458:
515:
17:
38:
of the formula PRR′R″, where R, R′, R″ = H, alkyl, aryl, etc. They are a subset of chiral phosphines, a broader class of compounds where the stereogenic center can reside at sites other than phosphorus. P-chirality exploits the high barrier for inversion of phosphines, which ensures that
358:
363:
499:
224:
556:
492:
217:
605:
396:
610:
580:
600:
485:
381:
210:
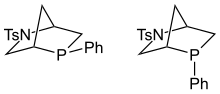
549:
391:
180:
Mislow, Kurt; Baechler, Raymond D. (1971). "Effect of ligand electronegativity on the inversion barrier of phosphines".
43:
readily. The inversion barrier is relatively insensitive to substituents for triorganophosphines. By contrast, most
234:
353:
35:
585:
279:
79:
542:
417:
331:
248:
427:
412:
263:
105:
93:
60:
595:
325:
48:
575:
386:
284:
373:
162:
590:
422:
348:
258:
189:
152:
144:
465:
432:
320:
300:
274:
109:
202:
526:
469:
157:
132:
569:
315:
437:
310:
253:
457:
514:
67:
305:
522:
166:
16:
40:
193:
148:
100:-Chiral phosphines have been investigated for two main applications, as
101:
71:
75:
44:
15:
206:
82:
and related reactions. DIPAMP is prepared by coupling the
530:
473:
78:. These chelating ligands support catalysts used in
405:
372:
341:
293:
241:
133:"Chiral Phosphines in Nucleophilic Organocatalysis"
364:Ultraviolet–visible spectroscopy of stereoisomers
92:-Chiral phosphines are of particular interest in
550:
493:
218:
131:Xiao, Y.; Sun, Z.; Guo, H.; Kwon, O. (2014).
8:
557:
543:
500:
486:
225:
211:
203:
156:
182:Journal of the American Chemical Society
120:
137:Beilstein Journal of Organic Chemistry
126:
124:
86:-chiral methylphenylanisylphosphine.
7:
511:
509:
454:
452:
14:
359:NMR spectroscopy of stereoisomers
47:of the type NRR′R″ undergo rapid
513:
456:
397:Diastereomeric recrystallization
1:
39:enantiomers of PRR'R" do not
529:. You can help Knowledge by
472:. You can help Knowledge by
392:Chiral column chromatography
59:Most chiral phosphines are
627:
508:
451:
354:Chiral derivatizing agents
235:enantioselective synthesis
36:organophosphorus compounds
24:-chiral "Kwon phosphines".
280:Supramolecular chirality
80:asymmetric hydrogenation
606:Chemical reaction stubs
108:and as nucleophiles in
70:. Famous examples are
611:Chemical process stubs
581:Coordination chemistry
25:
601:Stereochemistry stubs
418:Chiral pool synthesis
332:Diastereomeric excess
106:homogeneous catalysts
19:
428:Asymmetric catalysis
413:Asymmetric induction
94:asymmetric catalysis
326:Enantiomeric excess
194:10.1021/ja00732a036
149:10.3762/bjoc.10.218
49:pyramidal inversion
423:Chiral auxiliaries
387:Kinetic resolution
285:Inherent chirality
270:-symmetric ligands
32:-Chiral phosphines
26:
538:
537:
481:
480:
446:
445:
382:Recrystallization
374:Chiral resolution
618:
559:
552:
545:
517:
510:
502:
495:
488:
460:
453:
349:Optical rotation
294:Chiral molecules
259:Planar chirality
227:
220:
213:
204:
198:
197:
177:
171:
170:
160:
128:
626:
625:
621:
620:
619:
617:
616:
615:
586:Stereochemistry
566:
565:
564:
563:
507:
506:
466:stereochemistry
449:
447:
442:
433:Organocatalysis
401:
368:
337:
321:Racemic mixture
289:
275:Axial chirality
269:
242:Chirality types
237:
231:
201:
179:
178:
174:
130:
129:
122:
118:
110:organocatalysis
104:for asymmetric
64:
57:
55:Research themes
12:
11:
5:
624:
622:
614:
613:
608:
603:
598:
593:
588:
583:
578:
568:
567:
562:
561:
554:
547:
539:
536:
535:
518:
505:
504:
497:
490:
482:
479:
478:
461:
444:
443:
441:
440:
435:
430:
425:
420:
415:
409:
407:
403:
402:
400:
399:
394:
389:
384:
378:
376:
370:
369:
367:
366:
361:
356:
351:
345:
343:
339:
338:
336:
335:
329:
323:
318:
313:
308:
303:
297:
295:
291:
290:
288:
287:
282:
277:
272:
267:
261:
256:
251:
245:
243:
239:
238:
232:
230:
229:
222:
215:
207:
200:
199:
188:(3): 773–774.
172:
119:
117:
114:
62:
56:
53:
13:
10:
9:
6:
4:
3:
2:
623:
612:
609:
607:
604:
602:
599:
597:
594:
592:
589:
587:
584:
582:
579:
577:
574:
573:
571:
560:
555:
553:
548:
546:
541:
540:
534:
532:
528:
525:article is a
524:
519:
516:
512:
503:
498:
496:
491:
489:
484:
483:
477:
475:
471:
468:article is a
467:
462:
459:
455:
450:
439:
436:
434:
431:
429:
426:
424:
421:
419:
416:
414:
411:
410:
408:
404:
398:
395:
393:
390:
388:
385:
383:
380:
379:
377:
375:
371:
365:
362:
360:
357:
355:
352:
350:
347:
346:
344:
340:
333:
330:
327:
324:
322:
319:
317:
316:Meso compound
314:
312:
309:
307:
304:
302:
299:
298:
296:
292:
286:
283:
281:
278:
276:
273:
271:
266:
262:
260:
257:
255:
252:
250:
247:
246:
244:
240:
236:
228:
223:
221:
216:
214:
209:
208:
205:
195:
191:
187:
183:
176:
173:
168:
164:
159:
154:
150:
146:
143:: 2089–2121.
142:
138:
134:
127:
125:
121:
115:
113:
111:
107:
103:
99:
95:
91:
87:
85:
81:
77:
73:
69:
66:
54:
52:
50:
46:
42:
37:
33:
31:
23:
18:
531:expanding it
520:
474:expanding it
463:
448:
438:Biocatalysis
311:Diastereomer
301:Stereoisomer
264:
254:Stereocenter
233:Concepts in
185:
181:
175:
140:
136:
97:
89:
88:
83:
68:diphosphines
58:
29:
28:
27:
21:
596:Phosphanes
570:Categories
306:Enantiomer
116:References
65:-symmetric
576:Catalysis
523:catalysis
406:Reactions
249:Chirality
342:Analysis
167:25246969
41:racemize
591:Ligands
158:4168899
102:ligands
165:
155:
72:DIPAMP
45:amines
521:This
464:This
76:BINAP
527:stub
470:stub
334:(de)
328:(ee)
163:PMID
74:and
34:are
20:Two
190:doi
153:PMC
145:doi
572::
186:93
184:.
161:.
151:.
141:10
139:.
135:.
123:^
112:.
96:.
51:.
558:e
551:t
544:v
533:.
501:e
494:t
487:v
476:.
268:2
265:C
226:e
219:t
212:v
196:.
192::
169:.
147::
98:P
90:P
84:P
63:2
61:C
30:P
22:P
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.