1970:
297:
848:
1377:
1389:
1945:
380:
1857:
395:
1705:
42:
1468:
1644:
low vapour pressures, and require substantially higher temperatures to boil. Boiling points exhibit similar trends to melting points in terms of the size of ions and strength of other interactions. When vapourized, the ions are still not freed of one another. For example, in the vapour phase sodium chloride exists as diatomic "molecules".
1915:, one or more of the ionic components has a significant mobility, allowing conductivity even while the material as a whole remains solid. This is often highly temperature dependent, and may be the result of either a phase change or a high defect concentration. These materials are used in all solid-state
1631:
show molecule-like structures in the liquid phase). Inorganic compounds with simple ions typically have small ions, and thus have high melting points, so are solids at room temperature. Some substances with larger ions, however, have a melting point below or near room temperature (often defined as up
1438:
beneficial, they occur in greater concentration at higher temperatures. Once generated, these pairs of defects can diffuse mostly independently of one another, by hopping between lattice sites. This defect mobility is the source of most transport phenomena within an ionic crystal, including diffusion
1643:
Even when the local structure and bonding of an ionic solid is disrupted sufficiently to melt it, there are still strong long-range electrostatic forces of attraction holding the liquid together and preventing ions boiling to form a gas phase. This means that even room temperature ionic liquids have
2672:
will have a stoichiometry that depends on which oxidation states are present, to ensure overall neutrality. This can be indicated in the name by specifying either the oxidation state of the elements present, or the charge on the ions. Because of the risk of ambiguity in allocating oxidation states,
2493:
have described and widely used metal-containing salts as sources of colour in fireworks. Under intense heat, the electrons in the metal ions or small molecules can be excited. These electrons later return to lower energy states, and release light with a colour spectrum characteristic of the species
823:
can be applied, whereby the compounds with the most ionic character are those consisting of hard acids and hard bases: small, highly charged ions with a high difference in electronegativities between the anion and cation. This difference in electronegativities means that the charge separation, and
859:
The lattice energy is the summation of the interaction of all sites with all other sites. For unpolarizable spherical ions, only the charges and distances are required to determine the electrostatic interaction energy. For any particular ideal crystal structure, all distances are geometrically
723:
486:. In this method, the reactants are repeatedly finely ground into a paste and then heated to a temperature where the ions in neighboring reactants can diffuse together during the time the reactant mixture remains in the oven. Other synthetic routes use a solid precursor with the correct
798:
of the two interacting bodies is affected by the presence of one another, covalent interactions (non-ionic) also contribute to the overall energy of the compound formed. Salts are rarely purely ionic, i.e. held together only by electrostatic forces. The bonds between even the most
439:
of the opposite charges. To ensure that these do not contaminate the precipitated salt, it is important to ensure they do not also precipitate. If the two solutions have hydrogen ions and hydroxide ions as the counterions, they will react with one another in what is called an
3526:
This structure type can accommodate any charges on A and B that add up to six. When both are three the charge structure is equivalent to that of corrundum. The structure also has a variable lattice parameter c/a ratio, and the exact
Madelung constant depends on
815:. Conversely, covalent bonds between unlike atoms often exhibit some charge separation and can be considered to have a partial ionic character. The circumstances under which a compound will have ionic or covalent character can typically be understood using
860:
related to the smallest internuclear distance. So for each possible crystal structure, the total electrostatic energy can be related to the electrostatic energy of unit charges at the nearest neighboring distance by a multiplicative constant called the
1417:
consist of one vacancy of each type, and are generated at the surfaces of a crystal, occurring most commonly in compounds with a high coordination number and when the anions and cations are of similar size. If the cations have multiple possible
1443:. When vacancies collide with interstitials (Frenkel), they can recombine and annihilate one another. Similarly, vacancies are removed when they reach the surface of the crystal (Schottky). Defects in the crystal structure generally expand the
1583:, being able to react with either an acid or a base. This is also true of some compounds with ionic character, typically oxides or hydroxides of less-electropositive metals (so the compound also has significant covalent character), such as
1412:
consist of a cation vacancy paired with a cation interstitial and can be generated anywhere in the bulk of the crystal, occurring most commonly in compounds with a low coordination number and cations that are much smaller than the anions.
1696:. For example, halides with the caesium chloride structure (coordination number 8) are less compressible than those with the sodium chloride structure (coordination number 6), and less again than those with a coordination number of 4.
819:, which use only charges and the sizes of each ion. According to these rules, compounds with the most ionic character will have large positive ions with a low charge, bonded to a small negative ion with a high charge. More generally
2757:
root of the name, to give special names for the low and high oxidation states. For example, this scheme uses "ferrous" and "ferric", for iron(II) and iron(III) respectively, so the examples given above were classically named
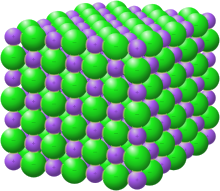
2019:
in the ultraviolet part of the spectrum). In compounds with less ionic character, their color deepens through yellow, orange, red, and black (as the absorption band shifts to longer wavelengths into the visible spectrum).
1796:, the cohesive forces between these ions within a solid, determines the solubility. The solubility is dependent on how well each ion interacts with the solvent, so certain patterns become apparent. For example, salts of
1407:
Within any crystal, there will usually be some defects. To maintain electroneutrality of the crystals, defects that involve loss of a cation will be associated with loss of an anion, i.e. these defects come in pairs.
1430:, a free electron occupying an anion vacancy. When the compound has three or more ionic components, even more defect types are possible. All of these point defects can be generated via thermal vibrations and have an
2745:, has uranium in an oxidation state of +6, so would be called a dioxouranium(VI) ion in Stock nomenclature. An even older naming system for metal cations, also still widely used, appended the suffixes
1769:
is higher. When the oppositely charged ions in the solid ionic lattice are surrounded by the opposite pole of a polar molecule, the solid ions are pulled out of the lattice and into the liquid. If the
435:
Insoluble salts can be precipitated by mixing two solutions, one with the cation and one with the anion in it. Because all solutions are electrically neutral, the two solutions mixed must also contain
249:
Individual ions within a salt usually have multiple near neighbours, so they are not considered to be part of molecules, but instead part of a continuous three-dimensional network. Salts usually form
2404:, water conditioning, for de-icing roads, and many other uses. Many salts are so widely used in society that they go by common names unrelated to their chemical identity. Examples of this include
1900:, they can conduct electricity because the ions become completely mobile. For this reason, molten salts and solutions containing dissolved salts (e.g., sodium chloride in water) can be used as
1607:
Electrostatic forces between particles are strongest when the charges are high, and the distance between the nuclei of the ions is small. In such cases, the compounds generally have very high
5158:
Rebelo, Luis P. N.; Canongia Lopes, José N.; Esperança, José M. S. S.; Filipe, Eduardo (2005-04-01). "On the
Critical Temperature, Normal Boiling Point, and Vapor Pressure of Ionic Liquids".
5472:
2554:, the common name is written using two words. The name of the cation (the unmodified element name for monatomic cations) comes first, followed by the name of the anion. For example, MgCl
6292:
1730:
and dispersed throughout the resulting solution. Salts do not exist in solution. In contrast, molecular compounds, which includes most organic compounds, remain intact in solution.
1568:. If it is the result of a reaction between a strong acid and a strong base, the result is a neutral salt. Weak acids reacted with weak bases can produce ionic compounds with both the
4278:
3317:
COO. Most group 1 and 2 metals form strong salts. Strong salts are especially useful when creating conductive compounds as their constituent ions allow for greater conductivity.
5680:
5218:
288:, because the ions become mobile. Some salts have large cations, large anions, or both. In terms of their properties, such species often are more similar to organic compounds.
2677:
integer followed by the sign (... , 2−, 1−, 1+, 2+, ...) in parentheses directly after the name of the cation (without a space separating them). For example, FeSO
5728:
2550:, salts are named according to their composition, not their structure. In the most simple case of a binary salt with no possible ambiguity about the charges and thus the
824:
resulting dipole moment, is maintained even when the ions are in contact (the excess electrons on the anions are not transferred or polarized to neutralize the cations).
6328:
3567:
1785:
is usually positive for most solid solutes like salts, which means that their solubility increases when the temperature increases. There are some unusual salts such as
1619:. Trends in melting points can be even better explained when the structure and ionic size ratio is taken into account. Above their melting point, salts melt and become
4483:
4357:
482:
of complex salts from solid reactants, which are first melted together. In other cases, the solid reactants do not need to be melted, but instead can react through a
1447:, reducing the overall density of the crystal. Defects also result in ions in distinctly different local environments, which causes them to experience a different
2459:. Because the solutes are charged ions they also increase the electrical conductivity of the solution. The increased ionic strength reduces the thickness of the
5418:
6525:
835:, for example, has led some philosophers of science to suggest that alternative approaches to understanding bonding are required. This could be by applying
5379:
D. Chasseau; G. Comberton; J. Gaultier; C. Hauw (1978). "Réexamen de la structure du complexe hexaméthylène-tétrathiafulvalène-tétracyanoquinodiméthane".
791:. The balance between these forces leads to a potential energy well with minimum energy when the nuclei are separated by a specific equilibrium distance.
772:
attraction between the net negative charge of the anions and net positive charge of the cations. There is also a small additional attractive force from
3833:
2633:(note that in both the empirical formula and the written name, the cations appear in alphabetical order, but the order varies between them because the
2537:
5115:
1708:
The aqueous solubility of a variety of salts as a function of temperature. Some compounds exhibiting unusual solubility behavior have been included.
2729:
respectively. For simple ions the ionic charge and the oxidation number are identical, but for polyatomic ions they often differ. For example, the
2613:, ...) are often required to indicate the relative compositions, and cations then anions are listed in alphabetical order. For example, KMgCl
2023:
The absorption band of simple cations shifts toward a shorter wavelength when they are involved in more covalent interactions. This occurs during
285:
1422:, then it is possible for cation vacancies to compensate for electron deficiencies on cation sites with higher oxidation numbers, resulting in a
320:
for each atom, demonstrating that the constituents were not arranged in molecules or finite aggregates, but instead as a network with long-range
2489:
Solid salts have long been used as paint pigments, and are resistant to organic solvents, but are sensitive to acidity or basicity. Since 1801
1789:, where this entropy change is negative, due to extra order induced in the water upon solution, and the solubility decreases with temperature.
1669:
6308:
883:
Using an even simpler approximation of the ions as impenetrable hard spheres, the arrangement of anions in these systems are often related to
448:
to form water. Alternately the counterions can be chosen to ensure that even when combined into a single solution they will remain soluble as
6485:
6466:
6443:
6422:
6401:
6382:
6356:
6337:
6302:
6281:
6260:
6241:
6220:
6201:
5925:
5864:
5807:
5722:
5592:
5466:
5212:
5109:
5035:
4975:
4905:
4621:
4566:
4467:
4341:
4091:
3827:
6128:
4217:
328:
were also found to have similar structural features. These compounds were soon described as being constituted of ions rather than neutral
4649:
Verwey, E. J. W. (1947). "Physical
Properties and Cation Arrangement of Oxides with Spinel Structures I. Cation Arrangement in Spinels".
4267:
5687:
6168:
6138:
1452:
4532:
2598:
from these names, the stoichiometry can be deduced from the charges on the ions, and the requirement of overall charge neutrality.
685:
where two different salts are mixed in water, their ions recombine, and the new salt is insoluble and precipitates. For example:
5621:
4005:
Hannay, N. Bruce; Smyth, Charles P. (February 1946). "The Dipole Moment of
Hydrogen Fluoride and the Ionic Character of Bonds".
2618:
1781:
provides a thermodynamic drive to remove ions from their positions in the crystal and dissolve in the liquid. In addition, the
6515:
4937:
1889:
1440:
884:
868:. When a reasonable form is assumed for the additional repulsive energy, the total lattice energy can be modelled using the
6373:
The nature of the chemical bond and the structure of molecules and crystals: an introduction to modern structural chemistry
2400:(sodium chloride) for over 8000 years, using it first as a food seasoning and preservative, and now also in manufacturing,
776:
which contributes only around 1–2% of the cohesive energy for small ions. When a pair of ions comes close enough for their
1355:, particularly with mixtures of anions or cations, can be cooled rapidly enough that there is not enough time for crystal
5202:
2630:
4033:
2641:
is K). When one of the ions already has a multiplicative prefix within its name, the alternate multiplicative prefixes (
1805:
1640:
like hydrocarbon chains, which also play a role in determining the strength of the interactions and propensity to melt.
3214:
Salts with varying number of hydrogen atoms replaced by cations as compared to their parent acid can be referred to as
6505:
2662:
1888:. This is achieved to some degree at high temperatures when the defect concentration increases the ionic mobility and
1778:
1766:
1423:
827:
Although chemists classify idealized bond types as being ionic or covalent, the existence of additional types such as
6291:
International Union of Pure and
Applied Chemistry, Division of Chemical Nomenclature (2005). Neil G. Connelly (ed.).
367:. Born predicted crystal energies based on the assumption of ionic constituents, which showed good correspondence to
2528:
occur (often with a reducing agent such as carbon) such that the metal ions gain electrons to become neutral atoms.
869:
5414:
2893:
773:
483:
6251:
Brown, Theodore L.; LeMay, H. Eugene Jr; Bursten, Bruce E.; Lanford, Steven; Sagatys, Dalius; Duffy, Neil (2009).
768:
between the charge distribution of these bodies, and in particular, the ionic bond resulting from the long-ranged
754:. The oppositely charged ions – typically a great many of them – are then attracted to each other to form a solid.
2452:
2376:) of the component ions. That slow, partial decomposition is usually accelerated by the presence of water, since
1431:
900:
788:
3456:
This structure type has a variable lattice parameter c/a ratio, and the exact
Madelung constant depends on this.
1660:, because the strict alignment of positive and negative ions must be maintained. Instead the material undergoes
5486:
West, Anthony R. (1991). "Solid electrolytes and mixed ionic?electronic conductors: an applications overview".
3487:
3406:
2136:
1809:
1723:
682:
460:
873:
5235:
Johnston, T. L.; Stokes, R. J.; Li, C. H. (December 1959). "The ductile–brittle transition in ionic solids".
1904:. This conductivity gain upon dissolving or melting is sometimes used as a defining characteristic of salts.
4756:
Alberti, A.; Vezzalini, G. (1978). "Madelung energies and cation distributions in olivine-type structures".
2460:
2456:
1969:
1877:
1873:
1719:
1402:
445:
428:
and the solid compound nucleates. This process occurs widely in nature and is the means of formation of the
296:
281:
273:
5063:
Pauling, Linus (1928-04-01). "The
Influence of Relative Ionic Sizes on the Properties of Ionic Compounds".
1511:
1243:
In some cases, the anions take on a simple cubic packing and the resulting common structures observed are:
513:
441:
5977:
Gibbons, Cyril S.; Reinsborough, Vincent C.; Whitla, W. Alexander (January 1975). "Crystal
Structures of K
4497:
Brackett, Thomas E.; Brackett, Elizabeth B. (1965). "The
Lattice Energies of the Alkaline Earth Halides".
3470:
3325:
3295:
2472:
2444:
2349:
2172:
1880:
to any significant extent when the substance is solid. In order to conduct, the charged particles must be
903:) of cations and anions, a variety of structures are commonly observed, and theoretically rationalized by
847:
747:
455:
If the solvent is water in either the evaporation or precipitation method of formation, in many cases the
313:
5513:
Boivin, J. C.; Mairesse, G. (October 1998). "Recent
Material Developments in Fast Oxide Ion Conductors".
3466:
2502:
1920:
877:
479:
4134:
Pearson, Ralph G. (October 1968). "Hard and soft acids and bases, HSAB, part II: Underlying theories".
506:
6086:
5388:
5314:
5271:
5240:
5000:
4765:
4730:
4697:
4658:
4413:
4143:
3732:
3653:
3606:
3234:
2338:
2318:
2077:
1893:
1762:
1448:
1089:
795:
498:
421:
3692:
Sherman, Jack (August 1932). "Crystal Energies of Ionic Compounds and Thermochemical Applications".
1451:, especially in the case of different cations exchanging lattice sites. This results in a different
4725:
Thompson, P.; Grimes, N. W. (27 September 2006). "Madelung calculations for the spinel structure".
3262:
2381:
2200:
2005:
1995:
1953:
1881:
1841:
1786:
1693:
1588:
896:
765:
628:
620:
317:
309:
5305:
Stillwell, Charles W. (January 1937). "Crystal chemistry. V. The properties of binary compounds".
4789:
4477:
4351:
4198:
3622:
3507:
3287:
3248:
2710:
2634:
2559:
2498:
2483:
2154:
2116:
2001:
1908:
1845:
1665:
1628:
1624:
333:
325:
227:
115:
4967:
904:
6144:
5856:
2702:
2682:
2015:
The anions in compounds with bonds with the most ionic character tend to be colorless (with an
6481:
6462:
6439:
6418:
6412:
6397:
6378:
6352:
6333:
6298:
6277:
6256:
6237:
6216:
6197:
6164:
6134:
5921:
5860:
5803:
5799:
5718:
5712:
5588:
5462:
5456:
5287:
5208:
5183:
5175:
5105:
5080:
5031:
4971:
4901:
4617:
4562:
4463:
4337:
4087:
4053:
3823:
3817:
3396:
3103:
3017:
2726:
2595:
2326:
2238:
1885:
1782:
1495:
1444:
888:
861:
836:
572:
539:
535:
502:
411:
321:
250:
123:
84:
6433:
6271:
6094:
6002:
5848:
5791:
5522:
5495:
5396:
5322:
5279:
5248:
5167:
5099:
5072:
5008:
4875:
4831:
4781:
4773:
4738:
4705:
4666:
4594:
4506:
4421:
4190:
4151:
4116:
4045:
4014:
3740:
3701:
3661:
3614:
3581:
3337:
3083:
2722:
2714:
2513:
2448:
2413:
2365:
1861:
1837:
1829:
1750:
1573:
1507:
1315:
1056:
816:
808:
769:
616:
608:
599:
520:
364:
360:
243:
239:
104:
3822:(Reprinted with corrections. ed.). New York: Cambridge University Press. p. 351.
6435:
Solid State Chemistry Synthesis, Structure, and Properties of Selected Oxides and Sulfides
3186:
2759:
2674:
2669:
2298:
2234:
2046:
2016:
1928:
1832:, where the 2+/2− pairing leads to high lattice energies. For similar reasons, most metal
1825:
1689:
1592:
1459:, so that the optical absorption (and hence colour) can change with defect concentration.
1419:
1414:
1290:
969:
804:
800:
739:
425:
387:
368:
305:
171:
100:
50:
35:
4822:
Souquet, J (October 1981). "Electrochemical properties of ionically conductive glasses".
352:
6090:
5392:
5318:
5275:
5244:
5004:
4769:
4734:
4701:
4662:
4524:
4417:
4147:
3736:
3657:
3610:
1704:
6510:
6455:
6323:
6231:
6156:
5266:
Kelly, A.; Tyson, W. R.; Cottrell, A. H. (1967-03-01). "Ductile and brittle crystals".
4960:
3341:
2763:
2718:
2591:
2571:
2525:
2490:
2440:
2421:
2357:
2322:
2310:
2306:
2095:
1916:
1821:
1793:
1774:
1616:
1596:
1569:
1409:
1376:
1097:
1064:
832:
828:
780:
727:
665:
646:
612:
475:
449:
356:
175:
6520:
6499:
6371:
6366:
6191:
6183:
5849:
5792:
5609:
4879:
4835:
4793:
4202:
3411:
3207:
2997:
2721:(... , −II, −I, 0, I, II, ...). So the examples given above would be named
2673:
IUPAC prefers direct indication of the ionic charge numbers. These are written as an
2551:
2478:
The chemical identity of the ions added is also important in many uses. For example,
2417:
1738:
1612:
1608:
1352:
1339:
892:
812:
777:
759:
561:
487:
456:
261:
257:
151:
108:
3626:
3577:
1876:. Although they contain charged atoms or clusters, these materials do not typically
1856:
6187:
3725:
Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences
3646:
Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences
3599:
Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences
3145:
2706:
2543:
2521:
2314:
2182:
2060:
1944:
1746:
1673:
1657:
1633:
1487:
1475:
1220:
1216:
784:
657:
494:
407:
4933:
3597:
Bragg, W. H.; Bragg, W. L. (1 July 1913). "The Reflection of X-rays by Crystals".
3230:
salts refer to those with more than one hydrogen atom replaced. Examples include:
2949:
Common salt-forming anions (parent acids in parentheses where available) include:
2277:
tend not to be salts, because insolubility is required for fastness. Some organic
501:) and highly electronegative halogen gases, or water, the atoms can be ionized by
4777:
2730:
2668:
Compounds containing one or more elements which can exist in a variety of charge/
3572:
3417:
3349:
3321:
3283:
3124:
3063:
2976:
2709:
each have a charge of 3+, to balance the 2− on each of the three sulfate ions).
2601:
If there are multiple different cations and/or anions, multiplicative prefixes (
2436:
2429:
2401:
2397:
2290:
1912:
1901:
1677:
1637:
1620:
1580:
1557:
1545:
1526:
1360:
998:
852:
820:
650:
348:
340:
269:
2031:
salts with an anion absorbing in the infrared can become colorful in solution.
394:
5400:
5283:
5252:
4742:
4426:
4401:
3427:
3376:
3372:
3364:
2860:
2377:
1734:
1713:
1584:
1456:
1388:
1356:
887:
arrangements of spheres, with the cations occupying tetrahedral or octahedral
751:
436:
155:
5291:
5179:
5084:
4061:
3576:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
6133:(3rd ed.). Hoboken, New Jersey: John Wiley & Sons Inc. p. 68.
3585:
3401:
3171:
2982:
2845:
2830:
2638:
2425:
2294:
2039:
2028:
2024:
1950:
1924:
1833:
1801:
1770:
1727:
1565:
1561:
1553:
1549:
1541:
1515:
1499:
977:
865:
557:
468:
429:
231:
211:
72:
5187:
4598:
4057:
3745:
3720:
3666:
3641:
3618:
2261:
do not absorb light in the part of the spectrum that is visible to humans.
1692:
of an salt is strongly determined by its structure, and in particular the
379:
17:
5499:
5026:
Weller, Mark; Overton, Tina; Rourke, Jonathan; Armstrong, Fraser (2014).
4991:
Davidson, David (November 1955). "Amphoteric molecules, ions and salts".
4561:(3. print ed.). Washington: American Chemical Society. p. 121.
4336:(3. ed.). Boca Raton, Fla. : Taylor & Francis, CRC. p. 44.
4049:
3437:
3368:
3226:, identifying that one, two, or three hydrogen atoms have been replaced;
3089:
3003:
2773:
2517:
2479:
2468:
2393:
2266:
1813:
1758:
1661:
1636:. Ions in ionic liquids often have uneven charge distributions, or bulky
1530:
1471:
1427:
1158:
1130:
1031:
1006:
743:
735:
592:
579:
344:
265:
195:
167:
159:
119:
61:
5076:
4866:
Schmalzried, Hermann (1965). "Point defects in ternary ionic crystals".
4785:
4510:
4120:
4018:
3705:
1765:
are significantly stronger than ion-induced dipole interactions, so the
1668:. As the temperature is elevated (usually close to the melting point) a
490:
ratio of non-volatile ions, which is heated to drive off other species.
3414:(the method used to test for salt presence during coating applications)
3384:
3380:
3324:. These salts do not dissociate well in water. They are generally more
3192:
3130:
3109:
3069:
3023:
2953:
2789:
2587:
2464:
2409:
2373:
2361:
2352:
and often odorless, whereas salts of either weak acids or weak bases ("
2274:
2270:
2254:
2009:
1817:
1653:
1435:
1230:
588:
528:
464:
414:
332:, but proof of this hypothesis was not found until the mid-1920s, when
277:
179:
127:
46:
41:
6098:
5526:
5171:
5012:
4710:
4685:
4670:
4181:
Hendry, Robin Findlay (2008). "Two Conceptions of the Chemical Bond".
4155:
1804:
and ammonium are usually soluble in water. Notable exceptions include
5326:
5030:(Sixth ed.). Oxford: Oxford University Press. pp. 129–130.
3537:
3432:
3422:
2933:
2918:
2369:
2258:
1897:
1797:
1754:
731:
383:
336:
experiments (which detect the density of electrons), were performed.
163:
92:
57:
54:
6006:
4107:
Pearson, Ralph G. (November 1963). "Hard and Soft Acids and Bases".
1467:
6077:
Fernelius, W. Conard (November 1982). "Numbers in chemical names".
4958:
Whitten, Kenneth W.; Galley, Kenneth D.; Davis, Raymond E. (1992).
4194:
2686:
3291:
3165:
3151:
2908:
2754:
2547:
2405:
2392:
Salts have long had a wide variety of uses and applications. Many
2334:
2330:
2302:
2035:
1855:
1742:
1703:
1656:. Once they reach the limit of their strength, they cannot deform
1537:
1522:
1503:
1466:
846:
721:
672:
632:
549:
467:, and can have very different chemical properties compared to the
393:
378:
295:
235:
103:(electrically neutral). The constituent ions are held together by
96:
64:
40:
5920:(Sixth ed.). Belmont, CA: Thomson Brooks/Cole. p. 111.
2439:
solutions. This is a simple way to control the concentration and
1247:
Common ionic compound structures with simple cubic packed anions
4300:
4298:
3333:
3329:
2912:
2804:
1865:
1533:
1518:
1510:. Other ionic compounds are known as salts and can be formed by
1491:
1426:. Another non-stoichiometric possibility is the formation of an
787:) to overlap, a short-ranged repulsive force occurs, due to the
642:
553:
524:
329:
88:
31:
6294:
Nomenclature of inorganic chemistry: IUPAC recommendations 2005
2482:
containing compounds are dissolved to supply fluoride ions for
2038:, which arise either from their constituent anions, cations or
493:
In some reactions between highly reactive metals (usually from
2509:
2278:
722:
6196:(27th repr. ed.). New York: Holt, Rinehart and Winston.
5916:
Kotz, John C.; Treichel, Paul M; Weaver, Gabriela C. (2006).
3845:
3843:
1860:
Edge-on view of portion of crystal structure of hexamethylene
5681:"Water fluoridation: a manual for engineers and technicians"
5207:(2nd ed.). New York: Elsevier Science. pp. 63–67.
3721:"A Quantitative Study of the Reflexion of X-Rays by Sylvine"
3371:, but are not considered salts. Examples of zwitterions are
3290:. These salts dissociate completely or almost completely in
2689:
ions balancing the 2− charge on the sulfate ion), whereas Fe
1836:
are not soluble in water. Some soluble carbonate salts are:
390:, forms when salty water evaporates leaving the ions behind.
6255:(2nd ed.). Frenchs Forest, N.S.W.: Pearson Australia.
2508:
Many metals are geologically most abundant as salts within
3799:
3797:
3784:
3782:
3780:
3320:
Weak salts or weak electrolyte salts are composed of weak
911:
Common ionic compound structures with close-packed anions
5554:
5552:
5550:
5548:
4684:
Verwey, E. J. W.; de Boer, F.; van Santen, J. H. (1948).
4559:
Teaching general chemistry: a materials science companion
3961:
3959:
3957:
1434:
concentration. Because they are energetically costly but
30:"Ionic compound" redirects here. Not to be confused with
6377:(3rd ed.). Ithaca, N.Y.: Cornell University Press.
4758:
Zeitschrift für Kristallographie – Crystalline Materials
3988:
3986:
3547:
the larger cation occupies the smaller tetrahedral site.
1714:
Solubility § Solubility of ionic compounds in water
1544:. If the compound is the result of a reaction between a
67:, Cl. The yellow stipples show the electrostatic forces.
6110:
6108:
6048:
6046:
6044:
6042:
6040:
5362:
5360:
4402:"Berechnung von Madelung'schen Zahlen für den NiAs-Typ"
4383:
4381:
4379:
3872:
3870:
6417:(1st ed.). New York: Cambridge University Press.
4900:. New Delhi: S. Chand & Company Ltd. p. 554.
2281:
are salts, but they are virtually insoluble in water.
1761:). This contrast is principally because the resulting
3465:
This structure has been referred to in references as
3367:
contain an anionic and a cationic centre in the same
474:
Molten salts will solidify on cooling to below their
53:, NaCl, a typical salt. The purple spheres represent
6332:(8th ed.). Hoboken, NJ: John Wiley & Sons.
2372:)) or the conjugate base (e.g., ammonium salts like
6253:
Chemistry: the central science: a broad perspective
4218:"Do bond classifications help or hinder chemistry?"
876:, or in the absence of structural information, the
505:, a process thermodynamically understood using the
6454:
6370:
5098:Tosi, M. P. (2002). Gaune-Escard, Marcelle (ed.).
4959:
4587:Angewandte Chemie International Edition in English
2215:is made green by the hydrated nickel(II) chloride
6215:(8th ed.). Oxford: Oxford University Press.
5458:An Introduction to Electronic and Ionic Materials
3719:James, R. W.; Brindley, G. W. (1 November 1928).
2435:Soluble salts can easily be dissolved to provide
1050:half octahedral (alternate layers fully occupied)
764:Ions in salts are primarily held together by the
371:measurements, further supporting the assumption.
256:Salts composed of small ions typically have high
5270:. Vol. 15, no. 135. pp. 567–586.
5239:. Vol. 4, no. 48. pp. 1316–1324.
4616:. Meerut: Krishna Prakashan Media. p. 171.
4585:Hoppe, R. (January 1966). "Madelung Constants".
316:. This revealed that there were six equidistant
87:consisting of an assembly of positively charged
6351:(3rd ed.). New York: W.H. Freeman and Co.
5101:Molten Salts: From Fundamentals to Applications
2253:are colorless or white because the constituent
2169:is made blue by the hydrated copper(II) cation.
339:Principal contributors to the development of a
6461:(2nd ed.). Lexington, Mass.: D.C. Heath.
5834:
5822:
5790:Xu, Ruren; Pang, Wenqin; Huo, Qisheng (2011).
5666:
5654:
5642:
5104:. Dordrecht: Springer Netherlands. p. 1.
4806:
4729:. Vol. 36, no. 3. pp. 501–505.
4440:
4370:
4304:
3977:
1556:. If it is the result of a reaction between a
1182:one-eighth tetrahedral and one-half octahedral
6347:McQuarrie, Donald A.; Rock, Peter A. (1991).
5686:. Centers for Disease Control. Archived from
5050:
3642:"The Reflection of X-rays by Crystals. (II.)"
750:, and this electron enters the fluorine atom
8:
4580:
4578:
4482:: CS1 maint: multiple names: authors list (
4356:: CS1 maint: multiple names: authors list (
4082:Lalena, John. N.; Cleary, David. A. (2010).
2505:for high-temperature solid-state synthesis.
2443:. The concentration of solutes affects many
6411:Wenk, Hans-Rudolph; Bulakh, Andrei (2004).
5851:Chemical metallurgy principles and practice
5717:. Discovery Publishing House. p. 230.
5415:"Electrical Conductivity of Ionic Compound"
343:treatment of ionic crystal structures were
300:X-ray spectrometer developed by W. H. Bragg
114:The component ions in a salt can be either
99:), which results in a compound with no net
5570:
5140:
5128:
4891:
4889:
4636:
4332:Moore, Lesley E. Smart; Elaine A. (2005).
4086:(2nd ed.). Hoboken, N.J: John Wiley.
3900:
3888:
3816:Wenk, Hans-Rudolf; Bulakh, Andrei (2003).
3803:
3788:
3328:than strong salts. They may be similar in
2467:particles, and therefore the stability of
1245:
909:
864:that can be efficiently computed using an
738:atoms undergoing a redox reaction to form
6276:. Cambridge: Royal Society of Chemistry.
4709:
4453:
4451:
4449:
4425:
4038:Journal of the Chemical Society (Resumed)
3744:
3687:
3685:
3683:
3681:
3679:
3677:
3665:
2653:, ...) are used. For example, Ba(BrF
2538:IUPAC nomenclature of inorganic chemistry
2344:Salts of strong acids and strong bases ("
27:Chemical compound involving ionic bonding
6476:Zumdahl, Steven; Zumdahl, Susan (2015).
6396:(2nd ed.). Cambridge, UK: RSC Pub.
5065:Journal of the American Chemical Society
4552:
4550:
4327:
4325:
4109:Journal of the American Chemical Society
4084:Principles of inorganic materials design
4007:Journal of the American Chemical Society
2244:
2226:
2222:
2218:
2206:
2177:
2160:
2146:
2142:
2126:
2122:
2100:
2090:
2086:
2082:
2055:
2051:
2008:containing the constituent ions, or the
6414:Minerals: Their Constitution and Origin
6236:(5th ed.). New York: McGraw-Hill.
6211:Atkins, Peter; de Paula, Julio (2006).
5777:
5765:
5753:
5741:
5558:
5539:
5439:
4557:Ellis, Arthur B. ; et al. (1995).
4462:. Boston: Academic Press. p. 123.
4316:
4253:
3992:
3965:
3936:
3912:
3861:
3849:
3819:Minerals: their constitution and origin
3771:
3759:
3560:
3449:
2516:materials, these ores are processed by
5204:Inorganic Chemistry a Unified Approach
4920:
4475:
4458:Dienes, Richard J. Borg, G.J. (1992).
4387:
4349:
4334:Solid state chemistry: an introduction
4241:
4168:
3948:
3924:
3506:The reference lists this structure as
3486:The reference lists this structure as
1820:are water-soluble. Exceptions include
1363:is formed (with no long-range order).
60:, Na, and the green spheres represent
6297:(New ed.). Cambridge: RSC Publ.
6114:
6064:
6052:
6031:
6019:
5964:
5952:
5940:
5903:
5891:
5879:
5450:
5448:
5366:
5351:
5339:
5230:
5228:
5153:
5151:
5149:
4861:
4859:
4857:
4855:
4853:
4851:
4849:
4847:
4845:
4817:
4815:
4216:Seifert, Vanessa (27 November 2023).
3876:
3301:Strong salts start with Na__, K__, NH
3286:salts are chemical salts composed of
2769:Common salt-forming cations include:
2384:equation of formation of weak salts.
420:Salts form upon evaporation of their
7:
5794:Modern inorganic synthetic chemistry
5624:from the original on 16 January 2016
4535:from the original on 27 January 2016
3473:, but both are now known as the RhBr
3340:they are derived from. For example,
1572:ion and conjugate acid ion, such as
6526:Chemical compounds by chemical bond
6438:. Dordrecht: Springer Netherlands.
6432:Wold, Aaron; Dwight, Kirby (1993).
6329:Introduction to Solid State Physics
6127:Voet, D. & Voet, J. G. (2005).
5160:The Journal of Physical Chemistry B
2705:(because the two iron ions in each
2631:magnesium dipotassium tetrachloride
272:. As solids they are almost always
6478:Chemistry: An Atoms First Approach
5618:General Chemistry Virtual Textbook
5455:Gao, Wei; Sammes, Nigel M (1999).
4966:(4th ed.). Saunders. p.
4612:Bhagi, Ajay; Raj, Gurdeep (2010).
3640:Bragg, W. H. (22 September 1913).
3573:Compendium of Chemical Terminology
3294:. They are generally odorless and
2713:, still in common use, writes the
2360:(e.g., acetates like acetic acid (
2135:is made red by the chromophore of
1676:becomes possible by the motion of
930:Hexagonal close packing of anions
25:
5918:Chemistry and Chemical Reactivity
5587:(1st ed.). London: Vintage.
5461:. World Scientific. p. 261.
4868:Progress in Solid State Chemistry
4531:. University of Liverpool. 2008.
2396:are ionic. Humans have processed
478:. This is sometimes used for the
6273:An introduction to ionic liquids
5855:. Weinheim: Wiley-VCH. pp.
5731:from the original on 2017-12-03.
5475:from the original on 2017-12-03.
5421:from the original on 21 May 2014
5381:Acta Crystallographica Section B
5221:from the original on 2017-12-03.
5201:Porterfield, William W. (2013).
5118:from the original on 2017-12-03.
4460:The physical chemistry of solids
3836:from the original on 2017-12-03.
3493:, which is now known as the RhBr
2663:barium bis(tetrafluoridobromate)
2345:
1975:Cobalt(II) chloride hexahydrate,
1968:
1943:
1892:is observed. When the salts are
1726:into individual ions, which are
1632:to 100 °C), and are termed
1387:
1375:
5847:Gupta, Chiranjib Kumar (2003).
5798:. Amsterdam: Elsevier. p.
4940:from the original on 2015-12-29
4690:The Journal of Chemical Physics
4686:"Cation Arrangement in Spinels"
4284:from the original on 2015-05-13
3536:However, in some cases such as
3513:, which is now known as the BiI
2619:magnesium potassium trichloride
2273:in water. Similarly, inorganic
899:(principally determined by the
839:to calculate binding energies.
587:A metal and a non-metal, e.g.,
463:, so the product is known as a
95:) and negatively charged ions (
5488:Journal of Materials Chemistry
4034:"The modern theory of valency"
2353:
2034:Salts exist in many different
1890:solid state ionic conductivity
1498:cations and basic anions ions
1441:solid state ionic conductivity
927:Cubic close packing of anions
230:. Salts containing basic ions
1:
6163:. Walker Publishing Company.
6079:Journal of Chemical Education
5995:Canadian Journal of Chemistry
5307:Journal of Chemical Education
4993:Journal of Chemical Education
4499:Journal of Physical Chemistry
4400:Zemann, J. (1 January 1958).
4136:Journal of Chemical Education
2269:are salts, some of which are
2006:colour of an aqueous solution
1872:Salts are characteristically
1623:(although some salts such as
6392:Russell, Michael S. (2009).
6270:Freemantle, Michael (2009).
5610:"Naming Chemical Substances"
4934:"Periodic Trends and Oxides"
4898:Advanced inorganic chemistry
4880:10.1016/0079-6786(65)90009-9
4836:10.1016/0167-2738(81)90198-3
4778:10.1524/zkri.1978.147.14.167
4525:"YCl3 – Yttrium trichloride"
2027:of metal ions, so colorless
2004:is often different from the
1884:rather than stationary in a
1806:ammonium hexachloroplatinate
1260:Interstitial sites occupied
398:Solid lead(II) sulfate (PbSO
6453:Zumdahl, Steven S. (1989).
5711:Satake, M; Mido, Y (1995).
4651:Journal of Chemical Physics
2685:(with the 2+ charge on the
2447:, including increasing the
2012:form of the same compound.
1779:enthalpy change of solution
1486:Ionic compounds containing
1424:non-stoichiometric compound
6542:
6394:The chemistry of fireworks
6230:Barrow, Gordon M. (1988).
6213:Atkins' physical chemistry
5835:Zumdahl & Zumdahl 2015
5823:Zumdahl & Zumdahl 2015
5667:Atkins & de Paula 2006
5655:Atkins & de Paula 2006
5643:Atkins & de Paula 2006
4807:Ashcroft & Mermin 1977
4441:Ashcroft & Mermin 1977
4371:Ashcroft & Mermin 1977
4305:Ashcroft & Mermin 1977
3978:Ashcroft & Mermin 1977
3235:Sodium phosphate monobasic
2535:
2501:, salts are often used as
2289:Salts can elicit all five
2094:is made red-orange by the
1993:
1927:, and in various kinds of
1783:entropy change of solution
1711:
1670:ductile–brittle transition
1603:Melting and boiling points
1400:
811:exhibit a small degree of
774:van der Waals interactions
757:
484:solid-state reaction route
150:). Each ion can be either
29:
5401:10.1107/S0567740878003830
5284:10.1080/14786436708220903
5253:10.1080/14786435908233367
5051:McQuarrie & Rock 1991
4743:10.1080/14786437708239734
4427:10.1107/S0365110X5800013X
3348:COONa, smells similar to
3305:__, or they end with __NO
3263:Sodium phosphate tribasic
2453:freezing-point depression
2380:is the other half of the
1828:(sparingly soluble), and
1749:, but tends to be low in
1579:Some ions are classed as
1529:, and salts that produce
1262:
1259:
1254:
1251:
929:
926:
923:
918:
915:
789:Pauli exclusion principle
742:. Sodium loses its outer
304:In 1913 the structure of
5583:Kurlansky, Mark (2003).
3407:Salt metathesis reaction
3249:Sodium phosphate dibasic
2621:to distinguish it from K
1810:potassium cobaltinitrite
683:salt metathesis reaction
461:water of crystallization
410:react directly with the
406:Many metals such as the
4896:Prakash, Satya (1945).
4614:Krishna's IAS Chemistry
4266:Carter, Robert (2016).
4032:Pauling, Linus (1948).
3586:10.1351/goldbook.S05447
3282:Strong salts or strong
2461:electrical double layer
2457:boiling-point elevation
1907:In some unusual salts:
1852:Electrical conductivity
1763:ion–dipole interactions
1737:of salts is highest in
1494:, and those containing
1403:crystallographic defect
807:pairs such as those in
783:(most simple ions have
446:neutralization reaction
424:. Once the solution is
274:electrically insulating
5571:Wenk & Bulakh 2004
5515:Chemistry of Materials
5268:Philosophical Magazine
5237:Philosophical Magazine
4727:Philosophical Magazine
4637:Wenk & Bulakh 2004
4599:10.1002/anie.196600951
4406:Acta Crystallographica
4275:CH370 Lecture Material
3901:Wold & Dwight 1993
3889:Wold & Dwight 1993
3804:Wold & Dwight 1993
3789:Wold & Dwight 1993
3746:10.1098/rspa.1928.0188
3667:10.1098/rspa.1913.0082
3619:10.1098/rspa.1913.0040
3471:chromium(III) chloride
2445:colligative properties
2356:") may smell like the
2181:is made violet by the
2173:potassium permanganate
2059:is made yellow by the
1869:
1709:
1506:(O) are classified as
1490:(H) are classified as
1478:
1449:crystal-field symmetry
856:
755:
748:electron configuration
514:salt-forming reactions
403:
391:
386:, the mineral form of
314:William Lawrence Bragg
301:
251:crystalline structures
238:(O) are classified as
68:
6516:Alchemical substances
6161:Salt: A World History
5608:Lower, Simon (2014).
5585:Salt: a world history
4183:Philosophy of Science
3467:yttrium(III) chloride
2590:, is an example of a
1896:or are melted into a
1894:dissolved in a liquid
1868:charge transfer salt.
1859:
1707:
1514:. Salts that produce
1470:
1148:two-thirds octahedral
1120:two-thirds octahedral
992:alternate tetrahedral
895:of the salt, and the
878:Kapustinskii equation
851:The unit cell of the
850:
725:
480:solid-state synthesis
459:formed also includes
397:
382:
299:
44:
6480:. Cengage Learning.
5500:10.1039/JM9910100157
4050:10.1039/JR9480001461
2364:) and cyanides like
2339:monosodium glutamate
2319:potassium bitartrate
2078:potassium dichromate
1652:Most salts are very
1198: = 0.2247,
1090:rhodium(III) bromide
1083:one-third octahedral
796:electronic structure
766:electrostatic forces
746:to give it a stable
512:Salts are formed by
292:History of discovery
105:electrostatic forces
6193:Solid state physics
6091:1982JChEd..59..964F
5780:, pp. 129–133.
5768:, pp. 108–117.
5714:Chemistry of Colour
5669:, pp. 163–169.
5657:, pp. 761–770.
5645:, pp. 150–157.
5393:1978AcCrB..34..689C
5354:, pp. 413–415.
5319:1937JChEd..14...34S
5276:1967PMag...15..567K
5245:1959PMag....4.1316J
5077:10.1021/ja01391a014
5028:Inorganic chemistry
5005:1955JChEd..32..550D
4770:1978ZK....147..167A
4735:1977PMag...36..501T
4702:1948JChPh..16.1091V
4663:1947JChPh..15..174V
4511:10.1021/j100894a062
4418:1958AcCry..11...55Z
4373:, pp. 382–387.
4148:1968JChEd..45..643P
4121:10.1021/ja00905a001
4019:10.1021/ja01206a003
3737:1928RSPSA.121..155J
3706:10.1021/cr60038a002
3658:1913RSPSA..89..246B
3611:1913RSPSA..88..428B
3288:strong electrolytes
2894:Quaternary ammonium
2382:reversible reaction
2309:, which will cause
2201:nickel(II) chloride
1996:Colour of chemicals
1954:cobalt(II) chloride
1909:fast-ion conductors
1878:conduct electricity
1842:potassium carbonate
1787:cerium(III) sulfate
1777:, the negative net
1773:energy exceeds the
1694:coordination number
1589:aluminium hydroxide
1552:, the result is an
1512:acid–base reactions
1457:d-electron orbitals
1248:
1237:site distributions
1213: = 0.4142
924:Interstitial sites
912:
891:. Depending on the
874:Born–Mayer equation
870:Born–Landé equation
326:inorganic compounds
310:William Henry Bragg
284:they become highly
6506:Chemical compounds
6233:Physical chemistry
5679:Reeves TG (1986).
4824:Solid State Ionics
4319:, p. 444–445.
4222:chemistryworld.com
3927:, p. 161–162.
3915:, p. 312–313.
3864:, p. 144–145.
3852:, p. 133–140.
2711:Stock nomenclature
2560:magnesium chloride
2499:chemical synthesis
2484:water fluoridation
2155:copper(II) sulfate
2117:cobalt(II) nitrate
1870:
1846:ammonium carbonate
1718:When simple salts
1710:
1629:iron(III) chloride
1625:aluminium chloride
1564:, the result is a
1536:when dissolved in
1521:when dissolved in
1479:
1445:lattice parameters
1276:Madelung constant
1263:Example structure
1246:
1227:site distributions
1163:Depends on charges
952:Madelung constant
946:Madelung constant
910:
857:
756:
442:acid–base reaction
404:
392:
324:order. Many other
318:nearest-neighbours
308:was determined by
302:
228:ammonium carbonate
69:
6487:978-1-305-68804-9
6468:978-0-669-16708-5
6445:978-94-011-1476-9
6424:978-1-107-39390-5
6403:978-0-85404-127-5
6384:978-0-8014-0333-0
6358:978-0-7167-2169-7
6349:General chemistry
6339:978-0-471-41526-8
6304:978-0-85404-438-2
6283:978-1-84755-161-0
6262:978-1-4425-1147-7
6243:978-0-07-003905-6
6222:978-0-19-870072-2
6203:978-0-03-083993-1
6184:Ashcroft, Neil W.
6099:10.1021/ed059p964
6067:, pp. 77–78.
6034:, pp. 76–77.
5955:, pp. 75–76.
5943:, pp. 36–37.
5927:978-0-534-99766-3
5866:978-3-527-60525-5
5809:978-0-444-53599-3
5724:978-81-7141-276-1
5594:978-0-09-928199-3
5527:10.1021/cm980236q
5521:(10): 2870–2888.
5468:978-981-02-3473-7
5342:, pp. 89–91.
5214:978-0-323-13894-9
5172:10.1021/jp050430h
5166:(13): 6040–6043.
5111:978-94-010-0458-9
5037:978-0-19-964182-6
5013:10.1021/ed032p550
4977:978-0-03-072373-5
4962:General Chemistry
4907:978-81-219-0263-2
4711:10.1063/1.1746736
4671:10.1063/1.1746464
4623:978-81-87224-70-9
4568:978-0-8412-2725-5
4505:(10): 3611–3614.
4469:978-0-12-118420-9
4343:978-0-7487-7516-3
4156:10.1021/ed045p643
4115:(22): 3533–3539.
4093:978-0-470-56753-1
3903:, pp. 79–81.
3829:978-0-521-52958-7
3397:Bonding in solids
3104:hydrofluoric acid
3018:hydrochloric acid
2727:iron(III) sulfate
2596:empirical formula
2594:). To obtain the
2542:According to the
2327:magnesium sulfate
2239:magnesium sulfate
1751:nonpolar solvents
1349:
1348:
1241:
1240:
1235:Depends on cation
1225:Depends on cation
862:Madelung constant
837:quantum mechanics
503:electron transfer
85:chemical compound
16:(Redirected from
6533:
6491:
6472:
6460:
6449:
6428:
6407:
6388:
6376:
6362:
6343:
6319:
6317:
6316:
6307:. Archived from
6287:
6266:
6247:
6226:
6207:
6188:Mermin, N. David
6149:
6148:
6143:. Archived from
6124:
6118:
6112:
6103:
6102:
6074:
6068:
6062:
6056:
6050:
6035:
6029:
6023:
6017:
6011:
6010:
5974:
5968:
5962:
5956:
5950:
5944:
5938:
5932:
5931:
5913:
5907:
5901:
5895:
5889:
5883:
5877:
5871:
5870:
5854:
5844:
5838:
5832:
5826:
5820:
5814:
5813:
5797:
5787:
5781:
5775:
5769:
5763:
5757:
5751:
5745:
5739:
5733:
5732:
5708:
5702:
5701:
5699:
5698:
5692:
5685:
5676:
5670:
5664:
5658:
5652:
5646:
5640:
5634:
5633:
5631:
5629:
5605:
5599:
5598:
5580:
5574:
5568:
5562:
5556:
5543:
5537:
5531:
5530:
5510:
5504:
5503:
5483:
5477:
5476:
5452:
5443:
5437:
5431:
5430:
5428:
5426:
5411:
5405:
5404:
5376:
5370:
5364:
5355:
5349:
5343:
5337:
5331:
5330:
5327:10.1021/ed014p34
5302:
5296:
5295:
5263:
5257:
5256:
5232:
5223:
5222:
5198:
5192:
5191:
5155:
5144:
5138:
5132:
5126:
5120:
5119:
5095:
5089:
5088:
5071:(4): 1036–1045.
5060:
5054:
5048:
5042:
5041:
5023:
5017:
5016:
4988:
4982:
4981:
4965:
4955:
4949:
4948:
4946:
4945:
4930:
4924:
4918:
4912:
4911:
4893:
4884:
4883:
4863:
4840:
4839:
4819:
4810:
4804:
4798:
4797:
4764:(1–4): 167–176.
4753:
4747:
4746:
4722:
4716:
4715:
4713:
4681:
4675:
4674:
4646:
4640:
4634:
4628:
4627:
4609:
4603:
4602:
4582:
4573:
4572:
4554:
4545:
4544:
4542:
4540:
4521:
4515:
4514:
4494:
4488:
4487:
4481:
4473:
4455:
4444:
4438:
4432:
4431:
4429:
4397:
4391:
4385:
4374:
4368:
4362:
4361:
4355:
4347:
4329:
4320:
4314:
4308:
4302:
4293:
4292:
4290:
4289:
4283:
4272:
4268:"Lattice Energy"
4263:
4257:
4251:
4245:
4239:
4233:
4232:
4230:
4228:
4213:
4207:
4206:
4178:
4172:
4166:
4160:
4159:
4131:
4125:
4124:
4104:
4098:
4097:
4079:
4073:
4072:
4070:
4069:
4060:. Archived from
4029:
4023:
4022:
4002:
3996:
3990:
3981:
3975:
3969:
3963:
3952:
3946:
3940:
3934:
3928:
3922:
3916:
3910:
3904:
3898:
3892:
3886:
3880:
3874:
3865:
3859:
3853:
3847:
3838:
3837:
3813:
3807:
3801:
3792:
3786:
3775:
3769:
3763:
3757:
3751:
3750:
3748:
3731:(787): 155–171.
3716:
3710:
3709:
3694:Chemical Reviews
3689:
3672:
3671:
3669:
3652:(610): 246–248.
3637:
3631:
3630:
3605:(605): 428–438.
3594:
3588:
3565:
3548:
3534:
3528:
3524:
3518:
3504:
3498:
3484:
3478:
3463:
3457:
3454:
3205:
3204:
3203:
3184:
3183:
3182:
3163:
3162:
3161:
3143:
3142:
3141:
3122:
3121:
3120:
3101:
3100:
3099:
3084:hydrocyanic acid
3081:
3080:
3079:
3061:
3060:
3059:
3051:
3050:
3043:
3042:
3034:
3033:
3015:
3014:
3013:
2995:
2994:
2993:
2974:
2973:
2972:
2965:
2964:
2945:
2944:
2943:
2930:
2929:
2928:
2906:
2905:
2904:
2890:
2889:
2888:
2881:
2880:
2872:
2871:
2857:
2856:
2855:
2842:
2841:
2840:
2827:
2826:
2825:
2816:
2815:
2814:
2801:
2800:
2799:
2786:
2785:
2784:
2744:
2743:
2742:
2723:iron(II) sulfate
2715:oxidation number
2703:iron(3+) sulfate
2683:iron(2+) sulfate
2670:oxidation states
2585:
2584:
2583:
2512:. To obtain the
2449:osmotic pressure
2414:milk of magnesia
2366:hydrogen cyanide
2252:
2230:
2214:
2196:
2195:
2194:
2191:
2180:
2168:
2150:
2134:
2112:
2111:
2110:
2107:
2093:
2073:
2072:
2071:
2068:
2058:
2002:colour of a salt
1972:
1947:
1929:chemical sensors
1838:sodium carbonate
1830:lead(II) sulfate
1767:heat of solution
1574:ammonium acetate
1482:Acidity/basicity
1420:oxidation states
1415:Schottky defects
1391:
1379:
1359:to occur, so an
1316:calcium fluoride
1249:
1057:cadmium chloride
913:
809:caesium fluoride
507:Born–Haber cycle
417:gases to salts.
365:Kazimierz Fajans
361:Paul Peter Ewald
334:X-ray reflection
244:sodium hydroxide
225:
224:
223:
209:
208:
207:
193:
192:
191:
149:
148:
147:
140:
139:
21:
6541:
6540:
6536:
6535:
6534:
6532:
6531:
6530:
6496:
6495:
6494:
6488:
6475:
6469:
6452:
6446:
6431:
6425:
6410:
6404:
6391:
6385:
6365:
6359:
6346:
6340:
6324:Kittel, Charles
6322:
6314:
6312:
6305:
6290:
6284:
6269:
6263:
6250:
6244:
6229:
6223:
6210:
6204:
6182:
6178:
6153:
6152:
6141:
6126:
6125:
6121:
6113:
6106:
6076:
6075:
6071:
6063:
6059:
6051:
6038:
6030:
6026:
6018:
6014:
6007:10.1139/v75-015
5992:
5988:
5984:
5980:
5976:
5975:
5971:
5963:
5959:
5951:
5947:
5939:
5935:
5928:
5915:
5914:
5910:
5902:
5898:
5890:
5886:
5878:
5874:
5867:
5846:
5845:
5841:
5837:, pp. 823.
5833:
5829:
5825:, pp. 822.
5821:
5817:
5810:
5789:
5788:
5784:
5776:
5772:
5764:
5760:
5752:
5748:
5740:
5736:
5725:
5710:
5709:
5705:
5696:
5694:
5690:
5683:
5678:
5677:
5673:
5665:
5661:
5653:
5649:
5641:
5637:
5627:
5625:
5617:
5607:
5606:
5602:
5595:
5582:
5581:
5577:
5569:
5565:
5557:
5546:
5538:
5534:
5512:
5511:
5507:
5485:
5484:
5480:
5469:
5454:
5453:
5446:
5438:
5434:
5424:
5422:
5413:
5412:
5408:
5378:
5377:
5373:
5365:
5358:
5350:
5346:
5338:
5334:
5304:
5303:
5299:
5265:
5264:
5260:
5234:
5233:
5226:
5215:
5200:
5199:
5195:
5157:
5156:
5147:
5143:, pp. 3–4.
5141:Freemantle 2009
5139:
5135:
5129:Freemantle 2009
5127:
5123:
5112:
5097:
5096:
5092:
5062:
5061:
5057:
5049:
5045:
5038:
5025:
5024:
5020:
4990:
4989:
4985:
4978:
4957:
4956:
4952:
4943:
4941:
4932:
4931:
4927:
4919:
4915:
4908:
4895:
4894:
4887:
4865:
4864:
4843:
4821:
4820:
4813:
4805:
4801:
4755:
4754:
4750:
4724:
4723:
4719:
4683:
4682:
4678:
4648:
4647:
4643:
4635:
4631:
4624:
4611:
4610:
4606:
4584:
4583:
4576:
4569:
4556:
4555:
4548:
4538:
4536:
4523:
4522:
4518:
4496:
4495:
4491:
4474:
4470:
4457:
4456:
4447:
4439:
4435:
4399:
4398:
4394:
4386:
4377:
4369:
4365:
4348:
4344:
4331:
4330:
4323:
4315:
4311:
4303:
4296:
4287:
4285:
4281:
4270:
4265:
4264:
4260:
4252:
4248:
4240:
4236:
4226:
4224:
4215:
4214:
4210:
4180:
4179:
4175:
4167:
4163:
4133:
4132:
4128:
4106:
4105:
4101:
4094:
4081:
4080:
4076:
4067:
4065:
4031:
4030:
4026:
4004:
4003:
3999:
3991:
3984:
3976:
3972:
3964:
3955:
3947:
3943:
3935:
3931:
3923:
3919:
3911:
3907:
3899:
3895:
3887:
3883:
3875:
3868:
3860:
3856:
3848:
3841:
3830:
3815:
3814:
3810:
3802:
3795:
3787:
3778:
3770:
3766:
3758:
3754:
3718:
3717:
3713:
3691:
3690:
3675:
3639:
3638:
3634:
3596:
3595:
3591:
3566:
3562:
3557:
3552:
3551:
3545:
3541:
3535:
3531:
3525:
3521:
3517:structure type.
3516:
3511:
3505:
3501:
3496:
3491:
3485:
3481:
3477:structure type.
3476:
3464:
3460:
3455:
3451:
3446:
3393:
3362:
3355:
3347:
3316:
3312:
3308:
3304:
3280:
3272:
3268:
3258:
3254:
3244:
3240:
3202:
3199:
3198:
3197:
3195:
3187:phosphoric acid
3181:
3178:
3177:
3176:
3174:
3160:
3158:
3157:
3156:
3154:
3140:
3137:
3136:
3135:
3133:
3119:
3116:
3115:
3114:
3112:
3098:
3096:
3095:
3094:
3092:
3078:
3076:
3075:
3074:
3072:
3058:
3055:
3054:
3053:
3049:
3047:
3046:
3045:
3041:
3038:
3037:
3036:
3032:
3030:
3029:
3028:
3026:
3012:
3010:
3009:
3008:
3006:
2992:
2989:
2988:
2987:
2985:
2971:
2969:
2968:
2967:
2963:
2960:
2959:
2958:
2956:
2942:
2940:
2939:
2938:
2936:
2927:
2925:
2924:
2923:
2921:
2903:
2900:
2899:
2898:
2896:
2887:
2885:
2884:
2883:
2879:
2876:
2875:
2874:
2870:
2867:
2866:
2865:
2863:
2854:
2852:
2851:
2850:
2848:
2839:
2837:
2836:
2835:
2833:
2824:
2822:
2821:
2820:
2818:
2813:
2811:
2810:
2809:
2807:
2798:
2796:
2795:
2794:
2792:
2783:
2780:
2779:
2778:
2776:
2760:ferrous sulfate
2741:
2738:
2737:
2736:
2734:
2700:
2696:
2692:
2680:
2660:
2656:
2628:
2624:
2616:
2582:
2579:
2578:
2577:
2575:
2569:
2565:
2557:
2546:recommended by
2540:
2534:
2526:redox reactions
2491:pyrotechnicians
2390:
2299:sodium chloride
2287:
2250:
2246:
2242:
2235:sodium chloride
2228:
2224:
2220:
2216:
2212:
2208:
2204:
2192:
2189:
2188:
2186:
2179:
2175:
2166:
2162:
2158:
2148:
2144:
2140:
2132:
2128:
2124:
2120:
2108:
2105:
2104:
2102:
2098:
2092:
2088:
2084:
2080:
2069:
2066:
2065:
2063:
2057:
2053:
2049:
2047:sodium chromate
2042:. For example:
2017:absorption band
1998:
1992:
1991:
1990:
1989:
1988:
1985:
1981:
1976:
1973:
1965:
1964:
1962:
1957:
1948:
1937:
1917:supercapacitors
1886:crystal lattice
1854:
1826:calcium sulfate
1716:
1702:
1690:compressibility
1686:
1684:Compressibility
1650:
1617:vapour pressure
1605:
1593:aluminium oxide
1496:electropositive
1484:
1465:
1410:Frenkel defects
1405:
1399:
1398:
1397:
1396:
1395:
1394:Schottky defect
1392:
1384:
1383:
1380:
1369:
1329:
1306:
1291:cesium chloride
1287:entirely filled
1272:
1271:Critical radius
1256:
1236:
1226:
1212:
1205:
1199:
1197:
1190:
1177:
1173:
1165:and structure
1164:
1143:
1114:
1110:
1077:
1025:all tetrahedral
1019:
970:sodium chloride
939:
938:Critical radius
920:
905:Pauling's rules
845:
805:electropositive
801:electronegative
781:electron shells
762:
740:sodium fluoride
720:
711:
708:↓ + 2 NaNO
707:
703:
699:
695:
691:
676:
669:
661:
654:
649:, e.g., 2
636:
624:
615:, e.g., 2
603:
596:
583:
576:
569:
565:
543:
532:
412:electronegative
401:
388:sodium chloride
377:
306:sodium chloride
294:
222:
219:
218:
217:
215:
206:
203:
202:
201:
199:
190:
187:
186:
185:
183:
172:sodium chloride
146:
144:
143:
142:
138:
135:
134:
133:
131:
101:electric charge
51:sodium chloride
39:
36:Sodium chloride
28:
23:
22:
15:
12:
11:
5:
6539:
6537:
6529:
6528:
6523:
6518:
6513:
6508:
6498:
6497:
6493:
6492:
6486:
6473:
6467:
6450:
6444:
6429:
6423:
6408:
6402:
6389:
6383:
6367:Pauling, Linus
6363:
6357:
6344:
6338:
6320:
6303:
6288:
6282:
6267:
6261:
6248:
6242:
6227:
6221:
6208:
6202:
6179:
6177:
6174:
6173:
6172:
6157:Mark Kurlansky
6151:
6150:
6147:on 2007-09-11.
6139:
6119:
6104:
6069:
6057:
6036:
6024:
6012:
6001:(1): 114–118.
5990:
5986:
5982:
5978:
5969:
5957:
5945:
5933:
5926:
5908:
5896:
5884:
5872:
5865:
5839:
5827:
5815:
5808:
5782:
5770:
5758:
5746:
5734:
5723:
5703:
5671:
5659:
5647:
5635:
5615:
5600:
5593:
5575:
5573:, p. 774.
5563:
5561:, p. 107.
5544:
5542:, p. 105.
5532:
5505:
5478:
5467:
5444:
5442:, p. 341.
5432:
5417:. 2011-05-22.
5406:
5371:
5369:, p. 422.
5356:
5344:
5332:
5297:
5258:
5224:
5213:
5193:
5145:
5133:
5121:
5110:
5090:
5055:
5053:, p. 503.
5043:
5036:
5018:
4983:
4976:
4950:
4925:
4923:, p. 376.
4913:
4906:
4885:
4841:
4811:
4809:, p. 384.
4799:
4748:
4717:
4676:
4657:(4): 174–180.
4641:
4639:, p. 778.
4629:
4622:
4604:
4574:
4567:
4546:
4516:
4489:
4468:
4445:
4443:, p. 386.
4433:
4392:
4375:
4363:
4342:
4321:
4309:
4307:, p. 383.
4294:
4258:
4256:, p. 509.
4246:
4234:
4208:
4195:10.1086/594534
4189:(5): 909–920.
4173:
4171:, p. 676.
4161:
4126:
4099:
4092:
4074:
4024:
4013:(2): 171–173.
3997:
3982:
3980:, p. 379.
3970:
3968:, p. 507.
3953:
3941:
3929:
3917:
3905:
3893:
3881:
3879:, p. 417.
3866:
3854:
3839:
3828:
3808:
3793:
3776:
3774:, p. 312.
3764:
3762:, p. 505.
3752:
3711:
3673:
3632:
3589:
3559:
3558:
3556:
3553:
3550:
3549:
3543:
3539:
3529:
3519:
3514:
3509:
3499:
3494:
3489:
3479:
3474:
3458:
3448:
3447:
3445:
3442:
3441:
3440:
3435:
3430:
3425:
3420:
3415:
3409:
3404:
3399:
3392:
3389:
3361:
3358:
3353:
3345:
3342:sodium acetate
3314:
3310:
3306:
3302:
3279:
3276:
3275:
3274:
3270:
3266:
3260:
3256:
3252:
3246:
3242:
3238:
3212:
3211:
3200:
3190:
3179:
3169:
3159:
3149:
3138:
3128:
3117:
3107:
3097:
3087:
3077:
3067:
3056:
3048:
3039:
3031:
3021:
3011:
3001:
2990:
2980:
2970:
2961:
2947:
2946:
2941:
2931:
2926:
2916:
2901:
2891:
2886:
2877:
2868:
2858:
2853:
2843:
2838:
2828:
2823:
2812:
2802:
2797:
2787:
2781:
2764:ferric sulfate
2739:
2719:Roman numerals
2698:
2694:
2690:
2678:
2658:
2654:
2626:
2622:
2614:
2592:polyatomic ion
2580:
2572:sodium sulfate
2567:
2563:
2555:
2533:
2530:
2451:, and causing
2441:ionic strength
2422:oil of vitriol
2389:
2386:
2358:conjugate acid
2313:if ingested),
2311:lead poisoning
2307:lead diacetate
2286:
2285:Taste and odor
2283:
2263:
2262:
2248:
2232:
2210:
2198:
2170:
2164:
2152:
2130:
2114:
2096:dichromate ion
2075:
1983:
1979:
1974:
1967:
1966:
1960:
1949:
1942:
1941:
1940:
1939:
1938:
1936:
1933:
1853:
1850:
1822:barium sulfate
1794:lattice energy
1775:lattice energy
1739:polar solvents
1701:
1698:
1685:
1682:
1649:
1646:
1613:boiling points
1604:
1601:
1597:lead(II) oxide
1570:conjugate base
1483:
1480:
1464:
1461:
1393:
1386:
1385:
1382:Frenkel defect
1381:
1374:
1373:
1372:
1371:
1370:
1368:
1365:
1347:
1346:
1344:
1342:
1337:
1334:
1331:
1327:
1323:
1322:
1320:
1318:
1313:
1310:
1307:
1304:
1300:
1299:
1296:
1293:
1288:
1285:
1282:
1278:
1277:
1274:
1269:
1265:
1264:
1261:
1258:
1253:
1252:Stoichiometry
1239:
1238:
1233:
1228:
1223:
1221:inverse spinel
1214:
1210:
1203:
1195:
1188:
1183:
1180:
1178:
1175:
1171:
1167:
1166:
1161:
1156:
1154:
1152:
1149:
1146:
1144:
1141:
1137:
1136:
1133:
1128:
1126:
1124:
1121:
1118:
1115:
1112:
1108:
1104:
1103:
1100:
1098:bismuth iodide
1095:
1092:
1087:
1084:
1081:
1078:
1075:
1071:
1070:
1067:
1065:cadmium iodide
1062:
1059:
1054:
1051:
1048:
1045:
1042:
1041:
1039:
1037:
1034:
1029:
1026:
1023:
1020:
1017:
1013:
1012:
1009:
1004:
1001:
996:
993:
990:
987:
984:
983:
980:
975:
972:
967:
964:
963:all octahedral
961:
958:
954:
953:
950:
947:
944:
941:
936:
932:
931:
928:
925:
922:
917:
916:Stoichiometry
844:
841:
833:metallic bonds
829:hydrogen bonds
758:Main article:
752:exothermically
728:electron shell
719:
716:
715:
714:
713:
712:
709:
705:
701:
697:
693:
689:
679:
674:
667:
659:
652:
647:base anhydride
639:
634:
622:
613:acid anhydride
605:
601:
594:
585:
581:
574:
567:
563:
546:
541:
530:
488:stoichiometric
476:freezing point
450:spectator ions
426:supersaturated
399:
376:
373:
369:thermochemical
357:Erwin Madelung
293:
290:
262:boiling points
242:, for example
220:
204:
188:
145:
136:
81:ionic compound
26:
24:
14:
13:
10:
9:
6:
4:
3:
2:
6538:
6527:
6524:
6522:
6519:
6517:
6514:
6512:
6509:
6507:
6504:
6503:
6501:
6489:
6483:
6479:
6474:
6470:
6464:
6459:
6458:
6451:
6447:
6441:
6437:
6436:
6430:
6426:
6420:
6416:
6415:
6409:
6405:
6399:
6395:
6390:
6386:
6380:
6375:
6374:
6368:
6364:
6360:
6354:
6350:
6345:
6341:
6335:
6331:
6330:
6325:
6321:
6311:on 2016-02-03
6310:
6306:
6300:
6296:
6295:
6289:
6285:
6279:
6275:
6274:
6268:
6264:
6258:
6254:
6249:
6245:
6239:
6235:
6234:
6228:
6224:
6218:
6214:
6209:
6205:
6199:
6195:
6194:
6189:
6185:
6181:
6180:
6175:
6170:
6169:0-14-200161-9
6166:
6162:
6158:
6155:
6154:
6146:
6142:
6140:9780471193500
6136:
6132:
6131:
6123:
6120:
6117:, p. 38.
6116:
6111:
6109:
6105:
6100:
6096:
6092:
6088:
6084:
6080:
6073:
6070:
6066:
6061:
6058:
6055:, p. 77.
6054:
6049:
6047:
6045:
6043:
6041:
6037:
6033:
6028:
6025:
6022:, p. 76.
6021:
6016:
6013:
6008:
6004:
6000:
5996:
5973:
5970:
5967:, p. 75.
5966:
5961:
5958:
5954:
5949:
5946:
5942:
5937:
5934:
5929:
5923:
5919:
5912:
5909:
5906:, p. 69.
5905:
5900:
5897:
5894:, p. 70.
5893:
5888:
5885:
5882:, p. 68.
5881:
5876:
5873:
5868:
5862:
5858:
5853:
5852:
5843:
5840:
5836:
5831:
5828:
5824:
5819:
5816:
5811:
5805:
5801:
5796:
5795:
5786:
5783:
5779:
5774:
5771:
5767:
5762:
5759:
5756:, p. 82.
5755:
5750:
5747:
5744:, p. 14.
5743:
5738:
5735:
5730:
5726:
5720:
5716:
5715:
5707:
5704:
5693:on 2017-02-08
5689:
5682:
5675:
5672:
5668:
5663:
5660:
5656:
5651:
5648:
5644:
5639:
5636:
5623:
5619:
5611:
5604:
5601:
5596:
5590:
5586:
5579:
5576:
5572:
5567:
5564:
5560:
5555:
5553:
5551:
5549:
5545:
5541:
5536:
5533:
5528:
5524:
5520:
5516:
5509:
5506:
5501:
5497:
5493:
5489:
5482:
5479:
5474:
5470:
5464:
5460:
5459:
5451:
5449:
5445:
5441:
5436:
5433:
5420:
5416:
5410:
5407:
5402:
5398:
5394:
5390:
5386:
5382:
5375:
5372:
5368:
5363:
5361:
5357:
5353:
5348:
5345:
5341:
5336:
5333:
5328:
5324:
5320:
5316:
5312:
5308:
5301:
5298:
5293:
5289:
5285:
5281:
5277:
5273:
5269:
5262:
5259:
5254:
5250:
5246:
5242:
5238:
5231:
5229:
5225:
5220:
5216:
5210:
5206:
5205:
5197:
5194:
5189:
5185:
5181:
5177:
5173:
5169:
5165:
5161:
5154:
5152:
5150:
5146:
5142:
5137:
5134:
5130:
5125:
5122:
5117:
5113:
5107:
5103:
5102:
5094:
5091:
5086:
5082:
5078:
5074:
5070:
5066:
5059:
5056:
5052:
5047:
5044:
5039:
5033:
5029:
5022:
5019:
5014:
5010:
5006:
5002:
4998:
4994:
4987:
4984:
4979:
4973:
4969:
4964:
4963:
4954:
4951:
4939:
4935:
4929:
4926:
4922:
4917:
4914:
4909:
4903:
4899:
4892:
4890:
4886:
4881:
4877:
4873:
4869:
4862:
4860:
4858:
4856:
4854:
4852:
4850:
4848:
4846:
4842:
4837:
4833:
4829:
4825:
4818:
4816:
4812:
4808:
4803:
4800:
4795:
4791:
4787:
4783:
4779:
4775:
4771:
4767:
4763:
4759:
4752:
4749:
4744:
4740:
4736:
4732:
4728:
4721:
4718:
4712:
4707:
4703:
4699:
4695:
4691:
4687:
4680:
4677:
4672:
4668:
4664:
4660:
4656:
4652:
4645:
4642:
4638:
4633:
4630:
4625:
4619:
4615:
4608:
4605:
4600:
4596:
4593:(1): 95–106.
4592:
4588:
4581:
4579:
4575:
4570:
4564:
4560:
4553:
4551:
4547:
4534:
4530:
4526:
4520:
4517:
4512:
4508:
4504:
4500:
4493:
4490:
4485:
4479:
4471:
4465:
4461:
4454:
4452:
4450:
4446:
4442:
4437:
4434:
4428:
4423:
4419:
4415:
4411:
4407:
4403:
4396:
4393:
4390:, p. 65.
4389:
4384:
4382:
4380:
4376:
4372:
4367:
4364:
4359:
4353:
4345:
4339:
4335:
4328:
4326:
4322:
4318:
4313:
4310:
4306:
4301:
4299:
4295:
4280:
4276:
4269:
4262:
4259:
4255:
4250:
4247:
4244:, p. 64.
4243:
4238:
4235:
4223:
4219:
4212:
4209:
4204:
4200:
4196:
4192:
4188:
4184:
4177:
4174:
4170:
4165:
4162:
4157:
4153:
4149:
4145:
4141:
4137:
4130:
4127:
4122:
4118:
4114:
4110:
4103:
4100:
4095:
4089:
4085:
4078:
4075:
4064:on 2021-12-07
4063:
4059:
4055:
4051:
4047:
4044:: 1461–1467.
4043:
4039:
4035:
4028:
4025:
4020:
4016:
4012:
4008:
4001:
3998:
3995:, p. 65.
3994:
3989:
3987:
3983:
3979:
3974:
3971:
3967:
3962:
3960:
3958:
3954:
3951:, p. 61.
3950:
3945:
3942:
3938:
3933:
3930:
3926:
3921:
3918:
3914:
3909:
3906:
3902:
3897:
3894:
3891:, p. 79.
3890:
3885:
3882:
3878:
3873:
3871:
3867:
3863:
3858:
3855:
3851:
3846:
3844:
3840:
3835:
3831:
3825:
3821:
3820:
3812:
3809:
3806:, p. 82.
3805:
3800:
3798:
3794:
3791:, p. 71.
3790:
3785:
3783:
3781:
3777:
3773:
3768:
3765:
3761:
3756:
3753:
3747:
3742:
3738:
3734:
3730:
3726:
3722:
3715:
3712:
3707:
3703:
3700:(1): 93–170.
3699:
3695:
3688:
3686:
3684:
3682:
3680:
3678:
3674:
3668:
3663:
3659:
3655:
3651:
3647:
3643:
3636:
3633:
3628:
3624:
3620:
3616:
3612:
3608:
3604:
3600:
3593:
3590:
3587:
3583:
3579:
3575:
3574:
3569:
3564:
3561:
3554:
3546:
3533:
3530:
3523:
3520:
3512:
3503:
3500:
3492:
3483:
3480:
3472:
3468:
3462:
3459:
3453:
3450:
3443:
3439:
3436:
3434:
3431:
3429:
3426:
3424:
3421:
3419:
3416:
3413:
3412:Bresle method
3410:
3408:
3405:
3403:
3400:
3398:
3395:
3394:
3390:
3388:
3386:
3382:
3378:
3374:
3370:
3366:
3359:
3357:
3351:
3343:
3339:
3335:
3331:
3327:
3323:
3318:
3299:
3297:
3293:
3289:
3285:
3277:
3264:
3261:
3250:
3247:
3236:
3233:
3232:
3231:
3229:
3225:
3221:
3217:
3209:
3208:sulfuric acid
3194:
3191:
3188:
3173:
3170:
3167:
3153:
3150:
3147:
3132:
3129:
3126:
3111:
3108:
3105:
3091:
3088:
3085:
3071:
3068:
3065:
3025:
3022:
3019:
3005:
3002:
2999:
2998:carbonic acid
2984:
2981:
2978:
2955:
2952:
2951:
2950:
2935:
2932:
2920:
2917:
2914:
2910:
2907:, R being an
2895:
2892:
2862:
2859:
2847:
2844:
2832:
2829:
2806:
2803:
2791:
2788:
2775:
2772:
2771:
2770:
2767:
2765:
2761:
2756:
2752:
2748:
2732:
2728:
2724:
2720:
2716:
2712:
2708:
2704:
2688:
2684:
2676:
2671:
2666:
2664:
2652:
2648:
2644:
2640:
2636:
2632:
2620:
2612:
2608:
2604:
2599:
2597:
2593:
2589:
2573:
2561:
2553:
2552:stoichiometry
2549:
2545:
2539:
2531:
2529:
2527:
2523:
2519:
2515:
2511:
2506:
2504:
2500:
2495:
2492:
2487:
2485:
2481:
2476:
2474:
2470:
2466:
2462:
2458:
2454:
2450:
2446:
2442:
2438:
2433:
2431:
2427:
2423:
2419:
2418:muriatic acid
2415:
2411:
2407:
2403:
2399:
2395:
2387:
2385:
2383:
2379:
2375:
2371:
2367:
2363:
2359:
2355:
2351:
2347:
2342:
2340:
2336:
2332:
2328:
2324:
2320:
2316:
2312:
2308:
2304:
2300:
2296:
2292:
2284:
2282:
2280:
2276:
2272:
2268:
2260:
2256:
2241:heptahydrate
2240:
2236:
2233:
2202:
2199:
2184:
2174:
2171:
2157:pentahydrate
2156:
2153:
2138:
2118:
2115:
2097:
2079:
2076:
2062:
2048:
2045:
2044:
2043:
2041:
2037:
2032:
2030:
2026:
2021:
2018:
2013:
2011:
2007:
2003:
1997:
1987:
1971:
1963:
1955:
1952:
1946:
1934:
1932:
1930:
1926:
1922:
1918:
1914:
1913:ionic glasses
1910:
1905:
1903:
1899:
1895:
1891:
1887:
1883:
1879:
1875:
1867:
1863:
1858:
1851:
1849:
1847:
1843:
1839:
1835:
1831:
1827:
1823:
1819:
1815:
1811:
1807:
1803:
1799:
1795:
1790:
1788:
1784:
1780:
1776:
1772:
1768:
1764:
1760:
1756:
1752:
1748:
1747:ionic liquids
1744:
1740:
1736:
1731:
1729:
1725:
1721:
1715:
1706:
1699:
1697:
1695:
1691:
1683:
1681:
1679:
1675:
1671:
1667:
1663:
1659:
1655:
1647:
1645:
1641:
1639:
1635:
1634:ionic liquids
1630:
1626:
1622:
1618:
1614:
1610:
1602:
1600:
1598:
1594:
1590:
1586:
1582:
1577:
1575:
1571:
1567:
1563:
1559:
1555:
1551:
1547:
1543:
1539:
1535:
1532:
1528:
1524:
1520:
1517:
1513:
1509:
1505:
1501:
1497:
1493:
1489:
1488:hydrogen ions
1481:
1477:
1473:
1469:
1462:
1460:
1458:
1454:
1450:
1446:
1442:
1437:
1433:
1429:
1425:
1421:
1416:
1411:
1404:
1390:
1378:
1366:
1364:
1362:
1358:
1354:
1353:ionic liquids
1345:
1343:
1341:
1340:lithium oxide
1338:
1335:
1332:
1325:
1324:
1321:
1319:
1317:
1314:
1311:
1308:
1302:
1301:
1297:
1294:
1292:
1289:
1286:
1283:
1280:
1279:
1275:
1270:
1267:
1266:
1257:coordination
1250:
1244:
1234:
1232:
1229:
1224:
1222:
1218:
1215:
1209:
1202:
1194:
1187:
1184:
1181:
1179:
1169:
1168:
1162:
1160:
1157:
1155:
1153:
1150:
1147:
1145:
1139:
1138:
1134:
1132:
1129:
1127:
1125:
1122:
1119:
1116:
1106:
1105:
1101:
1099:
1096:
1093:
1091:
1088:
1085:
1082:
1079:
1073:
1072:
1068:
1066:
1063:
1060:
1058:
1055:
1052:
1049:
1046:
1044:
1043:
1040:
1038:
1035:
1033:
1030:
1027:
1024:
1021:
1015:
1014:
1010:
1008:
1005:
1002:
1000:
997:
994:
991:
988:
986:
985:
981:
979:
976:
973:
971:
968:
965:
962:
959:
956:
955:
951:
948:
945:
942:
937:
934:
933:
921:coordination
914:
908:
906:
902:
898:
894:
893:stoichiometry
890:
886:
881:
879:
875:
871:
867:
863:
854:
849:
842:
840:
838:
834:
830:
825:
822:
818:
817:Fajans' rules
814:
810:
806:
802:
797:
792:
790:
786:
785:closed shells
782:
779:
775:
771:
767:
761:
760:Ionic bonding
753:
749:
745:
741:
737:
733:
729:
724:
717:
687:
686:
684:
680:
678:
670:
663:
655:
648:
644:
640:
638:
630:
626:
618:
614:
610:
606:
604:
597:
590:
586:
584:
577:
570:
559:
555:
551:
547:
545:
537:
533:
526:
522:
518:
517:
516:
515:
510:
508:
504:
500:
496:
491:
489:
485:
481:
477:
472:
470:
466:
462:
458:
457:ionic crystal
453:
451:
447:
443:
438:
433:
431:
427:
423:
418:
416:
413:
409:
408:alkali metals
396:
389:
385:
381:
374:
372:
370:
366:
362:
358:
354:
350:
346:
342:
337:
335:
331:
327:
323:
319:
315:
311:
307:
298:
291:
289:
287:
283:
279:
275:
271:
267:
263:
259:
254:
252:
247:
245:
241:
237:
233:
229:
213:
197:
181:
177:
173:
169:
165:
161:
157:
153:
129:
125:
121:
117:
112:
110:
106:
102:
98:
94:
90:
86:
82:
78:
74:
66:
63:
59:
56:
52:
49:structure of
48:
43:
37:
33:
19:
6477:
6456:
6434:
6413:
6393:
6372:
6348:
6327:
6313:. Retrieved
6309:the original
6293:
6272:
6252:
6232:
6212:
6192:
6176:Bibliography
6160:
6145:the original
6130:Biochemistry
6129:
6122:
6082:
6078:
6072:
6060:
6027:
6015:
5998:
5994:
5972:
5960:
5948:
5936:
5917:
5911:
5899:
5887:
5875:
5850:
5842:
5830:
5818:
5793:
5785:
5778:Russell 2009
5773:
5766:Russell 2009
5761:
5754:Russell 2009
5749:
5742:Russell 2009
5737:
5713:
5706:
5695:. Retrieved
5688:the original
5674:
5662:
5650:
5638:
5626:. Retrieved
5613:
5603:
5584:
5578:
5566:
5559:Pauling 1960
5540:Pauling 1960
5535:
5518:
5514:
5508:
5491:
5487:
5481:
5457:
5440:Zumdahl 1989
5435:
5423:. Retrieved
5409:
5384:
5380:
5374:
5347:
5335:
5310:
5306:
5300:
5267:
5261:
5236:
5203:
5196:
5163:
5159:
5136:
5131:, p. 1.
5124:
5100:
5093:
5068:
5064:
5058:
5046:
5027:
5021:
4996:
4992:
4986:
4961:
4953:
4942:. Retrieved
4928:
4916:
4897:
4871:
4867:
4827:
4823:
4802:
4786:11380/738457
4761:
4757:
4751:
4726:
4720:
4696:(12): 1091.
4693:
4689:
4679:
4654:
4650:
4644:
4632:
4613:
4607:
4590:
4586:
4558:
4537:. Retrieved
4528:
4519:
4502:
4498:
4492:
4459:
4436:
4412:(1): 55–56.
4409:
4405:
4395:
4366:
4333:
4317:Zumdahl 1989
4312:
4286:. Retrieved
4274:
4261:
4254:Pauling 1960
4249:
4237:
4225:. Retrieved
4221:
4211:
4186:
4182:
4176:
4164:
4139:
4135:
4129:
4112:
4108:
4102:
4083:
4077:
4066:. Retrieved
4062:the original
4041:
4037:
4027:
4010:
4006:
4000:
3993:Pauling 1960
3973:
3966:Pauling 1960
3944:
3939:, p. 6.
3937:Pauling 1960
3932:
3920:
3913:Zumdahl 1989
3908:
3896:
3884:
3862:Zumdahl 1989
3857:
3850:Zumdahl 1989
3818:
3811:
3772:Zumdahl 1989
3767:
3760:Pauling 1960
3755:
3728:
3724:
3714:
3697:
3693:
3649:
3645:
3635:
3602:
3598:
3592:
3571:
3563:
3532:
3522:
3502:
3482:
3461:
3452:
3363:
3322:electrolytes
3319:
3300:
3281:
3227:
3223:
3219:
3215:
3213:
3146:nitrous acid
2948:
2911:group or an
2768:
2750:
2746:
2707:formula unit
2667:
2650:
2646:
2642:
2610:
2606:
2602:
2600:
2544:nomenclature
2541:
2532:Nomenclature
2522:electrolysis
2507:
2496:
2488:
2477:
2434:
2391:
2346:strong salts
2343:
2291:basic tastes
2288:
2264:
2203:hexahydrate
2183:permanganate
2119:hexahydrate
2061:chromate ion
2033:
2022:
2014:
1999:
1977:
1958:
1906:
1902:electrolytes
1871:
1791:
1732:
1717:
1687:
1678:dislocations
1674:plastic flow
1672:occurs, and
1651:
1642:
1638:substituents
1621:molten salts
1606:
1578:
1527:alkali salts
1485:
1476:ionic liquid
1436:entropically
1406:
1350:
1255:Cation:anion
1242:
1207:
1200:
1192:
1185:
919:Cation:anion
901:radius ratio
897:coordination
885:close-packed
882:
858:
826:
793:
763:
726:A schematic
511:
492:
473:
454:
434:
419:
405:
353:Alfred Landé
338:
303:
255:
253:when solid.
248:
113:
80:
76:
70:
6085:(11): 964.
4999:(11): 550.
4921:Kittel 2005
4874:: 265–303.
4388:Kittel 2005
4242:Kittel 2005
4169:Barrow 1988
4142:(10): 643.
3949:Kittel 2005
3925:Barrow 1988
3428:Ionic bonds
3418:Carboxylate
3377:metabolites
3373:amino acids
3365:Zwitterions
3350:acetic acid
3296:nonvolatile
3284:electrolyte
3125:nitric acid
3064:citric acid
2977:acetic acid
2524:, in which
2473:suspensions
2437:electrolyte
2430:slaked lime
2402:agriculture
2398:common salt
2348:") are non-
2139:cobalt(II)
1648:Brittleness
1558:strong base
1546:strong acid
1540:are called
1525:are called
1432:equilibrium
1361:ionic glass
1336:half filled
1312:half filled
999:zinc blende
889:interstices
853:zinc blende
821:HSAB theory
730:diagram of
437:counterions
432:minerals.
349:Fritz Haber
341:theoretical
322:crystalline
276:, but when
158:), such as
109:ionic bonds
6500:Categories
6315:2023-02-05
6115:Brown 2009
6065:IUPAC 2005
6053:IUPAC 2005
6032:IUPAC 2005
6020:IUPAC 2005
5965:IUPAC 2005
5953:IUPAC 2005
5941:Brown 2009
5904:IUPAC 2005
5892:IUPAC 2005
5880:IUPAC 2005
5697:2016-01-18
5628:14 January
5494:(2): 157.
5425:2 December
5387:(2): 689.
5367:Brown 2009
5352:Brown 2009
5340:Brown 2009
4944:2015-11-10
4539:19 January
4529:ChemTube3D
4288:2016-01-19
4227:22 January
4068:2021-12-01
3877:Brown 2009
3555:References
3497:structure.
3360:Zwitterion
2861:Pyridinium
2731:uranyl(2+)
2536:See also:
2503:precursors
2378:hydrolysis
2354:weak salts
1994:See also:
1925:fuel cells
1874:insulators
1834:carbonates
1735:solubility
1724:dissociate
1712:See also:
1700:Solubility
1615:and a low
1585:zinc oxide
1581:amphoteric
1542:acid salts
1463:Properties
1401:See also:
1357:nucleation
935:Occupancy
471:material.
286:conductive
264:, and are
226:) ions in
178:, such as
176:polyatomic
156:simple ion
126:, such as
118:, such as
18:Ionic salt
6457:Chemistry
5313:(1): 34.
5292:0031-8086
5180:1520-6106
5085:0002-7863
4830:: 77–82.
4794:101158673
4478:cite book
4352:cite book
4203:120135228
3402:Ioliomics
3313:, or __CH
3228:polybasic
3216:monobasic
3172:Phosphate
2983:Carbonate
2846:Potassium
2831:Magnesium
2701:is named
2681:is named
2661:is named
2651:tetrakis-
2639:potassium
2617:is named
2570:is named
2558:is named
2514:elemental
2494:present.
2469:emulsions
2465:colloidal
2426:saltpeter
2237:NaCl and
2217:[NiCl
2141:[Co(H
2029:anhydrous
2025:hydration
1951:Anhydrous
1921:batteries
1816:and many
1802:potassium
1771:solvation
1753:(such as
1741:(such as
1658:malleably
1566:base salt
1562:weak acid
1554:acid salt
1550:weak base
1516:hydroxide
1500:hydroxide
1453:splitting
1298:1.762675
982:<1.73
978:nickeline
866:Ewald sum
855:structure
843:Structure
813:covalency
664:→ 2
627:→ 2
469:anhydrous
430:evaporite
422:solutions
375:Formation
282:dissolved
232:hydroxide
212:carbonate
166:(Na) and
162:(F), and
152:monatomic
122:(Cl), or
116:inorganic
73:chemistry
6369:(1960).
6326:(2005).
6190:(1977).
6159:(2002).
5729:Archived
5622:Archived
5473:Archived
5419:Archived
5219:Archived
5188:16851662
5116:Archived
4938:Archived
4533:Archived
4279:Archived
4058:18893624
3834:Archived
3627:13112732
3438:Salinity
3391:See also
3385:proteins
3381:peptides
3369:molecule
3326:volatile
3278:Strength
3224:tribasic
3090:Fluoride
3004:Chloride
2774:Ammonium
2562:, and Na
2518:smelting
2480:fluoride
2394:minerals
2350:volatile
2293:, e.g.,
2275:pigments
2267:minerals
2247:·7H
2209:·6H
2163:·5H
2137:hydrated
2129:·6H
2040:solvates
2010:hydrated
1818:sulfates
1814:nitrates
1759:gasoline
1728:solvated
1720:dissolve
1666:cleavage
1662:fracture
1531:hydrogen
1502:(OH) or
1428:F-center
1159:ilmenite
1135:25.0312
1131:corundum
1032:fluorite
1007:wurtzite
974:1.747565
744:electron
736:fluorine
556:, e.g.,
527:, e.g.,
415:halogens
345:Max Born
234:(OH) or
196:ammonium
170:(Cl) in
168:chloride
160:fluoride
154:(termed
120:chloride
62:chloride
6087:Bibcode
5389:Bibcode
5315:Bibcode
5272:Bibcode
5241:Bibcode
5001:Bibcode
4766:Bibcode
4731:Bibcode
4698:Bibcode
4659:Bibcode
4414:Bibcode
4144:Bibcode
3733:Bibcode
3654:Bibcode
3607:Bibcode
3375:, many
3332:to the
3309:, __ClO
3220:dibasic
3193:Sulfate
3131:Nitrite
3110:Nitrate
3070:Cyanide
3027:HOC(COO
3024:Citrate
2954:Acetate
2790:Calcium
2753:to the
2588:sulfate
2463:around
2410:calomel
2374:ammonia
2370:almonds
2362:vinegar
2329:), and
2271:soluble
2255:cations
1812:. Most
1722:, they
1654:brittle
1609:melting
1367:Defects
1231:olivine
1036:5.03878
794:If the
770:Coulomb
718:Bonding
681:In the
611:and an
552:and an
523:and an
499:Group 2
495:Group 1
465:hydrate
270:brittle
258:melting
194:), and
180:sulfate
128:acetate
124:organic
107:termed
93:cations
58:cations
47:crystal
6484:
6465:
6442:
6421:
6400:
6381:
6355:
6336:
6301:
6280:
6259:
6240:
6219:
6200:
6167:
6137:
5985:and Cs
5924:
5863:
5859:–365.
5806:
5721:
5591:
5465:
5290:
5211:
5186:
5178:
5108:
5083:
5034:
4974:
4904:
4792:
4620:
4565:
4466:
4340:
4201:
4090:
4056:
3826:
3625:
3433:Natron
3423:Halide
3383:, and
3356:COOH.
2934:Copper
2919:Sodium
2675:arabic
2635:symbol
2611:tetra-
2428:, and
2335:savory
2323:bitter
2259:anions
2185:anion
2036:colors
1935:Colour
1923:, and
1911:, and
1898:liquid
1882:mobile
1798:sodium
1755:petrol
1560:and a
1548:and a
1295:0.7321
1273:ratio
1217:spinel
1151:0.4142
1123:0.4142
1086:0.4142
1053:0.4142
1028:0.2247
1011:1.641
1003:1.6381
995:0.2247
966:0.4142
940:ratio
872:, the
732:sodium
704:→ PbSO
645:and a
384:Halite
363:, and
278:melted
210:) and
164:sodium
97:anions
65:anions
55:sodium
6511:Salts
5691:(PDF)
5684:(PDF)
4790:S2CID
4282:(PDF)
4271:(PDF)
4199:S2CID
3623:S2CID
3568:IUPAC
3527:this.
3444:Notes
3292:water
3222:, or
3166:water
3152:Oxide
2915:group
2909:alkyl
2755:Latin
2733:ion,
2647:tris-
2548:IUPAC
2406:borax
2331:umami
2303:sweet
2295:salty
2265:Some
2121:Co(NO
1745:) or
1743:water
1538:water
1523:water
1508:bases
1504:oxide
1492:acids
1474:, an
1351:Some
1268:Name
1102:8.26
1069:4.71
949:Name
943:Name
778:outer
688:Pb(NO
629:NaClO
550:metal
444:or a
330:atoms
240:bases
236:oxide
174:, or
83:is a
6521:Ions
6482:ISBN
6463:ISBN
6440:ISBN
6419:ISBN
6398:ISBN
6379:ISBN
6353:ISBN
6334:ISBN
6299:ISBN
6278:ISBN
6257:ISBN
6238:ISBN
6217:ISBN
6198:ISBN
6165:ISBN
6135:ISBN
5989:MgCl
5981:MgCl
5922:ISBN
5861:ISBN
5804:ISBN
5719:ISBN
5630:2016
5614:Chem
5589:ISBN
5463:ISBN
5427:2012
5288:ISSN
5209:ISBN
5184:PMID
5176:ISSN
5106:ISBN
5081:ISSN
5032:ISBN
4972:ISBN
4902:ISBN
4618:ISBN
4563:ISBN
4541:2016
4484:link
4464:ISBN
4358:link
4338:ISBN
4229:2024
4088:ISBN
4054:PMID
3824:ISBN
3578:salt
3538:MgAl
3508:FeCl
3488:MoCl
3469:and
3344:, CH
3338:base
3334:acid
3330:odor
3237:(NaH
3035:)(CH
2913:aryl
2817:and
2805:Iron
2762:and
2749:and
2747:-ous
2725:and
2643:bis-
2637:for
2625:MgCl
2607:tri-
2510:ores
2471:and
2455:and
2388:Uses
2315:sour
2279:dyes
2257:and
2243:MgSO
2205:NiCl
2176:KMnO
2159:CuSO
2000:The
1978:CoCl
1959:CoCl
1866:TCNQ
1844:and
1808:and
1792:The
1733:The
1688:The
1664:via
1627:and
1611:and
1595:and
1534:ions
1519:ions
1439:and
1094:6.67
1061:5.61
831:and
734:and
696:+ Na
666:NaNO
643:acid
617:NaOH
609:base
600:CaCl
573:MgSO
554:acid
525:acid
521:base
312:and
268:and
266:hard
260:and
89:ions
77:salt
75:, a
45:The
32:Salt
6095:doi
6003:doi
5993:".
5857:359
5523:doi
5496:doi
5397:doi
5323:doi
5280:doi
5249:doi
5168:doi
5164:109
5073:doi
5009:doi
4968:128
4876:doi
4832:doi
4782:hdl
4774:doi
4762:147
4739:doi
4706:doi
4667:doi
4595:doi
4507:doi
4422:doi
4191:doi
4152:doi
4117:doi
4046:doi
4015:doi
3741:doi
3729:121
3702:doi
3662:doi
3615:doi
3582:doi
3580:".
3336:or
3265:(Na
3255:HPO
3251:(Na
3073:C≡N
3044:COO
2966:COO
2751:-ic
2717:in
2693:(SO
2603:di-
2520:or
2497:In
2341:).
2333:or
2321:),
2301:),
2187:MnO
2064:CrO
2054:CrO
1982:·6H
1862:TTF
1455:of
1333:4:8
1309:8:4
1284:8:8
1140:ABO
1117:6:4
1080:6:2
1047:6:3
1022:8:4
989:4:4
960:6:6
651:HNO
641:An
536:HCl
497:or
280:or
141:COO
79:or
71:In
34:or
6502::
6186:;
6107:^
6093:.
6083:59
6081:.
6039:^
5999:53
5997:.
5802:.
5800:22
5727:.
5620:.
5612:.
5547:^
5519:10
5517:.
5490:.
5471:.
5447:^
5395:.
5385:34
5383:.
5359:^
5321:.
5311:14
5309:.
5286:.
5278:.
5247:.
5227:^
5217:.
5182:.
5174:.
5162:.
5148:^
5114:.
5079:.
5069:50
5067:.
5007:.
4997:32
4995:.
4970:.
4936:.
4888:^
4870:.
4844:^
4826:.
4814:^
4788:.
4780:.
4772:.
4760:.
4737:.
4704:.
4694:16
4692:.
4688:.
4665:.
4655:15
4653:.
4589:.
4577:^
4549:^
4527:.
4503:69
4501:.
4480:}}
4476:{{
4448:^
4420:.
4410:11
4408:.
4404:.
4378:^
4354:}}
4350:{{
4324:^
4297:^
4277:.
4273:.
4220:.
4197:.
4187:75
4185:.
4150:.
4140:45
4138:.
4113:85
4111:.
4052:.
4042:17
4040:.
4036:.
4011:68
4009:.
3985:^
3956:^
3869:^
3842:^
3832:.
3796:^
3779:^
3739:.
3727:.
3723:.
3698:11
3696:.
3676:^
3660:.
3650:89
3648:.
3644:.
3621:.
3613:.
3603:88
3601:.
3570:,
3387:.
3379:,
3352:CH
3298:.
3269:PO
3241:PO
3218:,
3196:SO
3175:PO
3134:NO
3113:NO
3007:Cl
2986:CO
2957:CH
2937:Cu
2922:Na
2897:NR
2882:NH
2834:Mg
2819:Fe
2808:Fe
2793:Ca
2777:NH
2766:.
2735:UO
2687:Fe
2665:.
2649:,
2645:,
2629:,
2609:,
2605:,
2586:,
2576:SO
2566:SO
2486:.
2475:.
2432:.
2424:,
2420:,
2416:,
2412:,
2408:,
2225:O)
2221:(H
2145:O)
2106:2−
2099:Cr
2085:Cr
2067:2−
2050:Na
1931:.
1919:,
1848:.
1840:,
1824:,
1800:,
1680:.
1599:.
1591:,
1587:,
1576:.
1472:+−
1303:MX
1281:MX
1219:,
1170:AB
1074:MX
1016:MX
957:MX
907:.
880:.
700:SO
671:+
658:Na
656:+
631:+
621:Cl
619:+
607:A
598:→
593:Cl
591:+
589:Ca
578:+
571:→
566:SO
560:+
558:Mg
548:A
544:Cl
540:NH
538:→
534:+
529:NH
519:A
509:.
452:.
359:,
355:,
351:,
347:,
246:.
216:CO
200:NH
184:SO
132:CH
111:.
6490:.
6471:.
6448:.
6427:.
6406:.
6387:.
6361:.
6342:.
6318:.
6286:.
6265:.
6246:.
6225:.
6206:.
6171:.
6101:.
6097::
6089::
6009:.
6005::
5991:4
5987:2
5983:4
5979:2
5930:.
5869:.
5812:.
5700:.
5632:.
5616:1
5597:.
5529:.
5525::
5502:.
5498::
5492:1
5429:.
5403:.
5399::
5391::
5329:.
5325::
5317::
5294:.
5282::
5274::
5255:.
5251::
5243::
5190:.
5170::
5087:.
5075::
5040:.
5015:.
5011::
5003::
4980:.
4947:.
4910:.
4882:.
4878::
4872:2
4838:.
4834::
4828:5
4796:.
4784::
4776::
4768::
4745:.
4741::
4733::
4714:.
4708::
4700::
4673:.
4669::
4661::
4626:.
4601:.
4597::
4591:5
4571:.
4543:.
4513:.
4509::
4486:)
4472:.
4430:.
4424::
4416::
4360:)
4346:.
4291:.
4231:.
4205:.
4193::
4158:.
4154::
4146::
4123:.
4119::
4096:.
4071:.
4048::
4021:.
4017::
3749:.
3743::
3735::
3708:.
3704::
3670:.
3664::
3656::
3629:.
3617::
3609::
3584::
3544:4
3542:O
3540:2
3515:3
3510:3
3495:3
3490:3
3475:3
3354:3
3346:3
3315:3
3311:4
3307:3
3303:4
3273:)
3271:4
3267:3
3259:)
3257:4
3253:2
3245:)
3243:4
3239:2
3210:)
3206:(
3201:4
3189:)
3185:(
3180:4
3168:)
3164:(
3155:O
3148:)
3144:(
3139:2
3127:)
3123:(
3118:3
3106:)
3102:(
3093:F
3086:)
3082:(
3066:)
3062:(
3057:2
3052:)
3040:2
3020:)
3016:(
3000:)
2996:(
2991:3
2979:)
2975:(
2962:3
2902:4
2878:5
2873:H
2869:5
2864:C
2849:K
2782:4
2740:2
2699:3
2697:)
2695:4
2691:2
2679:4
2659:2
2657:)
2655:4
2627:4
2623:2
2615:3
2581:4
2574:(
2568:4
2564:2
2556:2
2368:(
2337:(
2325:(
2317:(
2305:(
2297:(
2251:O
2249:2
2245:4
2231:.
2229:]
2227:4
2223:2
2219:2
2213:O
2211:2
2207:2
2197:.
2193:4
2190:−
2178:4
2167:O
2165:2
2161:4
2151:.
2149:]
2147:6
2143:2
2133:O
2131:2
2127:2
2125:)
2123:3
2113:.
2109:7
2103:O
2101:2
2091:7
2089:O
2087:2
2083:2
2081:K
2074:.
2070:4
2056:4
2052:2
1986:O
1984:2
1980:2
1961:2
1956:,
1864:/
1757:/
1330:X
1328:2
1326:M
1305:2
1211:O
1208:r
1206:/
1204:B
1201:r
1196:O
1193:r
1191:/
1189:A
1186:r
1176:4
1174:O
1172:2
1142:3
1113:3
1111:X
1109:2
1107:M
1076:3
1018:2
803:/
710:3
706:4
702:4
698:2
694:2
692:)
690:3
677:O
675:2
673:H
668:3
662:O
660:2
653:3
637:O
635:2
633:H
625:O
623:2
602:2
595:2
582:2
580:H
575:4
568:4
564:2
562:H
542:4
531:3
402:)
400:4
221:3
214:(
205:4
198:(
189:4
182:(
137:3
130:(
91:(
38:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.