28:
101:
182:, and suitably designed apparatus exploit this principle with a gradient that will yield different purities in particular temperature zones along the collection surface. Such techniques are especially helpful when the requirement is to refine or separate multiple products or impurities from the same mix of raw materials. It is necessary in particular when some of the required products have similar
154:, then the sublimed material can be collected from the cooled surface once heating ceases and the vacuum is released. Although this may be quite convenient for small quantities, adapting sublimation processes to large volume is generally not practical with the apparatus becoming extremely large and generally needing to be disassembled to recover products and remove residue.
112:
314:
157:
Among the advantages of applying the principle to certain materials are the comparatively low working temperatures, reduced exposure to gases such as oxygen that might harm certain products, and the ease with which it can be performed on extremely small quantities. The same apparatus may also be used
158:
for conventional distillation of extremely small quantities due to the very small volume and surface area between evaporating and condensing regions, although this is generally only useful if the cold finger can be cold enough to solidify the condensate.
123:
A typical sublimation apparatus separates a mix of appropriate solid materials in a vessel in which it applies heat under a controllable atmosphere (air,
204:
293:
107:
being purified on a sublimation apparatus. Note the white purified camphor on the cold finger, and the dark-brown crude product.
318:
31:
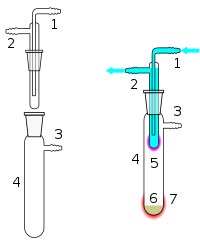
Simple sublimation apparatus. Water usually cold, is circulated in cold finger to allow the desired compound to be deposited.
344:
339:
334:
171:
349:
354:
179:
175:
81:
136:
132:
150:
which for very low-temperature sublimation may actually be cryogenically cooled. If the operation is a
167:
73:
289:
283:
237:
183:
77:
151:
187:
127:
or inert gas). If the material is not at first solid, then it may freeze under reduced
100:
27:
328:
17:
199:
85:
255:
166:
More sophisticated variants of sublimation apparatus include those that apply a
147:
116:
140:
241:
111:
313:
135:
and condenses as a purified compound on a cooled surface, leaving the non-
128:
104:
225:
124:
89:
119:, freshly sublimed on the cold finger of the sublimation apparatus.
99:
26:
84:. In principle, the operation resembles purification by
146:
The form of the cooled surface often is a so-called
226:"The Earliest Stages in the Evolution of the Still"
88:, except that the products do not pass through a
285:Chemical Process Equipment: Selection and Design
174:of different fractions along the cold surface.
8:
277:
275:
131:. Conditions are so chosen that the solid
205:List of purification methods in chemistry
288:. Butterworth-Heinemann. pp. 729–.
110:
216:
7:
25:
312:
170:so as to allow for controlled
1:
143:or solid products behind.
371:
282:James R. Couper (2012).
180:statistical distribution
176:Thermodynamic processes
115:Dark green crystals of
72:is equipment, commonly
224:Levey, Martin (1960).
120:
108:
76:, for purification of
61:
319:Sublimation apparatus
114:
103:
70:sublimation apparatus
30:
18:Sublimation apparatus
345:Separation processes
340:Laboratory glassware
335:Alchemical processes
321:at Wikimedia Commons
260:www.websters1913.com
168:temperature gradient
162:Temperature gradient
74:laboratory glassware
48:Sublimation chamber
350:Chemical equipment
184:sublimation points
121:
109:
62:
52:Sublimed compound
40:Cooling water out
355:Phase transitions
317:Media related to
295:978-0-12-396959-0
172:recrystallization
36:Cooling water in
16:(Redirected from
362:
316:
300:
299:
279:
270:
269:
267:
266:
256:"Webster's 1913"
252:
246:
245:
221:
60:External heating
44:Vacuum/gas line
21:
370:
369:
365:
364:
363:
361:
360:
359:
325:
324:
309:
304:
303:
296:
281:
280:
273:
264:
262:
254:
253:
249:
223:
222:
218:
213:
196:
188:pressure curves
164:
98:
56:Crude material
32:
23:
22:
15:
12:
11:
5:
368:
366:
358:
357:
352:
347:
342:
337:
327:
326:
323:
322:
308:
307:External links
305:
302:
301:
294:
271:
247:
215:
214:
212:
209:
208:
207:
202:
195:
192:
163:
160:
97:
94:
24:
14:
13:
10:
9:
6:
4:
3:
2:
367:
356:
353:
351:
348:
346:
343:
341:
338:
336:
333:
332:
330:
320:
315:
311:
310:
306:
297:
291:
287:
286:
278:
276:
272:
261:
257:
251:
248:
243:
239:
235:
231:
227:
220:
217:
210:
206:
203:
201:
198:
197:
193:
191:
189:
185:
181:
177:
173:
169:
161:
159:
155:
153:
152:batch process
149:
144:
142:
138:
134:
130:
126:
118:
113:
106:
102:
95:
93:
91:
87:
83:
80:by selective
79:
75:
71:
67:
59:
55:
51:
47:
43:
39:
35:
29:
19:
284:
263:. Retrieved
259:
250:
236:(1): 31–34.
233:
229:
219:
200:Distillation
165:
156:
145:
122:
90:liquid phase
86:distillation
69:
65:
63:
57:
53:
49:
45:
41:
37:
33:
148:cold finger
133:volatilizes
117:nickelocene
82:sublimation
66:sublimatory
329:Categories
265:2023-06-26
211:References
141:impurities
242:0021-1753
178:follow a
139:residual
78:compounds
194:See also
137:volatile
129:pressure
96:Overview
105:Camphor
292:
240:
125:vacuum
290:ISBN
238:ISSN
230:Isis
186:or
68:or
331::
274:^
258:.
234:51
232:.
228:.
190:.
92:.
64:A
298:.
268:.
244:.
58:7
54:6
50:5
46:4
42:3
38:2
34:1
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.