89:
54:
307:
Kawasaki T, Tanaka H, Tsutsumi T, Kasahara T, Sato I, Soai K (May 2006). "Chiral discrimination of cryptochiral saturated quaternary and tertiary hydrocarbons by asymmetric autocatalysis".
110:)-bis(dimethylamino)butane, whose chirality results solely due to the difference between N and N (7% isotopic mass difference), gave 45% ee when used as a stoichiometric ligand.
256:
Shibata T, Morioka H, Hayase T, Choji K, Soai K (1996). "Highly
Enantioselective Catalytic Asymmetric Automultiplication of Chiral Pyrimidyl Alcohol".
138:
36:
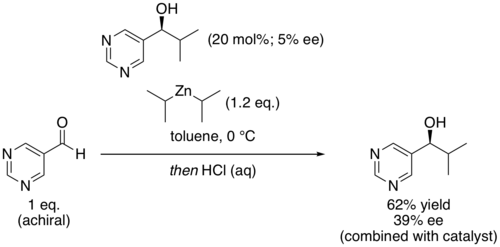
of the product. The product pyrimidyl alcohol is chiral and induces that same chirality in further catalytic cycles. Starting with a low
213:
Soai K, Shibata T, Morioka H, Choji K (1995). "Asymmetric autocatalysis and amplification of enantiomeric excess of a chiral molecule".
75:, with the major product of that first reaction being rapidly amplified. For example, Soai's group has demonstrated that even chiral
64:(1950–) discovered the reaction in 1995. For his work in "elucidating the origins of chirality and homochirality", Soai received the
479:
88:
40:("ee") produces a product with very high enantiomeric excess. The reaction has been studied for clues about the origin of
560:
449:
Micskei K, Rábai G, Gál E, Caglioti L, Pályi G (July 2008). "Oscillatory symmetry breaking in the Soai reaction".
95:
The chiral induction is believed to occur as a result of interactions between the C–H bonds of the alkane and the
65:
550:
283:
565:
83:
site for binding the nucleophile, are nonetheless capable of inducing asymmetric catalysis in the reaction.
29:
409:
222:
167:
72:
76:
37:
344:"Asymmetric Induction by a Nitrogen N/ N Isotopomer in Conjunction with Asymmetric Autocatalysis"
555:
238:
528:
466:
437:
396:
Islas JR, Lavabre D, Grevy JM, Lamoneda RH, Cabrera HR, Micheau JC, Buhse T (September 2005).
373:
324:
195:
134:
128:
518:
458:
427:
417:
363:
355:
316:
265:
230:
185:
175:
25:
342:
Matsumoto A, Ozaki H, Harada S, Tada K, Ayugase T, Ozawa H, et al. (December 2016).
494:
478:
Caglioti L, Hajdu C, Holczknecht O, Zékány L, Zucchi C, Micskei K, Pályi G (June 2006).
413:
226:
171:
432:
397:
368:
343:
190:
155:
544:
61:
41:
53:
242:
130:
Chirality and Life: A Short
Introduction to the Early Phases of Chemical Evolution
96:
80:
45:
402:
Proceedings of the
National Academy of Sciences of the United States of America
160:
Proceedings of the
National Academy of Sciences of the United States of America
156:"Asymmetric autocatalysis and its implications for the origin of homochirality"
33:
21:
422:
180:
532:
523:
506:
470:
441:
377:
359:
328:
199:
398:"Mirror-symmetry breaking in the Soai reaction: a kinetic understanding"
462:
320:
269:
234:
71:
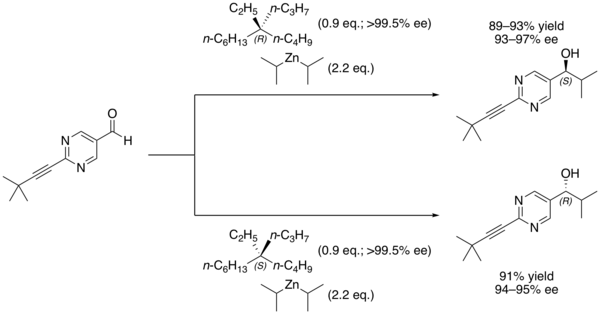
Other chiral additives can be used as the initial source of
102:
In another example, Soai and coworkers showed that even (
32:
and leads to rapidly increasing amounts of the same
507:"New aspects of Soai's asymmetric autocatalysis"
480:"The concept of racemates and the Soai reaction"
8:
302:
300:
522:
431:
421:
367:
189:
179:
309:Journal of the American Chemical Society
258:Journal of the American Chemical Society
505:Podlech J, Gehring T (September 2005).
119:
7:
451:The Journal of Physical Chemistry B
284:"CSJ Award 2010: Prof. Kenso Soai"
79:hydrocarbons, which have no clear
24:of pyrimidine-5-carbaldehyde with
14:
87:
52:
286:. The Chemical Society of Japan
127:Flügel RM (January 11, 2011).
1:
133:. Springer. pp. 16–17.
154:Blackmond DG (April 2004).
582:
16:In organic chemistry, the
66:Chemical Society of Japan
44:among certain classes of
493:: 62–80. Archived from
423:10.1073/pnas.0503171102
181:10.1073/pnas.0308363101
524:10.1002/anie.200501742
360:10.1002/anie.201608955
60:The Japanese chemist
73:asymmetric induction
414:2005PNAS..10213743I
354:(49): 15246–15249.
227:1995Natur.378..767S
172:2004PNAS..101.5732B
38:enantiomeric excess
500:on 12 August 2017.
28:. The reaction is
561:Organic reactions
511:Angewandte Chemie
463:10.1021/jp803334b
348:Angewandte Chemie
321:10.1021/ja061429e
270:10.1021/ja953066g
221:(6559): 767–768.
140:978-3-642-16977-9
99:of the aldehyde.
573:
536:
526:
501:
499:
484:
474:
457:(30): 9196–200.
445:
435:
425:
382:
381:
371:
339:
333:
332:
304:
295:
294:
292:
291:
280:
274:
273:
253:
247:
246:
235:10.1038/378767a0
210:
204:
203:
193:
183:
151:
145:
144:
124:
91:
56:
581:
580:
576:
575:
574:
572:
571:
570:
551:Stereochemistry
541:
540:
539:
504:
497:
482:
477:
448:
408:(39): 13743–8.
395:
391:
389:Further reading
386:
385:
341:
340:
336:
306:
305:
298:
289:
287:
282:
281:
277:
255:
254:
250:
212:
211:
207:
153:
152:
148:
141:
126:
125:
121:
116:
68:award in 2010.
26:diisopropylzinc
12:
11:
5:
579:
577:
569:
568:
566:Name reactions
563:
558:
553:
543:
542:
538:
537:
517:(36): 5776–7.
502:
475:
446:
392:
390:
387:
384:
383:
334:
315:(18): 6032–3.
296:
275:
264:(2): 471–472.
248:
205:
166:(16): 5732–6.
146:
139:
118:
117:
115:
112:
93:
92:
58:
57:
13:
10:
9:
6:
4:
3:
2:
578:
567:
564:
562:
559:
557:
554:
552:
549:
548:
546:
534:
530:
525:
520:
516:
512:
508:
503:
496:
492:
488:
481:
476:
472:
468:
464:
460:
456:
452:
447:
443:
439:
434:
429:
424:
419:
415:
411:
407:
403:
399:
394:
393:
388:
379:
375:
370:
365:
361:
357:
353:
349:
345:
338:
335:
330:
326:
322:
318:
314:
310:
303:
301:
297:
285:
279:
276:
271:
267:
263:
259:
252:
249:
244:
240:
236:
232:
228:
224:
220:
216:
209:
206:
201:
197:
192:
187:
182:
177:
173:
169:
165:
161:
157:
150:
147:
142:
136:
132:
131:
123:
120:
113:
111:
109:
105:
100:
98:
90:
86:
85:
84:
82:
78:
74:
69:
67:
63:
55:
51:
50:
49:
47:
43:
42:homochirality
39:
35:
31:
30:autocatalytic
27:
23:
19:
18:Soai reaction
514:
510:
495:the original
490:
486:
454:
450:
405:
401:
351:
347:
337:
312:
308:
288:. Retrieved
278:
261:
257:
251:
218:
214:
208:
163:
159:
149:
129:
122:
107:
103:
101:
97:pi electrons
94:
70:
59:
46:biomolecules
17:
15:
81:Lewis basic
545:Categories
290:2015-04-14
114:References
77:quaternary
62:Kensō Soai
34:enantiomer
22:alkylation
556:Catalysis
487:Viva Orig
533:16078286
471:18593153
442:16174731
378:27754589
329:16669661
200:15067112
433:1236534
410:Bibcode
369:5132014
243:4258847
223:Bibcode
168:Bibcode
20:is the
531:
469:
440:
430:
376:
366:
327:
241:
215:Nature
198:
191:395976
188:
137:
498:(PDF)
483:(PDF)
239:S2CID
529:PMID
467:PMID
438:PMID
374:PMID
325:PMID
196:PMID
135:ISBN
519:doi
459:doi
455:112
428:PMC
418:doi
406:102
364:PMC
356:doi
317:doi
313:128
266:doi
262:118
231:doi
219:378
186:PMC
176:doi
164:101
106:, 3
547::
527:.
515:44
513:.
509:.
491:34
489:.
485:.
465:.
453:.
436:.
426:.
416:.
404:.
400:.
372:.
362:.
352:55
350:.
346:.
323:.
311:.
299:^
260:.
237:.
229:.
217:.
194:.
184:.
174:.
162:.
158:.
104:2R
48:.
535:.
521::
473:.
461::
444:.
420::
412::
380:.
358::
331:.
319::
293:.
272:.
268::
245:.
233::
225::
202:.
178::
170::
143:.
108:S
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.