491:
475:
17:
520:
550:
193:
1153:
240:
1192:
288:
966:
temperature at which equilibrium will be reached in a reasonable amount of time. If needed, the selectivity can be increased by then slowly cooling the reaction mixture to shift the equilibrium further toward the most stable product. When the difference in product stability is very large, the thermodynamically controlled product can dominate even under relatively vigorous reaction conditions.
756:
well approximated as operating under kinetic control, due to negligibly slow equilibration. For example, many enantioselective catalytic systems provide nearly enantiopure product (> 99% ee), even though the enantiomeric products have the same Gibbs free energy and are equally favored thermodynamically.
121:
asymmetric transformations, the enantiomeric products are actually formed as a complex with the chirality source before the workup stage of the reaction, technically making the reaction a diastereoselective one. Although such reactions are still usually kinetically controlled, thermodynamic control
486:
If a much weaker base is used, the deprotonation will be incomplete, and there will be an equilibrium between reactants and products. Thermodynamic control is obtained, however the reaction remains incomplete unless the product enolate is trapped, as in the example below. Since H transfers are very
470:
between the base and the enolate is so large that the reaction is essentially irreversible, so the equilibration leading to the thermodynamic product is likely a proton exchange occurring during the addition between the kinetic enolate and as-yet-unreacted ketone. An inverse addition (adding ketone
755:
Unless equilibration is prevented (e.g., by removal of the product from the reaction mixture as soon as it forms), "pure" kinetic control is strictly speaking impossible, because some amount of equilibration will take place before the reactants are entirely consumed. In practice, many systems are
78:
The conditions of the reaction, such as temperature, pressure, or solvent, affect which reaction pathway may be favored: either the kinetically controlled or the thermodynamically controlled one. Note this is only true if the activation energy of the two pathways differ, with one pathway having a
563:
In principle, every reaction is on the continuum between pure kinetic control and pure thermodynamic control. These terms are with respect to a given temperature and time scale. A process approaches pure kinetic control at low temperature and short reaction time. For a sufficiently long time
1291:
Kseniya K. Borisova, Eugeniya V. Nikitina, Roman A. Novikov, Victor N. Khrustalev, Pavel V. Dorovatovskii, Yan V. Zubavichus, Maxim L. Kuznetsov, Vladimir P. Zaytsev, Alexey V. Varlamov and Fedor I. Zubkov. “Diels–Alder reactions between hexafluoro-2-butyne and bis-furyl dienes: kinetic versus
1278:
Kseniya K. Borisova, Elizaveta A. Kvyatkovskaya, Eugeniya V. Nikitina, Rinat R. Aysin, Roman A. Novikov, and Fedor I. Zubkov. “A Classical
Example of Total Kinetic and Thermodynamic Control. The Diels-Alder Reaction between DMAD and Bis-furyl Dienes.” J. Org. Chem., 2018, 83 (8), pp 4840-4850.
965:
is in the denominator in both cases. The ideal temperature to optimise the yield of the fastest-forming product will be the lowest temperature that will ensure reaction completion in a reasonable amount of time. The ideal temperature for a reaction under thermodynamic control is the lowest
748:
574:
A necessary condition for thermodynamic control is reversibility or a mechanism permitting the equilibration between products. Reactions are considered to take place under thermodynamic reaction control when the reverse reaction is sufficiently rapid that the
910:
490:
104:
is a field in which the distinction between kinetic and thermodynamic control is especially important. Because pairs of enantiomers have, for all intents and purposes, the same Gibbs free energy, thermodynamic control will produce a
515:
above room temperature leads predominantly to the thermodynamically more stable 1,4 adduct, 1-bromo-2-butene, but decreasing the reaction temperature to below room temperature favours the kinetic 1,2 adduct, 3-bromo-1-butene.
93:
Prevalence of thermodynamic or kinetic control determines the final composition of the product when these competing reaction pathways lead to different products. The reaction conditions as mentioned above influence the
1387:
Khopade, Tushar; Mete, Trimbak; Arora, Jyotsna; Bhat, Ramakrishna (2018). "An
Adverse Effect of Higher Catalyst Loading and Longer Reaction Time on Enantioselectivity in an Organocatalytic Multicomponent Reaction".
960:
In general, short reaction times favour kinetic control, whereas longer reaction times favour thermodynamic reaction control. Low temperatures will enhance the selectivity under both sets of conditions, since
540:
cation. The 1,4 adduct places the larger Br atom at a less congested site and includes a more highly substituted alkene moiety, while the 1,2 adduct is the result of the attack by the nucleophile (Br) at the
474:
192:
608:
582:
Under kinetic reaction control, one or both forward reactions leading to the possible products is significantly faster than the equilibration between the products. After reaction time
519:
786:
1081:
In the same way, one can detect the possibility of kinetic control if a temperature change causes a change in the product ratio that is inconsistent with equation 2, assuming that
1566:
456:
resulting from removal of the most accessible α-H while the thermodynamic product has the more highly substituted enolate moiety. Use of low temperatures and sterically demanding
1638:
1608:
1109:
239:
972:
In the same manner, if a reaction is under kinetic control at a given temperature, it will also be under kinetic control at any lower temperature for the same reaction time.
1044:
1536:
1074:
988:
changes with temperature but is not consistent with equation 1, that is a change in temperature (without changing the reaction time) causes a change in the product ratio
979:
under kinetic control, one can detect the presence of an equilibration mechanism (and therefore the possibility of thermodynamic control) if the product distribution:
969:
If a reaction is under thermodynamic control at a given temperature, it will also be under thermodynamic control at a higher temperature for the same reaction time.
1182:
in acetic acid was found to give a mixture of the gamma and the alpha acetate with the latter converting to the first by equilibration. This was interpreted as a
1644:, for instance if both were fragmentations of a molecule to produce a pair of molecules or if both were condensations of two molecules to give a single molecule.
1184:
case in the field of anionotropy of the phenomenon, familiar in prototropy, of the distinction between kinetic and thermodynamic control in ion-recombination
549:
1146:
considered in the light of the remarkable stability of the exo-compound on the one hand and the very facile dissociation of the endo isomer on the other.
35:
can decide the composition in a reaction product mixture when competing pathways lead to different products and the reaction conditions influence the
1142:
while the endo isomer is formed more rapidly, longer reaction times, as well as relatively elevated temperatures, result in higher exo / endo ratios
915:
In principle, "pure" thermodynamic control is also impossible, since equilibrium is only achieved after infinite reaction time. In practice, if
761:
Under pure thermodynamic reaction control, when the equilibrium has been reached, the product distribution will be a function of the stabilities
564:
scale, every reaction approaches pure thermodynamic control, at least in principle. This time scale becomes shorter as the temperature is raised.
545:
of the allylic cation bearing the greatest positive charge (the more highly substituted carbon is the most likely place for the positive charge).
1674:
Synthesen in der hydroaromatischen Reihe, IV. Mitteilung: Über die
Anlagerung von Maleinsäure-anhydrid an arylierte Diene, Triene und Fulvene
1267:
1191:
1152:
1480:
Unless one is content with an incomplete reaction, whence a separation of product from unreacted starting material may be necessary.
1251:
1232:
1216:
985:
shows one product to be dominant at one temperature while another dominates at a different temperature (inversion of dominance), or
1304:
471:
to the base) with rapid mixing would minimize this. The position of the equilibrium will depend on the countercation and solvent.
1748:
579:
establishes itself within the allotted reaction time. In this way, the thermodynamically more stable product is always favoured.
217:
1425:"Direction of Kinetically versus Thermodynamically Controlled Organocatalysis and Its Application in Chemoenzymatic Synthesis"
955:), or approximately five half-lives, and the system product ratio can be regarded as the result of thermodynamic control.
743:{\displaystyle \ln \left({\frac {_{t}}{_{t}}}\right)=\ln \left({\frac {k_{A}}{k_{B}}}\right)=-{\frac {\Delta E_{a}}{RT}}}
1170:
independently described a thermodynamic and kinetic reaction control model in 1948. They were reinvestigating a certain
1693:
Rearrangement and substitution in anionotropic systems. Part III. Mechanism of, and equilibrium in, anionotropic change
487:
fast, the trapping reaction being slower, the ratio of trapped products largely mirrors the deprotonation equilibrium.
1738:
101:
1262:
Advanced
Organic Chemistry Part A: Structure and Mechanisms, 5th ed., Francis A. Carey, Richard J. Sundberg, 2007
1423:
Rulli, Giuseppe; Duangdee, Nongnaphat; Baer, Katrin; Hummel, Werner; Berkessel, Albrecht; Gröger, Harald (2011).
1676:(Mitbearbeitet von Paul Pries). Berichte der deutschen chemischen Gesellschaft (A and B Series), 62: 2081–2087.
1293:
136:
905:{\displaystyle \ln \left({\frac {_{\infty }}{_{\infty }}}\right)=\ln \ K_{eq}=-{\frac {\Delta G^{\circ }}{RT}}}
1743:
272:
1541:
504:
36:
1654:
Studies on Diene-addition
Reactions. II.1 The Reaction of 6,6-Pentamethylenefulvene with Maleic Anhydride
1568:
is small, which would be the case if the rate-determining steps leading to each product were of the same
1171:
533:
16:
1613:
1583:
1084:
529:
1227:
A Guidebook to
Mechanism in Organic Chemistry, 6th Edition, Peter Sykes, Pearson Prentice Hall, 1986.
567:
In every reaction, the first product formed is that which is most easily formed. Thus, every reaction
1046:
that is larger or smaller than would be expected from the change in temperature alone, assuming that
766:
576:
163:
87:
991:
479:
248:
213:
114:
1640:
is small, which would be the case if the overall transformations to each product were of the same
1511:
1167:
1163:
1049:
1500:
rises and the proportion of the most stable product will tend toward 50% of the reaction mixture.
1454:
1175:
765:°. After an infinite amount of reaction time, the ratio of product concentrations will equal the
426:
1324:
1370:
Herbert O. House, Ben A. Tefertiller, Hugh D. Olmstead J. Org. Chem., 1968, 33 (3), pp 935–942
1344:
The
Chemistry of Carbanions. IX. The Potassium and Lithium Enolates Derived from Cyclic Ketones
1446:
1405:
1320:
1263:
1247:
1228:
1212:
1179:
776:
277:
95:
52:
40:
32:
1716:
1696:
1677:
1657:
1436:
1397:
1371:
1351:
1328:
1125:
508:
457:
438:
229:
221:
205:
186:
152:
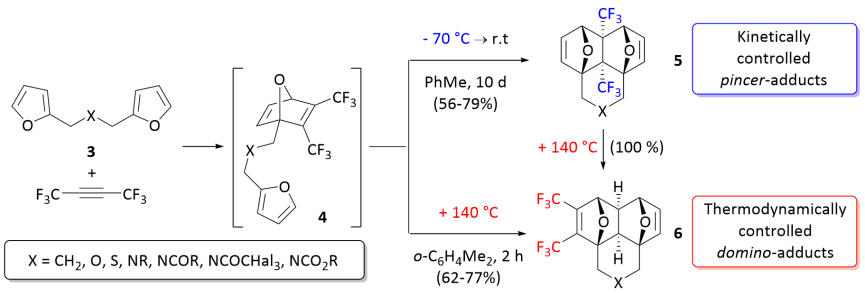
220:(DMAD) have been discovered and described in 2018. At low temperature, the reactions occur
140:
106:
1211:
Organic
Chemistry, 3rd ed., M. A. Fox & J. K. Whitesell, Jones & Bartlett, 2004
1713:Über die Verschiebung in der Allyl-Gruppe. 3. Mitteilung über Substitution und Addition
1347:
1280:
1246:
Introduction to
Organic Chemistry I, Seth Robert Elsheimer, Blackwell Publishing, 2000
1120:
The first to report on the relationship between kinetic and thermodynamic control were
937:, then for most practical purposes, the change in composition becomes negligible after
528:
The rationale for the differing selectivities is as follows: Both products result from
494:
The thermodynamic deprotonation of 2-methylcyclohexanone, with trapping of the enolate.
178:
1292:
thermodynamic control.” Chem. Commun., 2018, 54, pp 2850-2853. doi:10.1039/c7cc09466c
196:
Thermodynamic versus kinetic reaction control in reaction of cyclopanetdiene and furan
1732:
1332:
1121:
512:
445:
379:
1458:
441:, even in trace amounts, in this case mediated by the enolate or the proton source.
1641:
1569:
1159:
347:. The calculations showed that the first channel is more kinetically favourable (Δ
410:
406:
156:
287:
162:
is the main reaction product. At 81 °C and after long reaction times, the
1137:
1133:
167:
1720:
1681:
1294:
http://pubs.rsc.org/en/content/articlelanding/2018/cc/c7cc09466c#!divAbstract
371:
at elevated temperature. Indeed, the calculated activation barriers for the
1450:
1441:
1424:
1409:
1401:
299:
and hexafluoro-2-butyne. Relative energies are shown in kcal/mol for X = CH
75:
is the thermodynamic product and is favoured under thermodynamic control.
1700:
1695:
A. G. Catchpole, E. D. Hughes and C. K. Ingold J. Chem. Soc., 1948, 8-17
1471:
Only if a subsequent equilibration is as fast or faster is this not true.
422:
148:
20:
Energy profile diagram for kinetic versus thermodynamic product reaction.
1661:
1656:
R. B. Woodward, Harold Baer J. Am. Chem. Soc., 1944, 66 (4), pp 645–649
1375:
1355:
389:
followed by the intramolecular -cycloaddition in the chain intermediate
1129:
1124:
and Harold Baer in 1944. They were re-investigating a reaction between
537:
453:
1111:
is largely invariant with temperature over a modest temperature range.
1076:
is largely invariant with temperature over a modest temperature range.
523:
The addition of HBr to butadiene in ether. Data from
Elsheimer (2000).
238:
542:
449:
430:
418:
243:
Kinetic and thermodynamic control of the tandem Diels–Alder reaction.
276:
and represents the rate limiting step of the whole process with the
1572:, for instance if both involved collisions with the same reactant.
286:
144:
204:
kinetic and thermodynamic reaction control in the process of the
71:
is the kinetic product and is favoured under kinetic control and
434:
414:
259:
were performed. The reaction starting with cycloaddition of CF
208:
inter-/intramolecular Diels–Alder reaction of bis-furyl dienes
461:
1317:
Ketone enolates: regiospecific preparation and synthetic uses
590:
and thus a function of the difference in activation energies
363:≈ 4.2-4.7 kcal/mol) and this fact may cause isomerization of
548:
518:
489:
473:
191:
1319:, Tetrahedron, Volume 32, Issue 24, 1976, Pages 2979-2990,
460:
increases the kinetic selectivity. Here, the difference in
267:
at one of the furan moieties occurs in a concerted fashion
1368:
Stereochemistry of alkylation of 4-tert-butylcyclohexanone
224:
leading exclusively to adducts of pincer- cycloaddition (
166:
can assert itself and the thermodynamically more stable
155:, kinetic reaction control prevails and the less stable
177:
product is more stable by virtue of a lower degree of
1616:
1586:
1544:
1514:
1087:
1052:
994:
789:
611:
247:
Theoretical DFT calculations of the reaction between
117:
is under at least partial kinetic control. (In many
1281:
https://pubs.acs.org/doi/abs/10.1021/acs.joc.8b00336
351:≈ 5.7–5.9 kcal/mol). Meanwhile, the domino products
337:
or resulting in the formation of the domino product
1715:. Justus Liebigs Annalen der Chemie, 479: 211–277.
586:, the product ratio is the ratio of rate constants
291:Gibbs free energy profile for the reaction between
1632:
1602:
1560:
1530:
1315:Jean d'Angelo, Tetrahedron report number 25 :
1103:
1068:
1038:
904:
742:
775:and therefore be a function of the difference in
478:The kinetic and thermodynamic deprotonation of 2-
1610:will be temperature-independent or nearly so if
1538:will be temperature-independent or nearly so if
1178:. Solvolysis of gamma-phenylallyl chloride with
98:of the reaction - i.e., which pathway is taken.
1242:
1240:
185:product is favoured by orbital overlap in the
8:
975:If one presumes that a new reaction will be
200:An outstanding and very rare example of the
113:reaction that provides product with nonzero
327:either leading to the pincer type products
43:. The distinction is relevant when product
1350:J. Org. Chem., 1965, 30 (5), pp 1341–1348
532:protonation at position 1, resulting in a
1624:
1615:
1594:
1585:
1552:
1543:
1522:
1513:
1440:
1095:
1086:
1060:
1051:
1029:
1018:
1013:
1006:
995:
993:
923:interconvert with overall rate constants
885:
875:
860:
831:
813:
800:
788:
723:
713:
695:
685:
679:
653:
635:
622:
610:
1305:Thermodynamic Product vs Kinetic Product
236:) is observed at elevated temperatures.
15:
1429:Angewandte Chemie International Edition
1204:
429:and their enols interchange rapidly by
355:are more thermodynamically stable than
1711:Meisenheimer, J. and Link, J. (1930),
122:is at least possible, in principle.)
7:
1561:{\displaystyle \Delta S^{\ddagger }}
912: (equation 2)
750: (equation 1)
553:butadiene hydrobromination mechanism
319:Further, the reaction could proceed
417:and the thermodynamic product is a
1617:
1587:
1545:
1515:
1088:
1053:
878:
832:
814:
716:
385:the retro-Diels–Alder reaction of
14:
1633:{\displaystyle \Delta S^{\circ }}
1603:{\displaystyle \Delta G^{\circ }}
1104:{\displaystyle \Delta G^{\circ }}
1672:Diels, O. and Alder, K. (1929),
1190:
1151:
218:dimethyl acetylenedicarboxylate
67:is more stable. In such a case
59:is lower than that for product
1026:
1019:
1003:
996:
828:
821:
810:
803:
650:
643:
632:
625:
228:). The exclusive formation of
25:Thermodynamic reaction control
1:
1390:Chemistry: A European Journal
1366:Chemistry of carbanions. XV.
1039:{\displaystyle {_{t}}/{_{t}}}
571:starts under kinetic control.
452:, the kinetic product is the
413:, the kinetic product is the
1531:{\displaystyle \Delta E_{a}}
1333:10.1016/0040-4020(76)80156-1
1279:doi:10.1021/acs.joc.8b00336
1069:{\displaystyle \Delta E_{a}}
1765:
1132:first reported in 1929 by
499:In electrophilic additions
47:forms faster than product
109:by necessity. Thus, any
1721:10.1002/jlac.19304790114
1682:10.1002/cber.19290620829
397:are 34.0–34.4 kcal/mol.
323:two competing channels,
131:In Diels–Alder reactions
29:kinetic reaction control
1749:Chemical thermodynamics
433:transfers catalyzed by
1634:
1604:
1562:
1532:
1442:10.1002/anie.201008042
1402:10.1002/chem.201800278
1105:
1070:
1040:
906:
744:
554:
524:
505:electrophilic addition
495:
483:
316:
284:≈ 23.1–26.8 kcal/mol.
244:
197:
21:
1635:
1605:
1563:
1533:
1172:allylic rearrangement
1140:. They observed that
1106:
1071:
1041:
907:
745:
552:
522:
493:
477:
290:
242:
195:
19:
1701:10.1039/JR9480000008
1614:
1584:
1542:
1512:
1174:reported in 1930 by
1085:
1050:
992:
787:
767:equilibrium constant
609:
448:of an unsymmetrical
401:In enolate chemistry
164:chemical equilibrium
137:Diels–Alder reaction
102:Asymmetric synthesis
88:energy of activation
1662:10.1021/ja01232a042
1496:will approach 1 as
1376:10.1021/jo01267a002
1356:10.1021/jo01016a001
777:Gibbs free energies
480:methylcyclohexanone
249:hexafluoro-2-butyne
214:hexafluoro-2-butyne
115:enantiomeric excess
1739:Chemical reactions
1630:
1600:
1558:
1528:
1346:Herbert O. House,
1176:Jakob Meisenheimer
1101:
1066:
1036:
982:changes over time,
902:
740:
555:
525:
496:
484:
427:Carbonyl compounds
317:
278:activation barrier
245:
198:
90:) than the other.
22:
1435:(34): 7944–7947.
1396:(23): 6036–6040.
1268:978-0-387-44899-2
900:
855:
838:
738:
701:
660:
303:(plain text), S (
179:steric congestion
53:activation energy
41:stereoselectivity
33:chemical reaction
1756:
1723:
1709:
1703:
1690:
1684:
1670:
1664:
1651:
1645:
1639:
1637:
1636:
1631:
1629:
1628:
1609:
1607:
1606:
1601:
1599:
1598:
1579:
1573:
1567:
1565:
1564:
1559:
1557:
1556:
1537:
1535:
1534:
1529:
1527:
1526:
1507:
1501:
1487:
1481:
1478:
1472:
1469:
1463:
1462:
1444:
1420:
1414:
1413:
1384:
1378:
1364:
1358:
1341:
1335:
1313:
1307:
1302:
1296:
1289:
1283:
1276:
1270:
1260:
1254:
1244:
1235:
1225:
1219:
1209:
1194:
1155:
1144:which had to be
1126:maleic anhydride
1110:
1108:
1107:
1102:
1100:
1099:
1075:
1073:
1072:
1067:
1065:
1064:
1045:
1043:
1042:
1037:
1035:
1034:
1033:
1017:
1012:
1011:
1010:
911:
909:
908:
903:
901:
899:
891:
890:
889:
876:
868:
867:
853:
843:
839:
837:
836:
835:
819:
818:
817:
801:
749:
747:
746:
741:
739:
737:
729:
728:
727:
714:
706:
702:
700:
699:
690:
689:
680:
665:
661:
659:
658:
657:
641:
640:
639:
623:
509:hydrogen bromide
222:chemoselectively
187:transition state
153:room temperature
147:can produce two
1764:
1763:
1759:
1758:
1757:
1755:
1754:
1753:
1729:
1728:
1727:
1726:
1710:
1706:
1691:
1687:
1671:
1667:
1652:
1648:
1620:
1612:
1611:
1590:
1582:
1581:
1580:
1576:
1548:
1540:
1539:
1518:
1510:
1509:
1508:
1504:
1495:
1488:
1484:
1479:
1475:
1470:
1466:
1422:
1421:
1417:
1386:
1385:
1381:
1365:
1361:
1342:
1338:
1314:
1310:
1303:
1299:
1290:
1286:
1277:
1273:
1261:
1257:
1245:
1238:
1226:
1222:
1210:
1206:
1201:
1118:
1091:
1083:
1082:
1056:
1048:
1047:
1025:
1002:
990:
989:
954:
947:
936:
929:
892:
881:
877:
856:
827:
820:
809:
802:
796:
785:
784:
774:
730:
719:
715:
691:
681:
675:
649:
642:
631:
624:
618:
607:
606:
596:
560:
558:Characteristics
501:
468:
403:
310:
302:
266:
262:
173:is formed. The
141:cyclopentadiene
133:
128:
107:racemic mixture
85:
12:
11:
5:
1762:
1760:
1752:
1751:
1746:
1744:Thermodynamics
1741:
1731:
1730:
1725:
1724:
1704:
1685:
1665:
1646:
1627:
1623:
1619:
1597:
1593:
1589:
1574:
1555:
1551:
1547:
1525:
1521:
1517:
1502:
1493:
1482:
1473:
1464:
1415:
1379:
1359:
1348:Barry M. Trost
1336:
1308:
1297:
1284:
1271:
1255:
1236:
1220:
1203:
1202:
1200:
1197:
1196:
1195:
1157:
1156:
1117:
1114:
1113:
1112:
1098:
1094:
1090:
1079:
1078:
1077:
1063:
1059:
1055:
1032:
1028:
1024:
1021:
1016:
1009:
1005:
1001:
998:
986:
983:
973:
970:
967:
957:
956:
952:
945:
934:
927:
913:
898:
895:
888:
884:
880:
874:
871:
866:
863:
859:
852:
849:
846:
842:
834:
830:
826:
823:
816:
812:
808:
805:
799:
795:
792:
781:
780:
772:
758:
757:
752:
751:
736:
733:
726:
722:
718:
712:
709:
705:
698:
694:
688:
684:
678:
674:
671:
668:
664:
656:
652:
648:
645:
638:
634:
630:
627:
621:
617:
614:
603:
602:
594:
580:
572:
565:
559:
556:
547:
546:
500:
497:
466:
402:
399:
308:
300:
264:
260:
132:
129:
127:
124:
119:stoichiometric
83:
63:, yet product
13:
10:
9:
6:
4:
3:
2:
1761:
1750:
1747:
1745:
1742:
1740:
1737:
1736:
1734:
1722:
1718:
1714:
1708:
1705:
1702:
1698:
1694:
1689:
1686:
1683:
1679:
1675:
1669:
1666:
1663:
1659:
1655:
1650:
1647:
1643:
1625:
1621:
1595:
1591:
1578:
1575:
1571:
1553:
1549:
1523:
1519:
1506:
1503:
1499:
1492:
1486:
1483:
1477:
1474:
1468:
1465:
1460:
1456:
1452:
1448:
1443:
1438:
1434:
1430:
1426:
1419:
1416:
1411:
1407:
1403:
1399:
1395:
1391:
1383:
1380:
1377:
1373:
1369:
1363:
1360:
1357:
1353:
1349:
1345:
1340:
1337:
1334:
1330:
1326:
1322:
1318:
1312:
1309:
1306:
1301:
1298:
1295:
1288:
1285:
1282:
1275:
1272:
1269:
1265:
1259:
1256:
1253:
1252:0-632-04417-9
1249:
1243:
1241:
1237:
1234:
1233:0-582-44695-3
1230:
1224:
1221:
1218:
1217:0-7637-2197-2
1214:
1208:
1205:
1198:
1193:
1189:
1188:
1187:
1185:
1181:
1177:
1173:
1169:
1165:
1161:
1154:
1150:
1149:
1148:
1147:
1143:
1139:
1135:
1131:
1127:
1123:
1122:R.B. Woodward
1115:
1096:
1092:
1080:
1061:
1057:
1030:
1022:
1014:
1007:
999:
987:
984:
981:
980:
978:
974:
971:
968:
964:
959:
958:
951:
944:
940:
933:
926:
922:
918:
914:
896:
893:
886:
882:
872:
869:
864:
861:
857:
850:
847:
844:
840:
824:
806:
797:
793:
790:
783:
782:
778:
771:
768:
764:
760:
759:
754:
753:
734:
731:
724:
720:
710:
707:
703:
696:
692:
686:
682:
676:
672:
669:
666:
662:
654:
646:
636:
628:
619:
615:
612:
605:
604:
600:
593:
589:
585:
581:
578:
573:
570:
566:
562:
561:
557:
551:
544:
539:
535:
531:
527:
526:
521:
517:
514:
513:1,3-butadiene
510:
506:
498:
492:
488:
481:
476:
472:
469:
465:
459:
455:
451:
447:
446:deprotonation
442:
440:
436:
432:
428:
424:
420:
416:
412:
408:
400:
398:
396:
392:
388:
384:
381:
380:isomerization
378:
374:
370:
366:
362:
358:
354:
350:
346:
343:
340:
336:
333:
330:
326:
322:
314:
307:) and NC(O)CF
306:
298:
294:
289:
285:
283:
279:
275:
274:
270:
258:
254:
250:
241:
237:
235:
231:
227:
223:
219:
215:
211:
207:
203:
194:
190:
188:
184:
180:
176:
172:
169:
165:
161:
158:
154:
151:products. At
150:
146:
142:
138:
130:
125:
123:
120:
116:
112:
108:
103:
99:
97:
91:
89:
82:
76:
74:
70:
66:
62:
58:
54:
50:
46:
42:
38:
34:
30:
26:
18:
1712:
1707:
1692:
1688:
1673:
1668:
1653:
1649:
1642:molecularity
1577:
1570:molecularity
1505:
1497:
1490:
1485:
1476:
1467:
1432:
1428:
1418:
1393:
1389:
1382:
1367:
1362:
1343:
1339:
1316:
1311:
1300:
1287:
1274:
1258:
1223:
1207:
1183:
1168:G. Catchpole
1164:E. D. Hughes
1160:C. K. Ingold
1158:
1145:
1141:
1119:
976:
962:
949:
942:
938:
931:
924:
920:
916:
769:
762:
598:
591:
587:
583:
568:
536:-stabilized
507:reaction of
502:
485:
463:
443:
404:
394:
390:
386:
382:
376:
372:
368:
364:
360:
356:
352:
348:
344:
341:
338:
334:
331:
328:
324:
320:
318:
312:
304:
296:
292:
281:
271:
268:
256:
252:
246:
233:
225:
209:
201:
199:
182:
181:, while the
174:
170:
159:
134:
118:
110:
100:
92:
80:
77:
72:
68:
64:
60:
56:
55:for product
51:because the
48:
44:
28:
24:
23:
577:equilibrium
530:Markovnikov
411:enolate ion
407:protonation
251:and dienes
157:endo isomer
96:selectivity
37:selectivity
1733:Categories
1489:At worst,
1199:References
1138:Kurt Alder
1134:Otto Diels
232:-adducts (
168:exo isomer
1626:∘
1618:Δ
1596:∘
1588:Δ
1554:‡
1546:Δ
1516:Δ
1325:0040-4020
1097:∘
1089:Δ
1054:Δ
887:∘
879:Δ
873:−
851:
833:∞
815:∞
794:
717:Δ
711:−
673:
616:
534:resonance
111:catalytic
1459:42971817
1451:21744441
1410:29465758
977:a priori
569:a priori
423:aldehyde
393:to give
295:-dienes
149:isomeric
1130:fulvene
1116:History
941:~ 3.5/(
538:allylic
454:enolate
444:In the
405:In the
1457:
1449:
1408:
1323:
1266:
1250:
1231:
1215:
1128:and a
854:
543:carbon
450:ketone
431:proton
419:ketone
409:of an
305:italic
230:domino
206:tandem
79:lower
1455:S2CID
1162:with
458:bases
439:bases
435:acids
367:into
263:C≡CCF
212:with
145:furan
143:with
126:Scope
31:in a
1447:PMID
1406:PMID
1321:ISSN
1264:ISBN
1248:ISBN
1229:ISBN
1213:ISBN
1180:AcOK
1166:and
1136:and
930:and
919:and
597:or Δ
503:The
415:enol
345:TS2t
335:TS2k
325:i.e.
313:bold
297:3a-c
202:full
183:endo
135:The
1717:doi
1697:doi
1678:doi
1658:doi
1437:doi
1398:doi
1372:doi
1352:doi
1329:doi
511:to
437:or
421:or
383:via
342:via
332:via
321:via
293:bis
273:TS1
269:via
216:or
189:.
175:exo
139:of
39:or
27:or
1735::
1494:eq
1453:.
1445:.
1433:50
1431:.
1427:.
1404:.
1394:24
1392:.
1327:,
1239:^
1186:.
948:+
848:ln
791:ln
773:eq
670:ln
613:ln
425:.
375:→
359:(Δ
315:).
253:3a
1719::
1699::
1680::
1660::
1622:S
1592:G
1550:S
1524:a
1520:E
1498:T
1491:K
1461:.
1439::
1412:.
1400::
1374::
1354::
1331::
1093:G
1062:a
1058:E
1031:t
1027:]
1023:B
1020:[
1015:/
1008:t
1004:]
1000:A
997:[
963:T
953:r
950:k
946:f
943:k
939:t
935:r
932:k
928:f
925:k
921:B
917:A
897:T
894:R
883:G
870:=
865:q
862:e
858:K
845:=
841:)
829:]
825:B
822:[
811:]
807:A
804:[
798:(
779:,
770:K
763:G
735:T
732:R
725:a
721:E
708:=
704:)
697:B
693:k
687:A
683:k
677:(
667:=
663:)
655:t
651:]
647:B
644:[
637:t
633:]
629:A
626:[
620:(
601::
599:G
595:a
592:E
588:k
584:t
482:.
467:b
464:K
462:p
395:6
391:4
387:5
377:6
373:5
369:6
365:5
361:G
357:5
353:6
349:G
339:6
329:5
311:(
309:3
301:2
282:G
280:Δ
265:3
261:3
257:c
255:-
234:6
226:5
210:3
171:1
160:2
86:(
84:a
81:E
73:B
69:A
65:B
61:B
57:A
49:B
45:A
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.