66:
58:
40:
47:
35:
73:
Antimycins are produced by a non-ribosomal peptide synthetase (NRPS)/polyketide synthase (PKS) assembly complex which acts as an assembly line for antimycin production. The assembly is genetically coded for by the ant gene family. The assembly requires 14 proteins, AntBCDEFGHIJKLMNO, which shuttle
136:
5. 3-Aminosalicylate is presented to the NRPS, AntC. AntC has two modules which are organized
Condensation1 (C1) -Adenylation1 (A1) -Thiolation1 (T1) -Condensation2 (C2) -Adenylation2 (A2) -Ketoreduction (KR) -Thiolation2 (T2). The A1 domain activates and loads threonine, an amino acid, onto T1,
269:
Yan, Yan; Zhang, Lihan; Ito, Takuya; Qu, Xudong; Asakawa, Yoshinori; Awakawa, Takayoshi; Abe, Ikuro; Liu, Wen (2012). "Biosynthetic
Pathway for High Structural Diversity of a Common Dilactone Core in Antimycin Production".
94:. The last two steps involving AntB and AntO are tailoring steps. The following steps describe chemically what the Ant Enzymes do in order to synthesize Antimycin. Synthesis begins with tryptophan, an amino acid.
169:
homologue, AntB, catalyze the installation of the N-formyl group and the transesterification of the C-8 hydroxyl group, respectively, resulting in the backbone for the
Antimycin family.
25:
bacteria, a soil bacteria. These specialized metabolites likely function to kill neighboring organisms in order to provide the streptomyces bacteria with a competitive edge.
101:
of tryptophan, an amino acid, is opened by a pathway-specific tryptophan-2.3-dioxygnease, AntN, to make N-formyl-L-kynurenine.
159:
7. AntM catalyses the reduction of the β-keto group, which precedes the AntD TE domain – promoted release of the nine-membered
369:
320:
Sandy, Moriah; Rui, Zhe; Gallagher, Joe; Zhang, Wenjun (2012). "Enzymatic
Synthesis of Dilactone Scaffold of Antimycins".
123:
149:
137:
followed by a C1 promoted condensation of 3-aminosalicylate and threonine. The A2 domain activates and loads
156:
attached to AntC T2 domain and the 2-carboxy-acyl moiety attached to AntD Acetyl
Carrier Protein domain.
87:
374:
75:
79:
345:
337:
295:
287:
251:
233:
145:
329:
279:
241:
225:
105:
166:
91:
246:
213:
65:
57:
363:
183:
138:
109:
39:
21:
141:
onto T2. Pyruvate is reduced by the KR domain and condensed with threonine by C2
178:
341:
291:
237:
153:
130:
349:
299:
255:
46:
116:
229:
160:
34:
333:
283:
129:
4. Anthranilate is converted to 3-aminosalicylate by a multicomponent
119:
98:
83:
64:
56:
74:
the intermediates along the assembly line through a series of
207:
205:
203:
201:
199:
214:"The regulation and biosynthesis of antimycins"
315:
313:
311:
309:
212:Seipke, Ryan F; Hutchings, Matthew I (2013).
8:
122:protein, AntF and loaded onto its cognate
104:2. N-formyl-L-kynurenine is converted to
245:
86:(addition of a sulfur containing group),
19:are produced as secondary metabolites by
195:
218:Beilstein Journal of Organic Chemistry
7:
115:3. Anthranilate is activated by the
14:
165:8. A lipase homologue, AntO, and
126:, AntG, for further processing.
45:
38:
33:
1:
150:decarboxylative condensation
148:domain of PKS catalyses the
391:
108:by the pathway-specific
152:between the aminoacyl
70:
62:
370:Secondary metabolites
68:
60:
322:ACS Chemical Biology
76:transesterifications
29:Chemical structures
230:10.3762/bjoc.9.290
71:
63:
334:10.1021/cb300416w
328:(12): 1956–1961.
284:10.1021/ol301785x
278:(16): 4142–4145.
382:
354:
353:
317:
304:
303:
266:
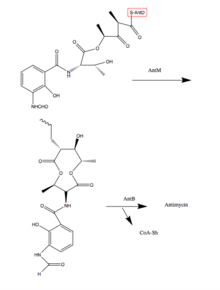
260:
259:
249:
209:
49:
42:
37:
390:
389:
385:
384:
383:
381:
380:
379:
360:
359:
358:
357:
319:
318:
307:
272:Organic Letters
268:
267:
263:
211:
210:
197:
192:
175:
167:acyltransferase
124:carrier protein
80:keto reductions
55:
31:
12:
11:
5:
388:
386:
378:
377:
372:
362:
361:
356:
355:
305:
261:
194:
193:
191:
188:
187:
186:
181:
174:
171:
54:
51:
30:
27:
13:
10:
9:
6:
4:
3:
2:
387:
376:
373:
371:
368:
367:
365:
351:
347:
343:
339:
335:
331:
327:
323:
316:
314:
312:
310:
306:
301:
297:
293:
289:
285:
281:
277:
273:
265:
262:
257:
253:
248:
243:
239:
235:
231:
227:
224:: 2556–2563.
223:
219:
215:
208:
206:
204:
202:
200:
196:
189:
185:
182:
180:
177:
176:
172:
170:
168:
163:
162:
157:
155:
151:
147:
142:
140:
134:
132:
127:
125:
121:
118:
113:
111:
107:
102:
100:
95:
93:
89:
88:condensations
85:
81:
77:
67:
59:
52:
50:
48:
43:
41:
36:
28:
26:
24:
23:
18:
325:
321:
275:
271:
264:
221:
217:
184:Urauchimycin
164:
158:
146:Ketosynthase
143:
135:
133:, AntHIJKL.
128:
114:
110:kynureninase
106:anthranilate
103:
96:
92:adenylations
72:
53:Biosynthesis
44:
32:
22:Streptomyces
20:
16:
15:
375:Antibiotics
179:Antimycin A
99:indole ring
84:thiolations
364:Categories
190:References
17:Antimycins
342:1554-8929
292:1523-7060
238:1860-5397
161:dilactone
154:thioester
131:oxygenase
350:22971101
300:22861048
256:24367419
173:See also
139:pyruvate
117:acyl-CoA
112:, AntP.
247:3869250
144:6. The
97:1. The
348:
340:
298:
290:
254:
244:
236:
120:ligase
90:, and
69:Part 2
61:Part 1
346:PMID
338:ISSN
296:PMID
288:ISSN
252:PMID
234:ISSN
330:doi
280:doi
242:PMC
226:doi
366::
344:.
336:.
324:.
308:^
294:.
286:.
276:14
274:.
250:.
240:.
232:.
220:.
216:.
198:^
82:,
78:,
352:.
332::
326:7
302:.
282::
258:.
228::
222:9
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.