683:Me
661:Me
646:Me
630:Me
608:Me
596:Me
574:Me
559:Me
518:Ln
373:Me
345:Me
332:Me
320:Me
304:Me
292:Me
260:Me
244:Me
232:Me
211:Me
195:Me
178:Me
149:.
57:.
2119:9
2079:6
1725:5
1705:7
1406:-
1307:3
1305:)
1303:5
1299:5
1267:2
1262:)
1254:5
1241:5
1237:C
1233:(
1227:2
1224:1
1220:+
1214:+
1209:]
1198:2
1193:)
1185:5
1172:5
1168:C
1164:(
1159:[
1153:+
1143:e
1127:3
1122:)
1114:5
1101:5
1097:C
1093:(
1079:+
1074:]
1063:2
1058:)
1050:5
1037:5
1033:C
1029:(
1024:[
1018:+
1008:e
992:2
987:)
979:5
966:5
962:C
958:(
903:6
899:2
895:3
893:)
891:5
889:F
887:6
858:3
856:)
854:3
850:4
848:H
846:5
820:3
818:)
816:5
812:5
795:3
793:)
791:5
787:5
783:3
781:)
779:5
775:5
761:x
754:3
752:)
750:5
746:5
742:3
740:)
738:5
734:5
730:3
728:)
726:5
722:5
708:3
706:)
704:5
700:5
693:x
689:3
687:)
685:5
681:5
667:3
665:)
663:5
659:5
652:3
650:)
648:5
644:5
640:2
636:2
634:)
632:5
628:5
614:2
612:)
610:5
606:5
602:3
600:)
598:5
594:5
580:3
578:)
576:5
572:5
565:3
563:)
561:5
557:5
525:3
522:+
510:3
505:)
497:5
493:H
484:5
480:C
476:(
451:5
447:H
438:5
429:3
426:+
417:3
379:3
377:)
375:5
371:5
367:3
365:)
363:2
359:3
357:H
355:5
351:3
349:)
347:5
343:5
338:3
336:)
334:5
330:5
326:3
324:)
322:5
318:5
314:2
310:2
308:)
306:5
302:5
298:3
296:)
294:5
290:5
286:5
284:H
282:5
278:2
274:5
272:H
270:5
266:2
264:)
262:5
258:5
254:3
250:3
248:)
246:5
242:5
238:3
236:)
234:5
230:5
226:2
217:2
215:)
213:5
209:5
205:2
201:2
199:)
197:5
193:5
183:5
176:5
172:n
167:3
165:)
163:5
161:H
159:5
120:)
114:(
109:)
105:(
95:·
88:·
81:·
74:·
51:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.